| Method no: |
PV2075 |
| |
| Control no: |
T-PV2075-01-8911-M |
|
|
| Matrix: |
Air |
| |
| Target concentration: |
0.5 ppm, 2 mg/m3 (OSHA TWA PEL) |
| |
| Procedure: |
Samples are collected by drawing a known volume
of air through a glass fiber filter impregnated with mercuric acetate.
Phenyl
mercaptan is regenerated from the mercuric phenyl mercaptide, formed
during
sampling, by treatment with hydrochloric acid. The phenyl mercaptan is
extracted into toluene and analyzed by gas chromatography with a flame
ionization detector. Samples should be protected from light after
sampling. |
| |
| Air volume and sampling rate: |
20L at 0.2 L/min
|
| |
| Status of method: |
Stop gap method. This method has
been only partially evaluated and is presented for information and trial
use. |
| |
| Date: November, 1989 |
Chemist: Mary E. Eide
|
Solvents Branch
OSHA Analytical Laboratory
Salt Lake City, UT 84115-1802
- General Discussion
1.1 Background
1.1.1 History of procedure
The OSHA PEL for phenyl mercaptan is 0.5 ppm. Several solid
sorbent
sampling tubes were tried for the collection of phenyl mercaptan,
but
phenyl mercaptan either did not desorb well from them, or it was not
stable on them. Phenyl mercaptan is readily oxidized by air.
Derivatizing the phenyl mercaptan appeared to be necessary for
stability
considerations. OSHA Method 26 derivatizes methyl mercaptan with
mercuric acetate (Ref. 5.1). This method of collection and analysis
was
tried. The collection, retention, extraction, and storage stability
were
all good using the mercuric acetate coated filters. The detection
limit
was the same using a flame ionization detector (FID) and a flame
photometric detector in the sulfur mode (FPD), so a FID was used for
this study.
1.1.2 Potential workplace exposure (Ref. 5.2)
Phenyl mercaptan is used as a chemical intermediate, solvent, and
as
an insect larvicide.
1.1.3 Toxic Effects (This section is for information purposes and
should not be taken as the basis for OSHA policy.)(Ref. 5.2)
Animal studies show phenyl mercaptan is metabolized to
methylphenylsulfone. Exposure to phenyl mercaptan causes
restlessness,
then increased respiration, incoordination, muscular weakness,
skeletal
muscle paralysis of the hind limbs, cyanosis, lethargy and/or
sedation,
respiratory depression, followed by coma and death. Phenyl mercaptan
is
an eye and skin irritant. Prolonged exposure causes kidney changes
along
with hyaline casts in the tubules and hyperemia of the adrenal
medulla.
In mice, lung, liver and kidney changes were seen following high
inhalation exposures.
1.1.4 Physical properties (Ref. 5.3):
|
Synonyms: |
benzenethiol; thiophenol |
|
Molecular weight: |
110.17 |
|
Density: |
1.0728 |
|
Freezing point: |
-15°C |
|
Boiling point: |
168°C |
|
Flash point: |
56°C (132°F) |
|
Odor: |
offensive mercaptan and garlic-like odor |
|
Color: |
colorless liquid |
|
Molecular formula: |
C6H6S |
|
CAS: |
108-98-5 |
|
IMIS: |
P105 |
|
RTECS: |
14447 (DC0525000) |
|
DOT: |
UN 2337 |
|
Compound: |
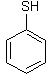
|
1.2 Limit defining parameters
1.2.1 The detection limit of the analytical procedure is 0.6 µg.
This
is the smallest amount that could be detected under normal operating
conditions.
1.2.2 The overall detection limit is 0.004 ppm, based on a 2 mL
extraction and a 20 liter air volume. (All ppm amounts in this study
are
based on a 20 liter air volume and a 2 mL extraction.)
1.3 Advantages
1.3.1 The sampling procedure is convenient.
1.3.2 The analytical method is reproducible and sensitive.
1.3.3 Reanalysis of samples is possible.
1.3.4 It may be possible to analyze other compounds at the same
time.
1.3.5 Interferences may be avoided by proper selection of column
and GC
parameters.
1.4 Disadvantages
1.4.1 The amount of sample that can be taken is limited by the
amount
of mercuric acetate on the filter.
1.4.2 Samples must be protected from light before analysis.
- Sampling procedure
2.1 Apparatus
2.1.1 A calibrated personal sampling pump, the flow of which can be
determined within ±5% at the recommended flow.
2.1.2 Glass fiber filters impregnated with mercuric acetate. The
filters are prepared by soaking 37 mm Gelman type A glass fiber
filters
(or equivalent) in a 5% (w/v) aqueous solution of mercuric acetate.
The
filters are allowed to dry, and then assembled in two piece filter
cassettes without backup pads. The filters may be yellowish in color,
which does not seem to affect their collection efficiency
2.2 Sampling technique
2.2.1 Immediately before sampling, remove the plugs from the filter
cassette.
2.2.2 Connect the cassette to the sampling pump with flexible
tubing.
2.2.3 Tubes should be placed in a vertical position to minimize
channeling, with the smaller section towards the pump.
2.2.4 Air being sampled should not pass through any hose or tubing
before entering the cassette.
2.2.5 Seal the cassette with the plugs immediately after sampling.
Seal
each sample with OSHA Form-21 sealing tape.
2.2.6 With each batch of samples, submit at least one blank, coated
filter from the same lot used for samples.
2.2.7 Transport the samples (and corresponding paperwork) to the
lab
for analysis.
2.2.8 Bulks submitted for analysis must be shipped in a separate
mailing container from the samples.
2.3 Extraction Efficiency
Six mercuric acetate filters were liquid spiked at each loading of
2.2 µg
(0.0488 ppm), 11 µg (0.244 ppm), and
2.2 µg (0.488 ppm). They were
allowed to equilibrate overnight at room temperature, placed into
separate
20 mL scintillation vials, extracted with 2 mL of toluene and 6 mL 25%
HCl
in water for 10 minutes with constant shaking, the toluene layer was
removed, and analyzed by GC/FID. The overall average was 99.5%
recovered
(Table 1).
Table 1
Extraction Efficiency
|
|
Filter#
% Recovered |
|
|
2.2 µg |
11 µg |
22 µg |
|
|
1 |
99.5 |
100 |
101 |
|
2 |
98.4 |
103 |
100 |
|
3 |
103 |
103 |
97.5 |
|
4 |
97.5 |
97.4 |
101 |
|
5 |
101 |
98.4 |
94.0 |
|
h
|
96.6 |
95.4 |
104 |
|
Average |
99.3 |
99.6 |
99.6 |
|
Overall Average |
99.5 |
|
|
|
Standard Deviation ± 2.81
|
2.4 Retention efficiency
2.4.1 Six mercuric acetate filters were liquid spiked
with
22 µg (0.488 ppm) phenyl mercaptan,
allowed to equilibrate overnight, and placed in a cassette with a
backup
filter coated with mercuric acetate. The cassettes had 20 liters of
humid
air (90% RH) pulled through them. They were opened, extracted and
analyzed
by GC/FID. There was no phenyl mercaptan found on the backup filters
(Table 2). The retention efficiency averaged 97.8%.
Table 2
Retention Efficiency
|
|
Filter #
|
% Recovered
'A'
|
% Recovered
'B'
|
Total
|
|
1 |
95.3 |
0.0 |
95.3 |
|
2 |
98.1 |
0.0 |
98.1 |
|
3 |
98.2 |
0.0 |
98.2 |
|
4 |
101 |
0.0 |
101 |
|
5 |
98.8 |
0.0 |
98.8 |
|
6 |
95.1 |
0.0 |
95.1 |
|
|
|
Average: |
97.8 |
|
2.4.2 A collection study was performed using three
cassettes, the first with a glass fiber filter followed by two with
mercuric acetate coated filters. The glass fiber filter was spiked
with
phenyl mercaptan, then immediately afterwards 20 liters of humid air
(85%
RH) was pulled through the cassettes. The phenyl mercaptan vaporized
off
the glass fiber filter and collected onto the mercuric acetate coated
filters. There was no residual phenyl mercaptan found on the glass
fiber
filters. The amount of phenyl mercaptan recovered off the mercuric
acetate
filters averaged 100% (Table 3).
Table 3
Collection Efficiency
|
|
|
% Recovered |
|
| Filter |
GFF |
'A' |
'B' |
Total |
| 1 |
0.0 |
100 |
0.0 |
|
| 2 |
0.0 |
103 |
0.0 |
|
| 3 |
0.0 |
98.3 |
0.0 |
|
|
Average |
|
|
100 |
|
2.5 Storage
Mercuric acetate coated filters were spiked with 44 µg
(0.976 ppm) phenyl mercaptan and stored at room temperature until opened
and analyzed. After day three the storage samples were covered in foil
for
the remainder of the storage period. The recoveries averaged 99.0%
for
the 17 days stored (Table 4).
Table 4
Storage Study
|
|
Day
|
% Recovered
|
|
|
3 |
102 |
|
3 |
99.0 |
|
10 |
97.9 |
|
10 |
96.8 |
|
10 |
97.0 |
|
17 |
99.6 |
|
17 |
101 |
|
17 |
98.9 |
|
Average
|
99.0
|
|
2.6 Precision
The precision was calculated using the area counts
from
six injections of each standard at concentrations of 1.1, 5.5, 11, and
22 µg/mL
phenyl mercaptan in toluene. The pooled coefficient of variation was
0.0105 (Table 5).
Table 5
Precision Study
|
|
Injection Number
|
1.1
µg/ml
|
5.5
µg/ml
|
11
µg/ml
|
22
µg/ml
|
|
|
1 |
21849 |
152180 |
325820 |
714720 |
|
2 |
21558 |
153360 |
322720 |
721980 |
|
3 |
21950 |
153540 |
322850 |
724130 |
|
4 |
21210 |
151170 |
323950 |
722910 |
|
5 |
21289 |
150720 |
329440 |
734120 |
|
6 |
21075 |
150590 |
329150 |
713360 |
|
Average |
21489 |
151927 |
325655 |
721870 |
|
|
SD |
± 357 |
± 1307 |
± 3032 |
± 7481 |
|
CV |
0.0166 |
0.0086 |
0.00931 |
0.0104 |
|
Pooled CV 0.0105
|
-
where:
|
|
CV(Coefficient of Variation) =
|
|
|
| StandardDeviation |
| Average |
|
-
|
POOLED CV |
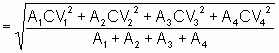 |
-
A1, A2, A3,
A4 = # of injections at each level
-
CV1, CV2, CV3
,CV4
= coefficients at each level
2.7 Air volume and sampling rate studied
2.7.1 The air volume studied is 20 liters.
2.7.2 The sampling rate studied is 0.2 liters per
minute.
2.8 Interferences
Suspected interferences should be listed on sample
data
sheets.
2.9 Safety precautions
2.9.1 Sampling equipment should be placed on an
employee
in a manner that does not interfere with work performance or
safety.
2.9.2 Safety glasses should be worn at all times.
2.9.3 Follow all safety practices that apply to the
workplace being sampled.
- Analytical method
3.1 Apparatus
3.1.1 Gas chromatograph equipped with a flame
ionization
detector. A Hewlett-Packard 5890 was used for this study.
3.1.2 GC column capable of separating the analyte and
an
internal standard from any interferences. The column used in this
study
was a 60-meter RTx-1 1.5-µm
df capillary column. The detection limit for the flame photometric
detector was performed using a 60-m DB-210 0.5-µm df capillary
column.
3.1.3 An electronic integrator or some other suitable
method
of measuring peak areas.
3.1.4 Two milliliter vials with Teflon-lined caps.
3.1.5 A 10 µL syringe or other convenient size for sample
injection.
3.1.6 Pipets for dispensing the toluene and hydrochloric
acid
solution.
3.1.7 Volumetric flasks - 5 mL and other convenient sizes
for
preparing standards.
3.1.8 20 mL scintillation vials for the extraction of the
filters.
3.2 Reagents
3.2.1 Purified GC grade nitrogen, hydrogen, and
air.
3.2.2 Phenyl mercaptan, Reagent grade
3.2.3 Toluene
3.2.4 Deionized water
3.2.5 Hydrochloric acid, a 25% v/v solution is made
with
deionized water
3.3 Sample preparation
3.3.1 Place into a scintillation vial 2 mL toluene and
6
mL 25% hydrochloric acid in water solution.
3.3.2 Remove the filter from the cassette. Fold the
filter
and push it into the vial with the cap as the vial is sealed.
3.3.3 Extract the filter for 10 minutes with
continuous
shaking. It is allowed to sit for 1 minute for the layers to separate.
The
upper toluene layer is removed and placed into a separate 2-mL vial
for
analysis.
3.4 Standard preparation
3.4.1 Standards are prepared by diluting a known
quantity
of phenyl mercaptan with toluene.
3.4.2 At least two separate stock standards should be
made. Dilutions of the stock standards are made to cover the range of
the
samples so that all samples are bracketed with standards. The range
used
in this study was from 0.2 µg/mL
to 44 µg/mL phenyl mercaptan in toluene.
3.5 Analysis
3.5.1 Gas chromatograph conditions
Gas Chromatograph Conditions
|
Flow rates (mL/min) |
Temperature (°C) |
|
Nitrogen(make-up):
|
30 |
Injector: |
200 |
|
Hydrogen(carrier): |
1 |
Detector: |
220 |
|
Hydrogen(detector): |
30 |
Column: |
90 |
|
Air: |
240 |
|
|
|
Injection size: |
3 µL |
|
|
|
Chromatogram: |
(See Figures 1 and 2) |
|
3.5.2 Peak areas are measured by an integrator or
other
suitable means.
3.6 Interferences (analytical)
3.6.1 Any compound having the general retention time
of
the analyte is an interference. Possible interferences should be
listed on
the sample data sheet. GC parameters should be adjusted if necessary
so
these interferences will pose no problems.
3.6.2 Retention time data on a single column is not
considered proof of chemical identity. Samples over the target
concentration should be confirmed by GC/Mass Spec or other suitable
means.
3.7 Calculations
3.7.1 A curve with area counts versus concentration is
calculated from the calibration standards.
3.7.2 The area counts for the samples are plotted with
the
calibration curve to obtain the concentration of phenyl mercaptan in
solution.
3.7.3 To calculate the concentration of analyte in the
air
sample the following formulas are used:
|
mass of analyte per sample
|
=
|
(µg/mL)(desorption
volume)
(desorption efficiency)
|
|
number of moles of
analyte
|
=
|
mass of analyte per sample
molecular weight
|
(number of moles of analyte)(molar volume at~25°C
and 760 mmHg) = (volume the analyte will occupy at~25°C
and 760 mmHg)
| ppm |
= |
(volume analyte occupies) (106) |
| air volume |
* All units must cancel.
3.7.4 The above equations can be consolidated to form
the
following formula. To calculate the ppm of analyte in the sample based
on
a 20 liter air sample:
ppm =
(µg/mL)(EV)(24.26)(106)(g)(mg)
(20L)(DE)(MW)(1000 mg)(1000
µg)
|
µg/mL |
= |
Concentration of analyte in sample or
standard |
|
24.46 |
= |
Molar volume (liters/mole) at 25 °C and 760 mm
Hg. |
|
MW |
= |
Molecular weight (g/mole) |
|
EV |
= |
Extraction volume of 2 mL |
|
20 L |
= |
20 liter air sample |
|
DE |
= |
Desorption efficiency |
3.8 Safety precautions
3.8.1 All handling of solvents should be done in a
hood.
3.8.2 Avoid skin contact with all solvents.
3.8.3 Wear safety glasses at all times.
-
Recommendations for further study
A vapor generated collection study should be
performed.
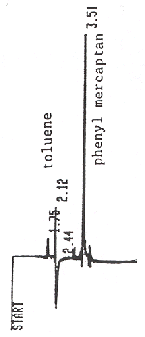
Figure 1. A standard of 11 µg/mL
phenyl mercaptan in toluene analyzed by GC/FPD on a DB-210 capillary
column at 100°C.
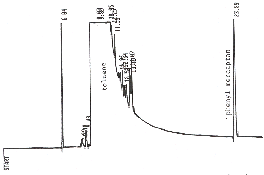
Figure 2. A standard of 11 µg/mL
phenyl mercaptan in toluene analyzed by GC/FID on a RTx-1 capillary
column at 90°C.
- References
5.1 Elskamp, C., Method 26, "Ethylenediamine,
diethylenetriamine,
and triethylenetetramine", Organic Methods Evaluation Branch,
OSHA
Analytical Laboratory, 1981.
5.2 "Documentation of the Threshold Limit Values and
Biological
Exposure Indices", Fifth Edition, American Conference of
Governmental
Industrial Hygienists Inc., Cincinnati, OH, 1986, p. 478.
5.3 Windholz, M., "The Merck Index", Tenth Edition, Merck
& Co., Rahway N.J., 1983, p. 1340.
|

