|
MIAK (METHYL ISOAMYL KETONE)
| Method number: |
PV2042 |
| |
| Matrix: |
Air |
| |
| Target Concentration: |
50 ppm (234 mg/m3) OSHA permissible exposure limit (PEL). |
| |
| Procedure: |
Samples are collected by drawing known volumes of air through glass sampling tubes containing
charcoal adsorbent. Samples are desorbed with a 99:1 (v/v) carbon disulfide/dimethyl formamide
solution and analyzed by gas chromatography (GC) using a flame ionization detector (FID). |
| |
Recommended air volume
and sampling rate: |
10 L at 0.2 L/min |
| |
Detection limit of the
overall procedure (based
on the recommended air
volume and the analytical
detection limit): |
0.21 ppm (0.985 mg/m3) |
| |
| Special requirement: |
Ship the samples to the laboratory for analysis immediately after sampling. If
delay is unavoidable, store the samples at reduced temperature. Store samples in a refrigerator
upon receipt at the laboratory. |
| |
| Status of method: |
Stopgap method. This method has been partially evaluated and is presented for
information and trial use only. |
| |
| Date: January 1992 (Final) |
Chemist: Ing-Fong Chan |
Organic Service Branch II
OSHA Technical Center
Salt Lake City, Utah
1. General Discussion
1.1. Background
1.1.1. History of procedure
This evaluation was undertaken to develop a sampling and analytical procedure for MIAK at 50 ppm
OSHA PEL (Ref. 5.1.).
1.1.2. Toxic effects (This section is for information only and should not be taken as the basis of OSHA policy.)
Eye and nose irritation are encountered at low levels. Narcosis and death can result at high concentrations.
The toxic behavior would be expected to resemble that of
methyl isobutyl ketone closely (Ref. 5.2.).
1.1.3. Potential workplace exposure
MIAK is used as a solvent for cellulose esters, acrylics
and vinyl copolymers (Ref. 5.2. and 5.3.). No data is
available on the extent of work place exposure.
1.1.4. Physical properties (Ref. 5.2., 5.3. and 5.4.)
| CAS number: |
110-12-3 |
| IMIS number: |
1776 |
| Molecular weight: |
114.19 |
| Molecular formula: |
C7H14O |
| Density: |
0.888 at 20°C |
| Boiling point: |
144°C at 101.3 kPa (760 mmHg) |
| Solubility: |
slightly soluble in water
soluble in alcohol and ether |
| Chemical name: |
MIAK (methyl isoamyl ketone) |
| Synonyms: |
5-Methyl-2-hexanone; 2-Methyl-5-hexanone; Methyl isopentyl ketone |
| Appearance: |
colorless, clear liquid with a pleasant odor |
| Structure: |
(CH3)2CHCH2CH2COCH3 |
1.2. Limit defining parameters
The detection limit of the analytical procedure, including a 9:1
split ratio, is 1.09 ng per injection. This is the amount of
analyte which will give a peak whose height is approximately five
times the baseline noise.
2. Sampling Procedure
2.1. Apparatus
2.1.1. Samples are collected by using a personal sampling pump
that can be calibrated to within ± 5% of the recommended
flow rate with the sampling device in line.
2.1.2. Samples are collected with 4-mm i.d. × 6-mm o.d. × 7.0 cm
glass sampling tubes packed with two sections of 20/40
mesh activated charcoal separated by a 2-mm portion of
urethane foam. The activated charcoal is prepared from
coconut shells and is fired at 600°C prior to packing.
The sampling section contains 100-mg and the back section
contains 50-mg of charcoal. A 3-mm portion of urethane
foam is placed between the outlet end of the tube and the
backup section. A plug of Silane treated glass wool is
placed in front of the sampling section.
2.2. Reagents
No sampling reagents are required.
2.3. Sampling technique
2.3.1. Immediately before sampling, break off the ends of the
charcoal tube. All tubes should be from the same lot.
2.3.2. Attach the sampling tube to the sampling pump with
flexible tubing. Position the tube so that sampled air first
passes through the 100-mg section.
2.3.3. Attach the tube vertically in the employee's breathing
zone in such a manner that it does not impede work
performance.
2.3.4. After sampling for the appropriate time, remove the
sampling tube and seal it with plastic caps.
2.3.5. Wrap each sample end-to-end with an OSHA seal (Form 21).
2.3.6. Record the air volume for each sample, and list any
possible interferences.
2.3.7. Submit at least one blank for each set of samples.
Handle the blank in the same manner as the samples, except
no air is drawn through it.
2.3.8. Ship the samples to the laboratory for analysis
immediately after sampling. If delay is unavoidable, store the
samples at reduced temperature.
2.3.9. Submit bulk samples for analysis in a separate container.
Do not ship them with air samples.
2.4. Desorption efficiency
Sixteen vials, each containing a 100-mg portion of charcoal, were
divided into four groups of four vials each. Vials of the first
group were liquid spiked with 2.6 µL of 10% MIAK in carbon
disulfide. Vials of the other three groups were liquid spiked with
1.4, 2.6 and 5.2 µL of neat MIAK (d=0.888), respectively. These
amount represent 0.1×, 0.5×, 1.0×, and 2.0× the target
concentration. The vials were stored overnight in a refrigerator (0°C),
desorbed with 1.0 mL of the desorbing solution, and analyzed as
in Section 3. The average desorption efficiency was 96.3%. The
results are listed in Table 2.4.
Table 2.4.
Desorption Efficiency
|
Sample # |
Amount
Spiked, µg |
Amount
Found, µg |
%
Recovered |
|
D1
D2
D3
D4 |
229
229
229
229 |
214
214
222
218 |
93.6
93.6
97.0
95.3 |
|
Average of 0.1× PEL = 94.9% |
|
D5
D6
D7
D8 |
1231
1231
1231
1231 |
1145
1193
1180
1298 |
92.5
96.9
95.9
97.4 |
|
Average of 0.5× PEL = 95.7% |
|
D9
D10
D11
D12 |
2286
2286
2286
2286 |
2232
2235
2226
2221 |
97.6
97.8
97.4
97.1 |
|
Average of 1× PEL = 97.5% |
|
D13
D14
D15
D16
D17 |
4571
4571
4571
4571
Blank |
4453
4429
4392
4438
0 |
97.4
96.9
96.1
97.1
Blank |
|
Average of 2× PEL = 96.9% |
|
2.5. Retention efficiency
Five charcoal tubes were each liquid spiked with 2.6 µL (1× PEL)
of neat MIAK. These were allowed to equilibrate for 2 hours and
then 10 L of humid air (~80% relative humidity) were drawn
through each tube at 0.2 L/min. Then the tubes were desorbed
with 1.0 mL of desorbing solution, and analyzed as in Section 3.
The results are listed in Table 2.5.
Table 2.5.
Retention Efficiency
|
Sample # |
Amount
Spiked, µg |
Amount
Found, µg |
%
Recovered |
|
R1
R2
R3
R4
R5 |
2286
2286
2286
2286
2286 |
2131
2111
2137
2146
2133 |
93.2
92.4
93.5
93.9
93.3 |
|
|
Average = 93.3% |
|
2.6. Sample storage
Twelve charcoal tubes were each liquid spiked with 2.6 µL (1×
PEL) of neat MIAK. These were allowed to equilibrate for 2 hours
and then 10 L of humid air (~80% relative humidity) were drawn
through each tube at 0.2 L/min. Half of the tubes were stored in
a drawer at ambient temperature, and the other half were stored
in a refrigerator (0°C). After ten days they were extracted and
analyzed as in Section 3. The results are given in Tables 2.6.1.
and 2.6.2.
Table 2.6.1.
Ambient Storage
|
Days
Stored |
Amount
Spiked, µg |
Amount
Found, µg |
%
Recovered |
|
10
10
10
10
10
10 |
2286
2286
2286
2286
2286
2286 |
1299
1460
1356
1526
1231
1291 |
56.8
63.9
59.3
66.8
53.8
56.5 |
|
|
Average = 59.5% |
|
Table 2.6.2
Refrigerator Storage
|
Days
Stored |
Amount
Spiked, µg |
Amount
Found, µg |
%
Recovered |
|
10
10
10
10
10
10 |
2286
2286
2286
2286
2286
2286 |
2183
2207
2166
2175
2178
2178 |
95.5
96.6
94.7
95.1
95.3
95.3 |
|
|
Average = 95.4% |
|
2.7. Recommended air volume and sampling rate
2.7.1. The recommended air volume is 10 L.
2.7.2. The recommended flow rate is 0.2 L/min.
2.8. Interferences (sampling)
It is not known if any compounds will interfere with the
collection of MIAK. Any suspected interferences should be reported to
the laboratory with submitted samples.
2.9. Safety precautions (sampling)
2.9.1. Attach the sampling equipment in such a manner that it
will not interfere with work performance or employee
safety.
2.9.2. Follow all safety practices that apply to the work area
being sampled.
3. Analytical Procedure
3.1. Apparatus
3.1.1. A GC equipped with an FID. A Hewlett-Packard 5890 Gas
Chromatograph equipped with a 7673A Autosampler and an
FID was used in this evaluation.
3.1.2. A GC column capable of separating MIAK and the internal
standard from any interferences. A 60 m × 0.32 mm i.d.
(0.5 µm film) DB-WAX capillary column was used in this
evaluation.
3.1.3. An electronic integrator or some other suitable means to
measure detector response. A Waters 860 Networking
Computer System was used in this evaluation.
3.1.4. Volumetric flasks, pipets, and syringes for preparing
standards, making dilutions and performing injections.
3.1.5. Vials, 2-mL, and 4-mL, with PTFE-lined caps.
3.2. Reagents
3.2.1. MIAK. 5-Methyl-2-hexanone, 99%, was obtained from
Aldrich Chemical Company.
3.2.2. Reagent grade solvent or better should be used.
3.2.3. Carbon disulfide. The carbon disulfide used in this
evaluation was purchased from EM Science.
3.2.4. Dimethyl formamide (DMF). The DMF was purchased from
Burdick and Jackson.
3.2.5. p-Cymene. The p-cymene used as internal standard was
purchased from Aldrich Chemical Company.
3.2.6. Desorbing solution. The desorbing solution is prepared
by adding 250 µL of p-cymene to 1 L of carbon
disulfide/DMF (99:1, v/v).
3.3. Standard preparation
Prepare standards at concentrations of 1 µL and 4 µL of
o-chloro-toluene per milliliter of desorbing solution. At least two
standards at 1 µL/mL are prepared. Standards must be used the day
they are prepared.
3.4. Sample preparation
3.4.1. Transfer the 100-mg section of the sampling tube to a
2-mL vial. Place the 50-mg backup section in a separate
2-mL vial.
3.4.2. Add 1.0 mL of desorbing solution to each vial and seal
with a Teflon-lined cap.
3.4.3. Shake the vials vigorously several times during the next
30 min.
3.5. Analysis
3.5.1. Instrument conditions
| Column: |
DB-WAX, 60 m × 0.32 mm i.d., 0.5 µm film |
| Injector temperature: |
180°C |
| Detector temperature: |
200°C |
| Column temperature: |
90°C (initial temp) |
| Temperature program: |
hold initial temp 3 min, increase temp at 10 °C/min to 160°C, hold final temp 1 min |
|
| Gas flow rates: |
|
column:
septum purge:
FID:
FID makeup:
FID: |
2.0 mL/min (hydrogen)
1.2 mL/min (hydrogen)
34 mL/min (hydrogen)
30 mL/min (nitrogen)
480 mL/min (air) |
| Injection volume: |
1 µL |
| Split ratio: |
9:1 |
| Retention time: |
6.48 min (MIAK)
8.60 min (p-cymene) |
3.5.2. Chromatogram (Figure 1.)
3.5.3. Measure detector response using a suitable method such as
electronic integration.
3.6. Interferences (analytical)
3.6.1. Any collected compound which produces an FID response and
has a similar retention time as MIAK or the internal
standard is a potential interference.
3.6.2. GC conditions may generally be varied to circumvent interferences.
3.6.3. Retention time on a single column is not proof of
chemical identity. Analysis by an alternate GC column, high
performance liquid chromatography (HPLC) and confirmation
by mass spectrometry are additional means of identification.
3.7. Calculations
3.7.1. An internal standard (ISTD) calibration method is used.
The linear nature of FID allows the use of a point
calibration, but the bracketing of samples with analytical
standards is a good practice.
3.7.2. Determine the µg/mL of MIAK in both sections of each
sample and blank from the calibration curve. If MIAK is found on the backup
section, it is added to the amount found on the front section. Blank
corrections should be performed before adding the results together.
3.7.3. Determine the air concentration by using the following formula.
| mg/m3 = |
(µg/mL, blank corrected) × (desorption volume, mL)
(air volume, L) × (desorption efficiency, decimal) |
| ppm = |
(mg/m)(24.46)
(114.19) |
| where |
24.46
114.19 |
=
= |
molar volume (liters) at 101.3 kPa (760 mmHg) and 25°C
molecular weight of MIAK |
3.8. Safety precautions (analytical)
3.8.1. Avoid skin contact and air exposure to MIAK.
3.8.2. Avoid skin contact with all solvents.
3.8.3. Wear safety glasses at all times.
4. Recommendation for Further Study
This method should be fully validated.
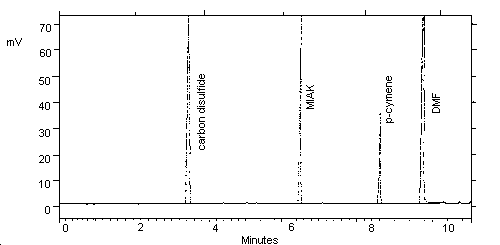
Figure 1. Chromatogram of MIAK at 0.5× target level
5. References
5.1. "Code of Federal Regulations", 29 CFR 1910.1000, Table Z-1-A.
Limits for Air Contaminants, U.S. Government Printing Office,
Washington, D.C., 1990.
5.2. Sitting, M., Handbook of Toxic and Hazardous Chemicals, Noyes
Publications, Park Ridge, N.J., 1981; p 456.
5.3. Documentation of the Threshold Limit Values and Biological
Exposure Indices, American Conference of Governmental Industrial
Hygienist INC., 5th ed., 1986; p 400.
5.4. Weast, RC., Astle, MJ., and Beyer, WH., Handbook of Chemistry and
Physics, 68th ed., CRC Press Inca, Boca Raton, Florida, 1987-88.
|

