|
CARBARYL (SEVIN)
| Method no.: |
63 |
|
| Matrix: |
Air |
|
| Target concentration: |
5 mg/m3 (OSHA PEL) |
|
| Procedure: |
Samples are collected by drawing known volumes of air through OSHA
versatile sampler (OVS-2) tubes, containing a glass fiber filter and two sections of XAD-2 adsorbent. Samples are extracted with acetonitrile and analyzed by HPLC using a UV detector. |
|
Recommended air volume
and sampling rate: |
60 L at 1.0 L/min |
|
| Reliable quantitation limit: |
0.028 mg/m3 |
|
Standard error of estimate
at the target concentration:
(Section 4.6.) |
5.1% |
|
| Status of method: |
Evaluated method. This method has been subjected to the established
evaluation procedures of the Organic Methods Evaluation Branch. |
|
| Date: March 1987 |
Chemist: Donald Burright |
Organic Methods Evaluation Branch
OSHA Analytical Laboratory
Salt Lake City, Utah
1. General Discussion
1.1. Background
1.1.1. History
This evaluation was undertaken to determine the effectiveness of the OSHA Versatile Sampler containing XAD-2 resin (OVS-2) as a sampling device for carbaryl. The OVS-2 was successfully evaluated for a number of organophosphorus pesticides (Ref. 5.1.). The
OVS-2 is a specially prepared glass tube containing a glass fiber filter and XAD-2 resin (Section 4.9.) which will collect both vapor and aerosol.
In the past, carbaryl was collected on a 37-mm glass fiber filter in a cassette. The analytical procedure developed by NIOSH (NIOSH Method 5006) required that the sample be derivatized and analyzed by absorption spectrophotometry (Ref. 5.2.). The analytical procedure used by OSHA required that the sample be extracted and analyzed by HPLC with a UV detector (Ref. 5.3.). The HPLC-UV procedure was evaluated in this work because it provided an analysis which was more specific for carbaryl than the spectrophotometric procedure. This evaluation is one of a series being performed to establish the OVS-2 as a common sampler for organic pesticides.
1.1.2. Toxic effects (This section is for information only and should not be taken as a basis for OSHA policy.)
Carbaryl inactivates cholinesterase, resulting in the accumulation of acetylcholine at the synapses in the nervous system, at neuromuscular junctions of the skeletal and smooth muscles, and secretory glands. Signs and symptoms of overexposure may include miosis, blurred vision, lacrimation, excessive nasal discharge or salivation, sweating, abdominal
cramps, nausea, vomiting, diarrhea, tremor, cyanosis and convulsions. (Ref. 5.4.)
The LD50 for rats (oral) is 500 mg/kg. A man who had consumed 0.5 L of an 80% solution of carbaryl died 6 h after exposure. (Ref. 5.5.)
1.1.3. Workplace exposure
Occupational exposure of carbaryl can occur in the manufacture, formulation, distribution, and application of carbaryl. This includes agricultural crop workers, farmers, plant nursery workers, spray pilots, and others engaged in spraying and dusting operations. NIOSH estimates that 100,000 U.S. workers are potentially exposed to carbaryl. (Ref.
5.5.)
1.1.4. Physical properties and other descriptive information (Ref. 5.5.)
| CAS no.: |
63-25-2 |
| molecular weight: |
201.22 |
| melting point: |
142°C |
| vapor pressure: |
5.5 × 10-3 Pa at 25°C
(4.1 × 10-5 mm Hg) |
| appearance: |
white crystals |
| specific gravity: |
1.232 at 20°C |
flash point:
(open cup) |
193°C |
solubility:
(in water) |
40 µg/mL at 30°C |
| molecular formula: |
C1 2H1 1NO2 |
| synonyms: |
Sevin; 1-naphthalenol N-methylcarbamate; Atoxan;
Caprolin; Gamonil; Panam; Sevidol |
| structure: |
 |
1.2. Limit defining parameters (The analyte air concentrations listed throughout this method are based on an air volume of 60 L and a solvent extraction volume of 2 mL.)
1.2.1. Detection limit of the analytical procedure
The detection limit of the analytical procedure is 6.8 ng per injection. This is the amount of analyte which will give a peak whose height is about 5 times the height of the baseline noise. (Section 4.1.)
1.2.2. Detection limit of the overall procedure
The detection limit of the overall procedure is 1.66 µg per sample (0.028 mg/m3). This is the amount of analyte spiked on the sampling device which allows recovery of an amount
equivalent to the detection limit of the analytical procedure. (Section 4.2.)
1.2.3. Reliable quantitation limit
The reliable quantitation limit is 1.66 µg per sample (0.028 mg/m3). This is the smallest
amount of analyte which can be quantitated within the requirements of a recovery of at least 75% and a precision (±1.96 SD) of ±25% or better. (Section 4.2.)
| |
The reliable quantitation limit and detection limits reported in
the method are based upon optimization of the instrument for the smallest
possible amount of the analyte. When the target concentration of the
analyte is exceptionally higher than these limits, they may not be
attainable at the routine operating parameters.
|
|
1.2.4. Instrument response to the analyte
The instrument response over the concentration range of 0.5 to 2 times the target concentrations is linear. (Section 4.4.)
1.2.5. Recovery
The recovery of carbaryl from samples used in a 16-day storage test remained above 92.6% when the samples were stored at about 22°C. (Section 4.6.) The recovery of analyte from the collection medium during storage must be 75% or greater.
1.2.6. Precision (analytical procedure)
The pooled coefficient of variation obtained from replicate determinations of analytical standards at 0.5, 1 and 2 times the target concentration is 0.003. (Section 4.3.)
1.2.7. Precision (overall procedure)
The precision at the 95% confidence level for the 16-day ambient temperature storage test is ±10.1%. (Section 4.6.) This includes an additional ±5% for sampling error. The overall procedure must provide results at the target concentration that are ±25% or better
at the 95% confidence level.
1.2.8. Reproducibility
Six samples, spiked by liquid injection with carbaryl, and a draft copy of this procedure were given to a chemist unassociated with this evaluation. The samples were analyzed after 20 days of storage at about 5°C. No individual sample deviated from its theoretical value by more than the precision reported in Section 1.2.7. (Section 4.7.)
1.3. Advantages
1.3.1. This sampling device has the capability to collect both vapors and aerosols of carbaryl without having to use two separate samplers in series.
1.3.2. The analytical procedure is more specific for the analysis of carbaryl than the NIOSH method.
1.4. Disadvantage
Currently, the OVS-2 tube is not commercially available.
2. Sampling Procedure
2.1. Apparatus
2.1.1. Samples are collected by use of a personal sampling pump that can be calibrated to within ±5% of the recommended flow rate with the sampling device in line.
2.1.2. Samples are collected with OVS-2 tubes, which are specially made 13 mm o.d. glass tubes that are tapered to
6-mm o.d., packed with a 140-mg backup section and a 270-mg sampling section of cleaned XAD-2 and a 13-mm diameter glass fiber filter. The backup
section is retained by two foam plugs and the sampling section is between one foam plug and the glass fiber filter. The glass fiber filter is held next to the sampling section by a polytetrafluoroethylene (PTFE) retainer. (Section 4.9., Figure 4.9.4.)
2.2. Reagents
No sampling reagents are required.
2.3. Sampling technique
2.3.1. Attach the small tubing adapter of the sampling tube to the sampling pump with flexible, plastic tubing such that the large, front section of the sampling tube is exposed directly to the atmosphere. Do not place any tubing in front of the sampler. The sampler should be attached vertically (large end down) in the worker's breathing zone in such a manner that it does not impede work performance.
2.3.2. After sampling for the appropriate time, remove the sampling device and seal the tube with plastic end caps.
2.3.3. Wrap each sample end-to-end with an OSHA seal (Form 21).
2.3.4. With each set of samples, submit at least one blank. The blank should be handled the same as the other samples except that no air is drawn through it.
2.3.5. Bulk samples should be submitted for analysis in a separate container. Do not ship with the air samples.
2.4. Retention efficiency
To test the sampler's ability to retain carbaryl, twice the target concentration amount of carbaryl (604 µg) was liquid spiked onto eight sampling tubes. Humid air (about 72% relative humidity) was
pulled through the tubes for 15 min to 4 h at 1 L/min. When the samples were analyzed, it was found that the analyte was present on the glass fiber filter and the front section of XAD-2 at levels
equal to 91-96% of the total amount spiked. No carbaryl was found on the back section. (Section 4.8.)
2.5. Extraction and desorption efficiencies (Section 4.5.)
2.5.1. The combined extraction and desorption efficiencies for carbaryl from the the glass fiber filter and the large XAD-2 section at the target concentration was 93.6%.
2.5.2. The extraction efficiency for carbaryl from glass fiber filters at the target concentration was 99.1%.
2.5.3. The average desorption efficiency for carbaryl from the lot of cleaned XAD-2 adsorbent used in this evaluation over the range of 0.5 to 2.0 times the target concentration was 92.8%.
2.5.4. Extracted/desorbed samples remain stable for at least 48 h.
2.6. Recommended air volume and sampling rate
2.6.1. The recommended air volume is 60 L.
2.6.2. The recommended air sampling rate is 1.0 L/min.
2.6.3. When short-term air samples are required, the recommended sampling rate is 1 L/min. The reliable quantitation limit is
0.11 mg/m3 for a 15-min sample.
2.7. Interferences (sampling)
Suspected interferences should be reported to the laboratory with submitted samples.
2.8. Safety precautions (sampling)
2.8.1. The sampling equipment should be attached to the worker in such a manner that it will not interfere with work performance or safety.
2.8.2. All safety practices that apply to the work area being sampled should be followed.
3. Analytical Procedure
3.1. Apparatus
3.1.1. High-performance liquid chromatograph equipped with an ultraviolet (UV) detector, and manual or automatic injector. A Waters M-6000A pump, Waters 710B autosampler, and Waters 440 UV detector were used in this evaluation.
3.1.2. HPLC column capable of separating carbaryl from any interferences. A 25-cm × 4.6-mm i.d. DuPont Zorbax ODS
(6-µm) column was used during this evaluation.
3.1.3. An electronic integrator or other suitable means of measuring detector response. A Hewlett-Packard 3357 data system was used in this evaluation.
3.1.4. Vials, 4-mL glass with PTFE-lined caps.
3.1.5. Volumetric flasks, pipets and syringes for preparing standards, making dilutions and performing injections.
3.2. Reagents
3.2.1. Acetonitrile, HPLC grade.
3.2.2. Water, HPLC grade. A Millipore Milli-Q system was used to prepare the water for this evaluation.
3.2.3. Carbaryl, 98% pure, (Chem Services Inc).
3.3. Standard preparation
Stock standard solutions are prepared by adding acetonitrile to preweighed amounts of carbaryl. Working range standard solutions are prepared by diluting the stock solutions with acetonitrile.
3.4. Sample preparation
3.4.1. Transfer the glass fiber filter and the 270-mg section of the sampling tube to a 4-mL glass vial. Place the first foam plug and the 140-mg section in a separate vial. A small glass funnel can be used to facilitate the transfer of the adsorbent. Discard the rear foam plug. Do not discard the glass sampling tube; it can be reused after it has been cleaned with surfactant or suitable solvent.
3.4.2. Add 2.0 mL of acetonitrile to each vial.
3.4.3. Seal the vials with PTFE-lined caps and extract/desorb the samples for 1 h. Shake the vials by hand with vigorous force several times during the extraction/desorption time.
3.5. Analysis
3.5.1. HPLC conditions
| column: |
25-cm × 4.6-mm i.d. stainless steel column packed with 6-µm DuPont Zorbax ODS |
| mobile phase: |
55:45 (v/v) acetonitrile/water |
| flow rate: |
1.0 mL/min |
| UV detector: |
280 nm |
| injection size: |
10 µL |
| retention time: |
6.5 min |
| chromatogram: |
Figure 3.5.1. |
3.5.2. Measure detector response using a suitable method, such as electronic integration.
3.5.3. Prepare a calibration curve using several solutions over a range of concentrations. The calibration curve is prepared daily. The samples are bracketed with analytical standards.
3.6. Interferences (analytical)
3.6.1. Any compound having a similar retention time as the analyte is a potential interference. Generally, chromatographic conditions can be altered to separate an interference from the analyte.
3.6.2. Retention time on a single column is not proof of chemical identity. Analysis by an alternate HPLC column, detection at another wavelength, comparison of absorbance response ratios, and confirmation by mass spectrometry are additional means of identification.
3.7. Calculations
3.7.1. Calibration curves are prepared by plotting detector response against concentration for each standard. The best fit line through the data points is determined by curve fitting.
3.7.2. The concentration, in µg/mL, for a particular sample is determined by comparing its detector response to the calibration curve. If carbaryl is found on the backup section, it is added to the amount found on the front section. Blank corrections for each section should be performed before adding the results together.
3.7.3. The air concentration of carbaryl can be expressed using the following equation.
mg/m3 = (A)(B) / (C)(D)
| where |
A = |
µg/mL from Section 3.7.2. |
|
B = |
extraction/desorption volume |
| C = |
liters of air sampled |
| D = |
combined extrac./desorp. efficiency (decimal) |
The combined extraction/desorption efficiency should be determined for the particular batch of resin and lot of filter used for the sample.
3.8. Safety precautions (analytical)
3.8.1. Avoid exposure to all standards.
3.8.2. Avoid exposure to all solvents.
3.8.3. Wear safety glasses at all times.
4. Backup Data
4.1. Detection limit of the analytical procedure
The detection limit of the analytical procedure is 6.8 ng per injection. This amount produced a peak whose height is about 5 times the height of the baseline noise. The injection volume recommended in the analytical procedure (10 µL) was used in the determination of the detection limit for the analytical procedure. (Figure 4.1.)
4.2. Detection limit of the overall procedure and reliable quantitation limit
The detection limit of the overall procedure and reliable quantitation limit are 1.66 µg per sample (0.028 mg/m3). The injection size recommended in the analytical procedure (10 µL) was used in this determination. Six samples were each liquid spiked with a solution containing carbaryl about equivalent to its analytical detection limit (1.4 µg per sample) and analyzed. The recovery of carbaryl from the samples was approximately equal to the detection limit of the analytical procedure. Since the recovery was above 75% and the precision (1.96 SD) better than ±25%, 1.66 µg per sample is also the reliable quantitation limit.
Table 4.2.
Reliable Quantitation Limit Data
|
| % recovered |
% recovered |
statistics |
|
| 95.7 |
93.3 |
 = = |
93.8 |
| 93.3 |
92.7 |
SD = |
1.3 |
| 95.1 |
92.7 |
1.96 SD = |
2.5 |
|
4.3. Precision (analytical method only)
The precision of the analytical method was evaluated by doing multiple injections of analytical standards. The results of this study are presented below.
Table 4.3.
Carbaryl Precision Data
|
× target conc.
µg/sample |
0.5×
151 |
1×
301 |
2×
603 |
|
| area |
217724 |
434879 |
869972 |
| counts |
218364 |
438644 |
869891 |
|
217953 |
436357 |
869537 |
| 218131 |
439434 |
867210 |
| 217875 |
439377 |
869231 |
| 218261 |
439468 |
868676 |
|
 |
218051 |
438027 |
869086 |
| SD |
243 |
1947 |
1034 |
| CV |
0.0011 |
0.0044 |
0.0012 |
|
 = 0.003 = 0.003 |
|
4.4. Instrument response to the analyte
The data in Table 4.3. are presented graphically in
Figure 4.4. This figure is a calibration curve over the concentration range of 0.5 to 2 times the target concentration. The instrument response is linear over this range. The slope of the line (2880 area counts per µg/mL) is a measure of the response of the instrument to the analyte.
4.5. Extraction/desorption efficiencies
4.5.1. Extraction from glass fiber filter
The extraction efficiency of carbaryl was determined by liquid spiking six glass fiber filters (GFF) with carbaryl at the target concentration
(302 µg/sample). These samples were stored overnight and then extracted with acetonitrile and analyzed.
Table 4.5.1.
Extraction Efficiency of Carbaryl
at 1× Target Conc. from GFF
|
extraction efficiency, % |
|
| 99.3 |
99.4 |
| 99.1 |
99.2 |
| 99.1 |
98.6 |
|
 = 99.1 = 99.1 |
|
4.5.2. Desorption from XAD-2 adsorbent
The desorption efficiency of carbaryl was determined by liquid spiking 270-mg portions of XAD-2 adsorbent with carbaryl at 0.5 to 2.0 times the target concentration. These samples were stored overnight and then desorbed with acetonitrileand analyzed. The results are listed below.
Table 4.5.2.
Desorption Efficiency
of Carbaryl from XAD-2
|
× target conc.
µg/sample |
0.5×
151 |
1×
302 |
2×
604 |
|
| desorption |
92.7 |
92.9 |
93.5 |
| efficiency, |
93.1 |
93.1 |
92.7 |
| % |
91.8 |
93.1 |
93.0 |
|
92.8 |
92.8 |
92.8 |
| 93.6 |
93.2 |
92.6 |
| 92.2 |
92.9 |
91.7 |
|
 |
92.7 |
93.0 |
92.7 |
|
The average desorption efficiency over the studied range was 92.8%.
4.5.3. The combined extraction/desorption efficiency of carbaryl was determined by liquid spiking glass fiber filters with the target concentration and placing the filter and the large section of
XAD-2 beads into a vial. The next day the samples were xtracted/desorbed with acetonitrile and analyzed. The samples were reanalyzed two days later with new standards to test the stability of extracted/desorbed samples.
Table 4.5.3.
Stability of Extracted/Desorbed Samples
|
|
original |
48 h later |
|
| extraction/ |
94.2 |
92.0 |
| desorption |
93.9 |
87.4 |
| efficiency, |
93.2 |
91.3 |
| % |
93.6 |
91.8 |
|
93.8 |
92.1 |
|
93.2 |
92.4 |
|
 |
93.6 |
91.2 |
|
|
% of original = 97.4 |
|
4.6. Storage data
Storage samples were generated by liquid spiking 36 sampling tubes with carbaryl (302 µg) and then pulling 30 L of humid air through them (about 85% relative humidity). One-half of the tubes were
stored in a freezer at -14°C and the other half were stored in a closed drawer at ambient temperature (about 22°C). Three samples from each group were analyzed periodically over a
16-day period. The results (percent recovered versus storage time) are given below and shown graphically in Figures
4.6.1. and 4.6.2. All samples analyzed in this evaluation were generated by liquid spiking the sampler with a solution containing carbaryl because no equipment was available
to produce an aerosol of carbaryl.
Table 4.6.
Storage Tests
|
| storage time |
|
% recovery |
| (days) |
|
(ambient) |
|
(refrigerated) |
|
| 0 |
|
91.2 |
90.6 |
92.0 |
|
91.2 |
90.6 |
92.0 |
|
93.3 |
93.4 |
94.7 |
93.3 |
93.4 |
94.7 |
| 3 |
93.2 |
92.9 |
93.3 |
93.6 |
94.6 |
94.0 |
| 7 |
94.3 |
94.4 |
94.2 |
95.3 |
95.5 |
94.5 |
| 10 |
93.2 |
93.0 |
95.0 |
94.8 |
95.0 |
94.2 |
| 13 |
91.9 |
93.8 |
93.7 |
95.2 |
93.8 |
95.6 |
| 16 |
96.5 |
95.3 |
95.6 |
98.4 |
95.9 |
95.9 |
|
4.7. Reproducibility data
Six samples, liquid spiked with carbaryl, were given to a chemist unassociated with this study. The samples were analyzed after being stored for 20 days at 5°C. The results were not corrected for extraction/desorption efficiency and are shown below. None of the data had a percent deviation greater than the precision of the overall procedure of ±10.1%.
Table 4.7.
Reproducibility Data
|
| sample |
µg spiked |
% recovered |
% deviation |
|
| 1 |
302 |
102.8 |
2.8 |
| 2 |
453 |
92.4 |
7.6 |
| 3 |
302 |
106.5 |
6.5 |
| 4 |
453 |
94.9 |
5.1 |
| 5 |
302 |
105.9 |
5.9 |
| 6 |
453 |
99.2 |
0.8 |
|
4.8. Retention efficiency
To test the ability of the sampler to retain the analytes, eight samplers were liquid spiked with twice the target concentration (604 µg) of carbaryl. Humid air (about 72% relative humidity) was pulled through the samplers for 15 min to 4 h at 1 L/min. The results show no carbaryl breakthrough onto the backup section of the sampling tube.
Table 4.8
Retention of Carbaryl
|
| air volume (L) |
µg recovered |
% recovered |
|
| 15.2 |
580.1 |
96.0 |
| 30.3 |
584.3 |
96.7 |
| 45.5 |
580.2 |
96.1 |
| 60.6 |
582.6 |
96.5 |
| 90.9 |
548.1 |
90.7 |
| 120.0 |
575.7 |
95.3 |
| 181.8 |
576.3 |
95.4 |
| 255.0 |
577.1 |
95.5 |
|
4.9. Preparation of the OVS-2 tube
It is anticipated that this sampler containing different adsorbents can be used to collect a broad range of airborne contaminants. For these applications the suffix will also reflect the type of resin contained in the sampler. For example, a sampler containing Tenax will be designated OVS-T and one containing XAD-7 will be called OVS-7.
4.9.1. Apparatus
4.9.1.1. Soxhlet extractor
4.9.1.2. Rotary evaporator
4.9.1.3. Miscellaneous glassware: vacuum flask, 2-L
round-bottom flask, Erlenmeyer flask, 250-mL Buchner funnel with course fritted disc, etc.
4.9.1.4. Urethane foam plugs, 3/8 in. × 1/2-in. diameter and 3/16 in. × 1/2-in. diameter.
4.9.1.5. Glass fiber filters, 1/2-in. diameter or 13-mm diameter.
4.9.1.6. PTFE retainer. The retainer is made by removing a 50° arc from a
piece of PTFE tubing, 1/8 in. × 1/2-in. o.d. ×
3/8-in. i.d.
4.9.1.7. Glass sampling tube. The sampling tube is constructed of two pieces of borosilicate glass tubing that have been joined together by a glass blower. One of the pieces is 50 mm × 13-mm o.d. × 11-mm i.d. The other piece is 25 mm × 6-mm o.d. × 4-mm i.d. (Figure 4.9.4.)
4.9.1.8. Plastic cap, 7/8 in. × 1/2-in. i.d. (Alliance Plastics, Inc., Erie PA).
4.9.1.9. Plastic cap, 3/4 in. × 7/32-in. i.d. (SKC, Inc, Eighty-Four, PA).
4.9.2. Reagents
4.9.2.1. Methanol, HPLC grade.
4.9.2.2. Toluene, HPLC grade.
4.9.2.3. Amberlite XAD-2 non-ionic polymeric adsorbent, 20/60 mesh (Aldrich Chemical, Milwaukee, WI).
4.9.3. Cleaning of adsorbent
Add 500 g of crude XAD-2 adsorbent to a large Erlenmeyer flask and pour in enough water to cover the adsorbent. Swirl the flask to wash the beads and discard the adsorbent that floats to the surface of the water. Filter the adsorbent using a Buchner funnel. Transfer the beads back to the Erlenmeyer flask and repeat the water wash and filtration.
Allow the adsorbent to air dry in the funnel for several minutes before removing the vacuum. Transfer the dried adsorbent to a Soxhlet extractor and extract the material with methanol for 24 h. Replace the contaminated
methanol with toluene and continue extracting for another 24 h. Replace the toluene with fresh methanol and continue extracting for 4 h. Transfer the cleaned adsorbent to a round-bottom flask and remove the methanol with the rotary evaporator. If toluene is still present in the resin, place the resin in a heated vacuum oven (100°C) for 8 h. Do not fully close the vent valve so that air is being swept through the oven. The cleaned adsorbent is now ready to be packed into sampling tubes.
4.9.4. Assembly of the OVS-2 tube
Place a large foam plug in the bottom of the large end of the glass tube. Add 140 mg of cleaned XAD-2 adsorbent to the tube. With the beads level, place the small foam plug on the beads. Next add 270 mg of cleaned XAD-2 adsorbent to the tube, followed by the
glass fiber filter. The filter should form a small cup and touch the sides of the tube all around. The PTFE retainer is inserted inside the glass tube. Gently press the PTFE retainer against the glass fiber filter. Cap the ends of the sampling tube. (Figure 4.9.4.)

Figure 3.5.1. Chromatogram of carbaryl at 0.5× the target concentration.
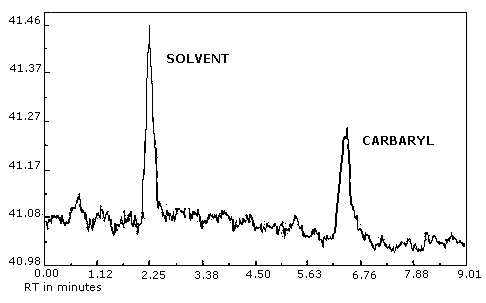
Figure 4.1. Analytical detection limit for carbaryl, 6.8 ng/injection.

Figure 4.4. Calibration curve for carbaryl, slope = 2880 area counts per microgram per milliliter.

Figure 4.6.1. Ambient storage test for carbaryl.
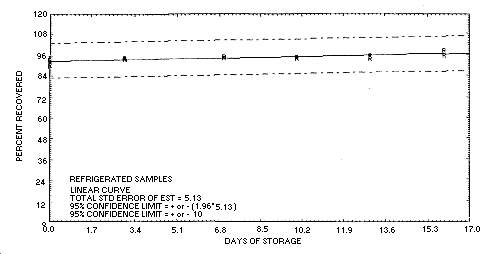
Figure 4.6.2. Refrigerated storage test for carbaryl.
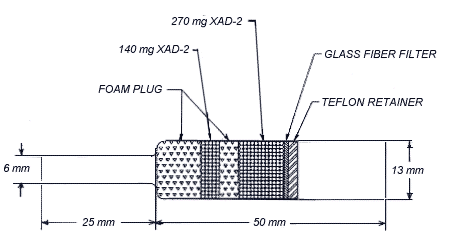
Figure 4.9.4. A drawing of an OVS-2 tube.
5. References
5.1. Burright, D., Method #62, "Chlorpyrifos, DDVP, Diazinon, Malathion, and Parathion", OSHA Analytical Laboratory, unpublished, 1986.
5.2. "NIOSH Manual of Analytical Methods", 3rd ed.; US Department of Health and Human Services, Centers for Disease Control, NIOSH; Cincinnati, OH, Feb. 1984; Vol. 1, Method 5006, Publ. No. 84-100.
5.3. "Chemical Information File", U.S. Department of Labor, Occupational Safety and Health Administration, Directorate of Technical Support, June 14, 1985.
5.4. "Occupational Health Guidelines for Chemical Hazards" NIOSH/OSHA, Jan. 1981, DHHS(NIOSH) Publ. No. 81-123.
5.5. "Criteria for a Recommended Standard ... Occupational Exposure of Carbaryl", U.S. Department of Health, Education, and Welfare, National Institute for Occupational Safety and Health; Cincinnati, OH; 1976; DHEW (NIOSH) Publ. No. 77-107.
|

