|
<<< Back to Sampling and Analytical Methods |
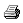 Printing Instructions
Printing Instructions |
For problems with accessibility in using figures, illustrations and PDFs in this method, please contact
the SLTC at (801) 233-4900. These procedures were designed and tested for internal use by OSHA personnel.
Mention of any company name or commercial product does not constitute endorsement by OSHA. |
Metal and Metalloid Particulates in Workplace Atmospheres (ICP Analysis)
[248
KB
PDF,
43 pages]
Related Information: Chemical Sampling -
Antimony & Compounds (as Sb),
Beryllium and Beryllium Compounds (as Be),
Cadmium,
Chromium, Metal and Insoluble Salts,
Cobalt, Metal, Dust & Fume (as Co),
Copper Dusts & Mists (as Cu),
Copper Fume (as Cu),
Iron Oxide Fume,
Lead, Inorganic (as Pb),
Manganese Compounds (as Mn),
Molybdenum (as Mo), Insoluble Compounds (Total Dust),
Nickel, Soluble Compounds (as Ni),
Vanadium,
Zinc Oxide Fume
|
| Method no.: |
ID-125G |
| |
|
| Control no.: |
T-ID125G-FV-03-0209-M |
| |
|
| Matrix: |
Air, Wipe, or Bulk |
| |
|
| OSHA Permissible Exposure
Limits: |
Permissible Exposure Limits (PELs) are
listed in Table 1 for elements commonly found in industrial environments.
This method has the capability of sampling and analyzing more than these
elements, the number being limited by instrumental capability, as well as
digestion solubility and stability. |
| |
|
| Collection Procedure: |
A calibrated personal sampling pump is
used to draw a known volume of air through a mixed-cellulose ester
membrane filter contained in a styrene cassette. |
| |
|
| Minimum Recommended Air
Volumes: |
Time Weighted Average Samples - 480 L
Short-Term Exposure Limit Samples - 30 L*
Ceiling Samples - 30 L |
| |
|
| Recommended Sampling Rate: |
2 L/min |
| |
|
| Analytical Procedure: |
Filters are digested with nitric acid,
sulfuric acid and hydrogen peroxide. Dissolution of the elements is
facilitated by addition of hydrochloric acid. Analysis is performed using
Inductively Coupled Argon Plasma-Atomic Emission Spectroscopy (ICAP-AES). |
| |
|
| Detection Limits: |
See
Table 2 |
| |
|
| Validation Level: |
See
Table 3 |
| |
|
| Precision and Accuracy: |
See
Table 3 |
| |
|
| Method Classification: |
Validated analytical method |
| |
|
| Chemist: |
Jerry Septon, Ray Abel, Michael Simmons |
| |
|
| Date (Revised): |
November, 1988 (September, 2002) |
| |
|
| * Take 60-L samples when
evaluating STEL exposures to beryllium. |
| |
|
| Commercial
manufacturers and products mentioned in this method are for descriptive
use only and do not constitute endorsements by USDOL-OSHA. Similar
products from other sources can be substituted. |
| |
|
Division of Physical Measurements and Inorganic Analyses
OSHA Technical Center
Sandy City, Utah
|
1. Introduction
1.1 Scope
1.1.1 This method describes the collection and subsequent
analysis of airborne metal and metalloid particulate by
Inductively Coupled Argon Plasma-Atomic Emission
Spectroscopy (ICAP-AES).
1.1.2 This method provides rapid simultaneous analysis and data
reduction for a wide range of elements, eliminating the
necessity of separate analyses by conventional atomic
absorption techniques.
1.1.3 This method was validated for 13 elements (Be, Cd, Co, Cr,
Cu, Fe, Mn, Mo, Ni, Pb, Sb, V, and Zn). Other elements can be added to or subtracted
from the method. The capability for expanding the
analysis to other elements is mainly dependent on
laboratory instrumentation and element solubility and
stability in the acid matrix used for digestion.
1.2 History
1.2.1.Previous to the introduction of ICAP-AES, samples
containing metallic particulates were digested in a
variety of ways and analyzed by Atomic Absorption
Spectroscopy (AAS) at the OSHA Analytical Laboratory.
1.2.2 A first generation plasma source and spectrometer
(Jarrell-Ash Model 975 Atomcomp) was then used by the OSHA
Analytical Laboratory. The analytical procedure for this
instrument is described in OSHA Method No. ID-125 (8.1).
1.2.3 Procurement of new inductively coupled plasma (ICP)
instruments, computers, and software allowed samples to be
determined using later technology. This technology
includes more sophisticated computer systems for data
reduction and instrument control.
1.2.4 When this method was originally written, three different ICP instruments at the OSHA
Salt Lake Technical Center (OSHA-SLTC) are used to apply
this method:
Jobin-Yvon (JY) Model 32 (Instruments SA, Edison, NJ)
Jarrell-Ash Model 975 Atomcomp* (Thermo Jarrell-Ash Corp., Franklin, MA)
Applied Research Lab. (ARL) Model 3560 (ARL, Sunland, CA)
These instruments are further referred to as ICP1, ICP2, or ICP3, respectively.
The Jarrell-Ash system was upgraded with a new computer,
generator, and software in 1989.
This method is applicable to any simultaneous
spectrometer. This method was validated using ICP1 and
the data is presented in a backup report (8.2). An
additional evaluation was performed using ICP3 (8.3).
2. Detection Limits and Working Ranges (8.2)
2.1 OSHA Permissible Exposure Limits (PELs) (8.4) for the elements
screened and validated are listed in Table 1. Detection limits
and working ranges are in Table 2. All reported detection limits
were calculated for 50-mL solution volumes.
2.2 The optimum working range for each element listed in
Table 2
extends several orders of magnitude above each detection limit.
3. Method Performance (8.2)
3.1 The precision and accuracy data for the 13 validated elements
using ICP1 are listed in Table 3.
These values are based on six
samples at each concentration level tested. Solutions of the 13
elements were spiked on mixed-cellulose ester filters. These
samples were then digested and analyzed using procedures mentioned
in this method and in reference 8.2.
3.2 Nine of the thirteen elements reported in Table 3 were spiked at
0.5, 1, and 2 times the PEL, assuming a 120-L air volume. Spikes for manganese were calculated
assuming a 30-L air volume. Approximately 200-L air volumes were assumed for Pb, Ni, and Sb.
3.3 The analytical error (AE) at 95% confidence for each element
listed in Table 3 was calculated as:
±AE% = 100 × [|Mean Bias| + 2(CV)]
Analytical errors for all elements tested were within ±25%; the
greatest value was ±18.1% for V. This element was validated near
it's detection limit.
4. Interferences (8.6)
High temperatures present in the plasma (5,000 to 8,000°C)
minimize most chemical and matrix interferences. Interferences do
exist, however, and can be categorized as follows:
4.1 Physical interferences such as nebulization and transport effects
are influences that determine the rate and particle size in which
analytes are delivered to the plasma. These effects are minimized
by matching the acid concentrations of samples and standards.
4.2 Chemical interferences are characterized by molecular compound
formation, ionization effects, and solute volatilization effects.
These effects are not severe in ICP analysis and are minimized by
matrix matching and careful selection of operating conditions such
as: incident plasma source power, sample uptake rate and plasma observation height.
4.3 Spectral interferences include:
a. Unresolved overlap of molecular band spectra.
b. Overlap of a spectral line from another element.
c. Background from continuous or recombination phenomena.
d. Background from stray light.
4.4 The first effect (a) can be minimized by a careful selection of
wavelengths for the reported elements. The
other types of spectral interferences (spectral overlap and
elevated background) are minimized by software which performs
interelement corrections. This software assumes a linear
relationship between the analyte and interference within the
working range limits. A spectral interference correction equation
typically used by ICP manufacturers is:
Corrected Concn = Calculated Concn - Ai × CPi
where:
| Ai |
is |
Correction factor |
| CPi |
is |
Concentration of the interfering element |
Samples having analyte concentrations above the working range
limits should be diluted into range; interelement corrections may
not be accurate above the working range. Experimentally
determined interelement corrections for the validated elements are
listed in reference 8.2.
4.5. If necessary, supplemental background correction can be performed
with additional software supplied by the instrument manufacturer.
5. Sampling
5.1 Equipment
5.1.1 Mixed cellulose ester (MCE) filters (0.8-µm pore size), cellulose backup pads, and cassettes, 37-mm diameter, part
no. MAWP 037 AO (Millipore Corp., Bedford, MA). Cassettes, filters (MCE) and backup pads of 25-mm diameter can also be used.
5.1.2 Gel bands (Omega Specialty Instrument Co., Chelmsford, MA) for sealing cassettes.
5.1.3 Sampling pumps capable of sampling at 2 L/min.
5.1.4 Assorted flexible tubing.
5.1.5 Stopwatch and bubble tube or meter for pump calibration.
5.1.6 Scintillation vials, 20-mL, part no. 74515 or 58515,
(Kimble, Div. of Owens-Illinois Inc., Toledo, OH) with
polypropylene or Teflon cap liners. If possible, submit
bulk or wipe samples in these vials for ICP analysis.
5.1.7 Smear tabs, part no. 225-24 (SKC Inc., Eighty Four, PA),
or Whatman no. 41 or no. 42 filters (Whatman LabSales
Inc. , Hillsboro, OR) for wipe sampling.
5.1.8 Gloves, disposable (for wipe sampling).
5.1.9 Ghost Wipes, 4" × 4" 1000/cs Wet with DI Water, part no. SC4250 (Environmental Express, Mt. Pleasant, SC) for wipe sampling.
5.2 Sampling Procedure - Air Samples
Welding fumes and samples requiring sample weights can be characterized using this method. Collect
samples on pre-weighed 37-mm polyvinyl chloride (PVC) filters at
2 L/min flow rate. Conduct the welding fume sampling with the
filter cassette located inside the welding helmet (8.6).
If the
free-space inside the hood precludes the use of 37-mm diameter
cassettes and filters, 25-mm sampling assemblies with pre-weighed
PVC filters can be used.
Desiccate and post-weigh each sample and then calculate total
welding fume exposure:
net weight (µg) - net
weight of blank (µg)
air volume (liters) |
= mg/m3 |
and determine compliance with the 5 mg/m3 TLV for welding fumes.
Submit the samples to the laboratory for welding fume/ICP analysis
to further characterize the samples.
5.2.1 Place a MCE filter and a cellulose backup pad in each two-
or three-piece cassette. Seal each cassette with a gel
band.
5.2.2 Calibrate each personal sampling pump with a prepared
cassette in-line to approximately 2 L/min flow rate.
5.2.3 Attach prepared cassettes to calibrated sampling pumps
(the backup pad should face the pump) and place in
appropriate positions on the employee or workplace area.
Collect the samples at about 2 L/min flow rates. Minimum
sampling times recommended are:
Recommended Sampling Times
|
| Time Weighted Average Samples |
240 min |
| Short-Term Exposure Limit Samples |
15 min* |
| Ceiling Samples |
15 min |
The analytical sensitivity of a specific analyte may
dictate using a larger sampling sampling time.
| * |
When determining compliance with the STEL for beryllium, take 30-min samples. |
| Note: |
If soluble compounds (i.e. Cr2+, Cr3+, soluble
salts of Al, Fe, Mo, Ni, ZnCl2, etc.) are suspected to be
present in the sampled air, take separate samples.
Request analysis for the specific compound(s). These
samples are analyzed using OSHA Method No.
ID-121 and not
by this method. |
5.2.4 If the filter becomes overloaded while sampling, another
filter cassette should be prepared. Consecutive samples
using shorter sampling periods should be taken if
overloading occurs.
5.2.5 Place plastic end caps on each cassette after sampling.
5.2.6 Attach an OSHA-21 seal around each cassette in such a way
as to secure the end caps.
5.3 Sampling Procedure - Wipe Samples
5.3.1 Wear clean, impervious, disposable
gloves when taking wipe samples to prevent sample contamination. Change
gloves between samples to reduce the possibility of cross contamination.
5.3.2 Moisten Smear Tabs and Whatman filters with deionized water
prior to use.
5.3.3 If using a Ghost Wipe remove it from its package and unfold it.
Next fold the Ghost Wipe in half and wipe a 10-cm × 10-cm area by
starting at the outside edge of the surface, applying firm pressure,
wipe the surface and progress towards the center by making concentric
squares of decreasing size. Fold wipe in half, with contaminant side in,
and wipe the surface again by making concentric squares of decreasing
size. Fold the wipe in half, contaminant side in, and wipe surface a
third time.
If using a Smear Tab or Whatman filter, wipe a 10-cm × 10-cm area by
starting at the outside edge of the surface, applying firm pressure,
wipe the surface and progress towards the center by making concentric
squares of decreasing size. If possible wipe the area at least 3 times.
5.3.4 Fold the wipe sample with exposed side in.
5.3.5 Transfer the wipe sample into a 20-mL scintillation vial and
seal with vinyl or electrical tape. Securely wrap an OSHA-21 seal
length-wise from vial top to bottom.
5.4 Sampling Procedure - Bulk Samples
In order of laboratory preference, bulk samples may be one of the
following:
1. a high-volume filter sample,
2. a representative settled dust (i.e. rafter) sample,
3. a sample of the bulk material in the workplace.
Transfer the bulk material into a 20-mL scintillation vial and
seal with vinyl or electrical tape. Securely wrap an OSHA-21 seal
length-wise from vial top to bottom.
5.5.Shipment
When other compounds or elements are known or suspected to be
present in the sampled air, such information should be transmitted
with the sample(s) to the laboratory.
5.5.1 Submit at least one blank sample with each set of air or
wipe samples. Blank filter samples should be handled in
the same manner as other samples, except no air is drawn
through the blank.
5.5.2 Send the samples to the laboratory with the OSHA 91A paperwork requesting ICP analysis.
5.5.3 Bulk samples should be shipped separately from air
samples. They should be accompanied by Material Safety
Data Sheets if available. Check current shipping
restrictions and ship to the laboratory by the appropriate
method.
6. Analysis
6.1 Safety Precautions
6.1.1 Prepare 1:1 H2SO4 in DI H2O cautiously.
1. Use a 1- or 2-L thick-walled, break- and heat-resistant bottle.
2. Wear thick rubber gloves, plastic apron, labcoat, and face shield.
3. Add 500 mL DI H2O to the bottle.
4. Place the bottle over the drain in a sink which has a slot vent to provide ventilation. Begin running cold tap water over the
side of the bottle, being careful not to get any tap water in the bottle. Let the level of water rise in the sink to provide
cooling of the bottle.
5. Carefully and slowly begin adding 500 mL concentrated H2SO4 to the DI H2O. Add a small quantity,
swirl to mix, and allow contents to cool. Do NOT allow boiling of solution within the container.
6. After the acid has been added, loosely cap the bottle and allow it to remain in the sink with the water running for at least 15 min.
Allow the solution to cool to room temperature.
7. A thick-walled beaker, Teflon-coated stirring bar, electronic stirrer and a ventilation hood can also be used to prepare 1:1
H2SO4 if precautions are taken to prevent solution overheating and splattering.
6.1.2 Digest all samples within a suitable exhaust hood.
6.1.3 To prevent splattering, add H2O2 (30%) to beakers in 2- to
3-drop groups.
6.1.4 Perchloric acid added to organic substances can produce
fires and/or explosions. If HClO4 solutions darken in
color while heating, immediately remove beakers from the
hotplate and carefully add a small amount of HNO3. Only
use HClO4 in exhaust hoods designed and reserved for HClO4
use.
6.1.5 Do not directly view the plasma.
6.1.6 Do not override the rf generator or torch box safety
interlocks.
6.2 Equipment
6.2.1 Inductively coupled argon plasma/atomic emission
direct-reading spectrometer, cooling unit for torch
assembly, computer, and radio-frequency (rf) generator.
6.2.2 Nebulizer.
6.2.3 Automatic sampler.
6.2.4 Peristaltic pumps (optional). Use one pump for automatic
sampler rinse. Use the other pump for sample introduction
into the nebulizer.
6.2.5 Mass Flow Controller (optional). Use the controller to
regulate nebulizer argon flow and sample uptake rate.
6.2.6 Borosilicate glass conical beakers, 125- and 250-mL.
6.2.7 Borosilicate glass volumetric flasks, 25-, 50-, 100-,
250-mL, and 1- or 2-L. Use the larger flasks for standard
preparation.
6.2.8 Thick walled, 1- or 2-L heat- and break-resistant bottle.
6.2.9 Mixed cellulose ester filters (0.45-µm pore size) and a
filtering apparatus. Use this system to remove any
insoluble particulates from sample solutions.
6.2.10 Hot plate capable of reaching 300°C.
6.2.11 Volumetric pipets, glass of various sizes.
6.2.12 Analytical balance (0.01 mg).
6.3 Reagents (reagent grade or better)
6.3.1 Deionized water (DI H2O).
6.3.2 Concentrated sulfuric (H2SO4), hydrochloric (HCl), nitric
(HNO3), and perchloric (HClO4) acids.
6.3.3 Prepare 1:1 H2SO4 (V/V) solutions as described in Section
6.1.1
6.3.4 Sample dilution solution or reagent blank
(8% HCl/4% H2SO4):
In an exhaust hood, slowly and carefully add 40 mL
concentrated H2SO4 to approximately 500 mL of DI H2O
contained in a thick-walled, heat- and break-resistant
bottle. Gently stir and allow the solution to cool to
room temperature. Slowly and carefully add 80 mL concentrated HCl, allow to cool, and dilute to 1 L with DI H2O.
6.3.5 Stock solutions of 1,000 µg/mL for standard preparation of
the various elements.
6.3.6 Hydrogen peroxide, (H2O2), 30%.
| Note: |
Some manufacturers use organotin compounds to stabilize H2O2.
Since Sn is one of the elements screened, use H2O2
that does not contain this type of stabilizer. |
6.3.7 Argon - quality as specified by the ICP manufacturer.
6.4 Standard Preparation
Prepare multielement calibration standards (STD) and
continuing calibration verification (CCV) solutions (see Addendum A for examples
of combinations) using 1,000 or 10,000 Fg/mL
stock solutions. An independent calibration verification (ICV) standard is a
mixture of elements whose concentrations are within their respective analytical
linear ranges and is used to verify the calibration standards. Whenever
possible, prepare the ICV standard from different stock solutions than those
used for calibration standards. The ICV standard should contain elements and
concentrations reflecting what is expected in the majority of the samples, or
problem elements. The final acid concentration of the STD, CCV and ICV standards
is 8% HCl/4% H2SO4. These standards should be stable for
at least 6 months.
6.5 Sample Preparation
The final acid concentration for the different sample matrices
should be 8% HCl/4% H2SO4. All of the elements validated are
soluble when using the following acid digestion procedures. Other
elements not included in the validated element list (Table 3)
should be evaluated for solubility and stability before using
these procedures.
| Note: |
Requests for analysis of compounds which have a PEL that
specifically addresses the soluble fraction (i.e., Fe, Ni, Mo,
etc.) are analyzed using OSHA Method No.
ID-121 and not by this
method. |
Filters, backup pads, wipes, and bulks are prepared by the following procedures:
6.5.1 Mixed-cellulose ester (MCE) membrane filters
1. If the beakers used for the digestion have not been cleaned using an
appropriate automated system, clean the insides of the 125-mL conical beakers by
refluxing 1:1 HNO3 using a hot plate in a ventilated hood. Carefully pour the
used 1:1 HNO3 into an appropriately labeled waste container. Allow the beakers
to cool, rinse them several times with DI H2O, and allow them to dry.
Carefully transfer any loose dust from the cassette into a labeled beaker. Using
forceps transfer the sample filter into the same digestion beaker. If the backup
pad appears contaminated, include it with the sample filter. If there is loose
dust present, rinse the cassette top (and ring, if present) with a small amount
of DI H2O and pour the water into the beaker with the sample filter. Wipe out
the cassette top (and ring, if present) interior surface with a clean Smear Tab
(or 1×2 inch section of Ghost Wipe) that has been moistened with DI H2O and
place it in the same digestion beaker with the rinse and sample filter.
Similarly wipe out the cassette bottom interior surface if the cassette contains
loose dust or if the backup pad is contaminated. Ensure that blank samples are
prepared and analyzed using the same materials and procedures as used for air
samples.
2. For samples with air volumes >200 L: Add 4 mL of 1:1 H2SO4, followed by 2 mL of concentrated HNO3 to each beaker containing the filter sample. For samples with air volumes
≤200 L: Add 2 mL of 1:1 H2SO4, followed by 2 mL of concentrated HNO3.
3. To facilitate the digestion, allow the filters to sit at least an hour in the 1:1 H2SO4. Add several drops of H2O2 (30%) to each beaker before placing
it on the hotplate.
4. Heat the beakers on a hot plate for approximately 10 min. The solutions should turn brown.
5. Cautiously add H2O2 in 2- to 3-drop groups until each solution becomes clear, colorless, or slightly yellow (the color is dependent on the concentration and type of analyte
present).
6. Heat several more minutes until dense, white fumes of SO3 just become evident. Remove the beakers from the hotplate and allow to cool.
7. Slowly and carefully add the following amount of concentrated HCl.
(CAUTION: SPLATTERING MAY OCCUR IF THE HCL IS ADDED TOO RAPIDLY OR THE H2SO4 SOLUTION IS STILL HOT):
| Acid Addition Data |
Acid Volume
|
Air Volume
|
| 2mL |
≤200 L |
| 4mL |
>200 L |
8. Rinse the sides of the beakers with DI H2O and return the beakers to the hotplate. Heat the beakers until near boiling to promote solubilization of all elements present. Remove the
beakers from the hotplate and allow to cool.
9. Quantitatively transfer the solutions into volumetric flasks using DI H2O. For samples having air volumes >200 L, dilute to 50 mL; volumes ≤200 L, dilute to 25 mL, if the filter
contains a lot of material, dilute to 50 mL.
6.5.2 Backup Pads
Contamination of the backup pad may be due to leakage of air around the filter
during sampling.
6.5.3 Wipe or Polyvinyl Chloride (PVC) Filter Samples
Polyvinyl chloride filters can be used for
sample collection and analysis by ICP. The industrial
hygienist may sample for gravimetric determinations of
total dust or total welding fumes using PVC filters and
also submit these samples for ICP analysis if a sample weight is required.
1. If the beakers used for the digestion have not
been cleaned using a appropriate automated system, reflux 1:1 HNO3 in 250-mL (for wipes) or 125-mL (for
PVC filters) conical beakers, empty and allow to
cool. Rinse the beakers several times with DI H2O
and allow to dry.
2. Place each filter or wipe in a separate washed
beaker.
3. Add the appropriate amount of 1:1 H2SO4 as listed
below:
Acid Addition Data
|
Acid Volume*
|
|
Sample Type
|
| 8 mL |
|
Wipe |
| 4 mL |
|
PVC filter (> 200 L Air Volume) |
| 2 mL |
|
PVC filter (>< or = 200 L Air Volume) |
|
* Concentrated HCl or 1:1 H2SO4
|
4. Add 10 mL concentrated HNO3 to each beaker. Place the beakers on the hotplate.
5. Add H2O2 in 2- to 3-drop groups. PVC filters and
wipes require more H2O2 for digestion than MCE
filters.
6. If HClO4 digestion precautions are followed (Section
6.1.4), 1 to 2 mL of HClO4 can also be used to
complete digestion. The PVC filter will not completely digest if
HClO4 is not added; therefore, thoroughly rinse the
filter residue with DI H2O during quantitative
transfer of the sample solution.
7. Allow digested samples to cool and carefully add the
same volume of concentrated HCl as the 1:1 H2SO4 used
in Section 6.5.3, Step 3. above.
8. Rinse the sides of the beaker with DI H2O and then
heat to near boiling.
9. After beakers have cooled to room temperature, dilute
digested wipe solutions to 100 mL. Dilute PVC filter
solutions to final volumes as stated in Section 6.5.1, Step 9.
6.5.4 Ghost Wipes
1.If the beakers used for the digestion have not been cleaned
using a appropriate automated system, reflux 1:1 HNO3 in
125-mL conical beakers, empty and allow to cool. Rinse the beakers
several times with deionized water and allow them to dry.
2. Place each Ghost Wipe in a separate washed beaker.
3. Add 4 mL of 1:1 H2SO4 to each beaker. Let
sample sit for 5 minutes.
4. Add 2 mL of deionized water followed by 2 mL concentrated HNO3
to each beaker. Let samples sit for 15 minutes. After 15 minutes, up
to 8 mL more of concentrated HNO3 can be added to
facilitate digestion, especially if samples are heavily loaded. Place
the beakers on the hotplate.
5. When solutions turns brown or black cautiously add H2O2
in 2- to 3-drop groups until each solution becomes clear, colorless,
or slightly yellow (the color is dependent on the concentration and
type of analyte present). Ghost Wipes may require up to 10 mL H2O2
before solution clears.
6. Heat solutions several more minutes until dense, white fumes of
SO3 just become evident. Remove beakers from hotplate and
allow them to cool.
7. After samples have cooled carefully add 4 mL of concentrated HCl
to each beaker.
8. Rinse the sides of the beaker with deionized water and then heat
again on hot plate until samples are near boiling.
9. After beakers have cooled to room temperature, dilute digested
Ghost Wipes solutions to 50 mL.
6.5.5 Bulks
1. Review any available material safety data sheets to
determine safe bulk handling. The safety data may
also offer a clue as to the aliquot amount needed for
adequate detection of the element(s) of interest.
2. Measure by volume or weight an appropriate aliquot of
any liquid bulk sample. Weigh the appropriate amount
of any solid bulk sample. Weigh an aliquot of any
paint bulk by placing a small amount on a MCE filter,
allow to air dry then take the dry weight.
| Note: |
Aliquot amounts of bulks are dependent on the
analytical sensitivity, detection limit, and
solubility of the material used. If uncertain, a 20-
to 50-mg aliquot of a solid material can be taken as
a starting point. Make sure the aliquot taken is
representative of the entire bulk sample. If needed,
use a mortar and pestle to grind any nonhomogeneous
particulate bulk samples in an exhaust hood. |
After measuring, transfer the aliquot to a previously cleaned or acid-washed 250-mL conical beaker.
3. Add 20 mL of 1:1 H2SO4 and digest on a hotplate.
Hydrogen peroxide (dropwise) and a few mLs of HNO3
can be carefully added to break up the matrix.
| Caution: |
Do not add the HNO3 to wet bulk materials containing
organic solvents. Significant reactions could occur. |
4. Remove the beaker from the hotplate and allow to
cool. Carefully add 20 mL of concentrated HCl and
then heat the solution to near boiling.
5. Allow to cool and quantitatively transfer to a 250-mL
volumetric flask. Dilute to volume with DI H2O.
Air, wipe, and bulk samples: If particulates are present
in any of the sample solutions, filter this solution
through a MCE filter (0.45-µm pore size) and then
re-digest the particulate and filter. Save the filtrates
for analysis.
6.6 Instrument Startup and Calibration
Follow the manufacturer's instructions for instrument start-up and
calibration. An example of ICP operating parameters is shown
below. These settings will vary from instrument to instrument:
| Gas Used |
Argon |
|
Gas Flow
(Rotameter settings) |
Plasma
Nebulizer
Auxiliary Plasma |
12 - 16 L/min
*
0.14 - 0.18 L/min |
| RF Power |
Incident
Reflected |
1.1 kW
< 5 W |
| Observation Height |
Plasma |
15 mm above work coil |
| Integration Time |
Peak Signal |
3 to 10 s |
| Wash Time |
Automatic Sampler
Without Automatic Sampler |
60 s
10 s |
| Number Of Exposures |
Standards & Samples |
2 to 10 |
| Nebulizer* |
Solution Uptake Rate
Pressure |
0.8 - 1.6 mL/min
~30 psig |
| Mass Flow Controller |
Flow Rate Range |
varies* |
| * This flow will vary depending on the type of nebulizer in use. |
6.6.1 Profile the instrument before beginning the calibration
and analysis. Follow the Standard Operating Procedure
(SOP) (8.7) or manufacturer's instructions for computer
initialization and profiling.
6.6.2 Obtain a two-point calibration curve by nebulizing the
working standards into the plasma and measuring atomic
emission intensities. For most instruments, a first-order
linear fit of the data is computer calculated and slope
and intercept coefficients are obtained. Perform
calibrations by following the instrument manufacturer's
guidelines.
6.6.3 See Addendum A for the proper sequence of
standards and samples during the analysis.
6.7 Analytical Procedure
For more details regarding analytical procedures, refer to the
instrument manufacturer's software manual(s) or the SOP (8.7).
6.7.1 If necessary, determine detection limits using the
manufacturer's software (if available). These limits
normally do not significantly change during short time
spans. A general rule is to recalculate detection limits
when an integral component (nebulizer, torch, mass flow
controller, etc.) of the ICP has been replaced or
adjusted. A typical calculation of detection limit (DL)
is shown:
| DL = |
(K × SDI × C)
(I - Io) |
× S |
where:
| S |
is |
Solution volume in mL |
| K |
is |
Degree of confidence (sigma value)* |
| SDI |
is |
Standard deviation of reagent blank intensity (Io.) |
| C |
is |
Concentration of the calibration standard in µg/mL |
| I |
is |
Total intensity of standard containing concentration C |
| Io |
is |
Background intensity (reagent blank) |
* In most cases, K=2 or 3 for qualitative and K=10 for quantitative determinations.
Reporting limits may be equal to or larger than the calculated detection limits.
Reporting limits should be verified annually for each matrix.
6.7.2 Analysis using an automatic sampler is described below:
1. Fill automatic sample vials to the minimum sample volume for one analysis and a potential rerun.
2. Load the automatic sampler with labeled standard and sample vials. A multielement working standard should
be analyzed after every 5 to 6 samples. A control standard should be occasionally analyzed to ensure
proper instrument operation. If an element or elements contained in the control standard are not
within specification (a general rule is to use a value less than ±10 to 15% of the known
concentration), the analyst should recalibrate before proceeding with the analysis.
3. Aspirate each sample or standard for approximately
1 min prior to initiating the exposure cycle. This
ensures equilibration in the plasma and minimizes
carry-over effects.
4. Dilute and reanalyze any samples containing elements
(both screened and validated) exceeding the working
range (Table 2). In particular, notice the reduced
upper limit (8 µg/mL) for lead. Interelement corrections may not be
accurate above the working range. Prepare the
dilutions by pipetting an appropriate aliquot from
the original solution and dilute with 8% HCl/4%
H2SO4.
5. Based on the calibration curve initially obtained,
convert the sample intensities to concentrations.
Then, using the air volume, solution volume, dilution
factor and sample weight, calculate the concentration
for each element analyzed as mg/m3 (air samples),
total micrograms (wipes), or percentage of total
weight (bulks) using the equations shown below.
6.8 Calculations
6.8.1 Total amount of analyte in the sample:
| µg A = (µg/mL A) × (mL S) × (DF) |
(1) |
where:
| µg A |
is |
Total µg of analyte in the sample |
| µg/mL A |
is |
Measured concentration of analyte in sample
solution (derived from calibration curve) |
| mL S |
is |
total volume of the solution analyzed |
| DF |
is |
amount of dilution applied to an aliquot of
the original solution (ratio of final
volume divided by the aliquot volume) |
6.8.2 The blank value, if any, is subtracted from each sample:
where:
| µgc A |
is |
µg of analyte, blank corrected |
| µgb A |
is |
µg of analyte in blank |
6.8.3 For air samples, the concentration of analyte in the sample is expressed in mg analyte per cubic meter for each
element or compound analyzed:
| mg/m3 = |
(µgc A) × (GF)
air
volume (L) |
|
(3) |
where:
GF is Gravimetric Factor
For those elements having a PEL listed as an oxide, the
gravimetric factors for the validated elements are:
1.4298 for Fe2O3 1.2447 for ZnO 1.7852
for V2O5
6.8.4 Convert bulk sample analytes to % composition using:
| %(w/w) = |
(µgc A) (100%)
(sample weight) (1,000 µg/mg) |
|
(4) |
where:
| µgc A |
is |
analyte amount (µg) |
| Sample wt |
is |
aliquot (in mg) of bulk taken in Section
6.5.4 |
7. Reporting Results
7.1 Air sample results are reported as mg/m3. Results for analytes
having a PEL as an oxide are reported as mg/m3 of the oxide.
7.2 Wipe sample concentrations are calculated and reported as total
micrograms for each element.
7.3 Bulk sample results are calculated and reported as elemental
percent by weight (or volume if liquid aliquots were used). Due
to differences in sample matrices between bulks and standards,
bulk results are approximate for each element determined. (Elemental values are
to be reported for all bulk analyses, do not use a gravimetric factor.)
7.4 Determinations of the screened elements or compounds are not
routinely reported. Spectral interference corrections for these
analytes are not included and validations have not been performed.
If a sample has a screened analyte over the PEL, the analyst
should contact her/his supervisor. Additional sampling, or if
possible, additional analysis of the original sample should be
performed to quantitate the potential overexposure.
8. References
8.1 Occupational Safety and Health Administration Analytical
Laboratory: OSHA Analytical Methods Manual (USDOL/OSHA-SLCAL
Method No. ID-125). Cincinnati, OH: American Conference of
Governmental Industrial Hygienists (Pub. No. ISBN: 0-936712-66-X),
1985.
8.2 Occupational Safety and Health Administration Technical Center:
ICP Backup Data Report (ID-125G) by J.C. Septon. Salt Lake City,
UT. Revised 1991.
8.3 Occupational Safety and Health Administration Technical Center:
ICP Backup Data Report (ARL 3560) by J.C. Septon. Salt Lake City,
UT. In progress.
8.4 "Toxic and Hazardous Substances," Code of Federal Regulations
Title 29, Pt. 1910.1000, Subpart Z. 1987. pp 676-682.
8.5 National Institute for Occupational Safety and Health: NIOSH
Manual of Analytical Methods, 2nd ed., Vol. 7 (DHEW/NIOSH Pub. No.
82-100). Cincinnati, OH, 1981. Method No. 351.
8.6 United States Department of Labor, OSHA: "Memorandum, Sampling for
Welding Fumes" by Patricia Clark, Director Designate, Directorate
of Compliance Programs. United States Department of Labor, OSHA,
Washington, DC, February 14, 1989. [Memo].
8.7 Occupational Safety and Health Administration Analytical
Laboratory: ICP Standard Operating Procedure by J.C. Septon. Salt
Lake City, UT. 1988 (unpublished).
Table 1
Air Contaminants - OSHA Permissible Exposure Limits* |
|
| Element |
Substance Exposed to |
PEL
(mg/m3)
|
|
|
|
TWA |
CEILING |
|
|
|
|
| Ag** |
Metal and soluble compounds
(as Ag) |
0.01 |
|
|
|
| As** |
Inorganic compounds (as As) |
See 29 CFR 1910.1018 for applications |
| Be# |
Beryllium and compounds
(as Be) |
0.002 |
0.005## |
|
|
|
| Ca** |
Calcium oxide |
5 |
|
|
|
| Cd |
Fume
Dust
(See 29 CFR 1910.1027) |
0.1
0.2 |
0.3
0.6 |
|
|
|
| Co |
Metal dust and fume (as Co) |
0.1 |
|
|
|
| Cr |
Cr metal (as Cr) |
1 |
|
|
|
| Cu |
Fumes (as Cu)
Dusts and mists (as Cu) |
0.1
1 |
|
|
|
| Fe |
Dicyclopentadienyl iron Total dust
Iron oxide fume (as Fe2O3) |
15
10 |
|
|
|
| Fe,V |
Ferrovanadium dust |
1 |
|
|
|
|
| Mg** |
Magnesium oxide fume Total particulate |
15 |
|
|
|
| Mn |
Mn compounds (as Mn)
Mn fume (as
Mn) |
|
5
5 |
|
|
|
| Mo |
Insoluble compounds (as Mo) Total dust |
15 |
|
|
|
| Ni |
Metal and insoluble
compounds (as Ni) |
1
1 |
|
|
|
| Pb |
Inorganic (see 29 CFR 1910.1025) |
| Sb |
Sb and compounds (as Sb) |
0.5 |
|
|
|
| Se** |
Se and compounds (as Se) |
0.2 |
|
|
|
| Sn** |
Inorganic compounds except oxides (as Sn) |
2 |
|
|
| Te** |
Te and compounds (as Te) |
0.1 |
|
|
|
| Zn |
Zinc oxide fume
Zinc oxide Total dust
Zinc stearate Total dust |
5
15
15 |
|
|
|
|
|
| * |
From reference 8.4 |
| ** |
Elements screened - PELs are listed for information only, because the
screened elements are not digested or analyzed using optimum conditions. |
| # |
Beryllium also has a Peak PEL of 0.025 µg/m³. |
| ## |
Both the Ceiling Limit and the STEL for beryllium are for a maximum 30-min duration. |
|
|
| Note: |
Compounds having total and respirable dust PELs of 15 and 5 mg/m³
respectively, are normally analyzed gravimetrically. Elements contained in
these dust samples can be identified by this or other methods, if necessary. |
|
Table 2
Detection Limits* and Upper Limits |
Analyte
|
Qual. Det.
Limit (µg)
|
Quan. Det.
Limit (µg)
|
Upper
Limit (µg/mL)
|
| Ag** |
1.1 |
3.8 |
** |
| Al** |
6.8 |
23. |
200 |
| As** |
1.7 |
5.7 |
20 |
| Be |
0.013 |
0.043 |
5 |
| Ca** |
0.79 |
2.6 |
20 |
| Cd |
0.14 |
0.47 |
50 |
| Co |
1.2 |
4.0 |
100 |
| Cr |
0.40 |
1.3 |
50 |
| Cu |
0.64 |
2.1 |
50 |
| Fe |
8.9 |
30. |
200 |
| Mg** |
2.1 |
7.1 |
20 |
| Mn |
0.061 |
0.20 |
15 |
| Mo |
0.52 |
1.7 |
100 |
| Pb |
2.1 |
7.0 |
8 |
| Ni |
0.59 |
2.0 |
100 |
| Sb |
4.2 |
14. |
100 |
| Se** |
3.2 |
11. |
20 |
| Si** |
2.4 |
8.1 |
20 |
| Sn** |
3.5 |
12. |
** |
| V |
0.57 |
1.9 |
100 |
| Zn |
0.14 |
0.47 |
50 |
| * |
Calculations are based on a 50-mL solution volume and equations listed in Section
6.7.1
Each detection limit is dependent on the spectral wavelength and order used for analysis. |
| ** |
Screened elements - Limits are approximate - the digestion and analysis are not optimized for these elements. |
Both the detection limits and upper limits were determined using ICP1
(JY-32 ICP). Performance may vary from instrument to instrument. Upper
limits are the upper linear range for each element. These were determined
using a linear model (8.2).
Table 3
Precision and Accuracy Data* for Spiked Samples |
|
Element
|
CV
|
Bias
|
Range (µg)
|
Analytical
Error (± %)
|
| Be |
0.010 |
0.010 |
0.12-0.48 |
2.9 |
| Cd1 |
0.014 |
-0.005 |
6-24 |
3.2 |
| Co |
0.020 |
-0.040 |
6-24 |
8.1 |
| Cr2 |
0.015 |
-0.074 |
60-240 |
10.3 |
| Cu3 |
0.017 |
0.054 |
60-240 |
8.7 |
| Fe |
0.018 |
-0.012 |
420-1680 |
4.8 |
| Mn |
0.032 |
0.077 |
75-300 |
14.1 |
| Mo4 |
0.047 |
-0.029 |
225-900 |
12.3 |
| Ni |
0.025 |
0.017 |
105-420 |
6.6 |
| Pb |
0.040 |
0.04 |
5-20 |
12.1 |
| Sb |
0.014 |
0.012 |
60-240 |
3.9 |
| V |
0.045 |
0.090 |
3.5-14 |
18.1 |
| Zn |
0.007 |
0.014 |
240-960 |
2.9 |
| * |
Reported values were obtained from ICP1 (ISA JY-32 ICP). Results may vary from instrument to instrument. |
The following exceptions were used when calculating spike amounts:
| 1 |
PEL for fume (0.1 mg/m3) used |
| 2 |
PEL for insoluble forms (1 mg/m3) used |
| 3 |
PEL for dust form (1 mg/m3) was used |
| 4 |
5 mg/m3 target concentration was used. This was performed because a very large amount of spike would be necessary if the 15 mg/m3
PEL for Mo was used. |
|
| CV |
Coefficient of Variation |
| Instrument Calibration & Quality Control
Sequence |
QC
Function |
Acronym |
Frequency |
Acceptance
Criteria |
Analytes & Nominal Concentrations (ppm) |
Initial
Calibration1 |
CB |
At the beginning of each analytical sequence
and
as required by failing performance checks |
n/a |
|
| STD-A |
RSD < 2%
(replicate
readings) |
Target analytes reported to clients:
1 - Be
5 - Cr, Cu, Pb, V
10 - Cd. Co, Mn, Mo, Ni, Sb, Zn
100 - Fe |
| STD-B |
Anticipated interfering analytes:
1 - Ag, Pt
10 - Al, As, Ce, Mg, Nb, Se, Sn, Ti |
Initial
Performance
Checks |
ICV |
Immediately following the initial calibration |
90-110%
Recovery |
0.5 - Be
2.5 - Cr, Cu, Pb, V
5 - Cd, Co, Mn, Mo, Ni, Sb, Zn
50 - Fe |
| CCV |
Immediately following each calibration event in
the analytical sequence
(IB must follow ICS) |
0.5 - Be
2.5 - Cr, Cu, Pb, V
5 - Cd, Co, Mn, Mo, Ni, Sb, Zn
50 - Fe |
| RLV |
75-125%
Recovery |
0.002 - Be
0.01 - Cd
0.03 - V
0.05 - Co, Cu, Mn
0.1 - Pb
0.2 - Cr
0.5 - Fe, Mo, Ni, Sb, Zn |
| ICS |
85-115%
Recovery
(Target
Analytes
Only) |
0.2 - Be, Cd
1 - Pb, V, Ag
5 - Co, Cr, Co, Mn, Mo, Ni, Sb, Zn, As, Se, Sn
10 - Ce, Nb, Pt
200 - Fe, Al, Mg, Ti |
| IB |
|X| < Reporting Limit
(Target
Analytes Only) |
|
Continuing
Performance
Checks |
CCV |
Every 10
field
samples |
90-110%
Recovery |
0.5 - Be
2.5 - Cr, Cu, Pb, V
5 - Cd, Co, Mn, Mo, Ni, Sb, Zn
50 - Fe |
| RLV |
75-125%
Recovery |
0.002 - Be
0.01 - Cd
0.03 - V
0.05 - Co, Cu, Mn
0.1 - Pb
0.2 - Cr
0.5 - Fe, Mo, Ni, Sb, Zn |
| IB |
|X| < Reporting Limit
(Target
Analytes Only) |
|
Sequence
Termination
Performance
Checks |
CCV |
At the end
of the
analytical
sequence |
90-110%
Recovery |
0.5 - Be
2.5 - Cr, Cu, Pb, V
5 - Cd, Co, Mn, Mo, Ni, Sb, Zn
50 - Fe |
| RLV |
75-125%
Recovery |
0.002 - Be
0.01 - Cd
0.03 - V
0.05 - Co, Cu, Mn
0.1 - Pb
0.2 - Cr
0.5 - Fe, Mo, Ni, Sb, Zn |
| IB |
|X| < Reporting Limit
(Target
Analytes Only) |
|
| Term: |
STD |
Calibration Standard(s) |
|
CB |
Calibration Blank |
|
ICV |
Independent Calibration Verification
(alternate source reference material) |
|
CCV |
Continuing Calibration Verification |
|
IB |
Instrument Blank (part of continuing
calibration verification) |
|
RVL |
Reporting Limit Verification |
|
ICS |
Interference Check Sample |
|
Target Analyte: any analyte that
is reported to clients in the final report of results |
1The linear calibration range for target analytes may be established/verified
for each analytical sequence by analyzing a high level calibration
verification standard at the end of the analytical sequence. In order to
report sample results that are measured at levels between this high standard
and the initial calibration standard, the high standard must exhibit a
recovery between 90 and 110%.
Addendum B
Ghost Wipe Backup Data
|
1. Backup Data
General background information about the determination of detection limits and reproducibility of
the overall procedure is found in the "Evaluation Guidelines for Surface Sampling
Methods".1 The Guidelines define analytical parameters, specific laboratory tests, statistical calculations and
acceptance criteria.
Sample preparation of Ghost Wipes was performed as described in Section 6.5.4 of OSHA Method
ID-125G. Instrument analytical conditions used are described in Section 1.9 of this document. Lot
number of Ghost Wipes used was 9901, manufactured on May 20, 1999.
1.1 Detection Limit of the overall procedure (DLOP) and reliable quantitation limit (RQL).
The DLOP is measured as mass per sample. Seven Ghost wipes were spiked with
descending increments of analyte. These spiked Ghost Wipes, and a sample blank, were
analyzed and the data obtained used to calculate the required parameters (standards error
of estimate and the slope) for the calculation of the DLOP.
The RQL is considered the lower limit for precise quantitative measurements. It is
determined from the regression line parameters obtained for the calculation of the
DLOP, providing 75% to 125% of the analyte is recovered.
The analyte target, calculated DLOPs, and RQLs for the 13 validated analytes are shown
below:
Table 1.1.1
Target Concentrations, DLOPs, and RQLs |
|
analyte and
wavelength |
target concn
(µg) |
DLOP
(µg) in 50 mL |
RQL
(µg) in 50 mL |
recovery at RQL*
(%) |
|
| BE 313.107 |
4 |
0.0052 |
0.017 |
112.8 |
| Cd 214.440 |
10 |
0.053 |
0.18 |
94.6 |
| Co 228.616 |
200 |
0.32 |
1.1 |
101.2 |
| Cr 267.716 |
1000 |
1.0 |
3.4 |
101.2 |
| Cu 324.752 |
200 |
0.45 |
1.5 |
106.0 |
| Fe 238.204 |
2000 |
1.5 |
5.1 |
105.5 |
| Mn 257.610 |
400 |
0.29 |
0.97 |
101.1 |
| Mo 202.031 |
1000 |
0.8 |
2.7 |
103.8 |
| Ni 232.003 |
1000 |
0.92 |
3.1 |
97.5 |
| Pb 220.353 |
100 |
0.55 |
1.8 |
81.9 |
| Sb 206.836 |
1000 |
1.5 |
5.1 |
105.8 |
| V 292.402 |
56 |
0.08 |
0.29 |
103.1 |
| Zn 213.857 |
800 |
2.2 |
7.3 |
95.4 |
|
*Percent recovery at or near the RQL.
Table 1.1.2
Detection Limit of the Overall Procedure for Beryllium |
 |
|
| mass per sample (µg) |
intensity |
|
| 0 |
-7.4 |
| 0.012 |
64.7 |
| 0.025 |
121.9 |
| Figure 1.1.2 Plot
of data to determine DLOP/RQL for Beryllium. (Y=44.36.7X +2.2) |
| 0.05 |
224.5 |
| 0.1 |
436.1 |
| 0.15 |
666.4 |
| 0.2 |
895.1 |
| 0.25 |
1110.4 |
|
Table 1.1.3
Detection Limit of the Overall Procedure for Cadmium |
 |
|
| mass per sample (µg) |
intensity |
|
| 0 |
0.4 |
| 0.031 |
4.5 |
| 0.062 |
4.6 |
| 0.12 |
9 |
| 0.25 |
15.8 |
| 0.38 |
24.6 |
| 0.5 |
32.5 |
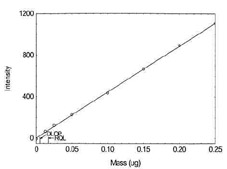
Figure 1.1.3. Plot of data to
determine the DLOP/RQL for Cadmium. (Y=65.2X + 0.7) |
| 0.63 |
43 |
|
Table 1.1.4
Detection Limit of the Overall Procedure for Cobalt |
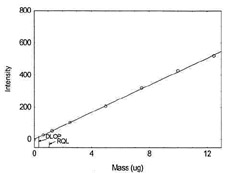 |
|
| mass per sample (µg) |
intensity |
|
| 0 |
-2.1 |
| 0.62 |
26.7 |
| 1.25 |
52.9 |
| 2.5 |
104.8 |
| 5 |
206.7 |
| 7.5 |
318.5 |
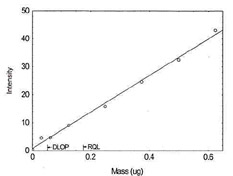
Figure 1.1.4. Plot of data to determine DLOP/RQL
for Cobalt. (Y=42.2X + 0.4) |
| 10 |
428.2 |
| 12.5 |
519.8 |
|
Table 1.1.5
Detection Limit of the Overall Procedure for Chromium |
 |
|
| mass per sample (µg) |
intensity |
|
| 0 |
46.3 |
| 3.1 |
673.8 |
| 6.2 |
1332.1 |
| 12.5 |
2749 |
| 25 |
5258 |
| 37.5 |
8015 |
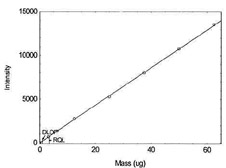
Figure 1.1.5. Plot of data to determine DLOP/RQL
for Chromium. (Y=215.6X - 6.5) |
| 50 |
10769.5 |
| 62.5 |
13546.3 |
|
Table 1.1.6
Detection Limit of the Overall Procedure for Copper |
 |
|
| mass per sample (µg) |
intensity |
|
| 0 |
1050.3 |
| 0.62 |
1404 |
| 1.3 |
1904.2 |
| 2.5 |
2687.7 |
| 5 |
4444.8 |
| 7.5 |
6128.2 |
| 10 |
7844.6 |
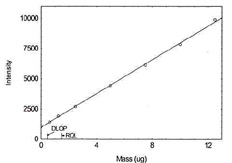
Figure 1.1.6. Plot of data to determine DLOP/RQL
for Copper. (Y=136.4X + 648.9) |
| 12.5 |
9893.2 |
|
Table 1.1.7
Detection Limit of the Overall Procedure for Iron |
 |
|
| mass per sample (µg) |
intensity |
|
| 0 |
699.2 |
| 6.2 |
1546.4 |
| 12.5 |
2371.5 |
| 25 |
4035.6 |
| 50 |
7338.3 |
| 75 |
10830.9 |
| 100 |
14309.3 |
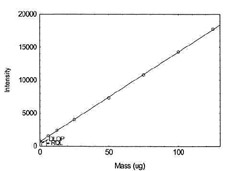
Figure 1.1.7. Plot of data to determine DLOP/RQL
for Iron. (Y= 136.4X + 648.9) |
| 125 |
17759.7 |
|
Table 1.1.8
Detection Limit of the Overall Procedure for Manganese |
 |
|
| mass per sample (µg) |
intensity |
|
| 0 |
166.6 |
| 1.2 |
2552.4 |
| 2.5 |
5015.1 |
| 5 |
9899.9 |
| 10 |
19028.4 |
| 15 |
28853.3 |
| 20 |
38573.7 |
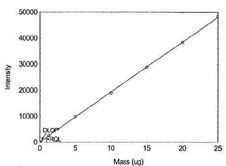
Figure 1.1.8. Plot of data to determine DLOP/RQL
for Manganese. (Y= 1924.2X + 124.5) |
| 25 |
48431.3 |
|
Table 1.1.9
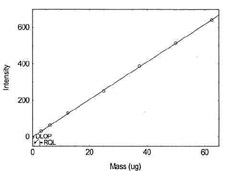
Detection Limit of the Overall Procedure for Molybdenum |
 |
|
| mass per sample (µg) |
intensity |
|
| 0 |
-2.7 |
| 3.1 |
32.3 |
| 6.2 |
63.2 |
| 12.5 |
130.3 |
| 25 |
251.6 |
| 37.5 |
388 |
| 50 |
515.2 |
Figure 1.1.9. Plot of data to determine DLOP/RQL
for Molybdenum. (Y= 10.3X - 1.1) |
| 62.5 |
641.4 |
|
Table 1.1.10
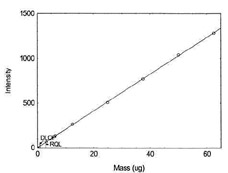
Detection Limit of the Overall Procedure for Nickel |
 |
|
| mass per sample (µg) |
intensity |
|
| 0 |
0.9 |
| 3.1 |
65.5 |
| 6.2 |
135.7 |
| 12.5 |
265.1 |
| 25 |
508.5 |
| 37.5 |
770.4 |
| 50 |
1040.4 |
Figure 1.1.10. Plot of data to determine
DLOP/RQL for Nickel. (Y= 20.6X + 3.0) |
| 62.5 |
1284 |
|
Table 1.1.11
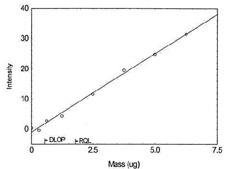
Detection Limit of the Overall Procedure for Lead |
 |
|
| mass per sample (µg) |
intensity |
|
| 0 |
0.3 |
| 0.31 |
-0.4 |
| 0.62 |
2.7 |
| 1.25 |
4.3 |
| 2.5 |
11.6 |
| 3.8 |
19.5 |
| 5 |
24.8 |
Figure 1.1.11. Plot of data to determine
DLOP/RQL for Lead. (Y= 5.2X - 1.0) |
| 3.2 |
31.5 |
|
Table 1.1.12
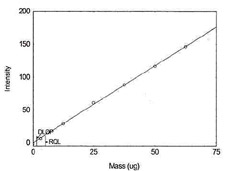
Detection Limit of the Overall Procedure for Antimony |
 |
|
| mass per sample (µg) |
intensity |
|
| 0 |
-1.3 |
| 3.1 |
6 |
| 6.2 |
15.2 |
| 12.5 |
28.8 |
| 25 |
60.8 |
| 37.5 |
88.1 |
| 50 |
116.6 |
Figure 1.1.12. Plot of data to determine
DLOP/RQL for Antimony. (Y= 2.4X - 0.4) |
| 62.5 |
146.3 |
|
Table 1.1.13
Detection Limit of the Overall Procedure for Vanadium |
 |
|
| mass per sample (µg) |
intensity |
|
| 0 |
5.7 |
| 0.17 |
39.8 |
| 0.35 |
64.5 |
| 0.7 |
114.6 |
| 1.4 |
230.2 |
| 2.1 |
352.4 |
| 2.8 |
472.4 |
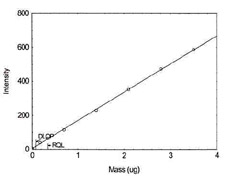
Figure 1.1.13 Plot of data to
determine DLOP/RQL for Vanadium. (Y= 165.9X + 4.6) |
| 3.5 |
586.7 |
|
Table 1.1.14
Detection Limit of the Overall Procedure for Zinc |
 |
|
| mass per sample (µg) |
intensity |
|
| 0 |
805.8 |
| 2.5 |
879.8 |
| 5 |
1088.1 |
| 10 |
1445.5 |
| 20 |
2201.2 |
| 30 |
3046.4 |
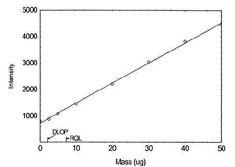
Figure 1.1.14. Plot of data to determine DLOP/RQL for Zinc (Y= 76.0X + 725.7) |
| 40 |
3825.4 |
| 50 |
4482.3 |
|
1.2 Storage Test
Storage samples were prepared by liquid-spiking Ghost Wipes at the
target concentration. Twelve storage samples were prepared. Three samples were analyzed
on the day prepared. Nine of the samples were stored at ambient temperature (about
22ºC). At 5-day intervals three samples were analyzed. Results were blank corrected.
Table 1.2.1
Storage Test for Beryllium |
 |
|
| time (days) |
recovery (%) |
|
| 0 |
90.2 |
91.3 |
92.8 |
| 5 |
90.7 |
93.7 |
91.2 |
| 10 |
90.1 |
88.9 |
92.0 |
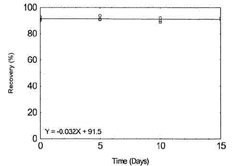
Figure 1.2.1. Storage test for Beryllium. |
| 15 |
91.1 |
91.3 |
91.9 |
|
Table 1.2.2
Storage Test for Cadmium |
 |
|
| time (days) |
recovery (%) |
|
| 0 |
91.0 |
92.6 |
93.3 |
| 5 |
95.8 |
97.9 |
96.8 |
| 10 |
96.8 |
94.7 |
97.7 |
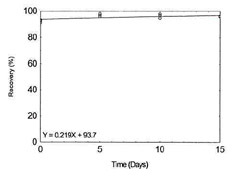
Figure 1.2.2. Storage test for Cadmium. |
| 15 |
96.0 |
96.0 |
96.4 |
|
Table 1.2.3
Storage Test for Cobalt |
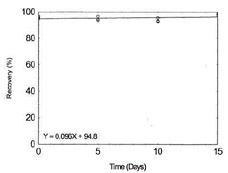 |
|
| time (days) |
recovery (%) |
|
| 0 |
94.8 |
95.9 |
96.7 |
| 5 |
93.6 |
96.6 |
94.3 |
| 10 |
93.2 |
92.7 |
95.6 |
Figure 1.2.3. Storage test for Cobalt. |
| 15 |
97.4 |
98.4 |
97.4 |
|
Table 1.2.4
Storage test for Chromium |
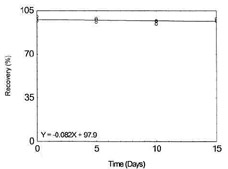 |
|
| time (days) |
recovery (%) |
|
| 0 |
96.7 |
100.1 |
98.1 |
| 5 |
95.6 |
98.7 |
97.6 |
| 10 |
96.4 |
94.1 |
97.0 |
Figure 1.2.4. Storage test for Chromium. |
| 15 |
96.7 |
98.5 |
97.0 |
|
Table 1.2.5
Storage Test for Copper |
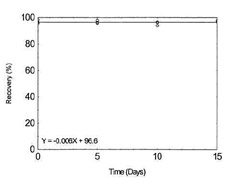 |
|
| time (days) |
recovery (%) |
|
| 0 |
95.7 |
96.6 |
98.2 |
| 5 |
95.9 |
97.8 |
96.3 |
| 10 |
96.3 |
93.8 |
96.6 |
Figure 1.2.5. Storage test for Copper. |
| 15 |
96.8 |
97.8 |
96.8 |
|
Table 1.2.6
Storage Test for Iron |
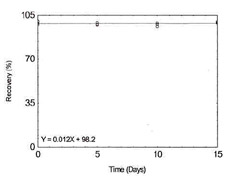 |
|
| time (days) |
recovery (%) |
|
| 0 |
97.5 |
98.6 |
100.3 |
| 5 |
97.1 |
99.0 |
97.7 |
| 10 |
97.7 |
95.9 |
98.8 |
Figure 1.2.6. Storage test for Iron. |
| 15 |
98.7 |
99.9 |
99.0 |
|
Table 1.2.7
Storage Test for Manganese |
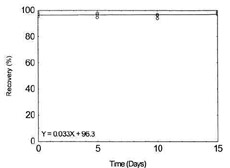 |
|
| time (days) |
recovery (%) |
|
| 0 |
95.2 |
98.5 |
96.8 |
| 5 |
94.6 |
97.5 |
96.5 |
| 10 |
96.0 |
93.8 |
96.6 |
Figure 1.2.7. Storage test for Manganese. |
| 15 |
96.9 |
98.7 |
97.2 |
|
Table 1.2.8
Storage Test for Molybdenum |
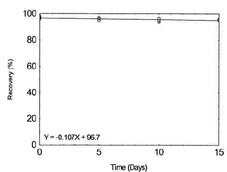 |
|
| time (days) |
recovery (%) |
|
| 0 |
96.0 |
97.0 |
98.0 |
| 5 |
96.3 |
97.0 |
95.0 |
| 10 |
94.7 |
93.6 |
96.5 |
Figure 1.2.8. Storage test for Molybdenum. |
| 15 |
95.3 |
95.8 |
95.8 |
|
Table 1.2.9
Storage Test for Nickel |
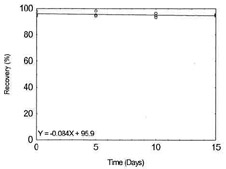 |
|
| time (days) |
recovery (%) |
|
| 0 |
94.5 |
97.7 |
95.1 |
| 5 |
94.2 |
98.1 |
95.0 |
| 10 |
94.5 |
93.3 |
96.2 |
Figure 1.2.9. Storage test for Nickel. |
| 15 |
94.4 |
94.6 |
95.3 |
|
Table 1.2.10
Storage Test for Lead |
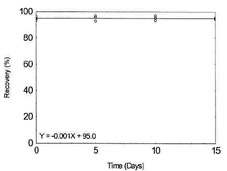 |
|
| time (days) |
recovery (%) |
|
| 0 |
93.6 |
94.8 |
96.3 |
| 5 |
93.0 |
96.6 |
95.7 |
| 10 |
95.2 |
93.4 |
96.7 |
Figure 1.2.10. Storage test for Lead. |
| 15 |
94.3 |
95.3 |
95.0 |
|
Table 1.2.11
Storage Test for Antimony |
 |
|
| time (days) |
recovery (%) |
|
| 0 |
85.8 |
85.5 |
90.1 |
| 5 |
86.9 |
88.2 |
85.6 |
| 10 |
78.2 |
81.7 |
86.6 |
Figure 1.2.11. Storage test for Antimony. |
| 15 |
85.5 |
89.6 |
89.0 |
|
Table 1.2.12
Storage Test for Vanadium |
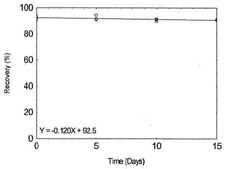 |
|
| time (days) |
recovery (%) |
|
| 0 |
91.3 |
93.6 |
92.4 |
| 5 |
91.2 |
94.4 |
91.7 |
| 10 |
91.0 |
89.5 |
91.6 |
Figure 1.2.12. Storage test for Vanadium. |
| 15 |
91.1 |
90.7 |
91.2 |
|
Table 1.2.13
Storage Test for Zinc |
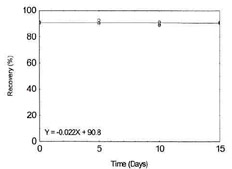 |
|
| time (days) |
recovery (%) |
|
| 0 |
90.0 |
90.9 |
91.4 |
| 5 |
89.9 |
92.8 |
90.7 |
| 10 |
89.6 |
88.7 |
91.4 |
Figure 1.2.13. Storage test for Zinc. |
| 15 |
90.3 |
91.4 |
90.9 |
|
1.3 Sampler Removal Efficiency
Six 100-cm2 glass plates were liquid-spiked at the target concentrations and allowed to
dry. Samples were collected from each surface by placing a Ghost Wipe folded in half at
an outside edge of the glass plate and progressing towards the center making concentric
squares of decreasing size while applying firm pressure. The Ghost Wipe was folded in
half with the contaminant side in and the glass plate was wiped a second time. The Ghost
Wipe was again folded in half and the surface wiped a third time. Results were blank
corrected.
Table 1.3.1
Sampler Removal Efficiency
Data for Beryllium on Ghost Wipes |
|
Table 1.3.2
Sampler Removal Efficiency
Data for Cadmium on Ghost Wipes |
|
|
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
|
|
| 4 |
3.770 |
94.2 |
|
10 |
9.300 |
93.0 |
| 4 |
3.755 |
93.9 |
|
10 |
9.168 |
91.6 |
| 4 |
3.938 |
98.4 |
|
10 |
9.625 |
96.2 |
| 4 |
3.880 |
97.0 |
|
10 |
9.535 |
95.4 |
| 4 |
3.848 |
96.0 |
|
10 |
9.595 |
96.0 |
| 4 |
3.785 |
84.4 |
|
10 |
8.330 |
83.3 |
|
|
|
Table 1.3.3
Sampler Removal Efficiency
Data for Cobalt on Ghost Wipes |
|
Table 1.3.4
Sampler Removal Efficiency
Data for Chromium on Ghost Wipes |
|
|
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
|
|
| 200 |
182.1 |
91.0 |
|
1000 |
964.5 |
96.4 |
| 200 |
182.0 |
91.0 |
|
1000 |
889.5 |
88.9 |
| 200 |
190.6 |
95.3 |
|
1000 |
1001.5 |
100.1 |
| 200 |
187.6 |
93.8 |
|
1000 |
917.5 |
91.7 |
| 200 |
187.8 |
93.9 |
|
1000 |
992.0 |
99.2 |
| 200 |
162.8 |
81.4 |
|
1000 |
927.0 |
92.7 |
|
|
|
Table 1.3.5
Sampler Removal Efficiency
Data for Copper on Ghost Wipes |
|
Table 1.3.6
Sampler Removal Efficiency
Data for Iron on Ghost Wipes |
|
|
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
|
|
| 200 |
194.1 |
97.0 |
|
2000 |
1914 |
95.7 |
| 200 |
193.6 |
96.8 |
|
2000 |
1913 |
95.6 |
| 200 |
200.7 |
100.4 |
|
2000 |
1987 |
99.4 |
| 200 |
195.6 |
97.9 |
|
2000 |
1940 |
97.0 |
| 200 |
197.3 |
98.6 |
|
2000 |
1957 |
97.8 |
| 200 |
172.5 |
86.2 |
|
2000 |
1704 |
85.2 |
|
|
|
Table 1.3.7
Sampler Removal Efficiency
Data for Manganese on Ghost Wipes |
|
Table 1.3.8
Sampler Removal Efficiency
Data for Molybdenum on Ghost Wipes |
|
|
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
|
|
| 400 |
385.1 |
96.3 |
|
1000 |
892.0 |
89.2 |
| 400 |
356.8 |
89.2 |
|
1000 |
698.5 |
69.8 |
| 400 |
400.0 |
100.0 |
|
1000 |
791.0 |
79.1 |
| 400 |
367.1 |
91.8 |
|
1000 |
780.5 |
78.0 |
| 400 |
395.7 |
98.9 |
|
1000 |
724.5 |
72.4 |
| 400 |
371.1 |
92.8 |
|
1000 |
749.0 |
74.9 |
|
|
|
Table 1.3.9
Sampler Removal Efficiency
Data for Nickel on Ghost Wipes |
|
Table 1.3.10
Sampler Removal Efficiency
Data for Lead on Ghost Wipes |
|
|
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
|
|
| 1000 |
913.5 |
91.4 |
|
100 |
90.95 |
91.0 |
| 1000 |
843.0 |
84.3 |
|
100 |
89.00 |
89.0 |
| 1000 |
955.0 |
95.5 |
|
100 |
93.60 |
93.6 |
| 1000 |
881.0 |
88.1 |
|
100 |
91.55 |
91.6 |
| 1000 |
941.5 |
94.2 |
|
100 |
92.75 |
92.8 |
| 1000 |
875.0 |
87.5 |
|
100 |
80.40 |
80.4 |
|
|
|
Table 1.3.11
Sampler Removal Efficiency
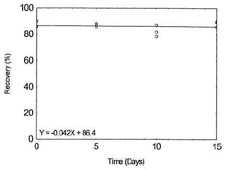
Data for Antimony on Ghost Wipes |
|
Table 1.3.12
Sampler Removal Efficiency
Data for Vanadium on Ghost Wipes |
|
|
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
|
|
| 1000 |
809.5 |
81.0 |
|
56 |
52.08 |
93.0 |
| 1000 |
654.5 |
65.4 |
|
56 |
49.41 |
88.2 |
| 1000 |
733.0 |
73.3 |
|
56 |
54.23 |
96.8 |
| 1000 |
717.5 |
71.8 |
|
56 |
50.23 |
89.7 |
| 1000 |
698.5 |
69.8 |
|
56 |
52.43 |
93.6 |
| 1000 |
690.5 |
69.0 |
|
56 |
50.98 |
91.0 |
|
|
|
Table 1.3.13
Sampler Removal Efficiency Data for Zinc on Ghost Wipes |
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
| 800 |
710.7 |
88.8 |
| 800 |
712.2 |
89.0 |
| 800 |
754.2 |
94.3 |
| 800 |
739.2 |
92.4 |
| 800 |
735.2 |
91.9 |
| 800 |
641.7 |
80.2 |
|
1.4 Analytical Method Recovery and Stability of Digested Samples
1.4.1 Analytical Method Recovery
Analytical method recovery (AMR) was determined by liquid-spiking Ghost Wipes
with the analytes at the RQL, 0.1, 1.0, and 10 times the target concentrations.
These samples were stored overnight at ambient temperature and then analyzed.
Results were blank corrected.
Table 1.4.1.1
Analytical Method Recovery
of Beryllium from Ghost Wipes |
|
| level |
sample number |
| × target concn |
µg per sampler |
1 |
2 |
3 |
4 |
mean |
|
| RQL |
0.017 |
114.7 |
82.4 |
94.1 |
82.4 |
93.4 |
| 0.1 |
0.4 |
95.6 |
99.5 |
96.1 |
97.4 |
97.2 |
| 1.0 |
4 |
94.5 |
95.1 |
90.6 |
94.4 |
93.6 |
| 10 |
40 |
92.2 |
92.9 |
93.6 |
94.4 |
93.3 |
| AMR |
|
|
|
|
|
94.4 |
|
Table 1.4.1.2
Analytical Method Recovery
of Cadmium from Ghost Wipes |
|
| level |
sample number |
| × target concn |
µg per sampler |
1 |
2 |
3 |
4 |
mean |
|
| RQL |
0.18 |
83.3 |
86.1 |
86.1 |
80.6 |
84.0 |
| 0.1 |
1 |
99.5 |
102.5 |
98.0 |
99.0 |
99.8 |
| 1.0 |
10 |
99.4 |
99.80 |
96.5 |
99.4 |
98.8 |
| 10 |
100 |
98.3 |
100.4 |
102.4 |
102.5 |
100.9 |
| AMR |
|
|
|
|
|
95.9 |
|
Table 1.4.1.3
Analytical Method Recovery
of Cobalt from Ghost Wipes |
|
| level |
sample number |
| × target concn |
µg per sampler |
1 |
2 |
3 |
4 |
mean |
|
| RQL |
1.1 |
105.9 |
92.7 |
94.1 |
97.7 |
97.6 |
| 0.1 |
20 |
99.2 |
101.9 |
100.2 |
101.1 |
100.6 |
| 1.0 |
200 |
98.2 |
98.1 |
93.7 |
97.7 |
96.9 |
| 10 |
2000 |
96.6 |
97.6 |
98.2 |
99.3 |
97.9 |
| AMR |
|
|
|
|
|
98.2 |
|
Table 1.4.1.4
Analytical Method Recovery
of Chromium from Ghost Wipes |
|
| level |
sample number |
| × target concn |
µg per sampler |
1 |
2 |
3 |
4 |
mean |
|
| RQL |
3.4 |
116.9 |
102.5 |
107.5 |
107.9 |
108.7 |
| 0.1 |
100 |
98.8 |
102.9 |
99.8 |
101.6 |
100.8 |
| 1.0 |
1000 |
99.3 |
98.9 |
95.3 |
99.1 |
98.2 |
| 10 |
10000 |
96.8 |
99.3 |
101.0 |
101.6 |
99.7 |
| AMR |
|
|
|
|
|
101.8 |
|
Table 1.4.1.5
Analytical Method Recovery
of Copper from Ghost Wipes |
|
| level |
sample number |
| × target concn |
µg per sampler |
1 |
2 |
3 |
4 |
mean |
|
| RQL |
1.5 |
110.0 |
111.7 |
100.0 |
89.7 |
102.8 |
| 0.1 |
20 |
99.8 |
102.7 |
101.4 |
101.0 |
101.2 |
| 1.0 |
200 |
98.0 |
97.4 |
97.4 |
97.4 |
96.6 |
| 10 |
2000 |
94.8 |
97.5 |
99.4 |
99.4 |
97.7 |
| AMR |
|
|
|
|
|
99.6 |
|
Table 1.4.1.6
Analytical Method Recovery
of Iron from Ghost Wipes |
|
| level |
sample number |
| × target concn |
µg per sampler |
1 |
2 |
3 |
4 |
mean |
|
| RQL |
5.1 |
131.9 |
112.9 |
107.4 |
93.0 |
111.3 |
| 0.1 |
200 |
102.3 |
106.2 |
102.9 |
104.2 |
103.9 |
| 1.0 |
2000 |
101.2 |
100.8 |
97.0 |
101.1 |
100.0 |
| 10 |
20000 |
98.5 |
101.7 |
103.8 |
103.8 |
102.0 |
| AMR |
|
|
|
|
|
104.3 |
|
Table 1.4.1.7
Analytical Method Recovery
of Manganese from Ghost Wipes |
|
| level |
sample number |
| × target concn |
µg per sampler |
1 |
2 |
3 |
4 |
mean |
|
| RQL |
0.97 |
121.0 |
107.4 |
137.7 |
165.6 |
132.9 |
| 0.1 |
40 |
99.2 |
103.0 |
100.3 |
101.9 |
101.1 |
| 1.0 |
400 |
99.1 |
98.6 |
95.1 |
98.7 |
97.9 |
| 10 |
4000 |
96.4 |
99.0 |
100.7 |
101.2 |
99.3 |
| AMR |
|
|
|
|
|
107.8 |
|
Table 1.4.1.8
Analytical Method Recovery
of Molybdenum from Ghost Wipes |
|
| level |
sample number |
| × target concn |
µg per sampler |
1 |
2 |
3 |
4 |
mean |
|
| RQL |
2.7 |
122.6 |
100.0 |
108.2 |
96.3 |
106.8 |
| 0.1 |
100 |
101.8 |
103.5 |
102.2 |
103.0 |
102.6 |
| 1.0 |
1000 |
98.9 |
98.7 |
94.6 |
98.5 |
97.7 |
| 10 |
1000 |
97.4 |
98.4 |
98.8 |
99.8 |
98.6 |
| AMR |
|
|
|
|
|
101.4 |
|
Table 1.4.1.9
Analytical Method Recovery
of Nickel from Ghost Wipes |
|
| level |
sample number |
| × target concn |
µg per sampler |
1 |
2 |
3 |
4 |
mean |
|
| RQL |
3.1 |
122.1 |
105.5 |
121.1 |
106.1 |
113.7 |
| 0.1 |
10 |
98.0 |
101.0 |
99.4 |
99.6 |
99.5 |
| 1.0 |
100 |
98.7 |
98.2 |
94.7 |
97.4 |
97.2 |
| 10 |
1000 |
96.9 |
97.7 |
97.8 |
99.1 |
97.9 |
| AMR |
|
|
|
|
|
102.1 |
|
Table 1.4.1.10
Analytical Method Recovery
of Lead from Ghost Wipes |
|
| level |
sample number |
| × target concn |
µg per sampler |
1 |
2 |
3 |
4 |
mean |
|
| RQL |
1.8 |
53.9 |
65.0 |
80.6 |
67.2 |
66.7 |
| 0.1 |
10 |
90.7 |
93.6 |
101.0 |
104.0 |
97.3 |
| 1.0 |
100 |
98.8 |
98.8 |
94.5 |
98.5 |
97.6 |
| 10 |
1000 |
96.6 |
99.8 |
101.3 |
102.4 |
100.0 |
| AMR |
|
|
|
|
|
90.4 |
|
Table 1.4.1.11
Analytical Method Recovery
of Antimony from Ghost Wipes |
|
| level |
sample number |
| × target concn |
µg per sampler |
1 |
2 |
3 |
4 |
mean |
|
| RQL |
5.1 |
76.9 |
59.8 |
37.1 |
47.8 |
55.4 |
| 0.1 |
100 |
90.9 |
90.6 |
90.6 |
92.5 |
91.2 |
| 1.0 |
1000 |
92.3 |
92.1 |
89.2 |
91.7 |
91.3 |
| 10 |
10000 |
88.7 |
85.9 |
93.2 |
94.5 |
90.6 |
| AMR |
|
|
|
|
|
82.1 |
|
Table 1.4.1.12
Analytical Method Recovery
of Vanadium from Ghost Wipes |
|
| level |
sample number |
| × target concn |
µg per sampler |
1 |
2 |
3 |
4 |
mean |
|
| RQL |
0.29 |
94.8 |
86.2 |
96.6 |
101.7 |
94.8 |
| 0.1 |
5.6 |
93.0 |
96.8 |
94.2 |
97.2 |
95.3 |
| 1.0 |
56 |
94.6 |
93.9 |
91.1 |
93.3 |
93.2 |
| 10 |
560 |
92.6 |
93.6 |
93.9 |
94.4 |
93.6 |
| AMR |
|
|
|
|
|
94.2 |
|
Table 1.4.1.13
Analytical Method Recovery
of Zinc from Ghost Wipes |
|
| level |
sample number |
| × target concn |
µg per sampler |
1 |
2 |
3 |
4 |
mean |
|
| RQL |
7.3 |
125.8 |
132.7 |
104.6 |
91.1 |
113.6 |
| 0.1 |
80 |
92.3 |
96.8 |
91.7 |
95.4 |
94.0 |
| 1.0 |
800 |
93.6 |
93.2 |
89.6 |
92.9 |
92.3 |
| 10 |
8000 |
93.5 |
92.6 |
93.2 |
94.1 |
93.4 |
| AMR |
|
|
|
|
|
98.3 |
|
1.4.2 Stability of Digested Samples
Stability of the digested samples were investigated by reanalyzing the four 1.0
times the target concentration samples seven days after initial analysis. After the
original analysis was performed the samples were stored at ambient temperature in the 50 mL volumetric flasks in which they were brought to volume in. For the
second analysis new aliquots were taken from each volumetric. Results were
blank corrected.
Table 1.4.2.1
Stability of Digested Samples
for Beryllium |
|
Table 1.4.2.2
Stability of Digested Samples
for Cadmium |
|
|
|
| initial (%) |
after 7 days (%) |
difference (%) |
|
initial (%) |
after 7 days (%) |
difference (%) |
|
|
|
| 94.5 |
91.9 |
2.9 |
|
99.4 |
97.2 |
2.2 |
| 95.1 |
92.3 |
2.8 |
|
99.8 |
98.4 |
1.4 |
| 90.6 |
87.3 |
3.3 |
|
96.5 |
94.0 |
2.5 |
| 94.4 |
9191 |
3.3 |
|
99.4 |
97.9 |
1.5 |
|
|
|
Table 1.4.2.3
Stability of Digested Samples
for Cobalt |
|
Table 1.4.2.4
Stability of Digested Samples
for Chromium |
|
|
|
| initial (%) |
after 7 days (%) |
difference (%) |
|
initial (%) |
after 7 days (%) |
difference (%) |
|
|
|
| 98.2 |
98.3 |
0.1 |
|
99.3 |
97.8 |
1.5 |
| 98.1 |
98.7 |
0.6 |
|
98.9 |
97.2 |
1.7 |
| 93.7 |
94.0 |
0.3 |
|
95.3 |
93.4 |
1.9 |
| 97.7 |
97.7 |
0.0 |
|
99.1 |
97.2 |
1.9 |
|
|
|
Table 1.4.2.5
Stability of Digested Samples
for Copper |
|
Table 1.4.2.6
Stability of Digested Samples
for Iron |
|
|
|
| initial (%) |
after 7 days (%) |
difference (%) |
|
initial (%) |
after 7 days (%) |
difference (%) |
|
|
|
| 98.0 |
96.8 |
1.2 |
|
101.2 |
99.2 |
1.0 |
| 97.4 |
95.9 |
1.5 |
|
100.8 |
98.6 |
2.2 |
| 93.4 |
91.9 |
1.5 |
|
97.0 |
94.1 |
2.9 |
| 97.4 |
95.8 |
1.6 |
|
101.1 |
98.7 |
2.4 |
|
|
|
Table 1.4.2.7
Stability of Digested Samples
for Manganese |
|
Table 1.4.2.8
Stability of Digested Samples
for Molybdenum |
|
|
|
| initial (%) |
after 7 days (%) |
difference (%) |
|
initial (%) |
after 7 days (%) |
difference (%) |
|
|
|
| 99.0 |
97.4 |
1.6 |
|
98.9 |
99.2 |
0.3 |
| 98.6 |
96.6 |
2.0 |
|
98.7 |
99.8 |
1.1 |
| 95.1 |
93.0 |
2.1 |
|
94.6 |
99.8 |
5.2 |
| 98.7 |
96.6 |
2.1 |
|
98.5 |
98.6 |
0.1 |
|
|
|
Table 1.4.2.9
Stability of Digested Samples
for Nickel |
|
Table 1.4.2.10
Stability of Digested Samples
for Lead |
|
|
|
| initial (%) |
after 7 days (%) |
difference (%) |
|
initial (%) |
after 7 days (%) |
difference (%) |
|
|
|
| 98.7 |
96.6 |
2.1 |
|
98.8 |
97.0 |
1.8 |
| 98.2 |
96.8 |
1.9 |
|
98.8 |
97.6 |
1.2 |
| 94.7 |
92.4 |
2.3 |
|
94.5 |
94.2 |
0.3 |
| 97.4 |
95.4 |
2.0 |
|
98.5 |
98.2 |
0.3 |
|
|
|
Table 1.4.2.11
Stability of Digested Samples
for Antimony |
|
Table 1.4.2.12
Stability of Digested Samples
for Vanadium |
|
|
|
| initial (%) |
after 7 days (%) |
difference (%) |
|
initial (%) |
after 7 days (%) |
difference (%) |
|
|
|
| 92.3 |
93.2 |
0.9 |
|
94.6 |
92.1 |
2.5 |
| 92.1 |
93.7 |
1.6 |
|
93.9 |
92.0 |
1.9 |
| 89.2 |
89.9 |
0.7 |
|
91.0 |
87.3 |
3.7 |
| 91.7 |
93.6 |
1.9 |
|
93.3 |
90.3 |
3.0 |
|
|
|
Table 1.4.2.13
Stability of Digested Samples for Zinc |
|
| initial (%) |
after 7 days (%) |
difference (%) |
|
| 93.6 |
93.5 |
0.1 |
| 93.2 |
93.9 |
0.7 |
| 89.6 |
89.6 |
0.0 |
| 92.9 |
92.7 |
0.2 |
|
1.5 Reproducibility
1.5.1 Sampling Reproducibility Six 100-cm2 glass plates were liquid-spiked at the target concentrations and
allowed to dry. Two chemist, other than the one developing the method,
conducted surface sampling. Results were blank corrected.
Table 1.5.1
Sampling Reproducibility Data for Beryllium on Ghost Wipes |
|
| |
Chemist 1 |
Chemist 2 |
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
recovered (µg/sample) |
recovery (%) |
|
| 4 |
3.821 |
95.5 |
3.621 |
90.5 |
| 4 |
3.788 |
94.7 |
3.646 |
91.2 |
| 4 |
3.687 |
92.2 |
3.678 |
91.9 |
| 4 |
3.973 |
99.3 |
3.640 |
91.0 |
| 4 |
4.023 |
100.6 |
3.711 |
92.8 |
| 4 |
3.899 |
97.5 |
3.599 |
90.0 |
|
Table 1.5.2
Sampling Reproducibility Data for Cadmium on Ghost Wipes |
|
| |
Chemist 1 |
Chemist 2 |
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
recovered (µg/sample) |
recovery (%) |
|
| 10 |
9.615 |
96.2 |
9.480 |
94.8 |
| 10 |
9.545 |
95.4 |
9.520 |
95.2 |
| 10 |
9.310 |
93.1 |
9.510 |
95.1 |
| 10 |
9.865 |
98.6 |
9.550 |
95.5 |
| 10 |
10.06 |
100.6 |
9.665 |
96.6 |
| 10 |
9.805 |
98.0 |
9.390 |
93.9 |
|
Table 1.5.3
Sampling Reproducibility Data for Cobalt on Ghost Wipes |
|
| |
Chemist 1 |
Chemist 2 |
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
recovered (µg/sample) |
recovery (%) |
|
| 200 |
182.1 |
91.1 |
183.5 |
91.8 |
| 200 |
179.5 |
89.7 |
185.5 |
92.9 |
| 200 |
173.3 |
86.6 |
185.4 |
92.7 |
| 200 |
187.6 |
93.8 |
185.0 |
92.5 |
| 200 |
190.5 |
95.2 |
188.2 |
94.1 |
| 200 |
184.0 |
92.0 |
182.5 |
91.2 |
|
Table 1.5.4
Sampling Reproducibility Data for Chromium on Ghost Wipes |
|
| |
Chemist 1 |
Chemist 2 |
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
recovered (µg/sample) |
recovery (%) |
|
| 1000 |
976.0 |
97.6 |
918.0 |
91.8 |
| 1000 |
981.0 |
87.1 |
956.0 |
95.6 |
| 1000 |
905.5 |
90.6 |
944.5 |
94.4 |
| 1000 |
991 |
99.1 |
935.0 |
93.5 |
| 1000 |
1025 |
102.5 |
980.5 |
98.0 |
| 1000 |
1012 |
101.2 |
954.5 |
95.4 |
|
Table 1.5.5
Sampling Reproducibility Data for Copper on Ghost Wipes |
|
| |
Chemist 1 |
Chemist 2 |
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
recovered (µg/sample) |
recovery (%) |
|
| 200 |
196.0 |
98.0 |
189.5 |
94.8 |
| 200 |
193.9 |
96.9 |
190.4 |
95.2 |
| 200 |
185.1 |
92.5 |
189.6 |
94.8 |
| 200 |
198.9 |
99.5 |
188.2 |
94.0 |
| 200 |
202.3 |
101.2 |
190.7 |
95.3 |
| 200 |
195.4 |
97.7 |
185.6 |
92.7 |
|
Table 1.5.6
Sampling Reproducibility Data for Iron on Ghost Wipes |
|
| |
Chemist 1 |
Chemist 2 |
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
recovered (µg/sample) |
recovery (%) |
|
| 2000 |
1961 |
98.1 |
1933 |
96.6 |
| 2000 |
1939 |
97.0 |
1949 |
97.5 |
| 2000 |
1838 |
91.9 |
1948 |
97.4 |
| 2000 |
2002 |
100.0 |
1936 |
96.8 |
| 2000 |
2038 |
101.9 |
1962 |
98.1 |
| 2000 |
1975 |
98.7 |
1909 |
95.5 |
|
Table 1.5.7
Sampling Reproducibility Data for Manganese on Ghost Wipes |
|
| |
Chemist 1 |
Chemist 2 |
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
recovered (µg/sample) |
recovery (%) |
|
| 400 |
390.5 |
97.6 |
369.0 |
92.3 |
| 400 |
392.4 |
98.1 |
383.7 |
95.9 |
| 400 |
363.5 |
90.9 |
379.0 |
94.8 |
| 400 |
396.2 |
99.1 |
374.8 |
93.7 |
| 400 |
409.1 |
102.2 |
391.8 |
98.0 |
| 400 |
403.3 |
100.8 |
382.6 |
95.6 |
|
Table 1.5.8
Sampling Reproducibility Data for Molybdenum on Ghost Wipes |
|
| |
Chemist 1 |
Chemist 2 |
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
recovered (µg/sample) |
recovery (%) |
|
| 1000 |
737.5 |
73.8 |
906.0 |
90.6 |
| 1000 |
862.0 |
86.2 |
932.0 |
93.2 |
| 1000 |
803.5 |
80.4 |
952.0 |
95.2 |
| 1000 |
934.0 |
93.4 |
908.0 |
90.7 |
| 1000 |
839.0 |
83.9 |
970.5 |
97.0 |
| 1000 |
929.5 |
93.0 |
938.5 |
93.8 |
|
Table 1.5.9
Sampling Reproducibility Data for Nickel on Ghost Wipes |
|
| |
Chemist 1 |
Chemist 2 |
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
recovered (µg/sample) |
recovery (%) |
|
| 1000 |
925.0 |
92.5 |
895.5 |
89.6 |
| 1000 |
928.0 |
92.8 |
932.5 |
93.2 |
| 1000 |
875.5 |
87.6 |
920.5 |
92.0 |
| 1000 |
945.0 |
94.5 |
915.0 |
91.5 |
| 1000 |
967.0 |
96.7 |
959.0 |
95.9 |
| 1000 |
959.0 |
95.9 |
936.0 |
93.6 |
|
Table 1.5.10
Sampling Reproducibility Data for Lead on Ghost Wipes |
|
| |
Chemist 1 |
Chemist 2 |
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
recovered (µg/sample) |
recovery (%) |
|
| 100 |
94.55 |
94.6 |
92.65 |
92.6 |
| 100 |
92.30 |
92.3 |
93.3 |
93.3 |
| 100 |
89.45 |
89.5 |
92.9 |
92.9 |
| 100 |
96.15 |
96.2 |
92.4 |
92.4 |
| 100 |
97.10 |
97.1 |
94.6 |
94.6 |
| 100 |
94.95 |
95.0 |
91.6 |
91.6 |
|
Table 1.5.11
Sampling Reproducibility Data for Antimony on Ghost Wipes |
|
| |
Chemist 1 |
Chemist 2 |
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
recovered (µg/sample) |
recovery (%) |
|
| 1000 |
757.0 |
75.7 |
751.5 |
75.2 |
| 1000 |
836.0 |
83.6 |
780.5 |
78.0 |
| 1000 |
813.0 |
81.3 |
797.5 |
79.8 |
| 1000 |
911.5 |
91.2 |
814.5 |
81.4 |
| 1000 |
823.0 |
82.3 |
882.5 |
88.2 |
| 1000 |
925.0 |
92.5 |
827.5 |
82.8 |
|
Table 1.5.12
Sampling Reproducibility Data for Vanadium on Ghost Wipes |
|
| |
Chemist 1 |
Chemist 2 |
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
recovered (µg/sample) |
recovery (%) |
|
| 56 |
52.58 |
93.9 |
48.54 |
86.7 |
| 56 |
51.68 |
92.3 |
50.88 |
90.9 |
| 56 |
49.37 |
88.2 |
50.78 |
90.7 |
| 56 |
53.28 |
95.2 |
50.48 |
90.2 |
| 56 |
54.38 |
97.1 |
52.48 |
93.7 |
| 56 |
53.83 |
96.1 |
51.23 |
91.5 |
|
Table 1.5.13
Sampling Reproducibility Data for Zinc on Ghost Wipes |
|
| |
Chemist 1 |
Chemist 2 |
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
recovered (µg/sample) |
recovery (%) |
|
| 800 |
725.5 |
90.7 |
729.2 |
91.2 |
| 800 |
720.7 |
90.1 |
727.7 |
91.0 |
| 800 |
697.7 |
87.2 |
742.2 |
92.8 |
| 800 |
750.7 |
93.8 |
731.2 |
91.4 |
| 800 |
754.7 |
94.3 |
751.7 |
94.0 |
| 800 |
732.7 |
91.6 |
728.2 |
91.0 |
|
1.5.2 Analytical Reproducibility
Six samples were prepared by spiking media in the same manner that was used in
the preparation of samples for the storage study. The samples were submitted to
the OSHA SLTC for analysis and the samples were analyzed by a chemist other than the one developing the method. The samples were
analyzed after being
stored for 15 days at ambient temperature. Results were blank corrected.
Table 1.5.2.1
Analytical Reproducibility Data for Beryllium on Ghost Wipes |
|
Table 1.5.2.2
Analytical Reproducibility Data for Cadmium on Ghost Wipes |
|
|
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
|
|
| 4 |
3.767 |
94.2 |
|
10 |
10.10 |
101.0 |
| 4 |
3.903 |
97.6 |
|
10 |
10.30 |
103.0 |
| 4 |
3.958 |
99.0 |
|
10 |
10.46 |
104.6 |
| 4 |
4.008 |
100.2 |
|
10 |
10.74 |
107.4 |
| 4 |
4.024 |
100.6 |
|
10 |
10.50 |
105.0 |
| 4 |
3.892 |
97.3 |
|
10 |
10.34 |
103.4 |
|
|
|
Table 1.5.2.3
Analytical Reproducibility Data for Cobalt on Ghost Wipes |
|
Table 1.5.2.4
Analytical Reproducibility Data for Chromium on Ghost Wipes |
|
|
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
|
|
| 200 |
193.8 |
96.9 |
|
1000 |
97.3 |
97.3 |
| 200 |
199.4 |
99.7 |
|
1000 |
1004 |
100.4 |
| 200 |
201.3 |
100.6 |
|
1000 |
1025 |
102.5 |
| 200 |
205.2 |
102.6 |
|
1000 |
1033 |
103.3 |
| 200 |
203.9 |
102.0 |
|
1000 |
1016 |
101.6 |
| 200 |
199.2 |
99.6 |
|
1000 |
1006 |
100.6 |
|
|
|
Table 1.5.2.5
Analytical Reproducibility Data for Copper on Ghost Wipes |
|
Table 1.5.2.6
Analytical Reproducibility Data for Iron on Ghost Wipes |
|
|
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
|
|
| 200 |
193.0 |
96.5 |
|
2000 |
2016 |
100.8 |
| 200 |
198.0 |
99.0 |
|
2000 |
2070 |
103.5 |
| 200 |
202.1 |
101.0 |
|
2000 |
2119 |
106.0 |
| 200 |
203.0 |
101.5 |
|
2000 |
2134 |
106.7 |
| 200 |
202.7 |
101.4 |
|
2000 |
2104 |
105.2 |
| 200 |
196.4 |
98.2 |
|
2000 |
2069 |
103.4 |
|
|
|
Table 1.5.2.7
Analytical Reproducibility Data for Manganese on Ghost Wipes |
|
Table 1.5.2.8
Analytical Reproducibility Data for Molybdenum on Ghost Wipes |
|
|
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
|
|
| 400 |
392.4 |
98.1 |
|
1000 |
982.4 |
98.2 |
| 400 |
404.4 |
101.1 |
|
1000 |
879.9 |
88.0 |
| 400 |
412.6 |
103.2 |
|
1000 |
1157 |
115.7 |
| 400 |
415.6 |
103.9 |
|
1000 |
1040 |
104.0 |
| 400 |
409.9 |
102.5 |
|
1000 |
1043 |
104.3 |
| 400 |
404.8 |
101.2 |
|
1000 |
1016 |
101.6 |
|
|
|
Table 1.5.2.9
Analytical Reproducibility Data for Nickel on Ghost Wipes |
|
Table 1.5.2.10
Analytical Reproducibility Data for Lead on Ghost Wipes |
|
|
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
|
|
| 1000 |
955.1 |
95.5 |
|
100 |
96.10 |
96.1 |
| 1000 |
983.6 |
98.4 |
|
100 |
97.75 |
97.8 |
| 1000 |
990.1 |
99.01 |
|
100 |
98.00 |
98.0 |
| 1000 |
1005 |
100.5 |
|
100 |
101.2 |
101.2 |
| 1000 |
997.6 |
99.8 |
|
100 |
99.6 |
99.6 |
| 1000 |
981.6 |
98.16 |
|
100 |
97.85 |
97.8 |
|
|
|
Table 1.5.2.11
Analytical Reproducibility Data for Antimony on Ghost Wipes |
|
Table 1.5.2.12
Analytical Reproducibility Data for Vanadium on Ghost Wipes |
|
|
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
|
|
| 1000 |
907.4 |
90.7 |
|
56 |
51.53 |
92.0 |
| 1000 |
729.9 |
73.0 |
|
56 |
53.55 |
95.6 |
| 1000 |
1107 |
111 |
|
56 |
53.85 |
96.2 |
| 1000 |
942.4 |
94.2 |
|
56 |
54.60 |
97.5 |
| 1000 |
943.4 |
94.3 |
|
56 |
54.80 |
97.9 |
| 1000 |
917.9 |
91.8 |
|
56 |
53.50 |
95.5 |
|
|
|
Table 1.5.2.13
Analytical Reproducibility Data for Zinc on Ghost Wipes |
|
| theoretical (µg/surface) |
recovered (µg/sample) |
recovery (%) |
|
| 800 |
733.4 |
91.7 |
| 800 |
751.9 |
94.0 |
| 800 |
761.4 |
95.2 |
| 800 |
771.9 |
96.5 |
| 800 |
770.9 |
96.4 |
| 800 |
750.9 |
93.9 |
|
1.6 Interferences
1.6.1 Media, Reagents, and Surface.
Six blank Ghost Wipes were analyzed, after each being used to wipe a separate cleaned glass plate, to determine interferences due to contamination from the
glass surface, media, and reagents.
Table 1.6.1.1
Contamination to Analysis from the Media and Reagents (µg found) |
|
| analyte |
wipe 1 |
wipe 2 |
wipe 3 |
wipe 4 |
wipe 5 |
wipe 6 |
average |
|
| Be |
0.009 |
0.004 |
0.000 |
0.008 |
0.008 |
0.008 |
0.006 |
| Cd |
0.000 |
0.015 |
0.070 |
0.000 |
0.000 |
0.000 |
0.014 |
| Co |
0.090 |
0.060 |
0.100 |
0.000 |
0.020 |
0.035 |
0.051 |
| Cr |
0.030 |
0.015 |
0.000 |
0.09 |
0.020 |
0.100 |
0.042 |
| Cu |
1.690 |
1.190 |
1.040 |
1.010 |
1.280 |
1.355 |
1.261 |
| Fe |
4.900 |
3.265 |
3.140 |
3.475 |
3.840 |
4.795 |
3.902 |
| Mn |
0.103 |
0.022 |
0.021 |
0.020 |
0.037 |
0.026 |
0.038 |
| Mo |
0.000 |
0.000 |
0.000 |
0.000 |
0.060 |
0.075 |
0.022 |
| Ni |
0.000 |
0.000 |
0.000 |
0.070 |
0.000 |
0.000 |
0.012 |
| Pb |
0.430 |
0.000 |
0.000 |
0.515 |
0.000 |
0.000 |
0.158 |
| Sb |
0.000 |
0.435 |
0.000 |
0.000 |
0.000 |
0.000 |
0.072 |
| V |
0.040 |
0.000 |
0.015 |
0.020 |
0.030 |
0.000 |
0.018 |
| Zn |
13.88 |
8.755 |
8.300 |
6.065 |
9.080 |
9.560 |
9.273 |
|
Six blank Ghost Wipes were analyzed, after each being used to wipe a separate cleaned glass plate, to determine interferences due to contamination from the
glass surface, media, and reagents.
Table 1.6.1.2
Contamination to Analysis from the Glass Surface, Media and Reagents
(µg found) |
|
| analyte |
wipe 1 |
wipe 2 |
wipe 3 |
wipe 4 |
wipe 5 |
wipe 6 |
average |
|
| Be |
0.000 |
0.000 |
0.000 |
0.000 |
0.002 |
0.000 |
0.000 |
| Cd |
0.030 |
0.020 |
0.020 |
0.005 |
0.005 |
0.020 |
0.017 |
| Co |
0.090 |
0.055 |
0.090 |
0.075 |
0.000 |
0.145 |
0.076 |
| Cr |
0.155 |
0.355 |
0.220 |
0.070 |
0.370 |
0.370 |
0.257 |
| Cu |
1.325 |
1.170 |
1.015 |
1.040 |
1.340 |
1.405 |
1.216 |
| Fe |
4.865 |
5.130 |
4.425 |
4.170 |
6.200 |
6.130 |
5.153 |
| Mn |
0.132 |
0.198 |
0.138 |
0.088 |
0.120 |
0.205 |
0.147 |
| Mo |
0.125 |
0.355 |
0.410 |
0.000 |
0.000 |
0.165 |
0.176 |
| Ni |
0.440 |
0.450 |
0.470 |
0.355 |
0.305 |
0.675 |
0.449 |
| Pb |
0.000 |
0.000 |
0.335 |
0.245 |
0.640 |
0.120 |
0.223 |
| Sb |
1.390 |
0.950 |
0.000 |
0.360 |
0.525 |
1.240 |
0.744 |
| V |
0.105 |
0.055 |
0.040 |
0.000 |
0.020 |
0.015 |
0.039 |
| Zn |
21.59 |
12.30 |
11.07 |
14.79 |
12.07 |
9.970 |
13.63 |
|
1.6.2 Spectral Interferences
The following Inter-Element Corrections (IEC) were used to correct for spectral
interferences:
Table 1.6.2.1
Radial ICP IEC Factors a,b,c |
| analyte & wavelength |
interfering analytes |
| Co |
Cr |
Cu |
Fe |
Mn |
Mo |
Ni |
Ti |
V |
| Cd 313.107 |
- |
- |
- |
0.0576 |
- |
- |
- |
- |
- |
| Co 214.44 |
- |
- |
- |
- |
- |
- |
0.1681 |
1.6467 |
- |
| Cr 228.616 |
- |
- |
- |
- |
0.2 |
- |
- |
- |
- |
| Cu 267.716 |
- |
- |
- |
- |
- |
0.4168 |
- |
- |
- |
| Fe 238.204 |
- |
0.0478 |
- |
- |
- |
- |
- |
- |
- |
| Ni 232.003 |
- |
47.35 |
- |
- |
- |
6.187 |
- |
- |
- |
| Pb 220.353 |
- |
- |
- |
- |
- |
- |
- |
- |
- |
| Sb 206.836 |
0.1 |
11.628 |
- |
- |
- |
- |
- |
- |
- |
| V 292.402 |
0.7907 |
- |
- |
- |
- |
- |
- |
0.8331 |
- |
| Zn 213.857 |
- |
- |
1.329 |
0.0518 |
- |
- |
4.691 |
0.2 |
- |
a. The IEC factor2
is the apparent analyte
concentration in ppb divided by the interferent concentration in ppm and
is calculated as follows:
| IEC Factor = |
Apparent Analyte Cocnc (ppb) |
| Interferent Concn (ppm) |
Where the apparent analyte concentration is the connection of the
interferent measured at the analyte wavelength in the absence of the
analyte.
The corrected analyte concentration is calculated as follows:
Corrected Analyte Concn (ppb)=
Measured Analyte Concn (ppb) - (Interferent Concn (ppm) * IEC Factor)
b. A dash indicates that no interference was observed
on the analyte from the interfering analyte. Interfering analytes were
analyzed at the following concentration:
200 ppm: Fe
100 ppm: Cr, Cu, Co, Mo, Ni
50 ppm: Mn, V, Ti
c. IEC factors are instrument dependent. Interferences
listed in this table do not represent all possible interferences.
1.7 Solubility and Stability of Lead Sulfate
The solubility of lead
sulfate was investigated by weighing out various amounts and placing it in a
conical beaker, along with a Ghost Wipe, and digested. The samples were
reanalyzed four days later to determine if any lead had precipitated out of
solution. Results were not blank corrected.
Table 1.7.1
Lead Sulfate Solubility and Stability |
|
| |
|
day 1 |
day 4 |
| PbSO4used (mg) |
theoretical for lead* (µg) |
lead found (µg) |
recovery (%) |
lead found (µg) |
recovery (%) |
|
| 0.025 |
16.73 |
15.92 |
95.2 |
16.82 |
100.5 |
| 0.078 |
52.23 |
49.82 |
95.4 |
50.30 |
96.3 |
| 0.162 |
108.5 |
105.9 |
97.6 |
107.8 |
99.3 |
| 0.230 |
154.0 |
153.2 |
99.5 |
155.2 |
100.7 |
| 0.252 |
168.7 |
162.7 |
96.4 |
167.0 |
98.9 |
| 0.477 |
319.4 |
313.7 |
98.2 |
319.8 |
100.1 |
| 0.595 |
398.4 |
395.9 |
99.4 |
402.7 |
101.1 |
| 0.698 |
467.4 |
472.6 |
101.1 |
472.6 |
101.1 |
| 0.821 |
549.7 |
544.1 |
99.0 |
555.7 |
101.1 |
| 1.125 |
753.3 |
739.1 |
98.1 |
751.2 |
99.7 |
| 1.254 |
839.6 |
826.1 |
98.4 |
838.7 |
99.9 |
| 1.715 |
1148 |
1121 |
97.6 |
1144 |
99.6 |
| 2.025 |
1356 |
1326 |
97.8 |
1346 |
99.3 |
| 2.738 |
1833 |
1821 |
99.3 |
1845 |
100.6 |
| 4.584 |
3069 |
2993 |
97.5 |
3048 |
99.3 |
|
*Purity of lead sulfate used was 98%
1.8 Analysis of three NIST Standard Reference Materials
Approximately 100 mg of material was placed in a conical beaker along a
Ghost Wipe. Samples were digested and then filtered through an MCE filter to
remove all particulate. The MCE filter for each sample, along with the
remaining particulate, were digested a second time. Results were not blank
corrected.
Table 1.8.1
Standard Reference Material 2580 Lead Paint |
| element |
sample 1
weight = 101.6 mg |
sample 2
weight = 100.9 mg |
sample 3
weight = 100.6 mg |
| first digest (µg) |
second digest (µg) |
theor (µg) |
recovery (%) |
first digest (µg) |
second digest (µg) |
theor (µg) |
recovery (%) |
first digest (µg) |
second digest (µg) |
theor (µg) |
recovery (%) |
| Pb |
2466 |
1653 |
4409 |
93.4 |
2345 |
992.0 |
4379 |
76.2 |
1730 |
1674 |
4366 |
78.0 |
| Fe* |
533.5 |
27.08 |
508.0 |
110 |
520.0 |
17.50 |
504.0 |
107 |
513.0 |
24.42 |
503.0 |
107 |
| Zn* |
2842 |
127.1 |
3048 |
97.4 |
2790 |
83.95 |
3027 |
94.9 |
2750 |
114.2 |
3018 |
94.9 |
*Results not certified by NIST
Table 1.8.2
Standard Reference Material 2583 Trace Elements in Indoor Dust |
| element |
sample 1
weight = 103.8 mg |
sample 2
weight = 100.3 mg |
sample 3
weight = 100.4 mg |
| first digest (µg) |
second digest (µg) |
theor (µg) |
recovery (%) |
first digest (µg) |
second digest (µg) |
theor (µg) |
recovery (%) |
first digest (µg) |
second digest (µg) |
theor (µg) |
recovery (%) |
| Cd |
0.565 |
0.000 |
0.757 |
74.6 |
0.695 |
0.000 |
0.732 |
94.9 |
0.465 |
0.000 |
0.733 |
63.4 |
| Cr |
5.215 |
0.900 |
8.304 |
73.6 |
5.585 |
0.905 |
8.024 |
80.8 |
5.395 |
0.755 |
8.032 |
76.6 |
| Pb |
6.820 |
0.365 |
8.916 |
80.6 |
6.425 |
0.050 |
8.616 |
75.2 |
6.710 |
0.160 |
8.624 |
79.7 |
Table 1.8.3
Standard Reference Material 1648 Urban Particulate Matter |
| element |
sample 1
weight = 101.4 mg |
sample 2
weight = 101.7 mg |
sample 3
weight = 101.1 mg |
| first digest (µg) |
second digest (µg) |
theor (µg) |
recovery (%) |
first digest (µg) |
second digest (µg) |
theor (µg) |
recovery (%) |
first digest (µg) |
second digest (µg) |
theor (µg) |
recovery (%) |
| Pb |
608.0 |
30.88 |
664.2 |
96.2 |
618.0 |
33.12 |
666.1 |
97.8 |
595.0 |
30.35 |
662.2 |
94.4 |
| Cd |
7.185 |
0.255 |
7.605 |
97.8 |
7.215 |
0.280 |
7.628 |
98.3 |
7.185 |
0.270 |
7.583 |
98.3 |
| Cr |
10.38 |
3.990 |
40.86 |
35.2 |
9.595 |
2.920 |
40.99 |
30.5 |
10.76 |
3.185 |
40.74 |
34.2 |
| Cu |
53.95 |
2.925 |
61.75 |
92.1 |
54.70 |
3.020 |
61.94 |
93.2 |
53.50 |
2.645 |
61.57 |
91.2 |
| Mn |
71.80 |
4.101 |
79.70 |
95.2 |
73.63 |
4.495 |
79.93 |
97.7 |
71.44 |
3.766 |
79.46 |
94.6 |
| Ni |
6.600 |
0.490 |
8.315 |
85.3 |
6.660 |
0.500 |
8.339 |
95.9 |
6.760 |
0.530 |
8.290 |
87.9 |
| V |
9.805 |
0.990 |
12.88 |
83.8 |
9.725 |
1.070 |
12.92 |
83.6 |
9.680 |
0.900 |
12.84 |
82.4 |
1.9 Instrument Analytical Conditions
| Instrument: |
Perkin-Elmer Optima 4300 DV ICP |
| Replicates: |
2 |
| Read Time: |
Auto, 5-20 sec |
| Sample Flow Rate: |
2.20 mL/min |
| Gas: |
Argon |
| Plasma Gas Flow: |
15 L/min |
| Auxiliary Gas Flow: |
0.20 L/min |
| Nebulizer Gas Flow: |
0.60 L/min |
| RF Power: |
1300 watts |
| Plasma View: |
Radial |
| View Distant: |
15.0 mm |
| Peak Algorithm: |
Peak Area |
| Overlap Correction: |
IEC |
| Background Correction: |
2-Point |
| Calibration: |
2-Point |
1 Lawrence, R. Evaluation Guidelines for Surface
Sampling Methods ; OSHA Salt Lake Technical Center, U.S. Department of
Labor: Salt Lake City, UT, 2001.
2 The Perkin-Elmer Corporation. WinLab32 Instrument Control Software
Guide ; Norwalk, Ct, 1999.
|
| |
|

