|
N-PHENYL-1-NAPHTHYLAMINE (N-PHENYL-a-NAPHTHYLAMINE)
N-PHENYL-2-NAPHTHYLAMINE (N-PHENYL-b-NAPHTHYLAMINE)
| Method number: | 96 | |
| |
| Matrix: | Air |
| |
| Procedure: |
Samples are collected closed face by drawing known volumes of air through sampling
devices consisting of three-piece opaque cassettes, each containing two
25-mm diameter extra thick glass fiber filters treated with 10 mg of
L-ascorbic acid (Vitamin C). Quantitation is performed by extracting the filters
with methyl alcohol and analyzing the extract by HPLC using a fluorescence detector. |
| |
Recommended air volume
and sampling rate: | 240 L at 1 L/min |
| |
|
|
|
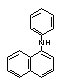
N-Phenyl-1-naphthylamine |
N-Phenyl-2-naphthylamine |
|
|
| Target concentration: |
1 ppb (9.0 µg/m3) |
1 ppb (9.0 µg/m3) |
| |
| Reliable quantitation limit: |
17 ppt (150 ng/m3) |
3.0 ppt (27 ng/m3) |
| |
Standard error of estimate at
the target concentration: |
5.2% | 5.3% |
|
|
| |
| Status of method: |
Evaluated method. This method has been subjected to the established evaluation procedures of the Organic Methods Evaluation Branch. |
| |
| Date: September 1992 | Chemist: Carl J. Elskamp |
Organic Methods Evaluation Branch
OSHA Salt Lake Technical Center
Salt Lake City, UT 84165-0200
1. General Discussion
1.1. Background
1.1.1. History
Previous to this evaluation there were no validated air sampling procedures for
N-phenyl-1-naphthylamine and N-phenyl-2-naphthylamine. Methods utilizing
collection on sulfuric acid-treated glass fiber filters have been validated by the
OSHA Salt Lake Technical Center for a number of other aromatic amines. (Ref. 5.1.-5.7.)
The sulfuric acid on the filters facilitates the collection and stabilization of the
amines by forming the corresponding amine salts. The salts are converted back to
the free amines and are either analyzed directly by HPLC or are derivatized and
analyzed by GC. For these two amines, it was found that acid-treated filters proved
to be an unacceptable collection medium because the amines were readily lost through oxidation.
An alternative sampling procedure using 25-mm extra thick glass fiber filters treated
with the antioxidant Vitamin C was developed and tested. The treated filter was
found to be highly effective in collecting the amines and the amines were stabilized,
even at room temperature storage, as long as the filters were protected from light.
This was accomplished by using opaque filter cassettes, which are the same as
used by OSHA for collecting airborne asbestos. A 10-mg loading of Vitamin C on
the filters provides for an acceptable collection efficiency for the amines without
causing an analytical interference.
No exposure limits have been established for these two amines by OSHA or ACGIH,
but ACGIH categorizes N-phenyl-2-naphthylamine as a suspected human carcinogen
(Ref. 5.8.), and N-phenyl-1-naphthylamine is also a suspected carcinogen
(Ref. 5.9.). Based on the carcinogenic potential of these two amines to humans, target
concentrations of 1 ppb were chosen for this evaluation.
The filters are extracted with methyl alcohol and the extract is analyzed directly by
HPLC using a fluorescence detector. The optimum excitation wavelength is 330 nm
for N-phenyl-1-naphthylamine and 300 nm for N-phenyl-2-naphthylamine, with the
latter fluorescing about six times more than the former. Although there was no
breakthrough detected in collection efficiency studies for either amine after sampling
for more than 360 L, a recommended air volume of 240 L (4 h sample at 1 L/min)
was chosen as a convenient sample size. This also makes the target concentration
about 50 times the reliable quantitation limit for N-phenyl-1-naphthylamine.
1.1.2. Toxic effects (This section is for information only and should not be taken as the basis of OSHA policy.)
N-Phenyl-1-naphthylamine is a skin, eye, and mucous membrane irritant. Exposure
by inhalation may cause respiratory irritation with sore nose, sore throat, and
cough. (Ref. 5.10.) The toxicity has not been fully characterized. It is a
questionable carcinogen based on experimental data. When heated to decomposition it emits
toxic fumes of NOx. (Ref. 5.9.)
N-Phenyl-2-naphthylamine is an eye, skin, and mucous membrane irritant. Exposure
through inhalation may cause sore throat, shortness of breath, headache, nausea,
dizziness, faintness, unconsciousness, bluish skin and methemoglobinemia.
Previously exposed persons may experience sensitization reactions. When heated
to decomposition it emits toxic fumes of NOx. (Ref. 5.10.) The International
Agency for Research on Cancer has concluded that there is limited evidence for
carcinogenicity of N-phenyl-2-naphthylamine to animals and inadequate evidence
for humans. (Ref. 5.11.) ACGIH considers N-phenyl-2-naphthylamine to be a
suspected human carcinogen because 2-naphthylamine is both an impurity in and
a human metabolite of it. (Ref. 5.8.)
1.1.3. Workplace exposure
N-Phenyl-1-naphthylamine is used in the production of dyes and other organic
chemicals, and also as a rubber antioxidant. (Ref. 5.12.)
N-Phenyl-2-naphthylamine is primarily used as an antioxidant in rubber processing.
It is also used as a stabilizer in electrical-insulating enamels and as an antioxidant
in other polymers, in greases, and in lubricating and transformer oils. It is used as
a chemical intermediate in the production of the rubber antioxidant,
N-phenyl-2-naphthylamine-acetone condensate, and in the production of
seven dyes. (Ref. 5.11.)
1.1.4. Physical properties and other descriptive information (Ref. 5.10.)
|
| Property |
N-Phenyl-1-naphthylamine |
|
N-Phenyl-2-naphthylamine |
|
CAS number:
molecular weight:
melting point:
boiling point:
vapor pressure:
specific gravity: |
90-30-2
219.29
55°C
335°C
0.1 mmHg at 20°C
1.23 |
|
135-88-6
219.29
108°C
395-399°C
15 mmHg at 235°C
1.24 |
| description: |
tan to purple crushed solidor crystals with an amine-like odor |
grey to tan flakes, rhombic crystals or powder |
| solubility: |
insoluble in water; soluble in acetone, benzene, alcohol |
insoluble in water; soluble in benzene, alcohol, ether, actic acid, acetone |
| synonyms: |
N-phenyl-a-naphthylamine;
1-anilinonaphthalene; N-(1-naphthyl)aniline;
a-naphthylphenylamine; phenyl-1-naphthylamine;
Aceto PAN; C.I. 44050; Neozone A; PANA; Naugaurd PANA |
N-phenyl-b-naphthylamine;
2-anilinonaphthalene; 2-naphthylphenylamine;
b-naphthylphenylamine; anilinonaphthalene;
2-(phenylamino)naphthalene; phenyl-b-naphthylamine;
N-(2-naphthyl)aniline; phenyl-2-naphthylamine; 2-naphthylamine,
N-phenyl-; 2-naphthalenamine, N-phenyl-;
Vanlube 82; PBNA; NCI-C02915; Antioxidant PBN; Agerite Powder; Antioxidant 116;
Nonox D; Neozone D; Stabilator AR |
| structural formula: |
 |
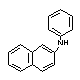 |
The analyte air concentrations throughout this method are based on the recommended sampling and analytical
parameters. Air concentrations listed in ppb and ppt are referenced to 25°C and 101.3 kPa (760 mmHg).
1.2. Limit defining parameters
1.2.1. Detection limit of the analytical procedure
The detection limits of the analytical procedure are 180 and 32.5 pg per injection for
N-phenyl-1-naphthylamine and N-phenyl-2-naphthylamine respectively. These
are the amounts of each analyte that will produce peaks with heights that are approximately
five times the baseline noise. (Section 4.1.)
1.2.2. Detection limit of the overall procedure
The detection limits of the overall procedure are 36.0 and 6.49 ng per sample for
N-phenyl-1-naphthylamine and N-phenyl-2-naphthylamine respectively. These are
the amounts of each analyte spiked on filters that, upon analysis, produce a peak
similar in size to that of the respective detection limit of the analytical procedure.
These detection limits correspond to air concentrations of 17 ppt (150 ng/m3 and
3.0 ppt (27 ng/m3 for N-phenyl-1-naphthylamine and
N-phenyl-2-naphthylamine respectively. (Section 4.2.)
1.2.3. Reliable quantitation limit
The reliable quantitation limits are 36.0 and 6.49 ng per sample for
N-phenyl-1-naphthylamine and N-phenyl-2-naphthylamine respectively.
These are the smallest amounts of each analyte spiked on sample filters that can be quantitated within the
requirements of a recovery of at least 75% and a precision (±1.96 SD) of ±25% or better. These reliable
quantitation limits correspond to air concentrations of 17 ppt (150 ng/m3 and
3.0 ppt (27 ng/m3 for N-phenyl-1-naphthylamine
and N-phenyl-2-naphthylamine respectively. (Section 4.3.)
The reliable quantitation limits and detection limits reported in this method are based upon
optimization of the instrumentation for the smallest possible amount of analyte. When the
target concentration of an analyte is exceptionally higher than these limits, they may not be
attainable at the routine operating parameters.
1.2.4. Instrument response to the analyte
The instrument response over concentration ranges representing 0.5 to 2 times the
target concentrations is linear for both analytes. (Section 4.4.)
1.2.5. Recovery
The recoveries of N-phenyl-1-naphthylamine and N-phenyl-2-naphthylamine
from samples used in 17-day ambient storage tests remained above 99% and 97%
respectively. (Section 4.5.)
1.2.6. Precision (analytical method only)
The pooled coefficients of variation obtained from replicate injections of analytical
standards at 0.5, 1, and 2 times the target concentrations are 0.0043 and 0.0019 for
N-phenyl-1-naphthylamine and N-phenyl-2-naphthylamine
respectively. (Section 4.6.)
1.2.7. Precision (overall procedure)
The precisions at the 95% confidence level for the 17-day ambient storage tests are ±10.2 and
±10.4% for N-phenyl-1-naphthylamine and N-phenyl-2-naphthylamine
respectively. These include an additional ±5% for sampling error. (Section 4.7.)
1.2.8. Reproducibility
Six samples for each analyte, spiked by liquid injection, and a draft copy of this
procedure were given to a chemist unassociated with this evaluation. The samples
were analyzed after 11 days of storage at approximately 0°C. No individual sample
result deviated from its theoretical value by more than the corresponding precision
of the overall procedure as reported in Section 1.2.7. (Section 4.8.)
1.3. Advantages
1.3.1. The Vitamin C treated glass fiber filter is not only a very effective air sampling
medium for these amines, the collected amines remain stable, even at room temperatures.
1.3.2. The analysis is rapid, sensitive, and precise.
2. Sampling Procedure
2.1. Apparatus
2.1.1. Samples are collected using a personal sampling pump that can be calibrated within
±5% of the recommended flow rate with the sampling device attached.
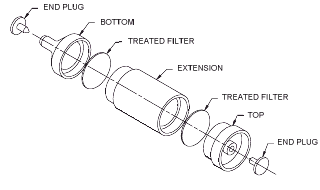 2.1.2. Samples are collected closed face using a sampling device consisting of two 25-mm
extra thick glass fiber filters treated with 10 mg of Vitamin C contained in a three-piece
opaque cassette. The filters and carbon-filled polypropylene cassettes used in
this evaluation were from Gelman Sciences, Ann Arbor, MI (product no. 66075 and 4376
respectively). The filters are treated with 0.6 mL of a 16.67 mg/mL solution of Vitamin C in methyl
alcohol. (Note: The dissolution of Vitamin C in methyl alcohol can be hastened by sonication.)
This is conveniently done by placing the untreated filters on a clean glass thin layer
chromatography plate and adding the Vitamin C solution to each filter using a dispenser or
pipetter. The filters are allowed to dry in a fume hood and can be stored in a closed container in a refrigerator for at least 3 months.
Before sampling is to begin, the treated filters are assembled into three-piece
25-mm cassettes without support pads. The top filter is separated from the bottom
filter by a 2-in. extension. (Note: The extension does not have to be this long, but
it is imperative that the two filters are not in contact with each other in the cassette.
This was the only type of opaque 25-mm cassette available for this evaluation.)
The cassette sections are held in place to form air-tight seals with shrink
bands or tape. Plastic plugs are inserted into the ends of the cassettes.
2.1.2. Samples are collected closed face using a sampling device consisting of two 25-mm
extra thick glass fiber filters treated with 10 mg of Vitamin C contained in a three-piece
opaque cassette. The filters and carbon-filled polypropylene cassettes used in
this evaluation were from Gelman Sciences, Ann Arbor, MI (product no. 66075 and 4376
respectively). The filters are treated with 0.6 mL of a 16.67 mg/mL solution of Vitamin C in methyl
alcohol. (Note: The dissolution of Vitamin C in methyl alcohol can be hastened by sonication.)
This is conveniently done by placing the untreated filters on a clean glass thin layer
chromatography plate and adding the Vitamin C solution to each filter using a dispenser or
pipetter. The filters are allowed to dry in a fume hood and can be stored in a closed container in a refrigerator for at least 3 months.
Before sampling is to begin, the treated filters are assembled into three-piece
25-mm cassettes without support pads. The top filter is separated from the bottom
filter by a 2-in. extension. (Note: The extension does not have to be this long, but
it is imperative that the two filters are not in contact with each other in the cassette.
This was the only type of opaque 25-mm cassette available for this evaluation.)
The cassette sections are held in place to form air-tight seals with shrink
bands or tape. Plastic plugs are inserted into the ends of the cassettes.
2.2. Reagents
None required.
2.3. Sampling technique
2.3.1. Remove the plastic end plugs from the sampling device immediately before sampling.
2.3.2. Attach the sampling device to the sampling pump with flexible, non-crimpable
tubing and place the device in the employee's breathing zone.
2.3.3. Immediately after sampling, insert the plastic end plugs into the sampling devices.
2.3.4. Seal and identify each sampling device with an OSHA Form 21.
2.3.5. Submit at least one blank with each sample set. Handle the blanks in the same manner
as the air samples, but draw no air through them.
2.3.6. Record the volume of air sampled (in liters) for each sample, along with any potential interferences.
2.4. Collection efficiency
Collection efficiency studies were conducted by drawing humid air through a sampling device
that was attached to a glass U-tube which was immersed in an oil bath heated to 50°C.
Microliter amounts of stock standards were injected into the U-tube. Tests were done
individually for each amine by injecting about 4.3 µg (approximately equal to two
times the target concentrations for a 240-L sample) into the U-tube.
The inlet of the U-tube was attached to a humid air generator so air at
approximately 80% relative humidity could be drawn through it and the sampling device at
1 L/min. The bottom filter in the cassette was replaced with a fresh filter every 30 minutes
and was analyzed to detect any breakthrough from the top filter. Similar tests were also done
by adding amounts approximately equal to ten times the target concentrations. None of the
amines were found on any of the bottom filters for any of the tests after sampling for more
than 360 min. The majority of the amine that was added to the U-tube was always found on the
top filter. Minor amounts were found in the U-tube, which was determined by
analyzing rinses of the U-tube after sampling was completed.
2.5. Extraction efficiency
2.5.1. The average extraction efficiencies from six filters for each amine spiked at the
target concentration are 94.9% and 96.7% for N-phenyl-1-naphthylamine
and N-phenyl-2-naphthylamine respectively. (Section 4.9.)
2.5.2. The stability of extracted samples was verified by reanalyzing the extraction
efficiency samples 24 h later using fresh standards. The average recoveries for the
reanalyzed samples are 92.8% and 92.9% for N-phenyl-1-naphthylamine and
N-phenyl-2-naphthylamine respectively. (Section 4.9.)
This same small loss of analyte was also observed in the analytical standards, indicating
that both analytical standards and samples should be analyzed as soon as feasible after being
prepared and extracted respectively.
2.5.3. Extraction efficiencies must be determined for each lot of treated filters. In order
to prevent loss of analyte through photo-oxidation, it is imperative that the filters
are immediately protected from light after they are spiked with the amine of interest.
2.6. Recommended air volume and sampling rate
2.6.1. The recommended air volume is 240 L.
2.6.2. The recommended sampling rate is 1 L/min.
2.6.3. When short-term samples are required, the reliable quantitation limits will be larger.
For example, the reliable quantitation limit for N-phenyl-2-naphthylamine
for a 15-L air sample would be 48 ppt (432 ng/m3).
2.7. Interferences (sampling)
2.7.1. Any compound in the sampled air that will react with the Vitamin C on the treated
filters or with the collected analyte is a potential sampling interference.
2.7.2. Suspected interferences should be reported to the laboratory with submitted samples.
2.8. Safety precautions (sampling)
2.8.1. Attach the sampling equipment to the employees so it will not interfere with work performance or safety.
2.8.2. Follow all safety procedures that apply to the work area being sampled.
3. Analytical Procedure
3.1. Apparatus
3.1.1. An HPLC equipped with an fluorescence detector. A Hewlett-Packard 1050 Series HPLC
system and a Kratos Analytical Spectroflow 980 Fluorescence Detector were used in this evaluation.
3.1.2. An HPLC column capable of separating the amines from the solvent, Vitamin C and
interferences. A Waters Nova-Pak™ C18 Radial-Pak™ (Millipore Corp.,
Milford, MA) 100-mm × 8-mm i.d. cartridge was used in conjunction
with a Waters RCM-100 radial compression module.
3.1.3. An electronic integrator or some other suitable means of measuring peak areas or
heights. A Waters 860 Networking Computer System was used in this evaluation.
3.1.4. Small resealable vials with Teflon-lined caps capable of holding at least 3 mL.
3.1.5. Dispensers or pipets capable of delivering 2.00 mL.
3.1.6. A rotator or rocker to extract the sample filters.
3.2. Reagents
3.2.1. N-Phenyl-1-naphthylamine and N-phenyl-2-naphthylamine,
reagent grade. The amines used in this evaluation were from Aldrich Chemical Company, Inc., Milwaukee, WI.
3.2.2. L-Ascorbic acid (Vitamin C), reagent grade. The Vitamin C used in this evaluation was from Aldrich Chemical Company, Inc.
3.2.3. Methyl alcohol, HPLC grade. The methyl alcohol used in this evaluation was from Fisher Chemical, Fair Lawn, NJ.
3.2.4. Standard preparation solution, consisting of 5.0 mg/mL of Vitamin C in methyl alcohol. (Note: The dissolution of Vitamin C in methyl alcohol can be hastened by sonication.)
3.2.5. Water, HPLC grade. The water used in this evaluation was from an in-house Millipore Milli-Q™ water purification system.
3.3. Standard preparation
3.3.1. CAUTION. THESE AROMATIC AMINES SHOULD BE CONSIDERED CARCINOGENIC.
Restrict the use of the pure amines and concentrated standards to regulated areas. All
standards are prepared and diluted with a preparation solution consisting of 5.0 mg/mL of
Vitamin C in methyl alcohol. Prepare concentrated stock standards by diluting 10 to 50 mg of
the pure amines to 10.00 mL. Prepare intermediate standards by diluting the appropriate
volume of concentrated stock standards to 25.00 or 50.00 mL. Prepare analytical standards by
injecting microliter amounts of intermediate standards into vials that contain 2.00 mL of
the preparation solution. For example, a 3.380 µg/µL concentrated stock
standard of N-phenyl-2-naphthylamine is prepared by dissolving 33.80 mg
into 10.00 mL of preparation solution. This is done by weighing the amine into a 10-mL
volumetric flask and adding about 5 to 8 mL of preparation solution. (Note: The flask can be
sonicated to hasten the dissolution of the amine.) After allowing the flask to cool to room
temperature if it is warm from sonication, the solution is diluted to the mark with the
preparation solution. An intermediate standard of 0.5408 µg/µL is made by
diluting 4.00 mL of the concentrated stock to 25.00 mL. An analytical standard of
2.163 µg/sample is prepared by adding 4.00 µL of this solution to 2.00 mL of
preparation solution. Stock and intermediate standards are stable for up to 3 months when
refrigerated in amber vials or bottles. Analytical standards must be prepared fresh daily.
3.3.2. Bracket sample concentrations with analytical standard concentrations. If sample
concentrations are higher than the upper range of prepared standards, prepare additional
standards to ascertain detector response.
3.4. Sample preparation
3.4.1. Transfer the sample filters to separate 4-mL vials.
3.4.2. Add 2.00 mL of methyl alcohol to each vial.
3.4.3. Recap and rotate or rock the vials for 10 min.
3.4.4. Analyze the methyl alcohol extract of each sample by HPLC.
3.5. Analysis
3.5.1. HPLC conditions and information
| injection volume: | 10 µL |
| column: | Waters Radial-Pak™ 100-mm × 8-mm i.d. Nova-Pak™ C18 cartridge in an RCM-100 radial compression module |
| mobile phase: | 85/15, methyl alcohol/water |
| flow rate: | 2 mL/min |
| retention times: | N-Phenyl-1-naphthylamine, 3.15 min
N-Phenyl-2-naphthylamine, 3.21 min
(Note: If better separation of the two amines is required, the strength of the mobile
phase can be changed. For example, when 68/32, methyl alcohol/water is used, the resulting
retention times are 12.0 and 12.8 min for N-phenyl-1-naphthylamine and
N-phenyl-2-naphthylamine respectively.) |
| excitation wavelength: | N-Phenyl-1-naphthylamine, 330 nm
N-Phenyl-2-naphthylamine, 300 nm |
| emission wavelength: | 370 nm standard long pass filter for both amines |
| response time: | 2 sec |
| photomultiplier voltage: | 1100 V |
| chromatogram at 1× the target concentration: |
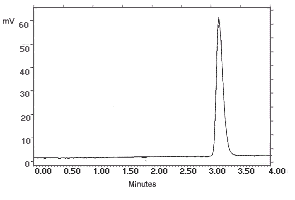
2.172 µg/sample of N-Phenyl-1-Naphthylamine
|
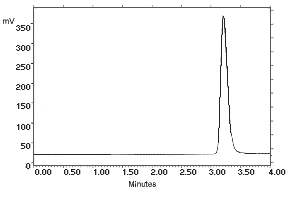
2.163 µg/sample of N-Phenyl-2-Naphthylamine
|
3.5.2. Construct acalibration curve by plotting response (peak areas or heights) of standard injections versus micrograms of
analyte per sample. Bracket sample concentrations with standards.
3.6. Interferences (analytical)
3.6.1. Any compound that elutes in the same general time as the amine of interest is a
potential interference. Suspected interferences reported to the laboratory with submitted
samples by the industrial hygienist must be considered before samples are extracted.
3.6.2. HPLC parameters may be changed to possibly circumvent interferences.
3.6.3. Retention time on a single column is not considered proof of chemical identity.
Analyte identity should be confirmed by mass spectrometry.
3.7. Calculations
The analyte concentration for samples is obtained from the proper calibration curve in
micrograms of analyte per sample. The bottom filter is analyzed to determine if there was any
breakthrough from the top filter during sampling. If any analyte is found on any bottom filter,
that amount is added to the amount found on the corresponding top filter. The combined
amount is then corrected by subtracting the total amount (if any) found on the corresponding
blank filters. The air concentrations are calculated using the following formulae.
|
µg/m3 =
|
(µg of analyte per sample) (1000)
(L of air sampled)(extraction efficiency)
|
ppb = (µg/m3) (24.46) / (219.29) = (µg/m3) (0.1115)
where 24.46 is the molar volume at 25°C and 101.3 kPa (760 mmHg) and 219.29 is the
molecular weight for N-phenyl-1-naphthylamine and N-phenyl-2-naphthylamine
3.8. Safety precautions (analytical)
3.8.1. CAUTION. THESE AROMATIC AMINES SHOULD BE CONSIDERED CARCINOGENIC.
Restrict the use of the pure amines and concentrated standards to regulated areas. Avoid skin
contact and inhalation of all chemicals.
3.8.2. Restrict the use of all chemicals to a fume hood if possible.
3.8.3. Wear safety glasses and a lab coat at all times while in the lab area.
4. Backup Data
4.1. Detection limit of the analytical procedure
The detection limits of 180 and 32.5 pg per injection were determined by making 10-µL
injections of dilute standards equivalent to 36.0 and 6.49 ng per sample for
N-phenyl-1-naphthylamine and N-phenyl-2-naphthylamine
respectively. These amounts were judged to give peaks with heights approximately five times
the baseline noise.
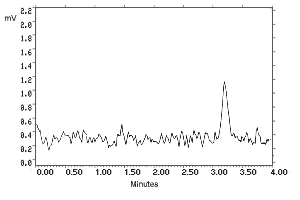
Figure 4.1.1. N-Phenyl-1-naphthylamine analytical detection limit chromatogram.
|
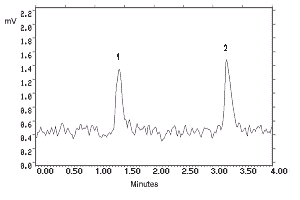
Figure 4.1.1. N-Phenyl-2-naphthylamine analytical detection limit chromatogram. Key: (1) matrix artifact, (2) N-phenyl-2-naphthylamine.
|
4.2. Detection limit of the overall procedure
The detection limits of the overall procedure were determined by analyzing filters spiked with
36.0 and 6.49 ng of N-phenyl-1-naphthylamine and N-phenyl-2-naphthylamine
respectively. These amounts correspond to air concentrations of 17 ppt
(150 ng/m3 and 3.0 ppt (27 ng/m3
for N-phenyl-1-naphthylamine and N-phenyl-2-naphthylamine respectively.
Table 4.2.1.
Detection Limit of the Overall Procedure
for N-Phenyl-1-naphthylamine
|
| sample no. | ng spiked | ng recovered |
|
1
2
3
4
5
6 |
36.0
36.0
36.0
36.0
36.0
36.0 |
V36.7
36.6
32.7
33.4
38.8
36.2 |
|
|
Table 4.2.2.
Detection Limit of the Overall Procedure
for N-Phenyl-2-naphthylamine
|
| sample no. | ng spiked | ng recovered |
|
1
2
3
4
5
6 |
6.49
6.49
6.49
6.49
6.49
6.49 |
6.28
6.26
6.08
6.64
6.32
6.28 |
|
|
4.3. Reliable quantitation limit
The reliable quantitation limits were determined by analyzing filters spiked with 36.0 and
6.49 ng of N-phenyl-1-naphthylamine and N-phenyl-2-naphthylamine
respectively. These amounts correspond to air concentrations of 17 ppt (150 ng/m3
and 3.0 ppt (27 ng/m3 for N-phenyl-1-naphthylamine
and N-phenyl-2-naphthylamine respectively.
Table 4.3.1.
Reliable Quantitation Limit
for N-Phenyl-1-naphthylamine
(Based on samples and data of Table 4.2.1.)
|
| percent recovered | statistics |
|
101.9
101.7
90.8
92.8
107.8
100.6 |
mean =
SD =
Precision =
= |
99.3
6.3
(1.96)(±6.3)
±12.3 |
|
|
Table 4.3.2.
Reliable Quantitation Limit
for N-Phenyl-2-naphthylamine
(Based on samples and data of Table 4.2.2.)
|
| percent recovered | statistics |
|
96.8
96.5
93.7
102.3
97.4
96.8 |
mean =
SD =
Precision =
= |
97.2
2.8
(1.96)(±2.8)
±5.5 |
|
|
4.4. Instrument response to the analyte
The instrument response to the analytes over the range of 0.5 to 2 times the target
concentrations was determined from multiple injections of analytical standards. The response
is linear for both analytes with slopes (in area counts per micrograms of analyte per sample)
of 208,500 and 1,289,000 for N-phenyl-1-naphthylamine and
N-phenyl-2-naphthylamine respectively.
Table 4.4.1.
Instrument Response to N-Phenyl-1-naphthylamine
|
× target concn
µg/sample
ppb |
0.5×
1.086
0.50 |
1×
2.172
1.01 |
2×
4.343
2.02 |
|
| area counts |
234947
235168
234277
234099
233284
232461
|
460583
455839
454370
457300
454967
454185
|
906939
902739
907653
901471
903653
902948
|
| mean | 234039 | 456207 | 904234 |
|
|
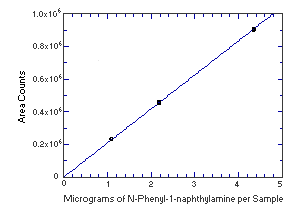
Figure 4.4.1. Instrument response to N-phenyl-1-naphthylamine.
| | |
Table 4.4.2.
Instrument Response to N-Phenyl-2-naphthylamine
|
× target concn
µg/sample
ppb |
0.5×
1.082
0.50 | 1×
2.163
1.01 | 2×
4.326
2.01 |
|
| area counts |
1402007
1403870
1401188
1404162
1400473
1395011
|
2808776
2801588
2802869
2795701
2792481
2798636
|
5573809
5566549
5569283
5569898
5564317
5560347
|
| mean | 1401118 | 2800008 | 5567367 |
|
|
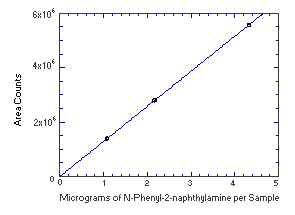
Figure 4.4.2. Instrument response to N-phenyl-2-naphthylamine.
|
4.5. Storage test
Thirty-six storage samples for each analyte were prepared by spiking microliter amounts of
intermediate standards onto Vitamin C treated glass fiber filters. The amounts of analytes
spiked (2.164 µg of N-phenyl-1-naphthylamine and 2.153 µg
of N-phenyl-2-naphthylamine) are equal to the target concentrations.
The filters were then assembled in cassettes and 240 L of air at approximately 80% relative
humidity and 21°C were drawn through each sampling device. Six samples for each amine
were analyzed immediately, fifteen were stored in a refrigerator at 0°C, and fifteen
were stored at approximately 21°C. Six samples for each amine, three from refrigerated
and three from ambient storage, were analyzed at intervals over a period of seventeen days.
The recoveries of N-phenyl-1-naphthylamine and N-phenyl-2-naphthylamine
from samples stored at ambient temperature remained above 99% and 97% respectively.
Table 4.5.1.
Storage Test for N-Phenyl-1-naphthylamine
|
days of
storage |
% recovery
(refrigerated) |
|
% recovery
(ambient) |
|
0
0
3
7
10
14
17 |
100.7
101.4
98.9
102.4
96.3
102.5
102.2 |
99.4
97.7
99.2
97.5
96.9
101.8
99.6 |
96.2
99.5
101.2
97.9
101.9
102.2
96.1 |
|
100.7
101.4
98.1
99.2
97.8
97.7
98.9 |
99.4
97.7
100.5
100.5
98.8
99.3
101.5 |
96.2
99.5
100.4
98.4
99.2
97.6
98.1 |
|
Table 4.5.2.
Storage Test for N-Phenyl-2-naphthylamine
|
days of
storage |
% recovery
(refrigerated) |
|
% recovery
(ambient) |
|
0
0
3
7
10
14
17 |
99.4
100.0
99.4
99.4
99.5
101.3
99.8 |
99.7
99.4
99.8
99.3
98.5
101.0
99.5 |
100.3
99.3
100.1
98.9
99.6
99.2
99.1 |
|
99.4
100.0
97.2
97.6
98.7
98.4
98.0 |
99.7
99.4
99.6
96.8
96.5
98.4
98.2 |
100.3
99.3
99.6
94.0
93.2
96.9
99.0 |
|
4.6. Precision (analytical method only)
The precision of the analytical method for each analyte is the pooled coefficient of variation
determined from replicate injections of standards. The coefficients of variation (CV) are
calculated from the data from Tables 4.4.1. and 4.4.2. The pooled coefficients of variation
are 0.0043 and 0.0019 for N-phenyl-1-naphthylamine and
N-phenyl-2-naphthylamine respectively.
Table 4.6.1.
Precision of the Analytical Method for N-Phenyl-1-naphthylamine
(Based on the Data of Table 4.4.1.)
|
× target concn
µg/sample
ppb |
0.5×
1.086
0.50 |
1×
2.172
1.01 |
2×
4.343
2.02 |
|
mean
CV |
234039
0.00437 |
456207
0.00532 |
904234
0.00275 |
|
|
Table 4.6.2.
Precision of the Analytical Method for N-Phenyl-2-naphthylamine
(Based on the Data of Table 4.4.2.)
|
× target concn
µg/sample
ppb |
0.5×
1.082
0.50 | 1×
2.163
1.01 | 2×
4.326
2.01 |
|
mean
CV |
1401118
0.00237 |
2800008
0.00205 |
5567367
0.00085 |
|
|
4.7. Precision (overall procedure)
The precision of the overall procedure is determined from the storage data. The determination
of the standard error of estimate (SEE) for a regression line plotted through the graphed storage
data allows the inclusion of storage time as one of the factors affecting overall precision.
The SEE is similar to the standard deviation, except it is a measure of dispersion of data
about a regression line instead of about a mean. It is determined with the following equation:
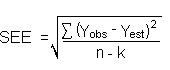
| where |
n =
k =
k = |
total no. of data points
2 for linear regression
3 for quadratic regression |
|
Yobs = |
observed percent recovery at a given time |
|
Yest = |
estimated percent recovery from the regression line at the same given time |
An additional 5% for pump error is added to the SEE by the addition of variances. The
precision at the 95% confidence level is obtained by multiplying the SEE (with pump error
included) by 1.96 (the z-statistic from the standard normal distribution at the
95% confidence level). The 95% confidence intervals are drawn about their respective
regression line in the storage graph as shown in Figure 4.5.1.1. The
data for Figures org096.html4.5.1.2. and 4.5.2.2. were
used to determine the SEEs of ±5.2% and ±5.3% and the precisions of the overall procedure of
±10.2% and ±10.4% for N-phenyl-1-naphthylamine and
N-phenyl-2-naphthylamine respectively.
4.8. Reproducibility
Samples were prepared by injecting microliter quantities of standards onto Vitamin C treated
filters, assembling the filters into cassettes, and drawing 240 L of 80% relative humidity air
through the samplers. The samples were stored for 11 days at 0°C before being analyzed
by another chemist. No individual sample result deviated from its theoretical value by more
than the corresponding precision of the overall procedure. The precisions of the overall
procedure are ±10.2% and ±10.4% for N-phenyl-1-naphthylamine and
N-phenyl-2-naphthylamine respectively.
Table 4.8.1.
Reproducibility Data for N-Phenyl-2-naphthylamine
|
| µg spiked | µg found | % found | % difference |
|
1.773
2.172
4.343
1.330
2.172
4.343 |
1.805
2.155
4.372
1.334
2.199
4.317 |
101.8
99.2
100.7
100.3
101.2
99.4 |
+1.8
-0.8
+0.7
+0.3
+1.2
-0.6 |
|
| |
Table 4.8.2.
Reproducibility Data for N-Phenyl-2-naphthylamine
|
| µg spiked | µg found | % found | % difference |
|
2.163
4.326
2.704
2.163
3.245
2.704 |
2.121
4.312
2.667
2.198
3.376
2.848 |
98.1
99.7
98.6
101.6
104.0
105.3 |
-1.9
-0.3
-1.4
+1.6
+4.0
+5.3 |
|
|
4.9. Extraction efficiency
Six Vitamin C treated filters for each analyte were spiked with the target concentration
amounts by liquid injection (2.164 and 2.153 µg of N-phenyl-1-naphthylamine
and N-phenyl-2-naphthylamine respectively). These samples were analyzed
to determine the extraction efficiencies. To determine the stability of extracted samples,
these same samples were allowed to remain at room temperature for 24 h and reanalyzed using fresh standards.
Table 4.9.1.
Extraction Efficiency for N-Phenyl-1-naphthylamine
|
sample
number |
% extraction
(initial) |
% extraction
(24 h later) |
|
1
2
3
4
5
6
mean |
94.0
90.9
96.3
98.2
92.7
97.5
94.9 |
92.1
89.8
94.5
95.4
89.6
95.7
92.8 |
|
| |
Table 4.9.2.
Extraction Efficiency for N-Phenyl-2-naphthylamine
|
sample
number |
% extraction
(initial) |
% extraction
(24 h later) |
|
1
2
3
4
5
6
mean |
97.0
98.4
95.5
97.7
95.3
96.4
96.7 |
92.4
93.5
91.6
94.6
92.2
92.9
92.9 |
|
|
5. References
5.1. "OSHA Analytical Methods Manual", 2nd ed.; U.S. Department of Labor, Occupational Safety
and Health Administration; OSHA Analytical Laboratory: Salt Lake City, UT, 1990; Method 57;
American Conference of Governmental Industrial Hygienists (ACGIH): Cincinnati, OH, Publication No. 4542.
5.2. ibid. Method 65.
5.3. ibid. Method 71.
5.4. ibid. Method 73.
5.5. ibid. Method 78.
5.6. Elskamp, C.J. "OSHA Method No. 87; m-, o-, and
p-Phenylenediamine", OSHA Salt Lake Technical Center, unpublished, Salt Lake City, UT 84165, February 1991.
5.7. Elskamp, C.J. "OSHA Method No. 93; 4-Aminobiphenyl, 1-Naphthylamine, and
2-Naphthylamine", OSHA Salt Lake Technical Center, unpublished, Salt Lake City, UT 84165,
January 1992.
5.8. "Documentation of the Threshold Limit Values and Biological Exposure Indices", 5th ed.;
American Conference of Governmental Industrial Hygienists Inc.: Cincinnati, OH, 1986; p 73.
5.9. Lewis, R.J., Sr., Ed. "Sax's Dangerous Properties of Industrial Materials", 8th ed.; Van
Nostrand Reinhold Co.: New York, NY, 1992.
5.10. Occupational Health Services, Inc.: Material Safety Data Sheets (MSDS) for
N-phenyl-1-naphthylamine and N-phenyl-2-naphthylamine, Jan. 1991.
5.11. "IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Overall Evaluations
of Carcinogenicity: An Updating of IARC Monographs", Volumes 1 to 42, Suppl 7, pp 318-319.
International Agency for Research on Cancer: Lyon, 1987.
5.12. Sax, N.I.; Lewis, R.J. Sr., Eds. "Hawley's Condensed Chemical Dictionary", 11th ed.; Van
Nostrand Reinhold Co.: New York, NY, 1987.
|

