Cosmic Rays
Cosmic-ray Composition
Because cosmic rays don't point back to their sources, we must use indirect
methods to determine their sources and the way they have traveled (or
"propagated") through the Galaxy. The chemical composition of the cosmic rays
provide a surprisingly rich source of such knowledge. The chemical
composition of the solar system has been determined from a combination
of spectroscopy on the Sun, studies of the solar wind, and by chemical
analysis of meteorites, which are presumed to have a purer sample of the
early solar system than terrestrial rocks. The composition of cosmic rays
is important because cosmic rays are a direct sample of matter from outside
the solar system and contain elements that are much too rare to be seen in
spectroscopic lines from other stars. Cosmic rays provide important
information on the chemical evolution of the universe.
If we look at the elemental composition measured for cosmic rays and compare
it to our best understanding of the composition of the solar system, we
quickly see some large differences.

|
Solar and
Galactic Cosmic Ray (GCR) Composition
|
Cosmic-ray Secondaries
In the figure above, we take the abundance of silicon as a "standard candle"
or reference point, and compare the abundances (relative to silicon)
of the elements in the solar system and in galactic cosmic rays. Silicon is
used as the reference because it is a common intermediate-weight element that
is easy to measure. We see that there is less hydrogen and helium in the
cosmic rays than in the solar system, we think because hydrogen and helium
are harder to accelerate to high energies than heavier elements. We also
see that some light elements (lithium, beryllium, and boron) that are rare in
the solar system (and in the rest of the universe) are quite common in cosmic
rays. We also see more cosmic ray elements between silicon and iron than in
the solar system.
The accepted reason for all the observed cosmic ray lithium, beryllium, and boron is that
these are pieces of heavier cosmic ray elements, especially carbon and
oxygen, that have had high speed collisions with the very tenuous gas in
interstellar space. Likewise, the elements between silicon and iron have been
supplemented by fragments of heavy cosmic rays such as iron and nickel. These
fragments are known as secondary cosmic rays or just secondaries.
From the number of secondaries observed at Earth, and with knowledge of the
probability of these collisions (which can be measured in particle accelerators
here on Earth), it is possible to calculate the amount of matter that the
cosmic rays have traveled through. More matter would breakup more primary
cosmic rays. If the cosmic rays have stayed in the Galaxy, the amount of
matter that they have passed through divided by the average density of
interstellar space (about one atom per cubic centimeter) gives the "age"
of cosmic rays. With this method, we determine an average cosmic ray age
of about two million years. But this turns out to be a bit wrong.
Radioactive Clocks
Another way to obtain the age of cosmic rays is to use radioactive isotopes
as clocks in a way very similar to the way carbon-14 is used by archaeologists.
There are several isotopes, beryllium-10, aluminum-26, chlorine-36, etc., which
are almost entirely secondaries. After they are created, they begin to decay,
and the fraction that reach us at Earth gives the age of the cosmic rays. With
this method, the average age of cosmic rays comes out to approximately ten million years.
The reason the two million year age from the previous paragraph is wrong is that
cosmic rays actually don't hang out just in the regions where the
density is one atom per cubic centimeter
(such as the galactic disk). Cosmic rays spend a large fraction of their time in the low
density
galactic halo, bouncing
back and forth through the galactic disk many times.
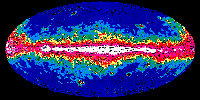
|
EGRET Gamma Ray All Sky Survey
|
As the cosmic rays interact with interstellar gas, they can produce gamma rays, which
can be seen in the EGRET gamma ray image of the Milky Way galaxy shown above.
|
