|
 |
 |
 |
 | |||||
| |||||||||
|
|
|
|
Annual Adverse Drug Experience Report: 1996October 30, 1997 Surveillance and Data Processing Branch TABLE OF CONTENTS
INTRODUCTIONThis report presents a descriptive overview of the 159,504 evaluable1, postmarket adverse drug experience (ADE) cases received by the US Food and Drug Administration (FDA) during calendar year 19962. A case consists of the original report of an ADE on a patient plus any follow-up information. At this time, October, 1997, the SRS has accumulated about 1.4 million cases. The primary purpose for maintaining the database is to serve as an early warning or signaling system for ADEs not detected during premarket testing. The ADE system depends upon detection of an adverse clinical event by a health professional or consumer, attribution of the clinical event to prior administration of a particular drug ("suspect" drug), and reporting of the ADE to the manufacturer of the suspected drug or directly to FDA. Data from these ADE cases are coded and entered into the computerized ADE database. Copies of the ADE cases are stored on microfilm or an imaging system. Up to five drugs per case may be entered into the computerized ADE database; the five can be a combination of "suspect" and "concomitant" drugs. Up to four adverse events per case and their associated body systems can by coded into the database, using FDA’s "Coding Symbols for Thesaurus of Adverse Reaction Terms" (COSTART). Reporting of postmarket ADEs by health professionals and consumers is voluntary. They may send their reports directly to FDA ("Direct" reports), to the drug manufacturer ("Manufacturer" reports), or both. Drug manufacturers are required by law and regulation to submit to FDA postmarket ADE reports received by any means from health professional or consumers. It is important to remember certain caveats when using data from FDA’s postmarket ADE database:
Over the next pages, various kinds of data and information are presented on the postmarket ADE cases computerized into the FDA ADE database during calendar year 1996. Due to rounding, the percentages in tables and graphs may not total to 100%. Figures 1 and 2 present copies of the postmarket ADE forms used by manufacturers and health professionals or consumers, respectively. ____________________ Standard MedWatch Form, front page
TYPES OF REPORTS There are three types of reports in the FDA computerized postmarket ADE database:
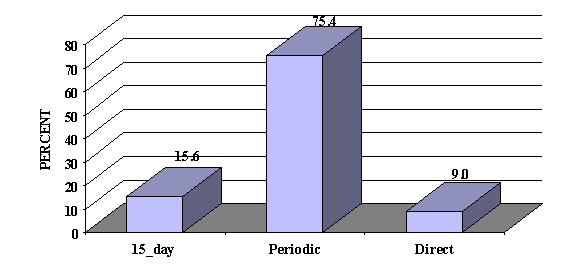
As shown in Figure 3, reports submitted to FDA via manufacturers accounted for 91.0%(145,021) of the 159,504 postmarket ADE cases. Only 9.0%(14,483) were submitted directly to FDA. 15-day report were 15.6%(24,815) of the total. Figure 3. Postmarket ADE Reports by Type of Report: 1996
N = 159,504
REPORTING BY HEALTH PROFESSIONALS AND CONSUMERS As shown in Figure 4, in 1996, there were 157,067 reporters for the 159,504 postmarket ADE cases, 64,752 (41.2%) reporters were consumers, 90,394 (57.6%) reporters were health professionals, and 1,921 (1.2%) were unknown sources. Figure 4 also shows that, over a four-year trend (1993-96), reports from consumers have increased both in absolute numbers and proportionally, whereas those from health professionals have gone up in absolute numbers.
Year: 1993 1994 1995 1996
GEOGRAPHIC LOCATION OF INITIAL REPORTER As shown in Table 1, the initial reporter for 81.2% (129,521) of the 159,504 postmarket ADE cases was located within the US census regions; 9.6% (15,260) of cases were missing location. There were 9.2% (14,723) of the postmarket ADE cases where the initial report source was foreign. There were four countries which each accounted for >= 9% of the foreign cases: France (31.2%), Japan (14.2%), United Kingdom (12.8%), Germany (9.2%).
Table 1. Postmarket ADE Reports by Geographic Location of Initial Reporter: 1996
____________________ As shown in Table 2, the ratio of female-to-male postmarket ADE cases was 1.7:1. For both females and males, the >= 60 year age group accounted for the greatest number of known sex-age cases. Table 2. Postmarket ADE Reports by Reports by Sex & Age of Patient: 1996
As shown in Figure 5, hospitalization was the most recorded serious outcome; congenital anomaly, the least. ( One case could have more than one outcome).
Figure 5. Postmarket ADE Reports by Type of Serious Report: 1996
Serious Outcome N = 105,599 LATENCY BETWEEN SUSPECT DRUG ADMINISTRATION AND ADE ONSET As shown in Figure 6, of the 159,504 postmarket ADE cases, 53.6% (85,517) had both a drug start date and an adverse experience onset date for the first-listed suspect drug and first-listed adverse experience, respectively, and the drug date was computerized as occurring before the adverse experience date. About half of these cases noted that the adverse event occurred within one week of drug initiation. Figure 6. Postmarket ADE Reports by Latency Period: 1996
Latency (days) N = 85,517 Table 3 presents the top-10 ranked drug classes associated with the 174,905 suspect drugs computerized from the 159.504 postmarket ADE cases. The top-ranked drug class, central nervous system agents, accounted for approximately little less than one-quarter of the drug class mentions3. Together with the second and third ranked drug classes, anti-infectives, and hormones and synthetic substitutes, these top three ranked drug classes comprised about half of the total drug class mentions.
Table 3. Postmarket ADE Reports by Top-10 Ranked Classes of Suspect Drugs: 1996
____________________ SUSPECT DRUGS BY ENTRY NAME AND NEW MOLECULAR ENTITY STATUS Table 4 shows the top-10 ranked suspect drugs as entered on the 159,504 postmarket ADE reporting forms. New Molecular Entities (NMEs) are defined as new drugs approved within the past three years. For this 1996 report, NMEs are new drugs approved during 1993-96. Of the 174,905 suspect drugs computerized from the 159,504 postmarket ADE cases, 30.2%(29,584) involved NMEs.
Table 4. Postmarket ADE Reports by Top-10 Ranked Suspect Drugs: 1996
_________________ DRUG CLASSES STRATIFIED BY HEALTH PROFESSIONALS OR CONSUMERS, TYPE OF REPORT, AND YEAR Table 5 shows the top-five ranked drug classes3 associated with suspect drugs, stratified by whether the initial reporter was a health professional or consumer, the type of report, and year the cases was computerized into the FDA postmarket ADE database. 1996 Data. In 1996, there were 155,529 drug class mentions where type of initial reporter and type of report were known. For consumers, only two of the top-five ranked drug classes were common to all report types: central nervous system agents and hormones and synthetic substitutes. For health professionals, there were four drug classes of the top-five ranked drug classes common to all report types: central nervous system agents, antineoplastic agents, anti-infective agents, and cardiovascular drugs. The only drug class in the top-five ranked drug classes common to both consumers and health professionals across report types was central nervous system agents.
Table 5. Top-5 Ranked Drug Classes Per Type of Reporter & Report: 1996
Table 6 presents the top-10 ranked routes of administration associated with the suspect drugs. There were 156,759 routes mentioned in conjunction with the 159,504 postmarket ADE cases. About three-fifths of the route mentions noted the oral route of administration.
Table 6. Postmarket ADE Reports by Top-10 Ranked Routes of Administration of Suspect Drugs: 1996
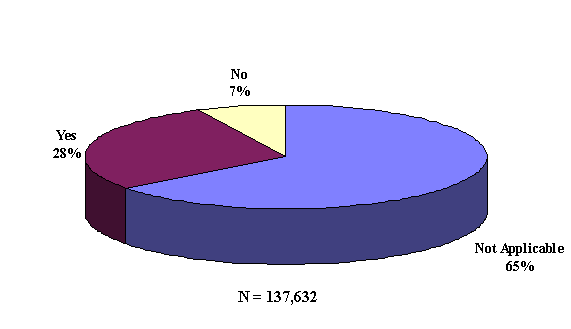
For the 174,905 suspect drug mentions, 78.7% (137,632) had an answer to the question of whether the adverse event abated after the suspect drug was stopped or the dose was reduced. Figure 7 shows the distribution of responses. About one-quarter of these 137,632 abate mentions indicated a positive dechallenge ("Yes" response).
Figure 7. Postmarket ADE Reports by Abate response: 1996
For the 174,905 suspect drug mentions, 76.2% (132,296) had an answer to the question of whether the adverse event reappeared after reintroduction of the suspect drug. Figure 8 shows the distribution of responses. Four percent (5,309) of these 132,296 reoccur mentions indicated a positive rechallenge ("Yes" response).
Figure 8. Postmarket ADE Reports by Reintroduction Response: 1996
N = 132,296 There were 159,515 body system mentions associated with the adverse events of the 159,504 postmarket ADE cases. The distribution of these mentions across the 12 body system mentions is presented in Figure 9. Four body systems each had > 10% of the 159,515 body system mentions: body as a whole (systemic adverse events) - 30.5%, skin and appendages system - 13%, nervous system - 11.7%, and digestive system - 11.4%. Figure 9. Postmarket ADE Reports by Body System: 1996 N = 159,515 ADVERSE EVENTS Table 7 shows the top-10 ranked adverse events reported with the 159,504 postmarket ADE cases. The top ranked ADE was " No drug effect: - 10% of the ADE cases reported this event.
Table 7. Top-10 Ranked Adverse Events: 1996
DRUG CLASSES ASSOCIATED WITH BODY SYSTEM ADVERSE EVENTS Table 8 presents the four body systems comprising the most adverse events, each of which has been crosstabulated by its top-five ranked suspect associated drug classes3. Three drug classes were in the top-five ranks for all four body systems, central nervous system agents, cardiovascular drugs, and anti-infective agents. Table 8. Top-4 Ranked Body Systems with Their Respective Top-5 Ranked Suspect Drug Classes: 1996
1996 In 1996, the Surveillance and Data Processing Branch (SDPB) received a total of 2,162 Freedom of Information (FOI) requests. These requests were for adverse reaction cases collected by the Food and Drug Administration’s Spontaneous Reporting System (SRS). All requests are logged in by the central FOI office and triaged to various responsive divisions throughout the Center for Drugs. SDPB processed FOI requests utilizing several forms of data accession. Compressed ASCII files were provided to mostly third-party businesses. Microfiche line listings or paper copies were also available depending on the preference of the requester. Case reports from the SRS database were obtained by people wanting a formalized version of the Medwatch form. Law firms comprised the most FOI requests, with third-party organizations ranking second. Third were the pharmaceutical companies and last were consumers. However, consumers made more inquiries in 1996 than in previous years. This could have been attributed to media reporting and those consumers wanting to establish a more significant role in their drug therapy. Hal Stepper Date created: March 08, 2001; last updated: June 23, 2005 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||