 |
 |
FDA Home Page | Search
FDA Site | FDA A-Z Index | Contact
FDA | FDA Centennial
![]()
Printable version (234 KB PDF)
FY 2005

PERFORMANCE REPORT
TO CONGRESS
for the
Animal Drug User Fee Act

Food and Drug Administration
Center for Veterinary Medicine
I am pleased to present the Food and Drug Administration FY 2005 Performance Report to Congress for the Animal Drug User Fee Act (ADUFA) of 2003. This report presents FDA's accomplishments for FY 2005, the second year operating under ADUFA, and also updates and finalizes the FY 2004 cohort data. It is my pleasure to report that FDA met or exceeded each performance goal for FY 2004 and is meeting or exceeding each performance goal targeted for FY 2005.
As part of ADUFA implementation, FDA continues to work to improve the new animal drug review process, hire additional FDA staff, and prepare guidance for the industry and staff. FDA continues to reach beyond the established ADUFA performance goals to work collaboratively with the industry to reduce the frequency of multiple review cycles in the new animal drug application process.
While FDA's first 2 years under ADUFA were highly productive and successful, much work still remains in order to complete the building process. In FY 2006, FDA plans to build on its accomplishments and continue to:
FDA is committed to improving the efficiency, quality, and predictability of the new animal drug review process. We are dedicated to exploring new approaches and technologies that offer high quality, cost-effective improvements in FDA's review of new animal drug applications and submissions. FDA looks forward to the continued success and significant improvements in the animal drug review process that ADUFA will make possible.
Andrew C. von Eschenbach, M.D.
Acting Commissioner of Food and Drugs
On November 18, 2003, the President signed the Animal Drug User Fee Act of 2003 (ADUFA) into law. ADUFA amends the Federal Food, Drug, and Cosmetic Act to authorize FDA to collect user fees from new animal drug sponsors. Under ADUFA, FDA agreed to pursue a comprehensive set of review performance goals to improve the timeliness and predictability of the review of new animal drug applications (NADAs) and investigational new animal drug (INAD) submissions. This report updates and finalizes FY 2004 accomplishments and describes FDA's accomplishments in FY 2005 toward meeting the performance goals.
FDA made steady progress in implementing ADUFA in FY 2005. Among the key activities and accomplishments during FY 2005 were:
FDA continues to improve the timeliness and efficiency of animal drug review programs and build FDA's capacity to meet the more rigorous goals in place for future years.
FY 2005 Activities and Accomplishments
Performance At-A-Glance for FY 2004 and FY 2005
Implementation Plans for FY 2006
Report on Final FY 2004 and Preliminary FY 2005 ADUFA Cohort Performance
Supplemental NADAs and Reactivations
Abbreviated New Animal Drug Applications
Appendices:
Appendix A: Secretary Thompson's Letter to Congress
Appendix B: Summary of the ADUFA Performance Goals
Appendix C: Summary of Footnotes
The Animal Drug User Fee Act of 2003 (ADUFA) was enacted on November 18, 2003. ADUFA authorized FDA to collect user fees for certain applications and supplements, establishments, products, and sponsors to support the review of new animal drugs. The Consolidated Appropriations Act of 2004, enacted on January 23, 2004, contained an appropriations provision enabling FDA's implementation of ADUFA. Under ADUFA, FDA agreed to meet specified performance goals for the review of certain submissions over 5 years. FDA agreed to review and act on submissions within shorter time periods for each new fiscal year. Information about ADUFA, including the text of then Secretary Thompson's November 13, 2003, letter to Congress, is located in Appendix A and can also be found at http://www.fda.gov/oc/adufa.
ADUFA requires that the Secretary submit two annual reports to Congress for each fiscal year fees are collected: 1) a performance report due within 60 days of the end of the fiscal year, and 2) a financial report within 120 days of the end of the fiscal year. This report is FDA's second annual performance report that updates and finalizes FY 2004 cohort data. The report also summarizes FDA's continuing progress in implementing ADUFA and in meeting quantifiable ADUFA review goals for FY 2005. In addition, this report describes FDA's implementation plans for FY 2006.
ADUFA was signed into law on November 18, 2003, amending the Federal Food, Drug, and Cosmetic Act and providing FDA with important new responsibilities, resources, and challenges. The goal of ADUFA is to better serve animal health and public health by providing additional funds to augment FDA resources devoted to “the process for review of new animal drug applications.”
Under ADUFA, FDA agreed to meet certain review performance goals. These goals aim to expedite the review of NADAs, supplemental NADAs, and INAD submissions.
This program is similar to the Prescription Drug User Fee Act (PDUFA) program for human drugs that has been in place for over 10 years. The expectation is that ADUFA, like PDUFA, will continue to help FDA expedite and improve its review of applications for new animal drugs so that safe and effective new products will be available more quickly. The guidelines and definitions below apply to FDA's implementation of ADUFA. Further information can be found in Appendix A and can also be found at http://www.fda.gov/oc/adufa.
Review and Act on Applications and Submissions. The term "review and act on" is understood to mean the issuance of a complete action letter after the complete review of an animal drug application, supplemental animal drug application, or investigational drug submission which either (1) approves an animal drug application or supplemental application, or notifies a sponsor that an INAD submission is complete, or (2) sets forth in detail the specific deficiencies in such animal drug application, supplemental animal drug application, or investigational animal drug submission in condition for approval.
Refuse to File Applications and Refuse to Review Submissions. Within 30 days of submission, FDA shall “refuse to file” an animal drug application, supplemental animal drug application, or their reactivation, which is determined to be insufficient on its face or otherwise of unacceptable quality for review upon initial inspection per 21 CFR 514.110. Thus, FDA will refuse to file an application containing numbers or types of errors, or flaws in the development plan, sufficient to cause the quality of the entire submission to be questioned to the extent that FDA cannot reasonably review it. Within 60 days of submission, FDA will refuse to review an INAD which is determined to be insufficient on its face or otherwise of unacceptable quality upon initial inspection using criteria and procedures similar to those found in 21 CFR 514.110. A decision to refuse to file an application or to refuse to review a submission as described above will result in the application or submission being excluded from the cohort upon which the relevant user fee goal is based. FDA will record the numbers and types of these exclusions and include them in its annual performance report.
ADUFA performance goals progress over a five-year period. FDA agreed to review and act on submissions within shorter periods of time each new year. With the fifth year of ADUFA ending on September 30, 2008, FDA has agreed to review and act on 90 percent of the following submissions within the specified times:
The 5-year progression of these goals is presented in Appendix B.
All FDA review performance statistics are based on a fiscal year receipt cohort. This methodology calculates performance statistics for submissions for the fiscal year FDA received them, regardless of when FDA ultimately acted on or approved the submissions. A result of this approach is that the statistics shown for a particular year may change from one report to the next. This is because as time passes, FDA completes work on more and more submissions in a receipt cohort. As more submissions are completed, the statistics for that year of receipt must be adjusted to reflect the new completions. Until all submissions in a cohort are completed, only a preliminary performance assessment can be provided for that cohort. In this report, FDA is providing the status of FY 2005 data, as of September 30, 2005, and final data for FY 2004.
As part of ADUFA implementation, FDA is addressing backlogs, hiring new employees, and developing policy and procedure documents to improve the new animal drug review process. These actions are intended to position FDA to meet the progressively challenging performance goals of ADUFA.
FDA made steady progress in implementing ADUFA in FY 2005. Among the key activities and accomplishments during FY 2005 were:
1. Format and Style Conventions for Letters
2. Preparing a Memorandum of Conference
3. Permissible Submission Tracking And Reporting System ( STARS) Action Codes for STARS Submissions
4. Review of Protocols
These are available on the CVM homepage on the FDA Web site at http://www.fda.gov/cvm.
FDA met or exceeded each of the ADUFA review performance goals (90 percent on time within the specified review times) for the FY 2004 cohort.
As of September 30, 2005, FDA is meeting or exceeding each of the designated ADUFA review performance goals (90 percent on time performance goals within the specified review times) for the FY 2005 cohort completed thus far. With submissions still pending, it is too early to make a final performance determination for FY 2005.
The table below summarizes FDA's review performance on the FY 2004 application submissions and the preliminary performance in reviewing FY 2005 application submissions.
During FY 2006, FDA will continue to expand its efforts, through employee hiring, training, and development of guidance. These efforts will improve the timeliness and efficiency of animal drug review programs and build FDA's capacity to meet the more rigorous goals in place for future years.
This report updates FDA’s review performance for FY 2004 and presents the
FY 2005 review performance for the ADUFA performance goals and commitments. All performance data are as of September 30, 2005, and calculated percentages are rounded to the nearest whole number. The following information refers to FDA performance presented in this report.
The table below summarizes the annual review time goals for original and administrative NADAs and reactivations. Over the 5-year period defined by ADUFA, the number of review days is incrementally reduced for each type of submission while the goal of reviewing 90 percent of submissions remains constant.
Submission Type |
Review Time Goal |
Performance Goal |
||||
|---|---|---|---|---|---|---|
| FY 04 | FY 05 | FY 06 | FY 07 | FY 08 | ||
Original NADAs and Reactivations |
295 days |
270 days |
230 days |
200 days |
180 days |
90% on time |
Administrative NADAs and Reactivations |
90 days |
85 days |
80 days |
70 days |
60 days |
90% on time |
The total number of NADAs and reactivations submitted to FDA and accepted for filing decreased from FY 2004.2 The decrease was primarily a result of fewer original NADAs and reactivations being submitted (see graph to the right and table below). |
 D D |
Filings |
|||||
|---|---|---|---|---|---|
Type |
FY 04 |
FY 05 |
FY 06 |
FY 07 |
FY 08 |
Original NADAs and Reactivations |
7 |
4 |
-- |
-- |
-- |
Administrative NADAs and Reactivations |
10 |
9 |
-- |
-- |
-- |
Total |
17 |
13 |
|||
FY 2004 Submissions
The 90 percent on-time ADUFA review goal was exceeded for all original and administrative NADAs and reactivations in FY 2004. FDA reviewed and acted on all original NADAs and reactivations within 295 days, and all administrative NADAs and reactivations within 90 days (see table below).
Submission Type |
Review Within |
Reviewed and Acted On |
Number On Time |
Percent on Time |
ADUFA Performance Goal |
|---|---|---|---|---|---|
Original NADAs and Reactivations |
295 days |
7 |
7 |
100% |
90% |
Administrative NADAs and Reactivations |
90 days |
10 |
10 |
100% |
90% |
FY 2005 Submissions
As of September 30, 2005, one-fourth (1 of 4) of the original NADAs and reactivations filed in FY 2005 have been reviewed and acted on within the 270-day ADUFA review goal. Two-thirds (6 of 9) of the administrative NADAs and reactivations filed in FY 2005 had been reviewed and acted on; and all had met the 85-day ADUFA review goal (see table below). With submissions still pending and not overdue, it is too early to make a final performance determination for FY 2005.
Submission |
Review Within |
Reviewed and Acted On |
Number On Time |
Percent on Time |
ADUFA Performance Goal |
|---|---|---|---|---|---|
Original NADAs and Reactivations |
270 days |
1 |
1 |
100% |
90% |
Administrative NADAs and Reactivations |
85 days |
6 |
6 |
100% |
90% |
The table below summarizes the annual review time goals for non-manufacturing and manufacturing supplemental NADAs and reactivations. Over the 5-year period defined by ADUFA, the number of review days is incrementally reduced for each type of submission while the goal of reviewing 90 percent of submissions remains constant.
Submission Type |
Review Time Goal |
Performance Goal |
||||
|---|---|---|---|---|---|---|
FY 04 |
FY 05 | FY 06 | FY 07 | FY 08 | ||
Non-manufacturing Supplemental NADAs and Reactivations |
320 days |
285 days |
235 days |
200 days |
180 days |
90% on time |
Manufacturing Supplemental NADAs and Reactivations |
225 days |
190 days |
140 days |
120 days |
120 days |
90% on time |
The total number of supplemental NADAs and reactivations filed increased by five percent from FY 2004. All of the increase was accounted for by manufacturing supplemental NADAs and reactivations (see graph to the right and table below).3 |
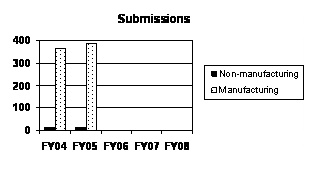 D D |
Submissions |
|||||
|---|---|---|---|---|---|
Type |
FY 04 |
FY 05 |
FY 06 |
FY 07 |
FY 08 |
Non-manufacturing Supplemental NADAs and Reactivations |
14 |
11 |
-- |
-- |
-- |
Manufacturing Supplemental NADAs and Reactivations |
3634 |
386 |
-- |
-- |
-- |
Total |
377 |
397 |
|||
FY 2004 Submissions
The 90 percent on-time ADUFA review goal was exceeded for all non-manufacturing and manufacturing supplemental NADAs and reactivations in FY 2004. FDA reviewed and acted on all of the non-manufacturing supplemental NADAs and reactivations within 320 days, and all but 4 (359 of 363) of the manufacturing supplemental NADAs and reactivations within 225 days (see table below).
Submission Type |
Review Within |
Reviewed and Acted On |
Number On Time |
Percent on Time |
ADUFA Performance Goal |
|---|---|---|---|---|---|
Non-manufacturing Supplemental NADAs and Reactivations |
320 days |
14 |
14 |
100% |
90% |
Manufacturing Supplemental NADAs and Reactivations |
225 days |
363 |
359 |
99% |
90% |
FY 2005 Submissions
As of September 30, 2005, 3 of the 11 non-manufacturing supplemental NADAs and reactivations submitted in FY 2005 had been reviewed and acted on within the 285-day ADUFA review goal. Three-fourths (297 of 386) of the manufacturing supplemental NADAs and reactivations submitted in FY 2005 had been reviewed and acted on; and all but one (296 of 297) had met the 190-day ADUFA review goal (see table below). With submissions still pending and not overdue, it is too early to make a final performance determination for FY 2005.
Submission Type |
Review Within |
Reviewed and Acted On |
Number On Time |
Percent on Time |
ADUFA Performance Goal |
|---|---|---|---|---|---|
Non-manufacturing Supplemental NADAs and Reactivations |
285 days |
3 |
3 |
100% |
90% |
Manufacturing Supplemental NADAs and Reactivations |
190 days |
297 |
296 |
100% |
90% |
The table below summarizes the annual review time goals for INAD studies and study protocol submissions. Over the 5-year period defined by ADUFA, the number of review days is incrementally reduced for each type of submission while the goal of reviewing 90 percent of submissions remains constant.
Submission Type |
Review Time Goal |
Performance Goal |
||||
|---|---|---|---|---|---|---|
| FY 04 | FY 05 | FY 06 | FY 07 | FY 08 | ||
INAD Studies |
320 days |
285 days |
235 days |
200 days |
180 days |
90% on time |
INAD Study Protocols |
125 days |
100 days |
80 days |
60 days |
50 days |
90% on time |
The total number of INAD submissions increased from FY 2004. All of the increase was a result of INAD studies that increased by 22 percent (see graph to the right and table below).5 |
 D D |
Submissions |
|||||
|---|---|---|---|---|---|
Type |
FY 04 |
FY 05 |
FY 06 |
FY 07 |
FY 08 |
INAD Studies |
2436 |
296 |
-- |
-- |
-- |
INAD Study Protocols |
1737 |
169 |
-- |
-- |
-- |
Total |
416 |
465 |
|||
FY 2004 Submissions
The 90 percent on-time ADUFA review goal was exceeded for INAD studies and study protocol submissions in FY 2004. FDA reviewed and acted on all of the INAD studies within 320 days and 99 percent (172 of 173) of the INAD study protocols within 125 days (see table below).
Submission Type |
Review Within |
Reviewed and Acted On |
Number On Time |
Percent on Time |
ADUFA Performance Goal |
|---|---|---|---|---|---|
INAD Studies |
320 days |
243 |
243 |
100% |
90% |
INAD Study Protocols |
125 days |
173 |
172 |
99% |
90% |
FY 2005 Submissions
As of September 30, 2005, over half (162 of 296) of the INAD studies submitted in FY 2005 had been reviewed and acted on; and all had met the 285-day ADUFA review goal. Most (148 of 169) of the INAD study protocols submitted in FY 2005 had been reviewed and acted on; and all but one (147 of 148) had met the 100-day ADUFA review goal (see table below). With submissions still pending and not overdue, it is too early to make a final performance determination for FY 2005.
Submission Type |
Review Within |
Reviewed and Acted On |
Number On Time |
Percent on Time |
ADUFA Performance Goal |
|---|---|---|---|---|---|
INAD Studies |
285 days |
162 |
162 |
100% |
90% |
INAD Study Protocols |
100 days |
148 |
147 |
99% |
90% |
Section 740(k) Abbreviated New Animal Drug Applications of the Federal Food, Drug, and Cosmetic Act provides:
The Secretary shall -
‘‘(1) to the extent practicable, segregate the review of abbreviated new animal drug applications from the process for the review of animal drug applications,” and
‘‘(2) adopt other administrative procedures to ensure that review times of abbreviated new animal drug applications do not increase from their current level, due to activities under the user fee program.''
FDA's CVM has established within its Office of New Animal Drug Evaluation (ONADE) a separate staff, the Generic Animal Drug Team, dedicated to the review of Abbreviated New Animal Drug Applications (ANADAs) and submissions. FDA also established a team within ONADE's Division of Manufacturing Technologies to handle related ANADA chemistry reviews.8
CVM maintains a separate review queue for ANADAs. It is important to emphasize that this queue is independent from the queue maintained for the process to review NADAs under ADUFA. This ensures that ANADAs are reviewed independently of applications under ADUFA by dedicated staff. Application management processes and adherence to them are being re-examined and continue to be worked on and improved within the Generic Animal Drug Team.
To ensure that review times for ANADAs and submissions do not increase due to activities under the user fee program, ONADE established a baseline of sentinel submission review times averaged over several fiscal years (2001 through 2003). FDA staff selected document and submission types for monitoring based on submission types that were analogous to the ADUFA sentinel submission types. FDA staff continually monitors current year completed review times for these submissions. The average review times increased during FY 2005, in large part due to staff turnover at FDA. Specifically, the increase in review times is attributed to: 1) work not accomplished while positions remained vacant and 2) the loss of productivity associated with the loss of experienced reviewers.
On November 13, 2003, the Department of Health and Human Services (HHS) Secretary Tommy G. Thompson sent identical performance goal letters to the following four members of Congress:
The Honorable Judd Gregg
Chairman
Committee on Health, Education, Labor and Pensions
United States Senate
The Honorable Edward Kennedy
Ranking Minority Member
Committee on Health, Education, Labor and Pensions
United States Senate
The Honorable W. J. (Billy) Tauzin
Chairman
Committee on Energy and Commerce
U.S. House of Representatives
The Honorable John Dingell
Ranking Minority Member
Committee on Energy and Commerce
U.S. House of Representatives
This appendix provides one copy of the four identical letters and a summary of the goals and procedures of CVM as agreed to under the "Animal Drug User Fee Act of 2003."
The generic animal drug review staff was brought up to full pre-ADUFA capacity by filling existing vacancies during FY 2005. In FY 2005, a number of the generic animal drug review staff left CVM and were subsequently replaced with new reviewers .
THE SECRETARY OF HEALTH AND HUMAN SERVICES
Washington, DC, November 13, 2003
The Honorable Judd Gregg
Chairman
Committee on Health, Education, Labor and Pensions
United States Senate
Washington, DC 20510
Dear Mr. Chairman:
As you are aware, the Food and Drug Administration has been working with representatives of the veterinary pharmaceutical industry and staff of your Committee to design a new animal drug "user fee" proposal. Under this proposal, the additional revenues generated from fees paid by this industry would be dedicated for use in expediting the process for the review of animal drug applications, in accordance with performance goals that have been developed by FDA in consultation with the industry. S.313, the "Animal Drug User Fee Act of 2003" reflects the fee mechanisms developed in these discussions. The performance goals are specific in the enclosure to this letter entitled, "Animal Drug User Fee Act Performance Goals and Procedures." I believe they represent a realistic projection of what FDA can accomplish with industry cooperation and the additional resources that would be provided by the bill and annual FDA appropriations that fully cover the costs of pay and inflation increases for the animal drug review process each year.
I appreciate the support of you and your staff, and the assistance of other Members of the Committee.
Sincerely,
TOMMY G. THOMPSON
Enclosure
The goals and procedures of the FDA Center for Veterinary Medicine (CVM) as agreed to under the “Animal Drug User Fee Act of 2003” are summarized as follows:
1. Review and act on 90 percent of complete animal drug applications (NADAs) and reactivations of such applications within 180 days after submission date.
2. Review and act on 90 percent of non-manufacturing supplemental animal drug applications (i.e. supplemental animal drug applications for which safety or effectiveness data are required) and reactivations of such supplemental applications within 180 days after submission date.
3. Review and act on 90 percent of manufacturing supplemental animal drug applications and reactivations of such supplemental applications within 120 days after submissions date.
4. Review and act on 90 percent of investigational animal drug study submissions within 180 days after submission date.
5. Review and act on 90 percent of investigational animal drug submissions consisting of protocols, that the Agency and the sponsor consider to be an essential part of the basis for making the decision to approve or not approve an animal drug application or supplemental animal drug applications, without substantial data within 50 days after submission date.
6. Review and act on 90 percent of administrative animal drug applications (NADAs submitted after all scientific decisions have been made in the investigational animal drug process, i.e., prior to the submission of the NADA) within 60 days after the submission date.
The term "review and act on" is understood to mean the issuance of a complete action letter after the complete review of an animal drug application, supplemental animal drug application, or investigational drug submission which either (1) approves an animal drug application or supplemental application of notifies a sponsor that an investigational new animal drug submission is complete or (2) sets forth in detail the specific deficiencies in such animal drug application, supplemental animal drug application, or investigational animal drug submission in condition for approval. Within 30 days of submission, FDA shall refuse to file and animal drug application, supplemental animal drug application, or their reactivation, which is determined to be insufficient on its face or otherwise of unacceptable quality for review upon initial inspection as per 21 CFR 514.110. Thus, the agency will refuse to file an application containing numbers or types of errors, or flaws in the development plan, sufficient to cause the quality of the entire submission to be questioned to the extent that it cannot reasonably be reviewed. Within 60 days of submission, FDA will refuse to review an investigational animal drug submission which is determined to be insufficient on its face or otherwise of unacceptable quality upon initial inspection using criteria and procedures similar to those found in 21 CFR 514.110. A decision to refuse to file an application or to refuse to review a submission as describe above will result in the application or submission not being entered into the cohort upon which the relevant user fee goal is based. The agency will keep a record of the numbers and types of such refusals and include them in its annual performance report.
FDA may request minor amendments to animals drug applications, supplemental animal drug applications, and investigational animal drug submissions. At its discretion, the Agency may extend an internal due date (but not a user fee goal) to allow for the complete review of an application or submission for which a minor amendment is requested. If a pending application is amended with significant changes, the amended application may be considered resubmitted, thereby effectively resetting the clock to the date FDA received the amendment. The Agency intends to establish the same policy for investigational animal drug submissions.
Sponsors are not required to submit study protocols for review. However, for each voluntarily submitted protocol for a study that the Agency and the sponsor considered to be an essential part of the basis for making the decision to approve or not approve an animal drug application or supplemental animal drug application, the Agency will issue an acknowledgment letter providing comments resulting from a complete review of the protocol. The acknowledgement letter will be as detailed as possible considering the quality and level of detail of the protocol submission; will include a succinct assessment of the protocol; and will state whether the Agency agrees, disagrees, or lacks sufficient information to reach a decision that the protocol design, execution plans and data analyses are adequate to achieve the objectives of the study. If the Agency determines that a protocol is acceptable, this represents an agreement that the data generated by the protocol can be used to support a safety or effectiveness decision regarding the subject animal drug. The fundamental agreement is that having agreed to the design, execution, or analyses proposed in protocols reviewed under this process, the Agency will not later alter its perspectives on the issues of design, execution or analyses unless public or animal health concerns unrecognized at the time of protocol assessment under this process are evident.
1. Review and act on pending animal drug applications, supplemental animal drug applications, and investigational animal drug submissions within 24 months of initiation of user fee payments.
1. Fifty percent of FDA incremental review staff recruited and on-board by first quarter of FY 2006. Total staff increment on-board by end of FY 2008.
2. FDA will review all submissions in accordance with procedures for working within a queue. An Application/submission that is not reviewed within the applicable interim Application/Submission Goal time frame (noted below) will be reviewed with the highest possible priority among those pending.
FY 04 Review and Act on 90 percent of:
FY 05 Review and Act on 90 percent of:
FY 06 Review and Act on 90 percent of:
FY 07 Review and Act on 90 percent of:
FY 08 Review and Act on 90 percent of:
Activity |
Performance Level |
FDA Review Time |
||||
|---|---|---|---|---|---|---|
FY 04 |
FY 05 |
FY 06 |
FY 07 |
FY 08 |
||
Application/Submission Goals |
||||||
Animal drug applications (NADAs) and reactivations of such applications |
90% |
295 |
270 |
230 |
200 |
180 |
Non-manufacturing supplemental animal drug applications and reactivations of such supplemental applications |
90% |
320 |
285 |
235 |
200 |
180 |
Manufacturing supplemental animal drug applications and reactivation of such supplemental applications |
90% |
225 |
190 |
140 |
120 |
120 |
Investigational animal drug study submissions |
90% |
320 |
285 |
235 |
200 |
180 |
Investigational animal drug submissions consisting of protocols, that the agency and the sponsor consider to be an essential part of the basis for making the decision to approve or not approve an animal drug application or supplemental animal drug application, without substantial data |
90% |
125 |
100 |
80 |
60 |
50 |
Administrative animal drug applications (administrative NADAs) |
90% |
90 |
85 |
80 |
70 |
60 |
Interim Backlog Goals |
||||||
Review and act on pending animal drug applications, supplemental animal drug applications, and investigational animal drug submissions within 24 months of initiation of user fee payments. |
||||||
Additional Interim Goals |
||||||
Fifty percent of FDA incremental review staff recruited and on-board by first quarter of FY 2006. Total staff increment on-board by end of FY 2008. |
||||||
FDA will review all submissions in accordance with procedures for working within a queue. An application/submission that is not reviewed within the applicable Interim Application/Submission Goal timeframe will be reviewed with the highest possible priority among those pending. |
||||||
1. Over the 5-year period defined by ADUFA, the number of review days is incrementally reduced for each of type of submission.
2. The count of FY 2005 submissions assumes that all submissions received in the last month of FY 2005 are filed.FDA makes a filing decision within 30 days of an original application's receipt. FDA calculates ADUFA review times, however, from the original receipt date of the filed application. In FY 2005, there was one administrative NADA that was refused to file.
3. In FY 2005, there were five manufacturing supplements that were withdrawn by the sponsor.
4. The FY 2004 report stated that 357 manufacturing supplements were received. However, 2 submissions were erroneously included and 16 supplements to "hybrids" (applications to copy a pioneer animal drug product and, at the same time, a request to make a change that differs from the pioneer animal drug product that requires data to demonstrate safety or effectiveness) were not previously included. Eight supplements were withdrawn, which brings the total to 363.
5. In FY 2005, there were four INAD studies and seven protocols that were refused to review because they were incomplete.
6. In FY 2004, 251 INAD studies were reported as having been received. However, 10 studies were filed without reply and 2 were refused to review, and therefore are not included in the cohort. These submissions were dropped from the cohort count. Four INAD studies were added that were inadvertently not included in FY 2004.
7. In FY 2004, 224 protocols were reported as having been received. Five were incorrectly counted because they were protocols with data submissions; 43 were filed without review; and 3 were subsequently classified as refused to review and, therefore, removed from the cohort.
8. The generic animal drug review staff was brought up to full pre-ADUFA capacity by filling existing vacancies during FY 2005. In FY 2005, a number of the generic animal drug review staff left CVM and were subsequently replaced with new reviewers .
 Department of Health and Human Services
Department of Health and Human Services 
Food and Drug Administration
This report was prepared by FDA's Center for Veterinary Medicine (CVM) in collaboration with the Office of Planning. For information on obtaining additional copies contact: Office of Planning (HFP-10) This report is available on the FDA Home Page at http://www.fda.gov. |
![]()