| Home
|
| Home
|
|
This chapter also available as PDF file. Final Report also is available as single PDF file (8.8 Mb) See also: Vision for the Program and Highlights of the Scientific Strategic Plan Authors, Reviewers, and Workshop Participants Graphics and Photography Source
Acronyms, Abbreviations,
|
CHAPTER 3.
|
| This chapter's contents... Question 3.1: What are the climate-relevant chemical, microphysical, and optical properties, and spatial and temporal distributions, of human-caused and naturally occurring aerosols? Question 3.2: What are the atmospheric sources and sinks of the greenhouse gases other than CO2 and the implications for the Earth's energy balance? Question 3.3: What are the effects of regional pollution on the global atmosphere and the effects of global climate and chemical change on regional air quality and atmospheric chemical inputs to ecosystems? Question 3.4: What are the characteristics of the recovery of the stratospheric ozone layer in response to declining abundances of ozone-depleting gases and increasing abundances of greenhouse gases? Question 3.5: What are the couplings and feedback mechanisms among climate change, air pollution, and ozone layer depletion, and their relationship to the health of humans and ecosystems? |
The composition of the atmosphere -- its gases and particles -- plays a critical role in connecting human welfare with global and regional changes because the atmosphere links all of the principal components of the Earth system. The atmosphere interacts with the oceans, land, terrestrial and marine plants and animals, and the frozen regions (see Figure 3-1). Because of these linkages, the atmosphere is a conduit of change. Emissions from natural sources and human activities enter the atmosphere at the surface and are transported to other geographical locations and often higher altitudes. Some emissions undergo chemical transformation or removal while in the atmosphere or interact with cloud formation and precipitation. Some natural events and human activities that change atmospheric composition also change the Earth's radiative (energy) balance. Subsequent responses to changes in atmospheric composition by the stratospheric ozone layer, the climate system, and regional chemical composition (air quality), create multiple environmental effects that can influence human health and natural systems.
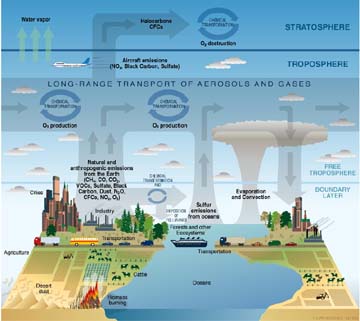 Figure 3-1: Schematic of chemical and transport processes related to atmospheric composition. These processes link the atmosphere with other components of the Earth system, including the oceans, land, and terrestrial and marine plants and animals. |
Atmospheric composition changes are indicators of many potential environmental issues. Observations of trends in atmospheric composition are among the earliest harbingers of global changes. For example, the decline of the concentrations of ozone-depleting substances, such as the chlorofluorocarbons (CFCs), has been the first measure of the effectiveness of international agreements to end production and use of these compounds.
A principal feature of the atmosphere is that it acts as a long-term "reservoir" for certain trace gases that can cause global changes. The long removal times of some gases, such as CO2 (>100 years) and perfluorocarbons (>1,000 years), imply that any associated global changes could persist over decades, centuries, and millennia -- affecting all countries and populations.
An effective program of scientific inquiry relating to changes in atmospheric composition must include two major foci. The first is a focus on Earth system interactions: How do changes in atmospheric composition alter and respond to the energy balance of the climate system? What are the interactions between the climate system and stratospheric ozone? What are the effects of regional pollution on the global atmosphere and the effects of global climate and chemical change on regional air quality? The second is a focus on Earth system and human system linkages: How is the composition of the global atmosphere, as it relates to climate, ozone depletion, ultraviolet radiation, and pollutant exposure, altered by human activities and natural phenomena? How quantitative is the knowledge of the major sources of emissions to the atmosphere? What are the atmospheric composition changes that could affect human health and natural ecosystems?
The overall research approach for understanding the role of atmospheric composition is an integrated application of long-term systematic observations, laboratory and field studies, and modeling, with periodic assessments of understanding and significance to decision making. Most of the activities related to atmospheric composition research are part of national and international partnerships, some of which are noted at the end of this chapter. Such partnerships are necessitated by the breadth and complexity of current issues and because the atmosphere links all nations. The overall research approach is based on the substantial body of knowledge and understanding available from the work of many international scientists. The status of understanding is reported as part of cooperative international assessment activities (e.g., IPCC, 2001a, b, d; WMO, 1999, 2003).
In looking ahead at what the specific policy-relevant information needs associated with atmospheric composition will be, five broad challenges are apparent, with goals and examples of key research objectives outlined below.
Question 3.1: What are the climate-relevant chemical, microphysical, and optical properties, and spatial and temporal distributions, of human-caused and naturally occurring aerosols? |
Research has demonstrated that atmospheric particles (aerosols) can cause a net cooling or warming tendency within the climate system, depending upon their physical and chemical characteristics. Sulfate-based aerosols, for example, tend to cool, whereas black carbon (soot) tends to warm the system (see Figure 3-2). In addition to these direct effects, aerosols can also have indirect effects on radiative forcing (e.g., changes in cloud properties). When climate models include the effects of sulfate aerosol, the simulation of global mean surface temperatures is improved. One of the largest uncertainties about the net impact of aerosols on climate is the diverse warming and cooling influences of the very complex mixture of aerosol types and their spatial distributions. Further, the poorly understood impact of aerosols on the formation of both water droplets and ice crystals in clouds also results in large uncertainties in the ability to project climate changes (see Figure 3-2). More detail is needed globally to describe the scattering and absorbing optical properties of aerosols from regional sources and how these aerosols impact other regions of the globe.
 Figure 3-2: Schematic comparing several factors that influence Earth's climate on the basis of their contribution to radiative forcing between 1750 and 2000. Two principal categories of radiative forcing factors are the greenhouse gases and the combination of aerosols and clouds. The rectangular bars represent a best estimate of the contributions of these forcings, some of which yield warming and some cooling, while the vertical line about the rectangular bars indicates the range of estimates. A vertical line without a rectangular bar denotes a forcing for which no best estimate can be given owing to large uncertainties. Scientific understanding of aerosol effects is very low, as shown on the horizontal axis. Source: IPCC (2001d). For more information, see Annex C. |
Illustrative Research Questions
The relationship of aerosols to climate change is complex because of the diverse formation and transformation processes involving aerosols (see Figure 3-3). This complexity underlies many of the important research questions related to aerosols.
 Figure 3-3: Schematic of the processes that cause the formation and transformation of aerosol particles in the atmosphere. Aerosols may be emitted directly into the atmosphere or be formed there from the emissions of gaseous precursors. Particles grow by condensation of gases and by coagulation with other particles, and their number and composition can influence the formation and radiative characteristics of clouds. For more information, see Annex C. |
Research Needs
Significant research is required to complete our understanding of aerosols and their role in climate change processes. The representation of aerosol properties and their distribution in the atmosphere is highly complex. The needs outlined below describe important future steps to improve our understanding.
|
Box 3-1. Atmospheric Composition CCRI Priority - Aerosols The Climate Change Research Initiative (CCRI) will leverage existing U.S. Global Climate Research Program (USGCRP) research to address major gaps in understanding climate change. Uncertainties related to the effects of aerosols on climate are large, with both warming and cooling effects possible depending on the nature and distribution of the aerosol. The CCRI will advance the understanding of the distribution of all major types of aerosols and their variability through time, the different contributions of aerosols from human activities, and the processes by which the different contributions are linked to global distributions of aerosols. The CCRI will support research to improve understanding of the processes by which trace gases and aerosols are transformed and transported in the atmosphere. Studies of how atmospheric chemistry, composition, and climate are linked will be emphasized, including those processes that control the abundance of constituents that affect the Earth's radiation budget, such as tropospheric methane, ozone, and aerosols. The global distributions of a limited number of atmospheric parameters (including climatically relevant parameters such as ozone and aerosols) and their variabilities will be obtained from satellite observations over long periods of time along with more comprehensive suites of observations over briefer time periods. Satellite data recently obtained and to become available for the first time for methane, tropospheric ozone, and tropospheric aerosols will be analyzed and interpreted in the context of global models and assimilation systems. The studies will provide an observational- and model-based evaluation of the radiative forcings associated with aerosol direct and indirect effects. These forcing results will contribute to the CCSP climate projections for research and assessment. |
These research needs are strongly linked to those of the Climate Variability and Change (Chapter 4), Water Cycle (Chapter 5), Land-Use/Land-Cover Change (Chapter 6), and Carbon Cycle (Chapter 7) elements.
MILESTONES, PRODUCTS AND PAYOFFS
Question 3.2: What are the atmospheric sources and sinks of the greenhouse gases other than CO2 and the implications for the Earth's energy balance? |
The increasing concentrations of chemically active greenhouse gases, such as methane (CH4), tropospheric ozone (O3), nitrous oxide (N2O), and halocarbons (e.g., CFCs, hydrochlorofluorocarbons (HCFCs), hydrofluorocarbons (HFCs), perfluorocarbons (PFCs), and sulfur hexafluoride (SF6)), represent primary contributions to the radiative forcing of the climate system (see Figure 3-2). Anthropogenic emissions of CO2 are addressed in Chapter 7. Water vapor, considered the most dominant greenhouse gas (albeit as a feedback), is discussed as part of the hydrological cycle (see Chapter 5).
The natural and anthropogenic emission sources leading to the observed growth rates of CH4 (the second-most influential anthropogenic greenhouse gas) and N2O abundances are qualitatively understood but poorly quantified (e.g., the amount of CH4 emitted by rice agriculture). Trends in tropospheric ozone (the third-most influential anthropogenic greenhouse gas) are not well determined because they are driven by a mix of natural and anthropogenic emissions, including CH4 and regional pollutants such as the reactive nitrogen oxides (NOx). The atmospheric concentrations and sources of the CFCs and HCFCs have been well studied because of their role in stratospheric ozone depletion. Some important greenhouse gases are removed from the troposphere and stratosphere in reaction with the hydroxyl radical (OH) (e.g., CH4, HFCs, HCFCs) or by photolysis (e.g., N2O). Reactions that remove a greenhouse gas control its lifetime in the atmosphere and, hence, its contribution to radiative forcing of climate.
Illustrative Research Questions
Driven by the need to have a better predictive understanding of the relationship between the emission sources of these radiatively active gases and their global distributions and radiative forcing, several questions continue to face the research community. More quantitative information is needed to help answer these questions, which include:
Field and laboratory studies, satellite observations, and diagnostic transport/chemical modeling are needed to fully address these questions. Examples of these activities are:
MILESTONES, PRODUCTS AND PAYOFFS
Question 3.3: What are the effects of regional pollution on the global atmosphere and the effects of global climate and chemical change on regional air quality and atmospheric chemical inputs to ecosystems? |
Emissions from rapidly industrializing regions of the world have the potential to impact air quality and ecosystem health in regions far from the sources. Paleo-chemical data from ice cores and snow document past perturbations and demonstrate that even remote areas, such as Greenland, are influenced by worldwide emissions. The anthropogenic contribution to the nitrogen cycle from fossil fuel combustion and fertilizer production now rivals in magnitude the natural input from nitrogen-fixing organisms and lightning. This additional nitrogen input to the biosphere illustrates how human activities could have important consequences for ecosystem structure and function. The importance of the effect of regional pollution on global tropospheric chemistry has been recognized for some time. Now, the importance of understanding the reverse effect -- that of global-scale transport of pollutants or global change on regional air quality -- is increasing. A well-recognized example is the enhancement of background global ozone concentrations by anthropogenic emissions.
Illustrative Research Questions
This emerging picture is shaping several questions of importance to society. Some examples of these are as follows:
These questions are being addressed by measurements of key tropospheric constituents from satellites, airborne platforms, and surface sites. Model analyses and simulations are used to provide a regional and global context for the measurement dataset and to address future scenarios. Near-term goals include the following:
MILESTONES, PRODUCTS AND PAYOFFS
Question 3.4: What are the characteristics of the recovery of the stratospheric ozone layer in response to declining abundances of ozone-depleting gases and increasing abundances of greenhouse gases? |
State of Knowledge
The primary cause of stratospheric ozone depletion observed over the last two decades is an increase in the concentrations of industrially produced ozone-depleting chemicals (e.g., CFCs). The depletion has been significant, ranging from a few percent per decade at mid-latitudes to greater than fifty percent seasonal losses at high latitudes. Notable is the annually recurring Antarctic ozone hole, as well as smaller, but still significant, winter/spring ozone losses recently observed in the Arctic. Reductions in atmospheric ozone levels lead to increased exposure to ultraviolet radiation at the surface, which has harmful consequences for plant and animal life, and human health. In response to these findings, the nations of the world ratified the Montreal Protocol on Substances That Deplete the Ozone Layer and agreed to phase out the production of most ozone-depleting chemicals.
Ground-based and satellite measurements show that concentrations of many of these compounds are now beginning to decrease in the atmosphere (WMO, 1999, 2003). As the atmospheric burden of ozone-depleting chemicals falls in response to international efforts, stratospheric ozone concentrations should begin to recover in coming decades (see Figure 3-4). However, because of the ongoing changes in atmospheric composition and climate parameters, which began before the onset of stratospheric ozone depletion, the exact course and timing of ozone recovery in the coming decades is not fully known.
|
Figure 3-4: Schematic illustrating the estimated recovery of stratospheric ozone in the coming decades. Observations show the decline of global total ozone (top panel) and minimum values of total ozone over Antarctica (lower panel) beginning in 1980. As the abundances of ozone-depleting gases decline in the atmosphere, ozone values are expected to increase and recover towards 1980 and earlier values. The estimated recovery of ozone is shown based on predictions by several atmospheric models which use different assumptions about the composition and meteorology of the future stratosphere. The research needs as outlined in Section 3.4 address reducing the range of uncertainty in ozone recovery estimates. Source: Adapted from WMO (2003). For more information, see Annex C. |
Illustrative Research Questions
Research Needs
Improving our understanding of the complex interaction between the ozone layer and the climate system requires further investigations of the processes that interconnect ozone, water vapor, reactive trace constituents (notably chlorine and bromine compounds and reactive nitrogen oxides), aerosols, and temperature. Research needs include the following:
MILESTONES, PRODUCTS AND PAYOFFS
Question 3.5: What are the couplings and feedback mechanisms among climate change, air pollution, and ozone layer depletion, and their relationship to the health of humans and ecosystems? |
State of Knowledge
This question is intended to underscore the explicit need to better understand the relationships that exist between research issues that historically have been treated separately. Understanding the potential consequences of changes in atmospheric composition cuts across a range of important societal issues and scientific disciplines. Atmospheric composition links climate change, air quality, and ozone depletion with human and ecosystem health. The links involve physical processes and feedback mechanisms -- some of which are newly recognized and most of which require further study.
Understanding these connections is important for understanding climate change on regional and global scales. For example, research has demonstrated that stratospheric ozone depletion not only causes increased exposure to ultraviolet radiation at the surface, but also exerts a cooling influence on the global climate. Conversely, climate-related changes may cool the lower stratosphere and increase ozone layer depletion in polar regions. Tropospheric ozone, of concern primarily as a component of smog, is not only a local health risk, but also exerts a warming influence on the global climate. Emissions of SO2 from fossil-fuel combustion not only lead to the formation of acid rain, but also contribute to sulfate-aerosol haze, which exerts a cooling influence on the global climate system.
It is clear that these issues, which initially may have been treated separately by scientists and policymakers, now require a more integrated approach that addresses multiple sources, stresses, and impacts (NASA, 2002).
Illustrative Research Questions
Research needs
Research needs, in addition to those given in Sections 3-1 to 3-4, linking atmospheric composition issues to the health of humans and ecosystems are:
MILESTONES, PRODUCTS AND PAYOFFS
National and International Partnerships |
The Atmospheric Composition research focus is linked via cooperation, co-planning, and joint execution to several national and international planning and coordinating activities. A few examples are:
Chapter 3 AuthorsLead Authors
Contributors
|
|