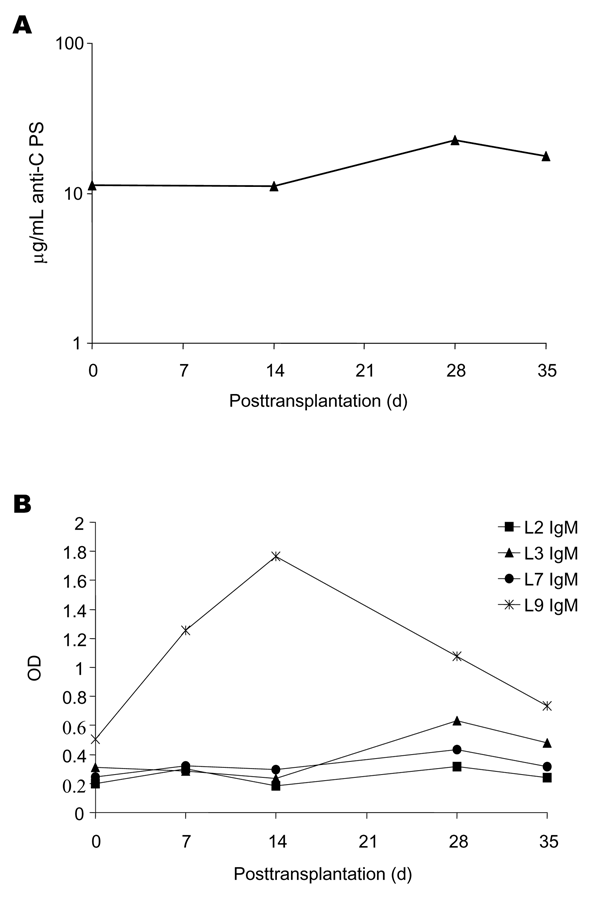
| |
Figure. Pre- and posttransplantation serum antibodies as measured
by enzyme-linked immunosorbent assay (ELISA). A) Immunoglobulin (Ig) G
antibodies to Neisseria meningitidis serogroup C capsular polysaccharide
(CPS) determined as described by Arakere and Frasch (7)
with minor modifications. Samples were run in duplicate at 8 serial dilutions,
and antibody concentrations were calculated relative to the standard reference
serum lot CDC 1992 (courtesy of G. Carlone, Centers for Disease Control
and Prevention, Atlanta, GA). B) IgM antibodies to N. meningitidis
lipooligosaccharide (LOS) immunotypes (L2, L3, L7, L9). ELISA to detect
antibodies to LOS immunotypes L2, L3, L7, or L9 was performed as described
(8), with minor modifications, by using goat
antihuman IgG (γ-specific, Kirkegaard & Perry, Gaithersburg,
MD, USA) or goat antihuman IgM (μ-specific, Sigma, St. Louis, MO, USA)
conjugated to alkaline phosphatase. Samples were run in duplicate at 4
serial dilutions. OD, optical density.
|