skip
navigation  |
800-411-3222 (Español)
Cole Crops and Other Brassicas: Organic Production |
| By Martin
Guerena NCAT Agriculture Specialist © 2006 NCAT ATTRA Publication #IP275 |
The
printable PDF version of the entire document is available
at: http://attra.ncat.org/attra-pub/PDF/cole.pdf 20 pages — 841K Download Acrobat Reader |
AbstractCole crops and other brassicas are grown throughout the U.S. These crops are an excellent choice for many organic farmers because of the variety of crops in this family, their nutritional qualities, health benefits, compatibility in planting rotations, and pest-suppressive qualities. This publication covers soils, fertility, planting, irrigation, pest management, harvesting and marketing. Table of Contents
IntroductionBrassicaceae (the mustard family, previously Cruciferae or the crucifers) include many food, forage, ornamental, and weed plants. The brassicas are classified as “cool season,” meaning that they are relatively resistant to frost and light freezes. Throughout the U.S. they are grown in the spring or fall, so that development takes place when temperatures are cool. The northern Midwest, Pacific Northwest, and New England produce brassicas in the summer, while winter production takes place in the Southwest and other Sunbelt states. California is able to produce brassicas year-round because of the moderating effect of the cold Pacific current. Cole crops are a group in Brassicaceae that includes varieties of the species Brassica oleracea such as broccoli, cabbage, cauliflower, and Brussels sprouts. Optimal growing temperatures for most cole crops are 60 to 65°F. (Maynard and Hochmuth, 1997) An important fact to keep in mind is that these plants are closely related and share similar climatic requirements as well as pests and diseases. However, though similar, they are not identical. There may be larger differences between varieties of broccoli than between broccoli and cauliflower. Many varieties from each group have been developed so that they vary in pest susceptibility, temperature tolerances, shape, color, and length of growing season. Check with other farmers in your area or your Extension agent to see which varieties are adapted to your local conditions. The genus and species Brassica oleracea was developed in western and central Europe from wild species found in the Mediterranean region. (Nieuwhof, 1969) Brassica rapa, a similar species developed in Asia, consists of turnips, Chinese cabbage, bok choi, rapini, canola, and the mustards. Other plants in Brassicaceae from other genera are: radishes (Raphanus), watercress (Nasturtium), and horseradish (Armoracia). Nutritionally, brassicas are high in carotenoids, vitamins C and A, calcium, iron, magnesium, and dietary fiber. Broccoli and broccoli-seed sprouts in particular contain high levels of antioxidant sulphoraphanes, which are anticarcinogenic compounds. The sulphoraphanes are also thought to protect eyes from the damage caused by UV light that can lead to macular degeneration (Xiangqun and Talalay, 2004) and to prevent high blood pressure, heart disease, and stroke. (Lingyun et al., 2004) Glucosinolates, chemical precursors to sulphoraphines, form isothiocyanates in the soil. Isothiocyanates are biologically active compounds that are of considerable interest to farmers because of their ability to suppress some insects, diseases, nematodes, and weeds in a process known as biofumigation. Ornamental crops in the mustard family make up about 50 genera, including Arabis, Erysimum (Cheiranthus), Hesperis, Iberis, Lobularia, Lunaria, and Matthiola. (Watson and Dallwitz, 1992) The number of crops in this family, their nutritional qualities, health benefits, compatibility in planting rotations, and pest suppressive qualities make these crops an excellent choice for many organic farmers. Table 1 includes the scientific and common
names of members of the mustard family and
Organic ProductionOrganic production of brassica crops, or any commodity, relies on management techniques that replenish and maintain long-term soil fertility by optimizing the soil's biological activity. This is achieved through crop rotation, cover cropping, composting, and by using organically accepted fertilizer products that feed the soil while providing plants with nutrients. Besides producing high quality crops, a healthy, well-balanced soil can help plants develop natural resistance to insect pests and diseases. When pest controls are needed, organic farmers manage insects, diseases, weeds, and other pests with cultural, mechanical, biological, and—as a last resort—organically accepted biorational and chemical controls. In 2002, the USDA implemented the National Organic Standards regulating organic production nationwide. All farmers and ranchers wishing to market their products as organic must be certified. An exception to this requirement is made for farmers who sell less than $5,000 annually. For more information on organic crop production and organic farm certification, see ATTRA's Organic Crop Production Overview and Organic Farm Certification and the National Organic Program. Soil and FertilityThe brassicas are heavy feeders that can grow on a variety of soils as long as the soils provide adequate nutrients and moisture and are well drained. The soil is where plant health begins and ends. A healthy soil will have a greater capacity to moderate the uptake of fertilizers and will allow a more balanced uptake of nutrients, creating a healthy plant that is less attractive to pests and more resistant to pest damage. Soil components—minerals, air, water, and organic matter—vary widely depending on geography and climate. The challenge on farmland is to maintain healthy soils with adequate levels of organic matter. Healthy soils will demonstrate the following characteristics: good tilth, good habitat for numerous and diverse microorganisms, absorption and retention of water, the ability to buffer salts and pH, an “earthy smell,” resistance to erosion by either wind or water, and production of healthy crops. Organic matter is the soil component primarily responsible for these traits. Organic matter is broken down by soil organisms, creating humus. Humus in turn provides nutrients to crop plants. Sustainable soil management maintains soil health and productivity by taking care of and increasing the soil's organic matter. Cultural practices, such as the application of manures and compost, using cover crops, and rotating crops are methods to achieve this. Healthy soil can be considered a living organism that must be nurtured in order to sustain its life and productivity. Throughout their life cycles, brassica crops require particular nutrients in varying quantities to support optimal growth and reproduction. Nitrogen is the nutritional element that most cultivated crops need in the greatest amounts. Plants use it to form proteins, chlorophyll, protoplasm, and enzymes. In cole crops, it's most important for overall growth, and adequate amounts are necessary for best yields. Usually the initial nitrogen available from green manure or compost is enough, but as the plant develops it may need supplemental nitrogen, and succeeding crops may also need a nitrogen boost. Organic sources of supplemental nitrogen include guano, pelleted compost, fish emulsion, blood meal, feather meal, cottonseed meal, alfalfa meal, and kelp, and they should be applied as soon as the plants are strong enough (usually about 6 inches tall) to withstand the side-dressing operation. The mineralization of nitrogen and its availability to plants varies greatly, depending on the nitrogen source, the temperature, humidity, texture of the material, and microbial activity. In a transitional or newly certified operation, growers should keep records of the materials they used and how the crops responded to them. Once growers learn how each material reacts to specific conditions, and as the soil's organic matter builds, fertility management usually improves. Composted manure and cover crop residues usually provide enough phosphorus for brassicas. If additional phosphorus is needed, rock phosphate may be an option. Potassium (potash) requirements for cole crops are high. Composted manures, composted straw and hay (especially animal bedding), granite dust, material derived from langbeinite, kelp meal, and wood ash (if not contaminated with colored paper, plastic, or other synthetic substances) are acceptable sources of potash. The macronutrients calcium and micronutrients boron, manganese, molybdenum, and iron are important for cole crop development. Biologically active soils with adequate organic matter usually supply enough of these nutrients. Compost and seaweed products are sources of supplemental micronutrients. For more information on soils and fertilizers see the ATTRA publications Sustainable Soil Management, Alternative Soil Amendments, and Sources of Organic Fertilizers and Amendments. Planting and TransplantingMost brassicas are direct-seeded into prepared seedbeds. The optimal time to plant is when soil temperatures are between 65 and 75°F, though some varieties can germinate in soils with temperatures as lowas 45°F and as high as 85°F. (Lorenz and Maynard, 1980) The seedbed should be pre-irrigated or solarized to reduce potential weed problems. Seeding machines such as the ICS vegetable precision planter, Earthway planter, Planet Junior, and Stanhay planters are suitable for both small and larger scale operations and can place seed at any desired space. If the bed is seeded too closely, thinning is necessary to achieve proper spacing. Good quality seed with a high germination percentage is important when establishing a direct-seeded stand. Some crops that have high seed costs, long growing seasons, and special growing requirements, such as cauliflower and Brussels sprouts, are usually transplanted from greenhouses to the field. Crops like cabbage and broccoli can either be direct-seeded or transplanted, depending on conditions such as season and costs. Direct-seeding broccoli during mid-summer for a fall crop is less expensive than using transplants. During late winter, using transplanted broccoli may open a marketing window for spring production that could be economically advantageous. Transplanting can overcome some problems, such as soil crusting and high or low soil temperatures, that can cause uneven seedling emergence. Factors like these should be taken into consideration when choosing the type of plant establishment. Growing transplants requires great experience and skill. Transplants can be purchased commercially and must be certified organic if they are used in a certified organic operation. For more information on transplant production, see the ATTRA publication Plug and Transplant Production for Organic Systems. Advantages in using transplants are uniform stand and quality, efficient use of seed, season extension, reduced weeding costs, reduced irrigation, shortened cropping period in the field, and less exposure to pests. Transplants should be free of pests, weather hardened, and not be long or leggy. Hardening is the process of gradually acclimating young greenhouse plants to the outside environment. Most transplants are hardened two weeks before planting in the field. Transplants should also be well irrigated prior to planting, so that the plants can survive until they are irrigated in the field. Also, transplanting should be done during cool weather and with minimal root disturbance to reduce transplant shock. Transplant shock is the stress every transplant experiences while adjusting to its new environment. In California most cole crops are grown on raised beds, making cultivation and irrigation easier. Broccoli and cabbage are planted in 2 rows per 40-inch bed. Broccoli is spaced at 8 inches and cabbage at 12 inches apart within the row. Cauliflower is usually grown on a single, narrower row (36 to 38 inches), off center along one side. As irrigation water evaporates, salts accumulate on the ridge of the mounded row. The seedlings are planted below the ridge to avoid salt accumulation in the root zone. Depending on the variety, cauliflower can also be grown on 2 rows per 40-inch bed, 12 to 14 inches apart. Irrigation
Soil texture, environmental conditions, and crop age are factors to consider when irrigating any crop. Cole crops are generally shallow-rooted, with roots ranging from 18 to 24 inches long. Some exceptions to this are mustard, rutabaga, and turnips, whose roots range from 36 to 48 inches. (Doneen and MacGillivray, 1943) Chinese cabbage and pak choi have shallow root systems that respond well to light, frequent irrigations. (Larkcom, 1991) Essentially, the art of irrigation is applying the right amount of water to the plants so that they produce an economically viable crop. Too much water is wasteful and can cause problems with diseases and weeds. Too little water causes plants to slow their development, eventually causing stress, pest susceptibility, and lower yields. So, how much is enough? A rule of thumb is that vegetables will need about 1 inch of water per week from rain or supplemental irrigation in order to grow vigorously. In arid regions about 2 inches are required. (Lorenz and Maynard, 1980) Sprinkler irrigation should be used for germinating seeds and establishing transplants. Once the plants are established, furrow or drip irrigation is recommended. Organic Integrated Pest ManagementIntegrated Pest Management (IPM) is a broad ecological approach to pest management using a variety of pest control techniques that target the entire pest complex of a crop ecosystem. Integrated management of pests ensures high-quality agricultural production in a sustainable, environmentally safe, and economically sound manner. (Bajwa and Kogan, 2002) Soil health is based on soil biology, which is responsible for the cycling of nutrients. The complex interactions of this biological community are known as the soil food web. The soil ecosystem is composed of bacteria, fungi, protozoa, nematodes, algae, arthropods (insects and mites), and large soil-dwelling mammals like moles, ground squirrels, and gophers. The photosynthesizers or primary producers in this system use the sun's energy to convert atmospheric carbon into sugars. Other organisms feed off these primary producers. Dead organisms and their byproducts decompose, becoming the soil's organic matter that stores nutrients and energy. Plants use these nutrients, preventing them from accumulating in soil and water. The life cycle of all these organisms improves the condition of soils by enhancing structure, water-infiltration and water-holding capacity, and aeration. This results in healthy plants that are more productive and resistant to pests. Larry Phelan and his colleagues from Ohio State University found that fewer corn borer eggs were laid on corn grown in organic soil. The researchers took soil from an organic farm and from a conventional farm and repeated the experiment in a greenhouse. They treated each soil with organic or chemical fertilizers to determine whether the results were due to short-term nutrient uptake. Again, corn grown in organic soil had fewer eggs on it, regardless of the fertilizer that was applied. Modern agricultural methods are not conducive to maintaining ecological equilibrium because of constant tilling and synthetic inputs to the soil. During their evolution, plants obtained nutrients solely from the soil food web. According to Phelan, it is the slow release of nitrogen in this system that ultimately causes the corn borer to lay fewer eggs. The plants in the conventional system were nutritionally out-of-balance, receiving too much nitrogen. The extra nitrogen formed free amino acids that were not tied up in proteins, stimulating the insects to feed and deposit eggs. Another plant protection phenomena attributed to soil microbial activity is induced and acquired systemic resistance, in which the plant's immune system is stimulated to resist pest attack. In one study, the soil fungus Trichoderma hamatum induced systemic resistance in cucumber against Phytophthora crown rot and leaf blight. (Khan et al., 2004) The concept of healthy soils being responsible for plant health has long been known to organic farmers, and scientists are just starting to document it. IPM is based on the following components: pest identification, monitoring, mechanical and physical controls, cultural controls, biological controls, and chemical controls. For a detailed description of integrated pest management concepts, see the ATTRA publication Biointensive Integrated Pest Management. The biological and cultural insect controls for cole crops involve understanding the ecology of agricultural systems. We invite pest problems by planting large expanses of a single, susceptible crop. When there is a diverse farmscape involving many types of plants and animals, the likelihood of severe insect pest outbreaks diminishes considerably. That is why farmers must create production methods that complement natural systems. The use of beneficial insect habitats along crop field borders increases the presence of beneficial insects. (Grez and Prado, 2000; White et al., 1995; Bugg, 1993) These habitats provide shelter, food (pollen and nectar), and act as refuges that attract pests' natural enemies to fields. When purchased beneficial insects are released, these field-edge habitats will encourage the beneficials to remain and continue their life cycle there, helping to reduce pest populations. Some pests may also inhabit the field-edge habitats; therefore, these habitats should be monitored along with the crop. For additional information, see ATTRA's Farmscaping to Enhance Biological Control. Aphids
The cabbage aphid, Brevicoryne brassicae, is a major pest of cole crops worldwide. It is small (1/8 inch long), dark green, and exudes a gray, waxy secretion. What it lacks in size it makes up in numbers, reaching adulthood in 8 to 12 days and producing 5 to 6 nymphs asexually per day for 30 days. (Hines and Hutchison, 2002) Aphids pierce plants and suck their juices, distorting leaves and growing points. Large colonies infest leaves, heads, and flower stalks, making products unmarketable. Other aphids such as the green peach aphid and turnip aphid will feed on brassicas, but they usually do not cause economic damage. Cabbage aphids have many natural enemies that can be attracted to fields with habitat plantings. These include aphid and syrphid flies, lacewings, and the predaceous midge (all of which produce larvae that consume aphids), minute pirate bugs, bigeyed bugs, lady beetles (the adults and larvae of which both consume aphids), soldier beetles, and parasitic wasps like Diaeretiella rapae. In some humid areas there are outbreaks of naturally existing fungi that cause epidemics among aphid colonies. The insect-consuming fungus Beauveria bassiana, which is sold commercially as a bioinsecticide, is not effective on cabbage aphid due to a fatty-acid secretion produced by the aphid that is likely involved in resistance to the fungus. (Szafranek et al., 2001) Cultural controls that reduce aphid populations include the use of sprinkler irrigation, where water at high pressure dislodges the insects from plants. This practice may work when plants are young and cupping or when inflorescence development has not yet occurred. Broccoli and cabbage plants interplanted in clover used as a living mulch showed a reduction in aphids, compared to plants in clean cultivated fields. (Costello and Altieri, 1994; Theunissen et al., 1995) In the broccoli trial, the clover mulches had to be mowed early in the cropping cycle to give the broccoli plants a growth advantage. Mowing of the cover crop may be limited by lack of labor and/or equipment. The cabbage trial was not mowed and produced smaller but more marketable heads than the clean monocrop. Other interplanting strategies to combat cabbage aphid include the use of mustard or collards as trap crops and the use of different varieties of the same crop in sequential plantings. Cabbage aphids exhibit a preference for certain species and will also discriminate among varieties and plants of varying age. (Altieri and Schmidt, 1987; Kloen and Altieri, 1990) Nitrogen management can have an effect on aphid infestations. Studies in Great Britain showed that Brussels sprouts treated with high nitrogen (3.2 mg/g plant fresh wt.) grew more rapidly than those with low nitrogen (0.64 mg/g plant fresh wt.), but the improved growth with high nitrogen was offset by the increased population of aphids. (Koritas and Garsed, 1984) However, too little nitrogen can also cause stress in plants and make them susceptible to insect attack. Organically accepted insecticides include insecticidal soap, neem, rotenone, and pyrethrum. The waxy leaf cuticle of brassicas and the white, waxy secretions of the cabbage aphid tend to repel water-based insecticides, so a spreader-sticker is recommended. Many growers use soap to overcome this problem. Water hardness will reduce the efficacy of insecticidal soap, because calcium, iron, and magnesium will precipitate the fatty acids and make the soap much less effective against the insects. The best way to determine how well your water will work is to use the soap-jar test. Let a jar full of spray solution sit for 20 minutes, then look for precipitates in the soap-water solution. Caterpillar pests
The cabbage looper (Trichoplusia ni), diamondback moth (Plutella xylostella), and imported cabbage worm (Pieris rapae) are the major caterpillar pests of cole crops. Other caterpillar pests can be regional or seasonal problems, like armyworms, cutworms, cabbage webworms, corn earworms, cross striped cabbageworms, gulf white cabbageworms, and southern cabbageworms. Caterpillars have many natural enemies that help keep their populations down. Predators such as ground beetles, spiders, damsel bugs, minute pirate bugs, assassin bugs, bigeyed bugs, and lacewing larvae attack caterpillars. The parasitic wasps Trichogramma spp., Copidosoma spp., Apanteles spp., Diadegma spp., and Hyposoter spp. sting and parasitize eggs and larvae. Some of these organisms are available commercially, or they may occur naturally in the environment. For information on suppliers of beneficial insects, contact your local Extension office or visit the Suppliers of Beneficial Organisms in North America Web site: www.cdpr.ca.gov/docs/ipminov/ben_supp/ben_sup2.htm.
Biopesticides or microbial controls consist of Bacillus thuringiensis (Bt), insect-consuming fungi, and viruses. Bt is a naturally occurring bacterium that produces a toxin that causes paralysis of a caterpillar's digestive tract. A caterpillar may continue to live for some hours after ingestion, but will not continue to feed. Bt strains are available in a number of commercial products, under various trade names. The following products have been approved for organic production by the Organic Materials Review Institute (OMRI): Prolong, from Cillus Technology Inc.; Britz BT Dust, from Britz Fertilizers Inc.; DiPel™ and Xantari™, from Valent Biosciences; Agree™, Deliver™, and Javelin™, from Certis USA.
Bt degrades rapidly in sunlight and requires careful timing or repeated applications. Bt must be ingested in sufficient amounts by the caterpillar to be effective. Consequently, growers must understand the feeding habits of the pests, so that proper formulations are used and timing of applications is optimal. Caterpillars in their early stages of development (first and second instars) are more susceptible to this toxin. Older and bigger worms are harder to kill. Entrust™ from Dow Agrosciences is derived from the soil organism, Saccharopolyspora spinosa. It is OMRI-approved and registered for control of armyworm, corn earworm, diamondback moth, imported cabbageworm, and loopers on cole crops.
Spod-X LC™ and Gemstar LC™ from Certis USA are nuclear polyhedrosis virus products available commercially and are OMRI-approved for the control of armyworm and corn earworm, respectively, on cabbage, cauliflower, and broccoli. Other naturally occurring granulosis viruses and nuclear polyhedrosis viruses sometimes occur in high density caterpillar populations.
Beauveria bassiana, the insect-eating fungus, will infect caterpillars if humidity and temperature are adequate. Commercial products include Naturalis L™, Mycotrol™, and Botanigard™. Botanical insecticides include neem products (Agroneem™ and Neemix™) that act as repellents, antifeedants, and insect growth regulators. Pyrethrin and rotenone-based products are broad spectrum and will kill beneficial insects as well as pests, so monitoring is important. Beneficial insect populations must also be considered when a pest population is present. Many times the beneficial population may be keeping the pest under the economic threshold, which is the level below economic injury to the crop. An application of a broad spectrum insecticide may damage both the pest and beneficial insect populations, and other minor pests may become a big problem. This is known as a secondary pest outbreak.
Other management practices to reduce caterpillar infestation include using floating row overs over a young crop to exclude egg-laying females, nocturnal overhead sprinkler irrigation, pheromone misters or emitters to disrupt mating, and pepper, garlic, and herbal repellents. Some of the control methods mentioned for cabbage looper, diamondback moth, and imported cabbage worm may work on armyworms, cutworms, cabbage webworm, corn earworm, cross striped cabbageworm, gulf white cabbageworm, and southern cabbageworm. If the problem is severe, contact your local farm advisor or the ATTRA project. Cabbage maggotThe cabbage maggot or cabbage fly (Delia redicum) will lay eggs in clusters near the stems of many cole crops or in the debris of a previous cole crop. Once hatched, the larvae bury themselves and start consuming feeder roots, eventually burrowing into the tap root. This provides entry sites for pathogens like clubroot (Plasmodiaphora brassicae). Maggots feed for three to five weeks, then pupate in the roots or in the surrounding soil. (Anon., 2003a) In Chinese cabbage, eggs laid on the surface of a maturing head hatch maggots that burrow into the head, making it unmarketable. Cabbage flies will have three to four generations per year starting in the spring through the early autumn. (Anon., 1998) A study in Denmark demonstrated the susceptibility of cabbage maggot
and pupae to Sternernema nematodes. (Neilsen,
2003) These insect-eating nematodes are available
Compost and straw mulches significantly reduce the population of root maggots infesting broccoli. The mulch acts as a barrier, preventing the flies from laying eggs directly in the soil. It also serves as a habitat for ground beetle and rove beetle that parasitize and prey on the cabbage maggot. (Prasad and Henderson, 2002) Other predators include spiders, harvestmen or daddy longlegs, and ants. Floating row covers will prevent cabbage flies from depositing eggs during the critical period after plant emergence or transplanting and will also reduce egg-laying on mature Chinese cabbage. Intercropping clovers or other legumes or letting non-brassica weeds fill in the spaces between crop rows, will keep root flies from finding open ground near a brassica stem. An experiment in England demonstrated that carboxylic acids (oxalic acid found in rhubarb, acetic acid, or vinegar) are potent inhibitors of egg laying by the cabbage fly. (Jones and Finch, 1989) Thus, a solution of crushed rhubarb leaves or a vinegar solution sprayed periodically around cole crop plants may deter the cabbage maggot. Symptoms of flea-beetle feeding are small, rounded, irregular holes. Heavy feeding makes leaves look as if they have been peppered with fine shot. Further damage may be done by the larvae, which feed on plant roots.
DiseasesDiseases in plants occur when a pathogen is present, the host is susceptible, and the environment is favorable for the disease to develop. Altering any one of these three factors may prevent the disease from occurring. Organisms responsible for plant diseases include fungi, bacteria, nematodes, and viruses. If these organisms are present, then manipulation of the environment and the host, to make it less susceptible, helps to more sustainably manage diseases on cole crops. Once again, soil health and management are the key for successful control of plant disease. A soil with adequate organic matter can house uncountable numbers of organisms such as bacteria, fungi, nematodes, protozoa, arthropods, and earthworms that deter harmful fungi, bacteria, nematodes, and arthropods from attacking plants. These beneficial organisms also help create a healthy plant that is able to resist pest attack. For more information, see the ATTRA publication Sustainable Management of Soil-Borne Plant Diseases. The leaf surface can also host beneficial organisms that compete with pathogens for space. A disease spore landing on a leaf has to find a suitable niche in order for it to germinate, penetrate, and infect the plant. The more beneficial organisms there are on the leaf, the harder it is for the disease spore to find its niche. Applying compost teas adds beneficial microorganisms to the leaf, making it more difficult for diseases to establish themselves. For more information on foliar disease controls, see the ATTRA publications Notes on Compost Teas, Use of Baking Soda as a Fungicide, Organic Alternatives for Late Blight Control on Potatoes, and Downy Mildew Control on Cucurbits. Clubroot
Clubroot is caused by the fungus Plasmodiophora brassicae. It infects cole crops through the root hairs or through wounds on larger roots. As the fungus spreads it distorts and disfigures the roots, causing them to swell and crack, allowing secondary organisms to invade and aid in decay. The disease is favored by acid soils; therefore, liming is recommended if the soil pH is lower than 7.2. (Anon., 2003b) Other methods to control clubroot include rotating out of cole crops for a couple of years, having good drainage, and controlling brassica-type weeds. Cole crops vary in their susceptibility to clubroot, with cabbage, Chinese cabbage, Brussels sprouts, and some turnips being very susceptible. Broccoli, cauliflower, collards, kale, kohlrabi, and some radishes have medium susceptibility. Garden cress, mustard, and some turnips and radishes are resistant. (Averre, 2000) Black leg
The fungus Phoma lingam causes black leg of cole crops. The fungus causes yellow to tan spots with black specks to form on leaves, and stem cankers form usually below the soil line. The fungus interferes with water conduction in tissues, wilting and debilitating plants. Seedlings can be killed, and surviving plants may be stunted. The disease can come in with the seed or be present on cole crop debris or brassica-type weeds. Controls include the use of clean, certified or hot-water treated seed, good soil drainage, rotation with non-brassica type crops, control of brassica-type weeds, deep incorporation of cole crop residues, and planting resistant varieties. To avoid blackleg, it is best to avoid planting near other cole crops or near fields that harbored cole crops during the past season. Fusarium yellowsFusarium yellows are caused by the soilborne fungus Fusarium oxysporum f. conglutinans. Symptoms are yellowing leaves, usually more pronounced on one side of the plant, the loss of lower leaves, curvature of petioles and midribs, and wilting. The ideal temperature range for the development of this disease is 75 to 85°F., with 60°F. being the lower limit (Anon., 1987), so it is observed from mid-spring through summer and is not a problem in early plantings. The fungus is persistent in the soil and has many plant hosts, so resistant varieties, good drainage, and soil-building practices such as cover crops and compost are recommended. Sclerotinia white rotThe fungi Sclerotinia sclerotiorum and Sclerotinia minor both can cause this rot favored by cool, wet conditions. The fungi have many hosts, including many commercial crops and cover crops that fit in a rotation with cole crops, though grasses are not affected. Good drainage and irrigation practices that reduce humidity in fields can reduce the disease. Deep plowing is often recommended, but the results are temporary and very disruptive to soil microorganisms. Biological controls include the fungus Coniothyrium minitans, which attacks sclerotia. Coniothyrium is available commercially in the product Contans™, from Sylvian Bioproducts, Inc. Black rot
Black rot is caused by the bacterium Xanthomonas campestris. This bacterium favors humid, rainy conditions, and is dispersed by the splashing of droplets of water. Xanthomonas enters the plant at leaf margins or through wounds. Leaf margins develop yellowish patches that turn brown with black veins. The infection works its way down the leaves, leaving a “V” pattern in its wake. The pathogen may eventually invade the vascular system, spreading throughout the plant. Controls include rotation, weed control, thorough debris incorporation, the use of clean seed, and application of approved copper products. Compost tea was successfully used in a study in the Willamette Valley of Oregon to suppress carrot bacterial leaf blight, Xanthomonas campestris pv. carotae. (Reinten and Salter, 2002) Downy mildew
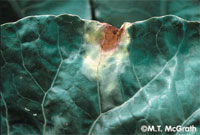
Downy mildew is a disease caused by the fungus Peronospora parasitica. Infection and development are favored by cool, wet weather, and the fungus attacks cole crops at all stages of growth. Once Peronospora invades a plant, it consumes the contents of the plant's cells and then sporulates, sending sporangia out to form cottony white masses, usually under leaves. The tops of leaves develop purplish spots that later turn yellow or brown. These spots correspond to the sporulating areas on the undersurfaces of the leaves. Infected young seedlings may die, while cauliflower curd, broccoli florets, radish roots, and cabbage heads may all become unmarketable. Management includes promoting good drainage, increasing spacing for better aeration, controlling brassica-type weeds, using resistant varieties, rotating with non-cole crops, incorporating plant debris, and avoiding the use of overhead irrigation. Alternaria leaf spot
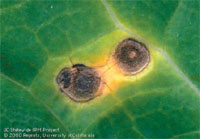
This disease is caused by the fungi Alternaria brassicae and/or A. brassicola. Small dark spots initially form on leaves, but later develop into tan spots with target-like concentric rings. When dried, these spots fall from the leaves, resulting in a “shot-hole” effect. Prolonged periods of high humidity, cool temperature, and rain favor its development. Infected cabbage heads will have spots on several leaves, and Brussels sprout buds will have several layers infected. It discolors broccoli and cauliflower heads, and Chinese cabbages are more susceptible than other cole crops. Management practices include using clean, certified seed, rotating with non-host crops, deeply incorporating plant debris, avoiding overhead irrigation, and promoting air circulation in the canopy. Physiological DisordersTipburn is the browning of internal leaf edges or tips within the heads of cabbage, Brussels sprouts, and cauliflower. These brown spots tend to break down during storage or transport, allowing secondary organisms to decay the product. The problem is related to rapid growth caused by excessive nitrogen, high temperature, water stress, and calcium deficiency. Calcium can be present in the soil but its translocation to the plant is limited, and it may not be available to accommodate rapid growth. Supplemental nitrogen applications should be timed to avoid rapid growth in the later stages of plant development. Riceyness of cauliflower causes the curds to become uneven and fuzzy, reducing marketability. Warm temperatures (> 68º F) during curd development are the cause of this disorder. Some newer hybrids can develop heads at 68 to 80ºF. (Dianello, 2003) Hollow stem in broccoli and other cole crops is caused by rapid growth, usually due to excessive nitrogen levels and high temperatures. The plant stem experiences rapid growth, and the core or pith cracks, leaving the stem hollow. Another factor that contributes to this disorder is plant spacing. The closer the plant spacing, the less likely this phenomenon will occur. Buttoning of broccoli and cauliflower occurs when immature
plants are exposed to consistently Bolting is caused by many factors and depends on the crop and the varieties grown. Stress caused by too much or too little water, transplant shock, day lengths of more than 12 hours, and low temperatures during the early stages of development are all contributing factors. WeedsThere are many weeds in Brassicaceae that are troublesome in cole crop plantings, because they compete for water, nutrients, and light, and they harbor insect and diseases that can affect the crop. Table 2 lists many of these weeds and includes both their common and scientific names. Weed control in organic systems, especially in vegetable production, relies heavily on crop rotations, cover crops, and cultivation. Of these, cultivation is the most critical to reduce weeds in an established cole crop stand. For cultivation to be successful, a straight, well-made bed, as well as straight seeding or transplant lines, is necessary in order for cultivating implements to remove most weeds while leaving the crop undisturbed. Cultivation implements will cut, bury, or turn over most young weeds, leaving the crop undisturbed and with reduced competition. In California, it usually takes two cultivations before a young cole crop starts to out-compete weeds. Hand hoeing may be necessary after the first cultivation to reduce weeds in the plant line. If the crop is direct-seeded, weeding and thinning take place after the first cultivation. For more information on weed control, check ATTRA's publications Principles of Sustainable Weed Management for Croplands, Alternative Control of Johnsongrass, Thistle Control Alternatives, and Field Bindweed Control Alternatives.
HarvestingCabbages are harvested when the heads are firm and solid. Sizes may vary, but firmness is the determining factor. If solid heads are left too long in the field to size up, they may crack or split. Cabbages should be sorted, packed, and stored according to size. Cauliflower heads with white firm curds 6 to 8 inches in diameter are what consumers prefer. Blanching or tying the outer leaves is done with certain varieties when the heads are about 3 to 4 inches in diameter, to keep sunlight from yellowing the curds. Curds should be handled carefully since they bruise easily and will develop discolored patches on these bruises. The surrounding trimmed leaves should be kept on the head for handling purposes to protect the curd. Some operations field-wrap the trimmed curds in cellophane or plastic bags before cooling and storing them in refrigeration. Broccoli is harvested when most heads are tight, 5 to 7 inches in diameter, and of a blue-green color. They are cut with a 6-inch stem. If left in the field, heads tend to loosen and expand, reducing quality. Fields should be harvested every three days, due to the rapid growth of this crop. Once the head is harvested, side shoots may develop from lateral axils on the stem, producing smaller inflorescences that may also be marketed. Brussels sprouts produce many small buds in leaf axils along the entire stem. The lowest sprouts on the stem are picked first, along with the leaves, and harvest progresses upward as the other sprouts mature. Sprouts may crack if left on the stem too long. Kale and collards are harvested leaves with petioles and are usually bunched together with a wire tie. Bunches are typically about 8 to 14 inches long. Kohlrabi should be harvested when the swollen stem is 2 to 3 inches in diameter. There are green and purple varieties. If left in the field for too long the swollen stem becomes woody. Chinese cabbage heads should feel solid and not collapse when pressed firmly with both hands. The wrapper leaves surrounding the cabbage should be stripped to leave only a couple to cover the firm head. Pak choi can be harvested a few leaves at a time, cutting the outer leaves when they reach a desired size, or by harvesting the whole head. Some varieties are very brittle and should be left to wilt slightly to avoid breaking before packing. PostharvestOnce a cole crop is harvested it continues its biological processes until it deteriorates to an unsaleable product. The key to successful postharvest handling is to delay these processes in order to get the product to the consumer in the best condition possible. Temperature is the most important factor affecting harvested produce. It directly affects the rates of all vital processes: respiration, ripening, moisture loss, and the development and spread of decaying organisms. The higher the temperature, the faster these processes occur. Therefore, proper temperature management is important throughout the supply chain, from harvesting to consumption. The sooner the field heat is removed from the product, the longer it will last, giving the producer more time to sell the product. Cooling methods vary according to the commodity; the most common are cold rooms, forced air cooling, hydrocooling, and icing. Cold rooms involve placing the product in containers in a cold room. The less field heat accumulated in the product, the quicker this system will cool the product. Forced air cooling is done in a cold room and requires containers with vents so that the cold air can flow through. The containers are stacked in rows placed on either side of a fan, leaving an aisle between the rows. The aisle and the open end are covered with a tarp to create a tunnel. The fan draws air from outside the tunnel through the openings in the containers, forcing cold air around the warm product. Hydrocooling uses cold water to rapidly cool commodities. Containers must be waterproof and allow water to enter to pass over product. Cartons or lugs are either submerged in cold water or showered from above with recirculated cold water. Icing consists of placing ice on the product in the container. This is usually done with broccoli, Brussels sprouts, and turnips. All brassica crops last the longest when stored at 32ºF, just above the freezing point. (Hardenburg et al., 1986) See ATTRA's publication Postharvest Handling of Fruits and Vegetables for more information. Economics and MarketingThe economics of growing cole crops vary enormously from crop to crop and in different regions. The following budgets were developed for growing organic cauliflower in the Northeast and organic broccoli in California. The organic cauliflower budget was prepared by Rutgers Cooperative Extension in cooperation with the Northeast Farm Management Committee in 1996. The organic broccoli budget was developed by the University of California Cooperative Extension in 2004. These budgets are intended as references in order for farmers to develop their own budgets based on local conditions. Large and some medium producers market their products to wholesalers, brokers, and terminal markets. These marketing options are not advantageous for some medium and smaller growers, because of low returns, uncertainty of prices, risk of rejection, and strict packing standards.
Alternative markets exist for smaller producers, such as marketing directly to consumers through farmers' markets, community supported agriculture (CSA), and roadside stands. Direct sales to restaurants and small, independent grocers are other choices. For more information on alternative marketing, see the following ATTRA publications: Selling to Restaurants, Community Supported Agriculture, Direct Marketing, Evaluating a Rural Enterprise, Farmers' Markets, Green Markets for Farm Products, and Organic Marketing Resources. SummaryThe number of crops in the brassica family, their nutritional qualities, health benefits, compatibility in planting rotations, and pest suppressive qualities make these crops an excellent choice for any organic farmers. They grow in all regions in different seasons and add diversity to a farmer's income and products. AcknowledgmentThe author wishes to thank Wyatt Brown, PhD, of the Horticulture and Crop Sciences Department at Cal Poly San Luis Obispo, for his thoughtful and thorough review of this publication. ReferencesAltieri, M.A., and L. L. Schmidt.
1987. Mixing broccoli cultivars reduces cabbage aphid numbers. California
Agriculture. Nov-Dec. p 24-26. Bugg, Robert. 1993. Habitat manipulation
to enhance the effectiveness of aphidophagous hover flies (Diptera:
Syrphidae). Sustainable Agriculture Technical Reviews. Vol. 5, No.
2. p.13-15.
Cole Crops and Other Brassicas: Organic Production
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Site Map | Comments | Disclaimer | Privacy Policy | Webmaster Copyright © NCAT 1997-2009. All Rights Reserved. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||






.jpg)

.jpg)