|
|
|
|||||||||||

|
|||||||||||||||||||||||||||||||||||||||||||||
Guide to the Application of Genotyping to Tuberculosis Prevention and ControlReturn to Genotyping Main MenuApplying Genotyping Results to Tuberculosis
|
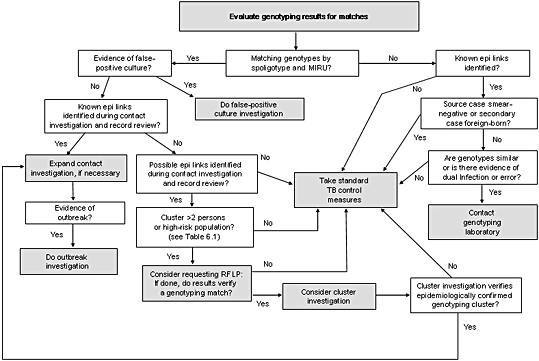
Summing Up: False-Positive CulturesSuspect that a genotyping match might represent a false-positive culture if any of the following are true:
|
Action Steps for Genotyping Clusters with Known Epidemiologic Links Identified
Known epidemiologic links (see Chapter 4, Combining Genotyping and Epidemiologic Data to Improve Our Understanding of Tuberculosis Transmission, for definitions of epidemiologic links) between at least some of the members of the same genotyping cluster suggest that they are part of the same chain of recent TB transmission. The evidence in favor of recent transmission is strong enough at this point in the decision analysis that it is usually not necessary to collect additional data by requesting RFLP analyses of the isolates or by conducting a cluster investigation for the purpose of deciding on appropriate action (see Chapter 5, Developing a Tuberculosis Genotyping Program, for exceptions to this rule).
Two possible action steps are described in this situation in Figure 6.1: a) expanding the contact investigations or b) initiating (or expanding) an outbreak investigation. The decision about which step to take is based on what is known about the cluster from previous investigations and whether the cluster of cases has grown to be an outbreak.
Deciding when an epidemiologically conformed genotyping cluster has become an outbreak is always a challenge. The following text lists characteristics of outbreaks that may help in this decision-making process; an outbreak is likely occurring if any one of the following criteria is met.
Three criteria for defining an outbreak
1. An increase in the expected number of TB cases.
Although this is a standard epidemiologic definition, it is at times a challenge to apply, since the expected number of TB cases in a specific setting is often difficult to know. This criterion is more useful if it is applied to specific subgroups of persons who share demographic characteristics or specific exposures rather than to the population as a whole.
For example, two unrelated children with TB who go to the same school and are in the same classroom would arguably meet the “unexpected increase” criterion for an outbreak, since under normal classroom circumstances the expected number of children with TB would be zero. Two homeless men with TB in the same city, on the other hand, would probably not meet this criterion, since TB in homeless men is more common than in school-age children.
Universal genotyping will help considerably in identifying outbreaks, since it will help to provide an answer not only to the question “is there an increase in the number of cases in a subgroup?” but also to the question “is there an increase in cases that belong to a specific genotyping cluster that involves recent transmission?” Another helpful aspect of having genotyping results available is that they will help define the scope of an outbreak, since genotyping results can identify persons with genotyping links to outbreak cases where no epidemiologic links were known.
2. Transmission continues despite adequate control efforts by the TB program.
For example, two TB patients who work at the same job might not be considered an outbreak. If each was well investigated, including thorough contact investigations with appropriate screening and treatment for LTBI, there might be little else to do. If, however, an additional person with TB appeared a month later with an isolate that was a genotype match to the isolates from the other two persons, this might meet the criterion of ongoing transmission despite an apparently adequate TB-program response.
3. The contact investigation has grown to a size that requires additional outside help.
This often happens when a TB program is devoting most of its resources to conducting a large contact investigation, but the demand for resources continues to increase to the point that the program cannot meet its routine obligations for basic TB control practices. At this point, declaring the situation to be an outbreak can lead to obtaining additional resources to conduct an outbreak investigation from the state program or from CDC. If the decision is made to conduct an expanded contact investigation or an outbreak investigation, see the respective sections for guidance about how to proceed.
Genotyping Clusters with Possible Epidemiologic Links Identified
The next decision point shown in Figure 6.1 focuses on genotyping clusters where known epidemiologic links have not been established. The next question to consider is whether previously collected data from contact investigations and record reviews show that persons in a genotyping cluster share possible epidemiologic links. If possible epidemiologic links are identified (e.g., the TB patients live in the same neighborhood or were homeless) (see Chapter 4, Combining Genotyping and Epidemiologic Data to Improve Our Understanding of Tuberculosis Transmission, for a definition of possible epidemiologic links), the patients might be involved in the same chain of recent transmission and further investigation might be helpful, especially if the genotyping cluster has more than two patients or if the patients are considered high-risk. If the RFLP results show that the presumed cluster is, in fact, made up of genetically distinct isolates (i.e., the isolates have non-matching RFLP patterns), there is no need to conduct a cluster investigation.
If, on the other hand, the RFLP results confirm that the isolates belong to the same PCR/RFLP cluster (i.e., if the RFLP patterns match), the TB program should consider conducting a cluster investigation
Whether to Launch a Cluster Investigation
Cluster investigations should be launched only when needed, since they can be labor intensive. If RFLP results confirm that the isolates belong to a single PCR/RFLP cluster, it is helpful to compare the characteristics of the cluster with the prioritization scheme described in Table 6.1. This prioritized list should not be interpreted as providing absolute instructions about when to conduct a cluster investigation and when not to; rather, it provides helpful guidance about when a cluster investigation is needed and when it might be wise to wait to see if additional TB patients are identified as belonging to the cluster. The information that this prioritization scheme is based on comes from the NTGSN study, and parts of it may have to be updated when our experience with the PCR genotyping methods grows.
Most commonly, clusters involve only two persons. One way to save valuable TB program resources is to investigate only clusters that involve at least three persons. Some programs will want to investigate clusters of only two persons; if resources are available to do so, this aggressive approach will identify episodes of recent transmission sooner. If resources are scarce, however, conducting investigations only of clusters with at least three persons is a reasonable policy. In fact, if resources are really scarce, conducting a cluster investigation only when the cluster grows to four persons may have to be adopted as an interim policy until more resources are identified.
The decision as to what cluster size should be investigated is influenced by whether the cluster contains high-risk persons. “High-risk” in this setting refers to a) characteristics of the persons in the cluster that might make them particularly infectious (e.g., having cavities on chest radiographs), b) characteristics of the M. tuberculosis strain that make it particularly dangerous (e.g., a multidrug-resistant strain), or c) characteristics of persons in the cluster that, if shared by their exposed contacts, would increase the risk of progression from LTBI to active TB (e.g., HIV infection or other immunocompromising conditions) or would increase transmission among a group (e.g., jail inmates, nursing home residents, the homeless). A TB program that would otherwise decide to conduct investigations only of clusters containing at least three persons might decide to conduct an investigation of a two-person cluster if one or the other persons with TB was considered “high-risk.”
Additional information that comes from future genotyping laboratory reports may tip the balance in favor of conducting an investigation. If one laboratory report identifies a two-person cluster, the TB program might decide not to begin a cluster investigation if neither of the two persons is “high-risk.” If the next laboratory report identifies a third person with a matching genotype, the TB program will probably want to initiate a cluster investigation of all involved persons. In general, the decision whether to launch a cluster investigation is a dynamic process, and a decision at an early stage not to do one should not inhibit a TB program from changing its mind should new information become available. If the decision is made to conduct a cluster investigation, details on how to do so are provided later in this chapter in the section titled, Cluster Investigations.
Table 6.1. Prioritizing genotyping cluster investigations.
| Priority (from high to low) |
Type of cluster | Rationale for priority |
|---|---|---|
| |
Suspected false-positive culture | Need to determine which patients do not have TB and stop treatment |
| Cluster of three or more high-risk* patients with possible epidemiologic links | Need to confirm or exclude recent transmission in large clusters of high-risk* patients | |
| Cluster of two high-risk* TB patients with possible epidemiologic links | Smaller clusters less likely to yield epidemiologic links, but presence of high-risk patients deserves attention | |
| Cluster of three or more low-risk TB patients with possible epidemiologic links | Investigation of low-risk patients less urgent than high-risk* patients, but larger clusters may deserve attention | |
| Cluster of two low-risk TB patients with possible epidemiologic links | Investigation of smaller clusters of low-risk
patients often does not yield helpful information Investigations can, however, provide data for monitoring program performance |
|
| Cluster of high-risk* TB patients who have not been found to have even possible epidemiologic links | Low yield for establishing new epidemiologic linksInvestigations can, however, provide data for monitoring program performance Reserved for programs with sufficient resources | |
| Cluster of low-risk TB patients who have not been found to have even possible epidemiologic links | Very low yield for establishing new epidemiologic links Investigations can, however, provide data for monitoring program performanceReserved for programs with sufficient resources |
* "High risk" is defined as patients living in congregate settings (e.g., correctional institutions and nursing homes), persons infected with HIV or having other immunocompromising conditions, children, patients with cavities on chest radiographs or with MDR TB, and the homeless.
Genotyping Clusters with No Epidemiologic Links
Not all genotyping clusters represent recent transmission, and conducting a cluster investigation when the chance of gaining new information is slim is often not a wise investment of resources. If information from adequately conducted contact investigations and case interviews does not reveal even possible epidemiologic links between patients in a genotyping cluster, it is probably sufficient for TB programs to simply ensure that standard TB control measures are completed, such as ensuring that all cases are completely treated and all infected contacts are identified and treated appropriately. Of course, if future genotyping matches are identified and new patients are added to a genotyping cluster, new epidemiologic links within the cluster may become apparent, and at that point the TB program may need to initiate a cluster or an outbreak investigation.
Last Reviewed: 05/18/2008
Content Source: Division of Tuberculosis Elimination
National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention
If you would like to order any of the DTBE publications please visit the online order form.
You will need Adobe Acrobat™ Reader v5.0 or higher to read pages that are in PDF format. Download the Adobe Acrobat™ Reader.
If you have difficulty accessing any material on the DTBE Web site because of a disability, please contact us in writing or via telephone and we will work with you to make the information available.
Division of Tuberculosis Elimination
Attn: Content Manager, DTBE Web site
Centers for Disease Control and Prevention
1600 Clifton Rd., NE Mailstop E-10
Atlanta, GA 30333
CDC-INFO at (1-800) 232-4636
TTY: 1 (888) 232-6348
E-mail: cdcinfo@cdc.gov
![]() Home | Site Map
| Contact Us
Home | Site Map
| Contact Us
Accessibility
| Privacy Policy Notice |
FOIA
| USA.gov
CDC Home |
Search |
Health Topics A-Z
Centers for Disease Control & Prevention
National Center for HIV/AIDS,
Viral Hepatitis, STD, and TB Prevention
Division of Tuberculosis Elimination
Please send comments/suggestions/requests to: CDCINFO@cdc.gov