|
|
|
|||||||||||

|
|||||||||||||||||||||||||||||||||||||||||||||
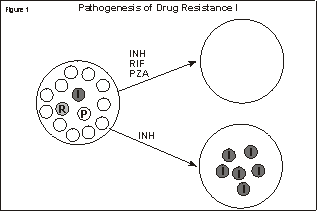
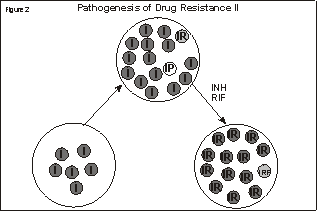
Multidrug-Resistant TuberculosisReturn to DTBE Publications Page This is an archived document. The links and content are no longer being updated. DTBE publications can be ordered using the online ordering system. Multidrug-Resistant Tuberculosis, 1994 Introduction Background In the United States, approximately 20% of deaths in the early 1800s were attributed to tuberculosis; this figure had decreased to 11% by midcentury. Mortality declined throughout the century probably because of improvements in living conditions and nutrition. The realization in the late 1800s that tuberculosis was infectious eventually led to the isolation of tuberculosis patients in treatment centers, or sanatoria. Treatment in sanatoria consisted of bed rest, enhanced nutrition, and various surgical procedures aimed at closing lung cavities. The discovery of antituberculosis medications in the mid–20th century further reduced tuberculosis incidence and mortality and rendered surgical resection unnecessary. Soon after antituberculosis medications were introduced, researchers recognized that tuberculosis was unique with respect to the frequency and importance of the emergence of drug resistance during therapy. They discovered that drug-resistant mutants existed in wild strains of Mycobacterium tuberculosis, that selection of these mutants could occur under certain treatment conditions, and that drug-resistant tuberculosis was more difficult to cure. Once additional drugs were available, controlled trials were conducted and demonstrated that combined regimens were more efficient than single-drug regimens in treating tuberculosis and preventing the emergence of drug resistance. Transmission and Pathogenesis of Drug-Resistant TuberculosisTransmission Pathogenesis Drug-resistant mutants are selected when therapy is inadequate, for example, when a single drug is used to treat a large population of bacilli. The treatment of a wild-type strain of M. tuberculosis with a single drug kills the majority of the bacilli in the population, but the small number of mutants resistant to the drug continue to multiply (Figure 1). After 2 weeks to several months of treatment with the single drug, the resistant bacilli will outgrow the susceptible bacilli, causing clinical drug resistance. Furthermore, in a large population of resistant mutants, additional mutations can occur resulting in doubly-resistant mutants (Figure 2). Click on image for more detailFigures 1 and 2. Empty circles represent tubercle bacilli susceptible to all antituberculosis drugs. A circle with a letter in it represents a tubercle bacillus with resistance to the drug indicated by the letter: I = isoniazid resistance; R = rifampin resistance; P = pyrazinamide resistance. Adapted with permission from Reichman LB: A looming public health nightmare. Lungs at Work 1992. Copyright © 1992 American Lung Association.
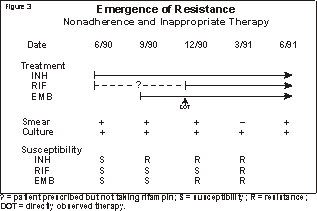
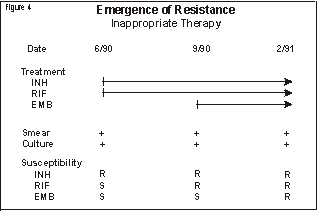
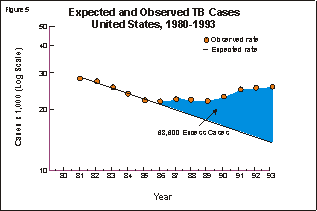
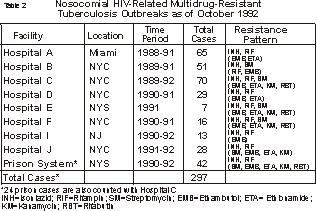
Click on image for more detailIn contrast, the emergence of drug resistance is far less likely in most types of extrapulmonary tuberculosis, in which the bacillary population is much smaller. The bacillary population is even smaller in latent tuberculosis infection, so the chances that drug resistance will emerge during preventive therapy are negligible. Drug resistance is divided into two types: primary resistance and secondary (or acquired) resistance. Primary resistance occurs in persons who do not have a history of previous treatment; these persons are initially infected with resistant organisms. Secondary resistance occurs during therapy for tuberculosis, either because the patient was treated with an inadequate regimen or because the patient did not take the prescribed regimen appropriately. Treatment with two or more drugs in combination can prevent the emergence of drug resistance, if the regimen includes at least two drugs to which the organisms demonstrate in vitro susceptibility. Multidrug resistance is defined as in vitro resistance of a strain of M. tuberculosis to two or more of the antituberculosis drugs. Clinically, the most important pattern of multidrug resistance is resistance to both isoniazid and rifampin. This combination has been associated with an overall cure rate of less than 60% in some reports, similar to the rate of cure before the discovery of antituberculosis drugs (4,5). The two main causes of drug resistance are nonadherence to therapy and the use of inadequate treatment regimens. When medications are not taken as prescribed, the infecting bacilli may be exposed to a single drug for long periods of time, which allows drug-resistant organisms to emerge (Figure 3). Click on image for more detailIn addition, some regimens may contain multiple drugs but only one drug to which the infecting bacilli are susceptible. This can happen when primary drug resistance is not suspected or when a single drug is added to a failing regimen (Figure 4). These regimens are equivalent to single-drug therapy, and they can select multidrug-resistant organisms. Acquired multidrug resistance usually results from a combination of nonadherence and inappropriate therapy. Click on image for more detailEpidemiologyTuberculosis in the United States Click on image for more detailDrug-resistant tuberculosis in the United States In a second survey of city and state laboratories that was conducted between 1975 and 1982, 6.9% of the cases involved resistance to one or more drugs, and 2.3% of cases involved resistance to two or more drugs (9-11). The third survey, conducted between 1982 and 1986, found that 9% of strains isolated in 31 health department laboratories were resistant to one or more drugs (12). The difference in primary resistance rates in the three surveys may be due to methodological differences; however, each survey showed a rate of primary drug resistance that was relatively low and that was stable or decreasing during the study period. Drug resistance did not appear to be an increasing problem, and the surveys were discontinued. Prompted by outbreaks of multidrug-resistant tuberculosis, CDC conducted an additional survey of drug resistance among tuberculosis cases reported from January through March 1991 (13). CDC then conducted a second survey of cases reported in the first quarter of 1992, and compared the findings from that survey with the findings from the 1991 survey (CDC, unpublished data, 1995). The proportion of culture-positive patients with drug susceptibility test results increased from 81.8% in 1991 to 84.1% in 1992. In 1991 resistance to at least one drug was found in 13.4% of new cases, 26.6% of recurrent cases, and 14.2% of cases overall; in comparison, in 1992 resistance to at least one drug was found in 12.8% of new cases, 19.4% of recurrent cases, and 13.1% of all cases. Resistance to both isoniazid and rifampin was reported in 3.2% of new cases, 6.9% of recurrent cases, and 3.5% of all cases in 1991, and was reported in 3.4% of new cases, 3.7% of recurrent cases, and 3.3% of all cases in 1992. The findings of these two surveys may not be comparable to the findings of the previous surveys because of significant methodological differences. In the previous surveys, susceptibility testing was performed at a single laboratory on isolates from only a sample of areas. In contrast, in the 1991 and 1992 surveys, susceptibility tests were performed at local laboratories, and researchers attempted to collect results from all areas for all of the cases reported during the study period. Furthermore, in the previous surveys, attempts were made to determine whether "new" cases had a history of previous treatment and if so, to exclude them from the study, whereas in the 1991 and 1992 surveys, classification of cases as "new" or "recurrent" was not verified, and a history of prior treatment was not sought. Although the full effect of these methodological differences cannot be determined, the most recent CDC survey suggests that drug resistance has increased in the United States after years of relative stability. Some regional data suggest that the increases in drug-resistant tuberculosis may be concentrated in large urban areas. In the 1991 CDC survey, 67% of the cases of drug resistance were reported from only five areas: New York City, California, Texas, New Jersey, and Florida. In both the 1991 and the 1992 survey, 61% of the cases of multidrug-resistant tuberculosis were reported from New York City alone. Frieden and colleagues conducted a survey in New York City of drug susceptibility results on specimens from all patients with a positive culture for M. tuberculosis during April 1991 (14). They found that of patients with no previous treatment, 23% had organisms resistant to at least one drug and 7% had organisms resistant to both isoniazid and rifampin. Beginning in 1993, results of drug susceptibility tests were reported to CDC from all 50 state health departments and from New York City, the District of Columbia, and Puerto Rico through SURVS-TB, CDC’s national TB surveillance system. In an analysis of areas reporting susceptibility results for at least 75% of culture-positive cases, New York City accounted for 80% of the cases of multidrug-resistant tuberculosis (CDC, unpublished data, 1995). Factors associated with drug resistance Click on image for more detailAnother risk factor for drug-resistant tuberculosis is contact with a person who has infectious, drug-resistant tuberculosis. Recent nosocomial outbreaks demonstrate a strong correlation between previous exposure to a patient who has infectious, multidrug-resistant tuberculosis and the subsequent development in the contact of multidrug-resistant tuberculosis (19-23). Drug resistance occurs more frequently in persons from areas of the world with a high prevalence of drug-resistant tuberculosis, such as Southeast Asia, Latin America, Haiti and the Phillippines. Several studies have found high rates of primary drug resistance in these persons, indicating transmission of drug resistant tuberculosis in the country of origin (24-27). However, because an accurate history of treatment may be difficult to obtain, some previously treated patients may be misclassified as having primary resistance. In the CDC survey conducted from 1982 to 1986, the rates of both primary and acquired drug resistance were two times higher among foreign-born persons than among persons born in the United States (12). Outbreaks of Drug-Resistant Tuberculosis From 1990 through August 1992, in collaboration with state and local health departments, CDC investigated outbreaks of multidrug-resistant tuberculosis in seven hospitals in Florida, New York, and New Jersey and in the New York State correctional system (Tables 2 and 3) (19-23, 34-40). The recent outbreaks differ considerably from previous outbreaks of drug-resistant tuberculosis in several ways. First, the recent outbreaks involved large numbers of patients; nearly 300 cases have been identified. In addition, in the recent outbreaks tuberculosis was transmitted not only from patient to patient but also from patient to health care worker. The epidemiologic evidence of nosocomial transmission was confirmed by DNA fingerprinting data: strains from epidemiologically linked cases were found to have identical patterns by restriction fragment length polymorphism (RFLP) analysis. RFLP analysis also suggested that several of the outbreaks in New York State were connected. Click on image for more detail
Click on image for more detailFinally, the recent outbreaks involved a large percentage of highly susceptible patients who became infected with highly drug-resistant organisms that are more difficult to treat. More than 80% of the cases occurred in persons infected with HIV, and all but six of the patients had organisms resistant to both isoniazid and rifampin. Mortality in these outbreaks was very high. In seven of the eight outbreaks, more than 70% of the patients died. The median interval between diagnosis and death ranged from 4 to 16 weeks. Factors associated with the outbreaks of multidrug-resistant
tuberculosis Prolonged infectiousness also promoted transmission. The diagnosis of tuberculosis in HIV-infected persons was sometimes delayed because of the unusual radiographic presentations of tuberculosis, coinfection with other pulmonary pathogens to which patients' symptoms were attributed, and the overgrowth of M. tuberculosis in the laboratory by other mycobacteria. Delays in diagnosis lead to delays in the initiation of isolation and treatment. In addition, drug resistance was not recognized promptly because of lengthy laboratory delays, which postponed the initiation of effective treatment and resulted in prolonged infectiousness. Inadequate infection control practices also facilitated transmission. Isolation rooms were found to be at positive pressure relative to other parts of the facility. Patients who had been assigned to isolation rooms were found in hallways, patient lounges, or other common areas. Furthermore, isolation precautions were discontinued prematurely, after an arbitrary number of days, rather than when there was clinical or laboratory evidence of decreased infectiousness. Finally, some patients who were given appropriate therapy in the hospital were lost to follow-up after discharge. A lack of consistent follow-up after discharge promotes lapses in therapy and heightens the potential for transmission both in the hospital and in the community. This problem was not specifically addressed in the outbreak investigations, but it was described in an unrelated report by Brudney and Dobkin. They studied 224 tuberculosis patients admitted to Harlem Hospital in 1988. Of 178 patients discharged on tuberculosis treatment, 89% were lost to follow-up and did not complete therapy. Forty-eight patients were readmitted to the hospital with infectious tuberculosis within 1 year (41). Treatment Second, initial regimens for treating tuberculosis should include four drugs: isoniazid, rifampin, pyrazinamide, and either ethambutol or streptomycin. When drug susceptibility results become available, the regimen can be adjusted accordingly. More drugs may be required for initial regimens in institutions where outbreaks of multidrug-resistant tuberculosis are occurring. In areas where resistance to isoniazid or rifampin is less than 4%, an initial regimen of three drugs may be adequate. The continued surveillance of drug resistance is necessary to detect changes in local drug resistance patterns and to evaluate the appropriateness of initial regimens in given areas. Third, therapy for tuberculosis should be directly observed by a health care provider. Nonadherence to therapy is a major cause of treatment failure, and it may contribute to the emergence of drug resistance. Directly observed therapy (DOT) is an effective way to ensure adherence, but the method must be tailored to each patient's needs and preferences. Tuberculosis that is resistant to isoniazid alone can be treated successfully by using rifampin and ethambutol for a minimum of 12 months, preferably supplemented by pyrazinamide for the first 2 months. In addition, a four-drug regimen has been shown to be highly effective for isoniazid-resistant organisms. Rifampin resistance, however, significantly reduces cure rates and increases the complexity and the required length of treatment (43). The treatment of multidrug-resistant tuberculosis should be based on the in vitro susceptibility test results and the patient's treatment history (5,44,45). The regimen should include at least three drugs — preferably drugs that the patient has not received before — to which the patient's organism is susceptible. The regimen should include an injectable medication whenever possible. If the regimen must be changed for any reason, two medications should be added simultaneously to avoid selecting for drug-resistant mutants. Medications used to treat multidrug-resistant tuberculosis are less effective, more costly, and more likely to cause adverse reactions than the five first-line drugs (44-48). Treatment is further complicated because of the length of therapy needed to prevent relapse: generally, therapy for multidrug-resistant tuberculosis should continue an additional 18 to 24 months after culture results convert to negative. Because of the high failure and relapse rates associated with multidrug-resistant tuberculosis, surgical resection has been used by some clinicians to supplement medical therapy. The surgical resection of a major pulmonary focus is best performed once aggressive medical therapy has achieved a clinical response. The administration of antituberculosis medications should continue for 18 to 24 months after culture results convert to negative. Iseman and associates reported that in a series of 99 patients being treated for multidrug-resistant tuberculosis, the combination of surgical resection and medical therapy produced lower rates of failure and relapse compared with that of historical controls (49). Preventive Therapy for Persons Exposed to Multidrug-Resistant
Tuberculosis In the outbreaks of multidrug-resistant tuberculosis, numerous tuberculin skin test conversions were documented among persons exposed to multidrug-resistant tuberculosis. Preventive therapy for persons exposed to multidrug-resistant tuberculosis has not been studied, but CDC has published guidelines for the management of such contacts (51). Decisions regarding preventive therapy should be based on the likelihood that the contact has been newly infected, that the infecting organisms are multidrug-resistant, and that active tuberculosis will develop. If the likelihood of these factors is low, standard preventive therapy is recommended. Alternative regimens should be considered for persons who are likely to be newly infected with a multidrug-resistant strain and who are at high risk for the development of active disease if infected. CDC recommends that alternative regimens include at least two drugs to which the infecting organism is known to be susceptible. Potential alternative regimens include pyrazinamide and ethambutol or pyrazinamide and a quinolone (e.g., ciprofloxacin) for 6 to 12 months. Control Measures Furthermore, appropriate infection control practices must be used to prevent the transmission of tuberculosis in health care settings (52). Different infection control measures interrupt the transmission of tuberculosis at different points in the transmission process. The most effective infection control measures curb transmission at the source by preventing the generation of infectious droplet nuclei. Examples of administrative control methods include the early diagnosis of disease and prompt initiation of effective therapy for persons with active tuberculosis, preventive therapy for persons with tuberculosis infection, and instruction of patients to cover their noses and mouths with a tissue when coughing. Booths and hoods used for aerosolized pentamidine or sputum induction are other examples of source control measures. Potentially infectious patients should be placed in tuberculosis isolation in a private room that has negative pressure relative to the rest of the facility. The air from the room should be exhausted directly to the outside or through a high-efficiency particulate air (HEPA) filter, if recirculation of air into the general ventilation system from the room is unavoidable. Isolation should be continued until the patient is no longer infectious. Patients known or suspected to have active tuberculosis should be considered infectious if they are coughing, if they are undergoing cough-inducing procedures, if their sputum smears contain acid-fast bacilli, if they are not receiving antituberculosis chemotherapy, if they recently started receiving chemotherapy, or if they are not responding to therapy. Most patients with drug-susceptible disease become noninfectious after 2 to 3 weeks of therapy, but the period of infectiousness varies from patient to patient. Patients with drug-susceptible disease who are receiving effective therapy and who show signs of response to therapy (i.e., a reduction in cough, the resolution of fever and a decreasing number of bacilli on sputum smears) are probably no longer infectious. However, patients with drug-resistant disease may take longer to respond to therapy, especially if drug resistance is not suspected and they are not receiving effective therapy. Therefore, patients should be considered infectious until their sputum smears are free of bacilli on 3 consecutive days. Ventilation and other engineering controls can be used to eliminate infectious droplet nuclei once they are released into the air, but these infection control measures are less efficient and less effective than administrative control methods. Ventilation reduces airborne contaminants by introducing uncontaminated air into the room, thereby diluting the concentration of contaminants. The contaminants are removed by exhausting the air from the room directly to the outside. Potential supplemental measures include HEPA filters and germicidal ultraviolet irradiation. These supplemental measures require proper installation and regular maintanence by qualified personnel, and their use in preventing tuberculosis transmission has not been well studied. Furthermore, exposure to ultraviolet irradiation may pose substantial health risks. Short-term exposure to germicidal ultraviolet irradiation is known to cause keratoconjunctivitis and erythema, and long-term exposure has been associated with an increased risk of basal cell carcinoma. Of all infection control methods, the use of personal protective equipment is the least efficient and least effective. These devices protect only the wearer, and they function only when they fit properly and are worn correctly. Administrative controls and engineering controls are necessary to protect all persons in the facility from airborne contaminants. Personal protective devices should be worn by health care workers in areas where the air is likely to be contaminated with a higher concentration of infectious droplet nuclei such as isolation rooms or rooms used for cough-inducing procedures. A tuberculosis screening and prevention program for health care workers is an essential component of infection control programs in health care facilities. All persons working in health care facilities should be skin tested upon employment. Persons who have negative skin test results should be retested periodically. Screening will detect tuberculosis infection in persons for whom preventive therapy may be appropriate. In addition, skin test results must be analyzed to evaluate the effectiveness of an infection control program by determining whether tuberculosis is being transmitted in the facility. Conclusion References
Last Reviewed: 05/18/2008 |
|
|
If you would like to order any of the DTBE publications please visit the online order form. You will need Adobe Acrobat™ Reader v5.0 or higher to read pages that are in PDF format. Download the Adobe Acrobat™ Reader. If you have difficulty accessing any material on the DTBE Web site because of a disability, please contact us in writing or via telephone and we will work with you to make the information available. Division of Tuberculosis Elimination
Centers for Disease Control & Prevention |