
| |  | |  |
Review
|
| Nanotoxicology: An Emerging Discipline Evolving from Studies of Ultrafine Particles Günter Oberdörster,1 Eva Oberdörster,2 and
Jan Oberdörster3
1Department of Environmental Medicine, University of Rochester,
Rochester, New York, USA; 2Department of Biology, Southern Methodist
University, Dallas, Texas, USA; 3Toxicology Department, Bayer CropScience,
Research Triangle Park, North Carolina, USA Abstract
Although humans have been exposed to airborne nanosized particles (NSPs ; < 100 nm) throughout their evolutionary stages, such exposure has increased dramatically over the last century due to anthropogenic sources. The rapidly developing field of nanotechnology is likely to become yet another source through inhalation, ingestion, skin uptake, and injection of engineered nanomaterials. Information about safety and potential hazards is urgently needed. Results of older biokinetic studies with NSPs and newer epidemiologic and toxicologic studies with airborne ultrafine particles can be viewed as the basis for the expanding field of nanotoxicology, which can be defined as safety evaluation of engineered nanostructures and nanodevices. Collectively, some emerging concepts of nanotoxicology can be identified from the results of these studies. When inhaled, specific sizes of NSPs are efficiently deposited by diffusional mechanisms in all regions of the respiratory tract. The small size facilitates uptake into cells and transcytosis across epithelial and endothelial cells into the blood and lymph circulation to reach potentially sensitive target sites such as bone marrow, lymph nodes, spleen, and heart. Access to the central nervous system and ganglia via translocation along axons and dendrites of neurons has also been observed. NSPs penetrating the skin distribute via uptake into lymphatic channels. Endocytosis and biokinetics are largely dependent on NSP surface chemistry (coating) and in vivo surface modifications. The greater surface area per mass compared with larger-sized particles of the same chemistry renders NSPs more active biologically. This activity includes a potential for inflammatory and pro-oxidant, but also antioxidant, activity, which can explain early findings showing mixed results in terms of toxicity of NSPs to environmentally relevant species. Evidence of mitochondrial distribution and oxidative stress response after NSP endocytosis points to a need for basic research on their interactions with subcellular structures. Additional considerations for assessing safety of engineered NSPs include careful selections of appropriate and relevant doses/concentrations, the likelihood of increased effects in a compromised organism, and also the benefits of possible desirable effects. An interdisciplinary team approach (e.g., toxicology, materials science, medicine, molecular biology, and bioinformatics, to name a few) is mandatory for nanotoxicology research to arrive at an appropriate risk assessment. Key words: biokinetics, central nervous system, engineered nanomaterials, environmental health, human health, nanosized particles, respiratory tract, risk assessment, skin, ultrafine particles. Environ Health Perspect 113:823-839 (2005) . doi:10.1289/ehp.7339 available via http://dx.doi.org/ [Online 22 March 2005]
Address correspondence to G. Oberdörster, University of Rochester, Department of Environmental Medicine, 575 Elmwood Ave., MRBx Building, Box 850, Rochester, NY 14642 USA. Telephone: (585) 275-3804. Fax: (585) 256-2631. E-mail: Gunter_Oberdorster@urmc.rochester.edu Supplemental Material is available online at http://ehp.niehs.nih.gov/members/2005/7339/supplemental.pdf We thank J. Havalack for excellent assistance in preparing the manuscript. This work was supported in part by the U.S. Environmental Protection Agency (EPA) STAR Program grant R827354, National Institute of Environmental Health Sciences grant ESO1247, U.S. Department of Defense MURI grant FA9550-04-1-430, and the National Science Foundation (SGER) BES-0427262. The views expressed by the authors are their own and do not necessarily reflect those of the EPA. J. Oberdörster is an employee of Bayer CropScience. The other authors declare they have no competing financial interests. Received 18 June 2004 ; accepted 22 March 2005. |
|
|
 |
Exposures to airborne nanosized particles (NSPs; < 100
nm) have been experienced by humans throughout their
evolutionary stages, but it is only with the advent
of the industrial revolution that such exposures have
increased
dramatically because of anthropogenic sources such
as internal combustion engines, power plants, and many
other
sources of thermodegradation. The rapidly developing
field of nanotechnology is likely to become yet another
source for human exposures to NSPs--engineered nanoparticles
(NPs)--by different routes: inhalation (respiratory
tract), ingestion [gastrointestinal (GI) tract], dermal
(skin), and injection (blood circulation). Table 1
summarizes some of the natural and anthropogenic sources
of NSPs,
the latter divided into unintentional and intentional
sources.
Table
1

|

Figure 1. Idealized
size distribution of traffic-related particulate
matter (U.S. EPA 2004). Dp, particle diameter.
The four polydisperse modes of traffic-related ambient
particulate matter span approximately four orders
of magnitude from < 1 nm to > 10 µm.
Nucleation- and Aitken-mode particles are defined
as UFPs (< approximately 100 nm). Source-dependent
chemical composition is not well controlled and varies
considerably. In contrast, NPs (1-100 nm) have well-controlled
chemistry and are generally monodispersed. |
Table 2
 |

Figure 2. Surface molecules
as a function of particle size. Surface molecules increase
exponentially when
particle size decreases < 100 nm, reflecting the
importance of surface area for increased chemical and
biologic activity of NSPs. The increased biologic activity
can be positive and desirable (e.g., antioxidant activity,
carrier capacity for therapeutics, penetration of cellular
barriers), negative and undesirable (e.g., toxicity,
induction of oxidative stress or of cellular dysfunction),
or a mix of both. Figure courtesy of H. Fissan (personal
communication). |
Biologically based or naturally occurring molecules
that are found inside organisms since the beginning
of life can serve as model nanosized materials. For example,
biogenic magnetite is a naturally occurring NSP that
occurs in many species ranging from bacteria to protozoa
to animals (Blakemore 1975; Kirschvink et al. 2001).
Biogenic magnetite has even been found in brains of
humans (Dunn et al. 1995; Kirschvink et al. 1992; Schultheiss-Grassi
et al. 1999) and has been associated with neurodegenerative
diseases (Dobson 2001; Hautot et al. 2003). A biologic
model of coated nanomaterials can be found in ferritin,
which is an approximately 12-nm-large iron storage
protein
that contains 5- to 7-nm-sized hydrous ferric oxide
phosphate inside a protective protein shell (Donlin
et al. 1998).
Nanosized materials, including fullerenes, occur naturally
from combustion processes such as forest fires and
volcanoes.
Obvious differences between unintentional and intentional
anthropogenic NSPs are the polydispersed and chemically
complex nature (elemental, soluble, and volatile carbon
compounds; soluble and poorly soluble inorganics; Cyrys
et al. 2003; Hughes et al. 1998) of the former, in contrast
to the monodisperse and precise chemically engineered
characteristics and solid form of the latter, generated
in gas or liquid phase [National Nanotechnology Initiative
(NNI) 2004]. However, despite these differences, the
same toxicologic principles are likely to apply for NPs,
because not only size but also a number of other particle
parameters determine their biologic activity. The multitude
of interactions of these factors has yet to be assessed,
and in this article we attempt to summarize our present
knowledge.
NSPs are variably called ultrafine particles (UFPs)
by toxicologists [U.S. Environmental Protection Agency
(EPA) 2004], Aitken mode and nucleation mode particles
by atmospheric scientists [Kulmala 2004; National Research
Council (NRC) 1983], and engineered nanostructured
materials by materials scientists (NNI 2004). Figure
1 depicts
the range of sizes of airborne ambient particulate
matter, including the nucleation-mode, Aitken-mode, accumulation-mode,
and coarse-mode particles. Ambient particles < 0.1 µm,
defined as UFPs in the toxicologic literature, consist
of transient nuclei or Aitken nuclei (NRC 1983). More
recently, even smaller particles in the nucleation mode
with peak diameters around 4 nm have been observed (McMurry
and Woo 2002). The distinction between NSPs generated
by internal combustion engines and NPs becomes further
clouded by the finding of nanotubes in diesel exhaust
(Evelyn et al. 2003). The use of the term “nano” in
this review reflects only particle size and not chemical
composition. For the purposes of this review, we use
the following terms: “Nanosized particle” (NSP)
includes all engineered and ambient nanosized spherical
particles < 100 nm. “Engineered nanoparticle” (NP)
includes only spherical NSPs specifically engineered
in the laboratory; other engineered nanosized structures
will be labeled according to their shape, for example,
nanotubes, nanofibers, nanowires, nanorings, and so on. “Ultrafine
particle” (UFP) includes ambient and laboratory-generated
NSPs that are not produced in a controlled, engineered
way.
Table 2 shows the tremendous differences in particle
number concentrations and particle surface areas for
particles of the four ambient modes, assuming an airborne
concentration of 10 µg/m3 of unit density
particles of each size. The extraordinarily high number
concentrations of NSPs per given mass will likely be
of toxicologic significance when these particles interact
with cells and subcellular components. Likewise, their
increased surface area per unit mass can be toxicologically
important if other characteristics such as surface chemistry
and bulk chemistry are the same. Although the mass of
UFPs in ambient air is very low, approaching only 0.5-2 µg/m3 at
background levels (Hughes et al. 1998), it can increase
several-fold during high pollution episodes or on highways
(Brand et al. 1991; Shi et al. 2001; Zhu et al. 2002).
Physicochemical characteristics as determinants
of biologic activity. The small size and
corresponding large specific surface area of solid
NSPs (Table 2) confer specific properties to them,
for example, making them desirable as catalysts for
chemical reactions. The importance of surface area
becomes evident when considering that surface atoms
or molecules play a dominant role in determining
bulk properties (Amato 1989); the ratio of surface
to total atoms or molecules increases exponentially
with decreasing particle size (Figure 2). Increased
surface reactivity predicts that NSPs exhibit greater
biologic activity per given mass compared with larger
particles, should they be taken up into living organisms
and provided they are solid rather than solute particles.
This increased biologic activity can be either positive
and desirable (e.g., antioxidant activity, carrier
capacity for therapeutics, penetration of cellular
barriers for drug delivery) or negative and undesirable
(e.g., toxicity, induction of oxidative stress or
of cellular dysfunction), or a mix of both. Not only
may adverse effects be induced, but interactions
of NSPs with cells and subcellular structures and
their biokinetics are likely to be very different
from those of larger-sized particles. For example,
more than 60 years ago virologists described the
translocation of 30- to 50-nm-sized virus particles along axons and dendrites of neurons and across epithelia
(Howe and Bodian 1940), whereas first reports about
increased inflammatory activity and epithelial translocation
of man-made 20- and 30-nm solid particles appeared
only more recently (Ferin et al. 1990; Oberdörster
et al. 1990).
The characteristic biokinetic behaviors of NPs are
attractive qualities for promising applications in medicine
as diagnostic and therapeutic devices and as tools to
investigate and understand molecular processes and structures
in living cells (Akerman et al. 2002; Foley et al. 2002;
Kreuter 2001; Li et al. 2003). For example, targeted
drug delivery to tissues that are difficult to reach
[e.g., central nervous system (CNS)], NPs for the fight
against cancer, intravascular nanosensor and nanorobotic
devices, and diagnostic and imaging procedures are presently
under development. The discipline of nanomedicine--defined
as medical application of nanotechnology and related
research--has arisen to design, test, and optimize
these applications so that they can eventually be used
routinely by physicians (Freitas 1999).
However, in apparent stark contrast to the many efforts
aimed at exploiting desirable properties of NPs for improving
human health are the limited attempts to evaluate potential
undesirable effects of NPs when administered intentionally
for medicinal purposes, or after unintentional exposure
during manufacture or processing for industrial applications.
The same properties that make NPs so attractive for development
in nanomedicine and for specific industrial processes
could also prove deleterious when NPs interact with cells.
Thus, evaluating the safety of NPs should be of highest
priority given their expected worldwide distribution
for industrial applications and the likelihood of human
exposure, directly or through release into the environment
(air, water, soil). Nanotoxicology--an emerging
discipline that can be defined as “science of engineered
nanodevices and nanostructures that deals with their
effects in living organisms”--is gaining increased
attention. Nanotoxicology research not only will provide
data for safety evaluation of engineered nanostructures
and devices but also will help to advance the field of
nanomedicine by providing information about their undesirable
properties and means to avoid them.
Human exposure to nanosized materials. In
addition to natural and anthropogenic sources of UFPs
in the ambient air, certain workplace conditions also
generate NSPs that can reach much higher exposure concentrations,
up to several hundred micrograms per cubic meter, than
is typically found at ambient levels. In contrast to
airborne UFP exposures occurring via inhalation at the
workplace and from ambient air, not much is known about
levels of exposure via different routes for NPs, whether
it is by direct human exposure or indirectly through
contamination of the environment. For example, are there
or will there be significant exposures to airborne singlet
engineered carbon nanotubes or C60 fullerenes?
First measurements at a model workplace gave only very
low concentrations, < 50 µg/m3, and
these were most likely in the form of aggregates (Maynard
et al. 2004). However, even very low concentrations of
nanosized materials in the air represent very high particle
number concentrations, as is well known from measurements
of ambient UFPs (Hughes et al. 1998). For example, a
low concentration of 10 µg/m3 of unit
density 20-nm particles translates into > 1  106 particles/cm3 (Table
2). Inhalation may be the major route of exposure for
NPs, yet ingestion and dermal exposures also need to
be considered during manufacture, use, and disposal of
engineered nanomaterials, and specific biomedical applications
for diagnostic and therapeutic purposes will require
intravenous, subcutaneous, or intramuscular administration
(Table l). It can be assumed, however, that the toxicology
of NPs can build on the experience and data already present
in the toxicology
literature of ambient UFPs. [Additional details provided in Supplemental Material
available online (http://ehp.niehs.nih.gov/members/2005/7339/supplemental.pdf).] 106 particles/cm3 (Table
2). Inhalation may be the major route of exposure for
NPs, yet ingestion and dermal exposures also need to
be considered during manufacture, use, and disposal of
engineered nanomaterials, and specific biomedical applications
for diagnostic and therapeutic purposes will require
intravenous, subcutaneous, or intramuscular administration
(Table l). It can be assumed, however, that the toxicology
of NPs can build on the experience and data already present
in the toxicology
literature of ambient UFPs. [Additional details provided in Supplemental Material
available online (http://ehp.niehs.nih.gov/members/2005/7339/supplemental.pdf).]
Manufactured nanomaterials in the environment. Manufactured
nanomaterials are likely to enter the environment for
several reasons. Some are and others will be produced
by the ton, and some of any material produced in such
mass quantities is likely to reach the environment from
manufacturing effluent or from spillage during shipping
and handling. They are being used in personal-care products
such as cosmetics and sunscreens and can therefore enter
the environment on a continual basis from washing off
of consumer products (Daughton and Ternes 1999). They
are being used in electronics, tires, fuel cells, and
many other products, and it is unknown whether some of
these materials may leak out or be worn off over the
period of use. They are also being used in disposable
materials such as filters and electronics and may therefore
reach the environment through landfills and other methods
of disposal.
Scientists have also found ways of using nanomaterials
in remediation. Although many of these are still in testing
stages (Chitose et al. 2003; Jaques and Kim 2000; Joo
et al. 2004; Nagaveni et al. 2004; Nghiem et al. 2004;
Tungittiplakorn et al. 2004), dozens of sites have already
been injected with various nanomaterials, including nano-iron
(Mach 2004). Testing to determine the safety of these
NPs used for remediation to environmentally relevant
species has not yet been done. Although most people are
concerned with effects on large wildlife, the basis of
many food chains depends on the benthic and soil flora
and fauna, which could be dramatically affected by such
NP injections. In addition, as noted by Lecoanet et al.
(2004), nanosized materials may not migrate through soils
at rapid enough rates to be valuable in remediation.
Future laboratory and field trials will help clarify
the line between remediation and contamination [Supplemental
Material available online (http://ehp.niehs.nih.gov/members/2005/7339/supplemental.pdf)].
Toxicology of Airborne UFPs
In recent years, interest in potential effects of exposure
to airborne UFPs has increased considerably, and studies
have shown that they can contribute to adverse health
effects in the respiratory tract as well as in extrapulmonary
organs. Results on direct effects of ambient and model
UFPs have been reported from epidemiologic studies and
controlled clinical studies in humans, inhalation/instillation
studies in rodents, or in vitro cell culture systems.
For example, several epidemiologic studies have found
associations of ambient UFPs with adverse respiratory
and cardiovascular effects resulting in morbidity and
mortality in susceptible parts of the population (Pekkanen
et al. 1997; Penttinen et al. 2001; Peters et al. 1997a,
1997b; von Klot et al. 2002; Wichmann et al. 2002), whereas
other epidemiologic studies have not seen such associations
(Pekkanen et al. 1997; Tiittanen et al. 1999). Controlled
clinical studies evaluated deposition and effects of
laboratory-generated UFPs. High deposition efficiencies
in the total respiratory tract of healthy subjects were
found, and deposition was even greater in subjects with
asthma or chronic obstructive pulmonary disease. In addition,
effects on the cardiovascular system, including blood
markers of coagulation and systemic inflammation and
pulmonary diffusion capacity, were observed after controlled
exposures to carbonaceous UFPs (Anderson et al. 1990;
Brown et al. 2002; Chalupa et al. 2004; Henneberger et
al., 2005; Jaques and Kim 2000; Pekkanen et al. 2002;
Pietropaoli et al. 2004; Wichmann et al. 2000).
Studies in animals using laboratory-generated model
UFPs or ambient UFPs showed that UFPs consistently induced
mild yet significant pulmonary inflammatory responses
as well as effects in extrapulmonary organs. Animal inhalation
studies included the use of different susceptibility
models in rodents, with analysis of lung lavage parameters
and lung histopathology, effects on the blood coagulation
cascade, and translocation studies to extrapulmonary
tissues (Elder et al. 2000, 2002, 2004; Ferin et al.
1991; Ferin and Oberdörster 1992; Kreyling et al.
2002; Li et al. 1999; Nemmar et al. 1999, 2002a, 2002b,
2003; Oberdörster et al. 1992a, 1995, 2000, 2002,
2004; Semmler et al. 2004; Zhou et al. 2003).

Figure 3. Hypothetical cellular interaction of
NSPs (adapted from Donaldson and Tran 2002). EGFR,
epidermal growth factor receptor. Inflammation and
oxidative stress can be mediated by several primary
pathways: a) the particle surface causes oxidative
stress resulting in increased intracellular calcium
and gene activation; b) transition metals released
from particles result in oxidative stress, increased
intracellular calcium, and gene activation; c) cell
surface receptors are activated by transition metals
released from particles, resulting in subsequent
gene activation; or d) intracellular distribution
of NSPs to mitochondria generates oxidative stress. |

Figure 4. Percentage of neutrophils in lung lavage
of rats (A,B) and mice (C,D) as indicators of inflammation
24 hr after intratracheal instillation of different
mass doses of 20-nm and 250-nm TiO2 particles in
rats and mice. (A,C) The steeper dose response of
nanosized TiO2 is obvious when the dose is expressed
as mass. (B,D) The same dose response relationship
as in (A,C) but with dose expressed as particle
surface area; this indicates that particle surface
area seems
to be a more appropriate dosemetric for comparing
effects of different-sized particles, provided they
are of the same chemical structure (anatase TiO2
in this case). Data show mean ± SD. |
In vitro studies using different cell systems
showed varying degrees of proinflammatory- and oxidative-stress-related
cellular responses after dosing with laboratory-generated
or filter-collected ambient UFPs (Brown et al. 2000,
2001; Li et al. 2003). Collectively, the in vitro results
have identified oxidative-stress-related changes
of gene expression and cell signaling pathways as underlying
mechanisms of UFP effects, as well as a role of transition
metals and certain organic compounds on combustion-generated
UFPs (Figure 3). These can alter cell signaling pathways,
including Ca 2+ signaling and cytokine signaling
(e.g., interleukin-8) (Donaldson et al. 2002; Donaldson
and Stone 2003). Effects were on a mass basis greater
for model UFPs than for fine particles, whereas effects
for ambient UFP cellular responses were sometimes greater
and sometimes less than those of fine and coarse particles.
The interpretation of the in vitro studies is
oftentimes difficult because particles of different
chemical compositions were used, target cells were
different,
and duration, end points, and generally high dose levels
also differed. Results from high doses in particular
should be viewed with caution if they are orders of
magnitude higher than predicted from relevant ambient
exposures
(see “Exposure dose-response considerations,” below).
[Supplemental Material available online ( http://ehp.niehs.nih.gov/members/2005/
7339/supplemental.pdf).]
Concepts of Nanotoxicology
Laboratory rodent studies. With respect
to potential health effects of NSPs, two examples should
serve to illustrate a) that NSPs have a higher
inflammatory potential per given mass than do larger
particles, provided they are chemically the same, and b)
that NSPs generated under certain occupational conditions
can elicit severe acute lung injury.
The first example involves studies with ultrafine and
fine titanium dioxide (TiO2) particles, which
showed that ultrafine anatase TiO2 (20 nm),
when instilled intratracheally into rats and mice, induced
a much greater pulmonary-inflammatory neutrophil response
(determined by lung lavage 24 hr after dosing) than did
fine anatase TiO2 (250 nm) when both types
of particles were instilled at the same mass dose (Figure
4A,C). However, when the instilled dose was expressed
as particle surface area, it became obvious that the
neutrophil response in the lung for both ultrafine and
fine TiO2 fitted the same dose-response
curve (Figure 4B,D), suggesting that particle surface
area for particles of different sizes but of the same
chemistry, such as TiO2, is a better dosemetric
than is particle mass or particle number (Oberdörster
G 2000). Moreover, normalizing the particle surface dose
to lung weight shows excellent agreement of the inflammatory
response in both rats and mice [Figure S-2 in Supplemental
Material available online (http://ehp.niehs.nih.gov/members/2005/7339/
supplemental.pdf)].
The better fit of dose-response relationships by
expressing the dose as surface area rather than mass
when describing toxicologic effects of inhaled solid
particles of different sizes has been pointed out repeatedly,
especially when particles of different size ranges--nano
to fine--were studied (Brown et al. 2001; Donaldson
et al. 1998, 2002; Driscoll 1996; Oberdörster and
Yu 1990; Oberdörster et al. 1992a; Tran et al. 1998,
2000) [Supplemental Material available online (http://ehp.niehs.nih.gov/members/2005/7339/supplemental.pdf)].
Particle chemistry, and specifically surface chemistry,
plays a decisive role in addition to particle size, as
shown in the second example: exposure of rats to polytetrafluoroethylene
(PTFE) fume. PTFE fume (generated by heating PTFE) has
long been known to be of high acute toxicity to birds
and mammals, including humans (Cavagna et al. 1961; Coleman
et al. 1968; Griffith et al. 1973; Nuttall et al. 1964;
Waritz and Kwon 1968). Analysis of these fumes revealed
the nanosized nature of the particles generated by heating
PTFE to about 480°C, with a count median diameter
(CMD) of 18 nm. They were highly toxic to rats, causing
severe acute lung injury with high mortality within 4
hr after a 15-min inhalation exposure to 50 µg/m3 (Oberdörster
et al. 1995). This short exposure resulted in an estimated
deposited dose in the alveolar regions of only approximately
60 ng. In humans, acute lung injury, known as polymer
fume fever, can result from exposures to PTFE fumes (Auclair
et al. 1983; Goldstein et al. 1987; Lee et al. 1997;
Williams et al. 1974; Woo et al. 2001). Additional rat
studies showed that the gas phase alone was not acutely
toxic and that aging of the PTFE fume particles for 3
min increased their particle size to > 100 nm because
of accumulation, which resulted in a loss of toxicity
(Johnston et al. 2000). However, it is most likely that
changes in particle surface chemistry during the aging
period contributed to this loss of toxicity, and that
this is not just an effect of the accumulated larger
particle size [Supplemental Material available online
(http://ehp.niehs.nih.gov/members/2005/7339/supplemental.pdf)].
These examples seem to represent the extremes of NSPs
in terms of pulmonary toxicity, one (TiO2)
being rather benign yet still inducing significantly
greater inflammatory effects on a mass basis than fine
particles of the same chemical makeup, the other (PTFE
fumes) inducing very high acute toxicity, possibly related
to reactive groups on the large surface per unit mass.
Engineered nanomaterials can have very different shapes,
for example, spheres, fibers, tubes, rings, and planes.
Toxicologic studies of spherical and fibrous particles
have well established that natural (e.g., asbestos) and
man-made (e.g., biopersistent vitreous) fibers are associated
with increased risks of pulmonary fibrosis and cancer
after prolonged exposures [Greim et al. 2001; International
Agency for Research on Cancer (IARC) 2002]. Critical
parameters are the three Ds: dose, dimension, and durability
of the fibers. Fibers are defined as elongated structures
with a diameter-to-length ratio (aspect ratio) of 1:
3 or greater and with a length of > 5 µm and
diameter ≤ 3 µm
[World Health Organization (WHO) 1985]. Carbon nanotubes
have aspect ratios of up to ≥ 100,
and length can exceed 5 µm with diameters ranging
from 0.7 to 1.5 nm for single-walled nanotubes, and 2
to 50 nm for multiwalled nanotubes. Results from three
studies using intratracheal dosing of carbon nanotubes
in rodents indicate significant acute inflammatory pulmonary
effects that either subsided in rats (Warheit et al.
2004) or were more persistent in mice (Lam et al. 2004;
Shvedova et al. 2004b). Administered doses were very
high, ranging from 1 to 5 mg/kg in rats; in mice doses
ranged from 3.3 to 16.6 mg/kg (Lam et al. 2004) or somewhat
lower, from 0.3 to 1.3 mg/kg (Shvedova et al. 2004a).
Granuloma formation as a normal foreign body response
of the lung to high doses of a persistent particulate
material was a consistent finding in these studies. Metal
impurities (e.g., iron) from the nanotube generation
process may also have contributed to the observed effects.
Although these in vivo first studies revealed
high acute effects, including mortality, this was explained
by the large doses of the instilled highly aggregated
nanotubes that caused death by obstructing the airways
and should not be considered a nanotube effect per se
(Warheit et al. 2004). Invitro studies
with carbon nanotubes also reported significant effects.
Dosing keratinocytes and bronchial epithelial cells invitro with
single-walled carbon nanotubes (SWNTs) resulted in oxidative
stress, as evidenced by the formation of free radicals,
accumulation of peroxidative products, and depletion
of cell antioxidants (Shvedova et al. 2004a, 2004b).
Multiwalled carbon nanotubes (MWNTs) showed proinflammatory
effects and were internalized in keratinocytes (Monteiro-Riviere
et al. 2005). Again, the relatively high doses applied
in these studies need to be considered when discussing
the relevancy of these findings for invivo exposures.
A most recent study in macrophages comparing SWNTs and
MWNTs with C60 fullerenes found a cytotoxicity
ranking on a mass basis in the order SWNT > MWNT > C60.
Profound cytotoxicity (mitochondrial function, cell morphology,
phagocytic function) was seen for SWNTs, even at a low
concentration of 0.38 µg/cm2. The possible
contribution of metal impurities of the nanotubes still
needs to be assessed. Therefore, whether the generally
recognized principles of fiber toxicology apply to these
nanofiber structures needs still to be determined (Huczko
et al. 2001).
Future studies should be designed to investigate both
effects and also the fate of nanotubes after deposition
in the respiratory tract, preferentially by inhalation
using well-dispersed (singlet) airborne nanotubes. In
order to design the studies using appropriate dosing,
it is necessary to assess the likelihood and degree of
human exposure. It is of utmost importance to characterize
human exposures in terms of the physicochemical nature,
the aggregation state, and concentration (number, mass,
surface area) of engineered nanomaterials and perform
animal and invitro studies accordingly.
If using direct instillation into the lower respiratory
tract, a large range of doses, which include expected
realistic exposures of appropriately prepared samples,
needs to be considered [Supplemental Material available
online (http://ehp.niehs.nih.gov/members/2005/7339/supplemental.pdf)].
Ecotoxicologic studies. Studies have
been carried out to date on only a few species that have
been accepted by regulatory agencies as models for defining
ecotoxicologic effects. Tests with uncoated, water-soluble,
colloidal fullerenes (nC60) show that the
48-hr LC50 (median lethal concentration) in Daphnia
magna is 800 ppb (Oberdörster E 2004b), using
standard U.S. EPA protocols (U.S. EPA 1994). In largemouth
bass (Micropterus salmoides), although no mortality
was seen, lipid peroxidation in the brain and glutathione
depletion in the gill were observed after exposure to
0.5 ppm nC60 for 48 hr (Oberdörster E
2004a). There are several hypotheses as to how lipid
damage may have occurred in the brain, including direct
redox activity by fullerenes reaching the brain via circulation
or axonal translocation (see also “Disposition
of NSPs in the respiratory tract,” below) and dissolving
into the lipid-rich brain tissue; oxyradical production
by microglia; or reactive fullerene metabolites may be
produced by cytochrome P450 metabolism. Initial follow-up
studies using suppressive subtractive hybridization of
pooled control fish versus pooled 0.5-ppm-exposed
fish liver mRNA were also performed. Proteins related
to immune responses and tissue repair were up-regulated,
and several proteins related to homeostatic control and
immune control were down-regulated. A cytochrome P450
(CYP2K4) involved in lipid metabolism was up-regulated
[Supplemental Material available online (http://ehp.niehs.nih.gov/members/2005/7339/supplemental.pdf)].
In addition to these biochemical and molecular-level
changes in fish, bactericidal properties of fullerenes
have also been reported and are being explored as potential
new antimicrobial agents (Yamakoshi et al. 2003). Engineered
nanomaterials used as antimicrobials may shift microbial
communities if they are released into the environment
via effluents. As we know from anthropogenic endocrine-disrupting
compounds, interference of signaling between nitrogen-fixing
bacteria and their plant hosts could be extremely harmful
both ecologically and economically in terms of crop production
(Fox et al. 2001).

Figure 5. Routes of exposure, uptake, distribution,
and degradation of NSPs in the environment. Solid
lines indicate routes that have been demonstrated
in the laboratory or field or that are currently
in use (remediation). Magenta lettering indicates
possible degradation routes, and blue lettering indicates
possible sinks and sources of NSPs. |

Figure 6. NPs have been shown to release oxyradicals
[pictured here is the mechanism of C60 as determined
by Yamakoshi et al. (2003)], which can interact with
the antioxidant defense system. Abbreviations: GPx,
glutathione peroxidase; GSH, reduced glutathione;
GSSG, oxidized glutathione; ISC, intersystem crossing;
R, any organic molecule; SOD, superoxide dismutase.
In addition to fullerenes, metals such as cadmium,
iron, or nickel quantum dots, or iron from SWNT manufacturing,
could also act in Fenton-type reactions. Phase II
biotransformation, ascorbic acid, vitamin E, beta
carotene, and other interactions are not shown. |
Aqueous fullerenes and coated SWNTs are stable in salt
solutions (Cheng et al. 2004; Warheit et al. 2004),
cell culture media (Lu et al. 2004; Sayes et al. 2004),
reconstituted
hard water, and MilliQ water (Dieckmann et al. 2003;
Oberdörster E 2004a, 2004b). NSPs will tend to
sorb onto sediment and soil particles and be immobilized
because
of their high surface area:mass ratio (Lecoanet and
Wiesner 2004). Biologic transport would occur from
ingested sediments,
and one would expect movement of nanomaterials through
the food chain (Figure 5).
To make engineered nanomaterials more biocompatible,
both surface coatings and covalent surface modifications
have been incorporated. Some studies have shown that
both the surface coating and the covalent modifications
can be weathered either by exposure to the oxygen in
air or by ultraviolet (UV) irradiation for 1-4
hr (Derfus et al. 2004; Rancan et al. 2002). Therefore,
although coatings and surface modifications may be critically
important in drug-delivery devices, the likelihood of
weathering under environmental conditions makes it important
to study toxicity under UV and air exposure conditions.
Even coatings used in drug delivery of NPs may not be
biopersistent or could be metabolized to expose the core
NP material [Supplemental Material available online (http://ehp.niehs.nih.gov/members/2005/7339/
supplemental.pdf)].
Reactive oxygen species mechanisms of NSP toxicity. Both in
vivo and in vitro, NSPs of various chemistries
have been shown to create reactive oxygen species (ROS).
ROS production has been found in NPs as diverse as
C60 fullerenes, SWNTs, quantum dots, and
UFPs, especially under concomitant exposure to light,
UV, or transition metals (Brown et al. 2000, 2001;
Derfus et al. 2004; Joo et al. 2004; Li et al. 2003;
Nagaveni et al. 2004; Oberdörster E 2004a; Rancan
et al. 2002; Sayes et al. 2004; Shvedova et al. 2004a,
2004b; Wilson et al. 2002; Yamakoshi et al. 2003).
It has been demonstrated that NSPs of various sizes
and various chemical compositions preferentially mobilize
to mitochondria (de Lorenzo 1970; Foley et al. 2002;
Gopinath et al. 1978; Li et al. 2003; Rodoslav et al.
2003). Because mitochondria are redox active organelles,
there is a likelihood of altering ROS production and
thereby overloading or interfering with antioxidant
defenses (Figure 3).
Figure 6 diagrams some of the antioxidant defense systems
that occur in animals, and possible areas where NSPs
may create oxyradicals. The C60 fullerene
is shown as a model NP producing superoxide, as has been
shown by Yamakoshi et al. (2003). The exact mechanism
by which each of these diverse NPs cause ROS is not yet
fully understood, but suggested mechanisms include a)
photo excitation of fullerenes and SWNTs, causing intersystem
crossing to create free electrons; b) metabolism
of NPs to create redox active intermediates, especially
if metabolism is via cytochrome P450s; and c)
inflammation responses in vivo that may cause
oxyradical release by macrophages. Other mechanisms will
likely emerge as studies on NP toxicity continue.
The small size and respective large specific surface
area of NPs, like those of ambient airborne UFPs, give
them unique properties with respect to a potential to
cause adverse effects. Certainly, as shown in studies
with UFPs, chemical composition and other particle parameters
are additional important effect modifiers. Results from
these studies will therefore serve as a basis for future
studies in the field of nanotoxicology, for example,
the propensity of NSPs to translocate across cell layers
and along neuronal pathways (see “Disposition of
NSPs in the respiratory tract” below).
Exposure dose-response considerations. A
careful evaluation of exposure-dose-response
relationships is critical to the toxicologic assessment
of NSPs. This includes not only questions about the dosemetric--mass,
number, or surface of the particles, as discussed above--but
most important, also the relevance of dose levels. For
example, it is tempting, and a continual practice, to
dose primary cells or cell lines in vitro with
very high doses without any consideration or discussion
of realistic in vivo exposures; for instance,
100 µg NSPs/mL culture medium--labeled as
a low dose--is extremely high and is unlikely to
be encountered in vivo. Likewise, intratracheal
instillations of several hundred micrograms into a rat
does not resemble a relevant in vivo inhalation
exposure; both dose and dose rate cause high bolus dose
artifacts. Although such studies may be used in a first
proof-of-principle approach, it is mandatory to follow
up and validate results using orders of magnitude lower
concentrations resembling realistic in vivo exposures,
including worst-case scenarios. The 500-year-old phrase “the
dose makes the poison” can also be paraphrased
as “the dose makes the mechanism.” The mechanistic
pathways that operate at low realistic doses are likely
to be different from those operating at very high doses
when the cell’s or organism’s defenses are
overwhelmed.

Figure 7. Some basic shapes of exposure-response
or dose-response relationships. Abbreviations:
H, hormetic (biphasic); L, linear (no threshold);
S, supralinear; T, threshold. Prerequisites for establishing
these relationships for NSPs from in vitro or in
vivo studies include a sufficient number of data
points, that is, over a wide range of exposure concentrations
or doses; knowledge about exposure levels; and information
about correlation of exposure with doses at the organismal
or cellular level (an exposure is not a dose). Dose-response
curves of different shapes can be extrapolated when
only response data at high dose levels (indicated
by dashed oval) are available. Lack of data in the
low--oftentimes the most relevant--dose
range can result in severe misinterpretation if a
threshold or even a hormetic response is present.
Consideration also needs to be given to the likelihood
that the shape or slope of exposure-dose-response
relationships change for susceptible parts of the
population. |

Figure 8. Predicted fractional deposition of
inhaled particles in the nasopharyngeal, tracheobronchial,
and alveolar region of the human respiratory tract
during nose breathing. Based on data from the International
Commission on Radiological Protection (1994). Drawing
courtesy of J. Harkema. |
Therefore, in vivo and in vitro studies
will provide useful data on the toxicity and mode of
action of NSPs only if justifiable concentrations/doses
are considered when designing such studies. This approach
is particularly important for the proper identification
of the dose-response curve. When data are generated
only at high concentrations/doses, it is difficult to
determine whether the dose-response curve in question
is best described by a linear (no threshold), supralinear,
threshold, or hormetic model (Figure 7). Study designs
should include doses that most closely reflect the expected
exposure levels. A critical gap that urgently needs to
be filled in this context is the complete lack of data
for human or environmental exposure levels of NSPs. Furthermore,
some knowledge about the biokinetics of NSPs is required
in order to estimate appropriate doses. Do specific NPs
reach certain target sites? If so, what are the doses,
dose rates, and their persistence? Further, although
it may be tempting to extrapolate from in vitro results
to an in vivo risk assessment, it is important
to keep in mind that in vitro tests are most useful
in providing information on mechanistic processes and
in elucidating mechanisms/mode of actions suggested by
studies in whole animals. A combination of in vitro and in
vivo studies with relevant dose levels will
be most useful in identifying the potential hazards
of NPs, and a thorough discussion and justification
of selected
dose levels should be mandatory.
Portals of Entry and
Target Tissues
Most of the toxicity research on NSPs in vivo has
been carried out in mammalian systems, with a focus on
respiratory system exposures for testing the hypothesis
that airborne UFPs cause significant health effects.
With respect to NPs, other exposure routes, such as skin
and GI tract, also need to be considered as potential
portals of entry. Portal-of-entry-specific defense
mechanisms protect the mammalian organism from harmful
materials. However, these defenses may not always be
as effective for NSPs, as is discussed below.
In order to appreciate what dose the organism receives
when airborne particles are inhaled, information about
their deposition as well as their subsequent fate is
needed. Here we focus on the fate of inhaled nanosized
materials both within the respiratory tract itself and
translocated out of the respiratory tract. There are
significant differences between NSPs and larger particles
regarding their behavior during deposition and clearance
in the respiratory tract [Supplemental Material available
online (http://ehp.niehs.nih.gov/members/2005/7339/
supplemental.pdf)].
Efficient deposition of inhaled NSPs. The
main mechanism for deposition of inhaled NSPs in the
respiratory tract is diffusion due to displacement when
they collide with air molecules. Other deposition mechanisms
of importance for larger particles, such as inertial
impaction, gravitational settling, and interception,
do not contribute to NSP deposition, and electrostatic
precipitation occurs only in cases where NSPs carry significant
electric charges. Figure 8 shows the fractional deposition
of inhaled particles in the nasopharyngeal, tracheobronchial,
and alveolar regions of the human respiratory tract under
conditions of nose breathing during rest, based on a
predictive mathematical model (International Commission
on Radiological Protection 1994). These predictions apply
to particles that are inhaled as singlet particles of
a given size and not as aggregates; the latter obviously
will have larger particle size and different deposition
site. In each of the three regions of the respiratory
tract, significant amounts of a certain size of NSPs
(1-100 nm) are deposited. For example, 90% of inhaled
1-nm particles are deposited in the nasopharyngeal compartment,
only approximately 10% in the tracheobronchial region,
and essentially none in the alveolar region. On the other
hand, 5-nm particles show about equal deposition of approximately
30% of the inhaled particles in all three regions; 20-nm
particles have the highest deposition efficiency in the
alveolar region (~ 50%), whereas in tracheobronchial
and nasopharyngeal regions this particle size deposits
with approximately 15% efficiency. These different deposition
efficiencies should have consequences for potential effects
induced by inhaled NSPs of different sizes as well as
for their disposition to extrapulmonary organs, as discussed
further below.
Disposition of NSPs in the respiratory tract. In
the preceding section we summarized data demonstrating
that inhaled NSPs of different sizes can target all three
regions of the respiratory tract. Several defense mechanisms
exist throughout the respiratory tract aimed at keeping
the mucosal surfaces free from cell debris and particles
deposited by inhalation. Several reviews describe the
well-known classic clearance mechanisms and pathways
for deposited particles (Kreyling and Scheuch 2000; Schlesinger
et al. 1997; U.S. EPA 2004), so here we only briefly
mention those mechanisms and point out specific differences
that exist with respect to inhaled NSPs [Supplemental
Material available online (http://ehp.niehs.nih.gov/members/2005/7339/supplemental.pdf)].
Once deposited, NSPs--in contrast to larger-sized
particles--appear to translocate readily to extrapulmonary
sites and reach other target organs by different transfer
routes and mechanisms. One involves transcytosis across
epithelia of the respiratory tract into the interstitium
and access to the blood circulation directly or via lymphatics,
resulting in distribution throughout the body. The other
is a not generally recognized mechanism that appears
to be distinct for NSPs and that involves their uptake
by sensory nerve endings embedded in airway epithelia,
followed by axonal translocation to ganglionic and CNS
structures.

Figure 9. Pathways of
particle clearance (disposition) in and out of the
respiratory tract. There are significant
differences between NSPs and larger particles for
some of these pathways (see “Disposition of
NSPs in the respiratory tract”). Drawing courtesy
of J. Harkema. |
Table 3
 |

Figure 10. In vivo retention of inhaled nanosized
and larger particles in alveolar macrophages (A)
and in exhaustively lavaged lungs (epithelial and
interstitial retention; B) 24 hr postexposure. The
alveolar macrophage is the most important defense
mechanism in the alveolar region for fine and coarse
particles, yet inhaled singlet NSPs are not efficiently
phagocytized by alveolar macrophages. |
Classical clearance pathways. The clearance
of deposited particles in the respiratory tract is basically
due to two processes (Table 3): a) physical translocation
of particles by different mechanisms and b) chemical
clearance processes. Leaching refers to loss of elements
from a particle matrix (e.g., loss of sodium from asbestos
fibers due to dissolution in intra- or extracellular
milieu). Chemical dissolution is directed at biosoluble
particles or components of particles that are either
lipid soluble or soluble in intracellular and extracellular
fluids. Solutes and soluble components can then undergo
absorption and diffusion or binding to proteins and other
subcellular structures and may be eventually cleared
into blood and lymphatic circulation. Chemical clearance
for biosoluble materials can happen at any location within
the three regions of the respiratory tract, although
to different degrees, depending on local extracellular
and intracellular conditions (pH). In contrast, a number
of diverse processes involving physical translocation
of inhaled particles exist in the respiratory tract and
are different in the three regions. Figure 9 summarizes
these clearance processes for solid particles. As discussed
further below, some of them show significant particle-size-dependent
differences, making them uniquely effective for a certain
particle size but very inefficient for other sizes.
The most prevalent mechanism for solid particle clearance
in the alveolar region is mediated by alveolar macrophages,
through phagocytosis of deposited particles. The success
of macrophage-particle encounter appears to be
facilitated by chemotactic attraction of alveolar macrophages
to the site of particle deposition (Warheit et al. 1988).
The chemotactic signal is most likely complement protein
5a (C5a), derived from activation of the complement cascade
from serum proteins present on the alveolar surface (Warheit
et al. 1986; Warheit and Hartsky 1993). This is followed
by gradual movement of the macrophages with internalized
particles toward the mucociliary escalator. The retention
half-time of solid particles in the alveolar region based
on this clearance mechanism is about 70 days in rats
and up to 700 days in humans. The efficacy of this clearance
mechanism depends highly on the efficiency of alveolar
macrophages to “sense” deposited particles,
move to the site of their deposition, and then phagocytize
them. This process of phagocytosis of deposited particles
takes place within a few hours, so by 6-12 hr after
deposition essentially all of the particles are phagocytized
by alveolar macrophages, to be cleared subsequently by
the slow alveolar clearance mentioned above. However,
it appears that there are significant particle-size-dependent
differences in the cascade of events leading to effective
alveolar macrophage-mediated clearance.
Figure 10 displays results of several studies in which
rats were exposed to different-sized particles (for the
3- and 10-µm particles, 10-µg and 40-µg
polystyrene beads, respectively, were instilled intratracheally)
(Kreyling et al. 2002; Oberdörster et al. 1992b,
2000; Semmler et al. 2004). Twenty-four hours later,
the lungs of the animals were lavaged repeatedly, retrieving
about 80% of the total macrophages as determined in earlier
lavage experiments (Ferin et al. 1991). As shown in Figure
10, approximately 80% of 0.5-, 3-, and 10-µm particles
could be retrieved with the macrophages, whereas only
approximately 20% of nanosized 15-20-nm and 80-nm
particles could be lavaged with the macrophages. In effect,
approximately 80% of the UFPs were retained in the lavaged
lung after exhaustive lavage, whereas approximately 20%
of the larger particles > 0.5 µm remained in
the lavaged lung. This indicates that NSPs either were
in epithelial cells or had further translocated to the
interstitium [Supplemental Material available online
(http://ehp.niehs.nih.gov/members/2005/7339/supplemental.pdf)].
Epithelial translocation. Because of the apparent
inefficiency of alveolar macrophage phagocytosis of NSPs,
one might expect that these particles interact instead
with epithelial cells. Indeed, results from several studies
show that NSPs deposited in the respiratory tract readily
gain access to epithelial and interstitial sites. This
was also shown in studies with ultrafine PTFE fumes:
shortly after a 15-min exposure, the fluorine-containing
particles could be found in interstitial and submucosal
sites of the conducting airways as well as in the interstitium
of the lung periphery close to the pleura (Oberdörster
G 2000). Such interstitial translocation represents a
shift in target site away from the alveolar space to
the interstitium, potentially causing direct particle-induced
effects there.
In a study evaluating the pulmonary inflammatory response
of TiO2 particles, ranging from NP TiO2 to
pigment-grade TiO2 (12-250 nm), a surprising
finding was that, 24 hr after intratracheal instillation
of different doses, higher doses induced a lower effect
(Oberdörster et al. 1992a). This was explained by
the additional finding that at the higher doses (expressed
as particle surface area) of the nanosized TiO2, ≥ 50%
had reached the pulmonary interstitium, causing a shift
of the inflammatory cell response from the alveolar space
to the interstitium [Supplemental Material available
online (http://ehp.niehs.nih.gov/members/2005/7339/supplemental.pdf)].
The smaller particle size of 12 and 20 nm versus 220
and 250 nm also means that the administered particle
number was more than three orders of magnitude higher
for the NSPs, a factor that seems to be an important
determinant for particle translocation across the alveolar
epithelium, as are the delivered total dose and the dose
rate (Ferin et al. 1992). Because interstitial translocation
of fine particles across the alveolar epithelium is more
prominent in larger species (dogs, nonhuman primates)
than in rodents (Kreyling and Scheuch 2000; Nikula et
al. 1997), it is reasonable to assume that the high translocation
of NSPs observed in rats occurs in humans as well [Supplemental
Material available online (http://ehp.niehs.nih.gov/members/2005/7339/supplemental.pdf)].
Translocation to the circulatory system. Once
the particles have reached pulmonary interstitial sites,
uptake into the blood circulation, in addition to lymphatic
pathways, can occur; again, this pathway is dependent
on particle size, favoring NSPs. Berry et al. (1977)
were the first to describe translocation of NSPs across
the alveolar epithelium using intratracheal instillations
of 30-nm gold particles in rats. Within 30 min postexposure,
they found large amounts of these particles in platelets
of pulmonary capillaries; the researchers suggested that
this is an elimination pathway for inhaled particles
that is significant for transporting the smallest air
pollutant particles--in particular, particles of
tobacco smoke--to distant organs. They also hypothesized
that this “might predispose to platelet aggregation
with formation of microthrombi atheromatous plaques” (Berry
et al. 1977).
Table
4
 |

Figure 11. Different forms of caveolae and cellular
tight junctions function as translocation mechanisms
across cell layers. Depending on particle surface
chemistry, NSPs have been shown to transcytose across
alveolar type I epithelial cells and capillary endothelial
cells (Table 4), but not via cellular tight junctions
in the healthy state (A). However, in a compromised
or disease state (e.g., endotoxin exposure; B) translocation
across widened tight junction occurs as well (Heckel
et al. 2004). This indicates that assessing potential
effects of NSPs in the compromised state is an important
component of nanotoxicology. Adapted from Cohen et
al. (2004). |
Table 5
 |

Figure 12. Close proximity
of olfactory mucosa to olfactory bulb of the CNS. Inhaled
NSP[s], especially
below 10 nm, deposit efficiently on the olfactory mucosa
by diffusion, similar to airborne “smell” molecules
which deposit in this area of olfactory dendritic cilia.
Subsequent uptake and translocation of solid NSP[s]
along axons of the olfactory nerve has been demonstrated
in non-human primates and rodents. Surface chemistry
of the particles may influence their neuronal translocation.
Copyright © the McGraw-Hill Companies, Inc. Reproduced
from Widmaier et al. (2004) with permission from McGraw-Hill. |
Table 6
 |
Since then, a number of studies with different particle
types have confirmed the existence of this translocation
pathway, as summarized in Table 4. Collectively, these
studies indicate that particle size and surface chemistry
(coating), and possibly charge, govern translocation
across epithelial and endothelial cell layers. In particular,
the studies summarized by Mehta et al. (2004) and those
performed by Heckel et al. (2004) using intravenous administration
of albumin-coated gold nanoparticles in rodents demonstrated
receptor-mediated transcytosis (albumin-binding proteins)
via caveolae (Figure 11). These 50-100 nm vesicles,
first described by Simionescu et al. (1975), form from
indentations of the plasmalemma and are coated with the
caveolin-1 protein. Albumin, as the most abundant protein
in plasma and interstitium, appears to facilitate NP
endocytosis, as does lecithin, a phospholipid: even 240-nm
polystyrene particles translocated across the alveolo-capillary
barrier when coated with lecithin, whereas uncoated particles
did not (Kato et al. 2003). The presence of both albumin
and phospholipids in alveolar epithelial lining fluid
may, therefore, be important constituents for facilitated
epithelial cell uptake of NSPs after deposition in the
alveolar space.
Rejman et al. (2004) reviewed a number of different
endocytic pathways for internalization of a variety of
substances, including phagocytosis, macropinocytosis,
clathrin-mediated endocytosis, and caveolae-mediated
endocytosis. They found in nonphagocytic cells in
vitro that internalization via clathrin-coated pits
prevailed for latex microspheres < 200 nm, whereas
with increasing size up to 500 nm, caveolae became the
predominant pathway. However, as shown in Table 4, surface
coating of NSPs with albumin clearly causes even the
smallest particles to be internalized via caveolae. The
presence of caveolae on cells differs: they are abundant
in lung capillaries and alveolar type l cells but not
in brain capillaries (Gumbleton 2001). In the lung, during
inspiratory expansion and expiratory contraction of the
alveolar walls, caveolae with openings around 40 nm disappear
and reappear, forming vesicles that are thought to function
as transport pathways across the cells for macromolecules
(Patton 1996). Knowledge from virology about cell entry
of biologic NSPs (viruses) via clathrin-coated pits and
caveolae mechanisms should also be considered (Smith
and Helenius 2004) and can shed light on the mechanism
by which engineered NPs may enter cells and interact
with subcellular structures.
Evidence in humans for the translocation of inhaled
NSPs into the blood circulation is ambiguous, with one
study showing rapid appearance in the blood and significant
accumulation of label in the liver of humans inhaling 99Tc-labeled
20-nm carbon particles (Nemmar et al. 2002a), whereas
another study using the same labeled particles reported
no such accumulation (Brown et al. 2002). Taking into
consideration all of the evidence from animal and human
studies for alveolar translocation of NSPs, it is likely
that this pathway also exists in humans; however, the
extent of extrapulmonary translocation is highly dependent
on particle surface characteristics/chemistry, in addition
to particle size. Translocation to the blood circulation
could provide a mechanism for a direct particle effect
on the cardiovascular system as an explanation for epidemiologic
findings of cardiovascular effects associated with inhaled
ambient UFPs (Pekkanen et al. 2002; Wichmann et al. 2000)
and for results of clinical studies showing vascular
responses to inhaled elemental carbon UFPs (Pietropaoli
et al. 2004). In addition to direct alveolar translocation
of NSPs, cardiovascular effects may also be the corollary
of a sequence of events starting with particle-induced
alveolar inflammation initiating a systemic acute phase
response with changes in blood coagulability and resulting
in cardiovascular effects (Seaton et al. 1995).
Once NSPs have translocated to the blood circulation,
they can be distributed throughout the body. The liver
is the major distribution site via uptake by Kupffer
cells, followed by the spleen as another organ of the
reticuloendothelial system, although coating with polyethylene
glycol (PEG) almost completely prevents hepatic and splenic
localization so that other organs can be targeted (Akerman
et al. 2002). Distribution to heart, kidney, and immune-modulating
organs (spleen, bone marrow) has been reported. For example,
several types of NPs, ranging from 10 to 240 nm, localized
to a significant degree in bone marrow after intravenous
injection into mice (Table 5). Such target specificity
may be extremely valuable for drug delivery; for example,
drug delivery to the CNS via blood-borne NPs requires
NP surface modifications in order to facilitate translocation
across the tight blood-brain barrier via specific
receptors (e.g., apolipoprotein coating for LDL-receptor-mediated
endocytosis in brain capillaries) (Kreuter 2001, 2004;
Kreuter et al. 2002). Such highly desirable properties
of NPs must be carefully weighed against potential adverse
cellular responses of targeted NP drug delivery, and
a rigorous toxicologic assessment is mandatory [Supplemental
Material available online (http://ehp.niehs.nih.gov/members/2005/7339/supplemental.pdf)].
Neuronal uptake and translocation. A translocation
pathway for solid particles in the respiratory tract
involving neuronal axons is apparently specific for NSPs.
Respective studies are summarized in Table 6. This pathway
was described > 60 years ago, yet it has received
little or no attention from toxicologists. This pathway,
shown in Figure 9 for the nasal and tracheobronchial
regions, comprises sensory nerve endings of the olfactory
and the trigeminus nerves and an intricate network of
sensory nerve endings in the tracheobronchial region.
These early studies concerned a large series of studies
with 30-nm polio virus intranasally instilled into chimpanzees
and rhesus monkeys (Bodian and Howe 1941a, 1941b; Howe
and Bodian 1940). Their studies revealed that the olfactory
nerve and olfactory bulbs are, indeed, portals of entry
to the CNS for intranasally instilled nanosized polio
virus particles, which could subsequently be recovered
from the olfactory bulbs. The close proximity of nasal
olfactory mucosa and olfactory bulb requires only a short
distance to be covered by neuronal transport (Figure
12). Bodian and Howe (1941b) determined the transport
velocity of the virus in the axoplasm of axons to be
2.4 mm/hr, which is very well in agreement with neuronal
transport velocities measured later by Adams and Bray
(1983) for solid particles (up to 500 nm) directly microinjected
into giant axons of crabs, and by de Lorenzo (1970) for
silver-coated colloidal gold (50 nm) in squirrel monkeys.
The de Lorenzo (1970) study demonstrated in squirrel
monkeys that intranasally instilled silver-coated colloidal
gold particles (50 nm) translocated anterogradely in
the axons of the olfactory nerves to the olfactory bulbs.
The 50-nm gold particles even crossed synapses in the
olfactory glomerulus to reach mitral cell dendrites within
1 hr after intranasal instillation. An interesting finding
in this study--and important for potential adverse
effects--was that the NSPs in the olfactory bulb
were no longer freely distributed in the cytoplasm but
were preferentially located in mitochondria (see also “Reactive
oxygen species mechanisms of NSP toxicity,” above).

Figure 13. Occlusion
of the right nostril of rats during 6-hr inhalation
of nanosized MnO2 particles
(~ 30 nm CMD, ~ 450 µg/m3) resulted in accumulation
of Mn only in the left olfactory bulb only at 24
hr after dosing. Data are mean ± SD. Data
from Feikert et al. (2004). |
Table 7
 |
Newer studies indicated that this translocation pathway
is also operational for inhaled NSPs. Inhalation of elemental 13C
UFPs (CMD = 35 nm) resulted in a significant increase
of 13C in the olfactory bulb on day 1, which
increased further throughout day 7 postexposure (Oberdörster
et al. 2004). Results of another inhalation study with
solid nanosized (CMD = 30 nm) manganese oxide (MnO 2)
particles in rats showed after a 12-day exposure a more
than 3.5-fold significant increase of Mn in the olfactory
bulb, compared with only a doubling of Mn in the lung.
When one nostril was occluded during a 6-hr exposure,
Mn accumulation in the olfactory bulb was restricted
to the side of the open nostril only (Figure 13) (Feikert
et al. 2004). This result contrasts with 15-day inhalation
of larger-sized MnO 2 particles in rats (1.3
and 18 µm mass median aerodynamic diameter) where
no significant increases in olfactory Mn was found
(Fechter et al. 2002). This was to be expected given
that the
individual axons of the fila olfactoria (forming the
olfactory nerve) are only 100-200 nm in diameter
(de Lorenzo 1957; Plattig 1989).
Collectively, these studies point out that the olfactory
nerve pathway should also be considered a portal of entry
to the CNS for humans under conditions of environmental
and occupational exposures to airborne NSPs. However,
there are important differences between rodents and humans.
The olfactory mucosa of the human nose comprises only
5% of the total nasal mucosal surface as opposed to 50%
in rats--which in addition are obligatory nose breathers
(Table 7). One can argue that the olfactory route may
therefore be an important transfer route to the CNS for
inhaled NSPs in animals with a well-developed olfaction
system, yet at the same time its importance for humans
with a more rudimentary olfactory system can be questioned.
However, estimates using a predictive particle deposition
model and data from Table 7 show that concentrations
of 20-nm translocated particles in the human olfactory
bulb can, indeed, be 1.6-10 times greater than
in rats [Supplemental Material available online (http://ehp.niehs.nih.gov/members/2005/7339/supplemental.pdf)].
Translocation into deeper brain structures may possibly
occur as well, as shown in rats for soluble Mn (Gianutsos
et al. 1997), but requires further confirmatory studies
with respect to solid NSPs. Further evidence for movement
of NSPs along axons and dendrites in humans is provided
by knowledge accumulated by virologists who have long
understood the movement of human meningitis virus through
olfactory and trigeminal neurons and, similarly, herpes
virus movement up and down the trigeminal neuron to trigger
outbreaks of herpes cold sores in humans (Kennedy and
Chaudhuri 2002; Terasaki et al. 1997).
There are additional neuronal translocation pathways
for solid NSPs via the trigeminus nerve and tracheobronchial
sensory nerves (Table 6). A study by Hunter and Dey (1998)
in rats demonstrated the translocation of intranasally
instilled rhodamine-labeled microspheres (20-200
nm) to the trigeminal ganglion inside the cranium via
uptake into the ophthalmic and maxillary branches of
the trigeminus nerve that supplies sensory nerve endings
throughout the nasal mucosa. In another study, Hunter
and Undem (1999) instilled the same microparticles intratracheally
into guinea pigs; they found neuronal translocation of
these solid microparticles to the ganglion nodosum in
the neck area that is networked into the vagal system.
This finding may be relevant for ambient UFPs because
it can be hypothesized that cardiovascular effects associated
with ambient particles in epidemiologic studies (Utell
et al. 2002) are in part due to direct effects of translocated
UFPs on the autonomic nervous system via sensory nerves
in the respiratory tract.
In the context of potential CNS effects of air pollution,
including ambient UFPs, two recent studies with exposures
of mice to concentrated ambient fine particles and UFPs
should be mentioned. Campbell et al. (2005) and Veronesi
et al. (in press) found significant increases of tumor
necrosis factor- or
decreases in dopaminergic neurons, supporting the hypothesis
of ambient PM causing neurodegenerative disease. A study
by Calderon-Garcidueñas et al. (2002) may also
point to an interesting link between air pollution and
CNS effects: these authors described significant inflammatory
or neurodegenerative changes in the olfactory mucosa,
olfactory bulb, and cortical and subcortical brain structures
in dogs from a heavily polluted area in Mexico City,
whereas these changes were not seen in dogs from a less-polluted
rural control city. However, whether direct effects of
airborne UFPs are the cause of these effects remains
to be determined. or
decreases in dopaminergic neurons, supporting the hypothesis
of ambient PM causing neurodegenerative disease. A study
by Calderon-Garcidueñas et al. (2002) may also
point to an interesting link between air pollution and
CNS effects: these authors described significant inflammatory
or neurodegenerative changes in the olfactory mucosa,
olfactory bulb, and cortical and subcortical brain structures
in dogs from a heavily polluted area in Mexico City,
whereas these changes were not seen in dogs from a less-polluted
rural control city. However, whether direct effects of
airborne UFPs are the cause of these effects remains
to be determined.
Although the existence of neuronal translocation of
NSPs has been well established, size alone is only one
particle parameter governing this process. Surface characteristics
of NSPs (chemistry, charge, shape, aggregation) are essential
determinants as well, and it should not be assumed that
all NSPs, when inhaled, will be distributed by the mechanism
described here. It should be kept in mind, however, that
the unique biokinetic behavior of NSPs--cellular
endocytosis, transcytosis, neuronal and circulatory translocation
and distribution--which makes them desirable for
medical therapeutic or diagnostic applications--may
be associated with potential toxicity. For example, NP-facilitated
drug delivery to the CNS raises the question of the fate
of NPs after their translocation to specific cell types
or to subcellular structures in the brain. For example,
does mitochondrial localization induce oxidative stress?
How persistent is the coating or the core of the NPs?
A respective safety evaluation is key [Supplemental Material
available online (http://ehp.niehs.nih.gov/members/2005/7339/supplemental.pdf)].
Exposure via GI Tract and Skin
NSPs cleared from the respiratory tract via the mucociliary
escalator can subsequently be ingested into the GI tract.
Alternatively, nanomaterials can be ingested directly,
for example, if contained in food or water or if used
in cosmetics or as drugs or drug delivery devices. Only
a few studies have investigated the uptake and disposition
of nanomaterials by the GI tract, and most have shown
that NSPs pass through the GI tract and are eliminated
rapidly. In rats dosed orally with radiolabeled functionalized
C60 fullerenes, water solubilized using PEG
and albumin (18 kBq in 100 µL), 98% were cleared
in the feces within 48 hr, whereas the rest was eliminated
via urine, indicating some uptake into the blood circulation
(Yamago et al. 1995). In contrast, in this same study,
90% of the same radiolabeled fullerenes administered
intravenously (9.6 kBq in ~ 50 µL or 14-18
kBq in 215 µL) were retained after 1 week, with
most (73-80%, depending on time course) found in
the liver. Studies by Kreyling and colleagues (Kreyling
et al. 2002; Semmler et al. 2004) using ultrafine 192Ir
did not show significant uptake in the GI tract, whereas
earlier studies with larger TiO2 particles
(150-500 nm) found uptake into the blood and movement
to the liver (Böckmann et al. 2000; Jani et al.
1994). Likely there are differences in GI tract uptake
dependent on both particle surface chemistry and particle
size. Indeed, after oral dosing in rats, Jani et al.
(1990) found a particle-size-dependent uptake of
polystyrene particles (ranging from 50 to 3,000 nm) by
the GI mucosa. This uptake (6.6% of the administered
50 nm, 5.8% of the 100 nm NSP, 0.8% of 1 µm particles,
and 0% for 3 µm particles) was mainly via the Peyer's
patches with translocation into the mesenteric lymph
and then to systemic organs (i.e., liver, spleen, blood,
bone marrow, and kidney).

Figure 14. The epidermis represents a tight barrier
against NSP penetration. Quantitatively, dermal translocation
will therefore be minimal or nonexistent under normal
conditions but increases in areas of skin flexing
(Tinkle et al. 2003) and broken skin. Once in the
dermis, lymphatic uptake is a major translocation
route, likely facilitated by uptake in dendritic
cells (epidermis) and macrophages; other potential
pathways may include the dense networks of blood
circulation and sensory nerves in the dermis. Adapted
from Essential Day Spa (2005) with permission from
www.essentialdayspa.com. |
A potentially important uptake route is through dermal
exposure. The epidermis, consisting of the outer horny
layer (stratum corneum), the prickle cell layer (stratum
spinosum), and basal cell layer (stratum basale), forms
a very tight protective layer for the underlying dermis
(Figure 14). The dermis has a rich supply of blood
and tissue macrophages, lymph vessels, dendritic cells
(Langerhans,
also in stratum spinosum of epidermis), and five different
types of sensory nerve endings. Broken skin represents
a readily available portal of entry even for larger
(0.5-7 µm)
particles, as evidenced by reports about accumulation
of large amounts of soil particles in inguinal lymph
nodes of people who often run or walk barefoot; this
can be associated with elephantiatic lymphedema (podoconiosis;
Corachan et al. 1988; Blundell et al. 1989). Tinkle et
al. (2003) hypothesized that unbroken skin when flexed--as
in wrist movements--would make the epidermis permeable
for NSPs. They demonstrated in a proof-of-concept experiment
that, indeed, flexing the skin, but not flat skin, resulted
in penetration of even 1 µm fluorescent beads to
the dermis. The follow-up question about access of particles
in the dermis to the circulation is answered by the aforementioned
reports of podoconiosis, that is, uptake into the lymphatic
system and regional lymph nodes. Subsequent translocation
of NSPs beyond lymph nodes to the blood circulation is
likely to occur as well, as shown in studies with small
asbestos fibers (Oberdörster et al. 1988).
Newer studies by Kim et al. (2004) in mice and pigs
with intradermally injected near-infrared quantum dots
confirmed that NPs, once in the dermis, will localize
to regional lymph nodes, which makes these particles
very useful for invivo imaging. Likely
transport mechanisms to the lymph nodes are skin macrophages
and dendritic (Langerhans) cells (Ohl et al. 2004; Sato
et al. 1998); this raises a question about potential
modulation of immune responses, after interaction of
these NP-containing macrophages and dendritic cells with
T lymphocytes. For example, Chen et al. (1998) were able
to raise antibodies in mice specific for C60 after
intraperitoneal injections of C60 conjugated
to thyroglobulin and serum albumin. Clearly, research
is needed to determine whether and under what conditions
NPs can be recognized by the immune system, following
any route of uptake into the organism.
Another question relates to the potential of sensory
skin nerves to take up and translocate NPs. Given that
this mechanism has been demonstrated for the nasal and
tracheobronchial regions of the respiratory tract, how
likely is this to occur in the dermis layer of the skin
with its dense supply of different types of sensory nerves?
It may be conceivable, considering data on neuronal uptake
and translocation of NSPs after intramuscular injection.
For example, nanosized ferritin and iron-dextran, after
injection into the tongue of mice, labeled the neurons
of the hypoglossal nuclei, and injection of both of these
NSPs into facial muscles of mice also resulted in synaptic
uptake; cationized ferritin was also detected in cell
bodies of facial neurons, indicating that electrical
charge is of importance for incorporation into axons
and axonal transport (Arvidson 1994; Malmgren et al.
1978; Olsson and Kristensson 1981). Other studies using
intramuscular injection of ferritin (~ 112 nm), iron-dextran
(11 or 21 nm), and gold protein (20-25 nm) NSPs
also showed rapid penetration through the basal lamina
into the synaptic clef of the neuromuscular junction,
but this was restricted to only the smaller nanoparticles,
implying that there may be a size-dependent penetration
of the basal lamina with a threshold somewhere between
10 and 20 nm (Oldfors and Fardeau 1983).
Neuronal transport of NSPs along sensory skin nerves
is well established for herpes virus. After passing through
the skin--especially broken skin--the viruses
are transported retrogradely along dendrites of sensory
neurons to the dorsal root ganglion, where they remain
dormant until a stress situation triggers anterograde
translocation along the dendrites back to the skin (Kennedy
and Chaudhuri 2002; Terasaki et al. 1997). Future studies
need to determine whether and to what degree such translocation
along sensory skin neurons also occurs with NPs penetrating
the epidermis.
The lack of toxicology data on engineered NPs does
not allow for adequate risk assessment. Because of this,
some may even believe that engineered NPs are so risky
that they call for a precautionary halt in NP-related
research. However, the precautionary principle should
not be used to stop research related to nanotechnology
and NPs. Instead, we should strive for a sound balance
between further development of nanotechnology and the
necessary research to identify potential hazards in order
to develop a scientifically defensible database for the
purpose of risk assessment. To be able to do this, a
basic knowledge about mammalian and ecotoxicologic profiles
of NPs is necessary, rather than attempting to assess
NP risks based on some popular science fiction literature.
Most important, sufficient resources should be allocated
by governmental agencies and industries to be able to
perform a scientifically based risk assessment and then
establish justifiable procedures for risk management.
The data needed for this risk assessment should be determined
a priori so that limited resources can be used efficiently
to develop useful and well-planned studies.
At this point, governmental regulation is not possible,
given the lack of needed information on which to base
such regulations. However, academia, industry, and regulatory
governmental agencies should seriously consider the view
that NPs have new and unique biologic properties and
that the potential risks of NPs are not the same as those
of the bulk material of the same chemistry. Assigning
a unique identifier to nanosized materials would indicate
that the toxicology profile of the material in question
may not be the same as the bulk material. Toxicologic
tests and the resulting database would provide information
for material safety data sheets for NPs as well as a
basis for potential NP risk assessments and risk management.
Obviously, this approach may not be appropriate for all
NPs, for example, when embedded in a matrix, and the
feasibility of this proposed strategy needs to be thoroughly
discussed and considered. For discussing this, and for
developing and deciding upon a reasonable battery of
tests for toxicologic profiling, it would be very useful
to convene international multidisciplinary workshops
of experts from industry, academia, and regulatory agencies
(including material scientists, chemists, chemical engineers,
toxicologists, physicians, regulators, statisticians,
and others) to establish an NP classification scheme
and testing guidelines. A multidisciplinary and multinational
collaborative team approach is critical. Respective efforts
have been initiated nationally by the American National
Standards Institute (ANSI 2004) and internationally by
the International Council on Nanotechnology (ICON 2004)
as well as the International Organization for Standardization
(Geneva, Switzerland).

Figure 15. Risk assessment
(NRC 1983) and risk management paradigm for NPs.
Risk assessment requires
answers to the following questions: Do NPs have adverse
effects? What are the dose-response relationships?
What are occupational/environmental levels in different
media? What is the calculated risk? Once a risk is
determined, a risk management decision can be established,
including exposure standards and regulations and
efforts for effective risk communication. Modified
from Oberdörster (1994). |
Because many regulatory agencies do not consider a
nanotechnologically manufactured substance different
from the conventional substance, the manufacture and
use of nanotechnology products are currently not specifically
regulated. Typically, nanosized substances are treated
as variations of the technical material or existing formulation
and thus do not require a separate registration. A main
reason for producing a nanosize form of a registered
substance, however, is that conversion of a substance
to a nanoparticle imparts new properties to the substance
(e.g., enhanced mechanical, electrical, optical, catalytic,
biologic activity). Thus, as stated above, although the
toxicology of the base material may be well defined,
the toxicity of the nanosize form of the substance may
be dramatically different from its parent form. As a
result, new toxicology data on the nanosize form of a
substance is likely to result in a different hazard assessment
for the NPs. Figure 15 shows a risk assessment/risk management
paradigm that points out different steps and data required
for this process.
As described in the preceding sections, the difference
in toxicologic profile of NPs compared with its parent
form is due to not only its intrinsic chemical properties
but also to a large degree to its differing kinetics in
vivo. Although larger particles may not enter the
CNS, the potential exists for inhaled NSPs to be translocated
to the CNS via the axons of sensory neurons in the upper
respiratory tract. Furthermore, although the toxicity
per unit mass of a particular substance may vary depending
on the nano versus larger form, it will be important
to take into account not only new biologic activities
but also potential new target organs and routes of exposure.
To what degree does the nanoform of a substance have
enhanced dermal penetration, or increased systemic uptake
via the lung or GI tract? What determines how many nanoparticles
that enter the systemic circulation will distribute throughout
the body, reach the bone marrow, cross the blood-brain
barrier, cross the placenta to affect the developing
offspring, or sequester effectively in the liver? Do
nanoparticles released into the environment affect species
that are important in food chain dynamics? What are the
long-term consequences of exposure to nanoparticles?
Changes in toxicity profile and new target organs can
be expected, and it will then be necessary to establish
new risk assessments for nanoparticles in addition to
the bulk material. Currently there exists a paucity of
data to effectively address these questions, but it will
be important to determine whether there exist common
modes of action/behavior of NPs to establish baseline
assumptions for use in risk assessments.
The use of nanotechnology products will likely increase
dramatically over the next decade. In fact, nanomaterials
are already being used in applications ranging from burn
and wound dressings to dental-bonding agents to sunscreens
and cosmetics to fuel cells, tires, optics, clothing,
and electronics. Although currently there exists little
public awareness of nanotechnology in everyday life (e.g.,
stain-free clothing), it would be prudent to examine
and address environmental and human health concerns before
the widespread adoption of nanotechnology. Both the societal
benefits and potential risks of nanotechnology should
be evaluated and clearly communicated to the general
public and regulators. This type of open communication
and risk/benefit evaluation will avoid the pitfalls encountered
with genetically modified organisms recently experienced
in the field of biotechnology. In that instance, the
benefits of the emerging field of biotechnology were
not communicated effectively before the introduction
of the technology. As the public’s awareness of
this new technology grew, regulators and producers of
biotechnology failed to effectively acknowledge public
concerns that genetically modified organisms could adversely
affect ecosystem balance. As a result, the public support
of genetically modified organisms, particularly in the
European Union, is low. For nanomaterial producers, it
will be important to demonstrate that what they may perceive
as a new and potentially harmless form of a familiar
material has, indeed, an acceptable risk profile. If
such proactive steps are not taken, nanomaterials may
be regarded as dangerous by the public and regulators,
which could lead to inappropriate categorization and
unnecessarily burdensome regulations. Such action (or
inaction on the side of producers), in turn, could result
in significant barriers to commercialization and the
widespread acceptance of otherwise useful nanotechnology
materials.

Figure 16. Biokinetics of NSPs. PNS, peripheral
nervous system. Although many uptake and translocation
routes have been demonstrated, others still are hypothetical
and need to be investigated. Translocation rates
are largely unknown, as are accumulation and retention
in critical target sites and their underlying mechanisms.
These, as well as potential adverse effects, largely
depend on physicochemical characteristics of the
surface and core of NSPs. Both qualitative and quantitative
changes in NSP biokinetics in a diseased or compromised
organism also need to be considered. |
Research on ambient UFPs has laid the foundation for
the emerging field of nanotoxicology, with the goal of
studying the biokinetics and the potential of engineered
nanomaterials (particles, tubes, shells, quantum dots,
etc.) to cause adverse effects. Major differences between
ambient UFPs and NPs are the polydisperse nature of the
former versus the monodisperse size of the latter, and
particle morphology, oftentimes a branched structure
from combustion particles versus spherical form of NPs,
although other shapes (tubes, wires, rings, planes) are
also manufactured. In addition, combustion-derived volatile
organic compounds and inorganic constituents (e.g., metals,
nitrates, sulfates) of different solubilities on UFPs
predict differences in the toxicologic profile between
UFPs and NPs. However, as far as the insoluble particle
is concerned, concepts of NSPs kinetics, including cell
interactions, will most likely be the same for UFPs and
NPs (Figure 16).
The introduction of nanostructured materials for biomedical
and electronics applications opens tremendous opportunities
for biomedical applications as therapeutic and diagnostic
tools as well as in the fields of engineering, electronics,
optics, consumer products, alternative energy, soil/water
remediation, and others. However, very little is yet
known about their potential to cause adverse effects
or humoral immune responses once they are introduced
into the organism--unintentionally or intentionally.
Nanomedicine products will be well tested before introduction
into the marketplace. However, for the manufacturers
of most current nanotechnology products, regulations
requiring nanomaterial-specific data on toxicity before
introduction into the marketplace are an evolving area
and presently under discussion (Bergeson and Auerbach
2004; Foresight and Governance Project 2003). During
a product’s life cycle (manufacture, use, disposal),
it is probable that nanomaterials will enter the environment,
and currently there is no unified plan to examine ecotoxicologic
effects of NPs. In addition, the stability of coatings
and covalent surface modifications need to be determined
both in ecologic settings and in vivo. [Supplemental
Material available online (http://ehp.niehs.nih.gov/members/2005/7339/supplemental.pdf).]
Results of older biokinetic studies and some new toxicology
studies with NSPs (mostly ambient UFPs) can be viewed
as the basis for the expanding field of nanotoxicology.
These studies showed that the greater surface area per
mass renders NSPs more active biologically than larger-sized
particles of the same chemistry, and that particle surface
area and number appear to be better predictors for NSPs-induced
inflammatory and oxidative stress responses. The following
emerging
concepts of nanotoxicology can be identified from these studies:
The biokinetics of NSPs are different from larger particles.
When inhaled, they are efficiently deposited in all regions
of the respiratory tract; they evade specific defense
mechanisms; and they can translocate out of the respiratory
tract via different pathways and mechanisms (endocytosis
and transcytosis). When in contact with skin, there is
evidence of penetration to the dermis followed by translocation
via lymph to regional lymph nodes. A possible uptake
into sensory nerves needs to be investigated. When ingested,
systemic uptake via lymph into the organism can occur,
but most are excreted via feces. When in blood circulation,
they can distribute throughout the organism, and they
are taken up into liver, spleen, bone marrow, heart,
and other organs. In general, translocation rates are
largely unknown; they are probably very low but are likely
to change in a compromised/diseased state.
The biologic activity and biokinetics are dependent
on many parameters: size, shape, chemistry, crystallinity,
surface properties (area, porosity, charge, surface modifications,
weathering of coating), agglomeration state, biopersistence,
and dose. These parameters are likely to modify responses
and cell interactions, such as a greater inflammatory
potential than larger particles per given mass, translocation
across epithelia from portal of entry to other organs,
translocation along axons and dendrites of neurons, induction
of oxidative stress, pro-oxidant and antioxidant activity
of NSPs in environmentally relevant species, binding
to proteins and receptors, and localization in mitochondria.
The principles of cellular and organismal interactions
discussed in this article should be applicable for both
ambient UFPs and NPs, even if the latter are coated with
a biocompatible material. Knowledge about the biopersistence
of this coating is as essential as is knowledge about
the bioavailability of the core material that could have
intrinsic toxic properties, for example, semiconductor
metal compounds in sub-10-nm quantum dots consisting
of cadmium and lead compounds. The very small size of
these materials makes them available to the same translocation
processes described here for polydisperse NSPs, possibly
even in a more efficient way because of their uniform
size. When studying biologic/toxicologic effects, new
processes of interactions with subcellular structures
(e.g., microtubuli, mitochondria) will likely be discovered.
The diversity of engineered nanomaterials and of the
potential effects represents major challenges and research
needs for nanotoxicology, including also the need for
assessing human exposure during manufacture and use.
The goal to exploit positive aspects of engineered nanomaterials
and avoid potential toxic effects can best be achieved
through a multidisciplinary team effort involving researchers
in toxicology, materials science, medicine, molecular
biology, bioinformatics, and their subspecialties.
Correction
The authors found additional
information on GI tract uptake of NSPs that
was not in
the original
manuscript published online. This information
has been included here in “Exposure via
GI Tract and Skin.”
|
|
|
 |
| [References Listed in PubMed] References
Adams RJ, Bray D. 1983. Rapid transport of foreign
particles microinjected into crab axons. Nature 303:
718-720.
Akerman MA, Chan WCW, Laakkonen P, Bhatia SN, Ruoslahti
E. 2002. Nanocrystal targeting in vivo. Proc Natl Acad
Sci USA 99:12617-12621.
Amato I. 1989. Making the right stuff. Sci News 136:108-110.
Anderson PJ, Wilson JD, Hiller FC. 1990. Respiratory
tract deposition of ultrafine particles in subjects with
obstructive or restrictive lung disease. Chest 97: 1115-1120.
ANSI. 2004. Home page. American National Standards
Institute. Available: http://www.ansi.org. [accessed
16 March 2005].
Arvidson B. 1994. A review of axonal transport of metals.
Toxicology 88:1-14.
Auclair F, Baudot P, Beiler D, Limasset
J. 1983. Accidents benines et mortetels dus aux “traitement” du
polytetrafluoroethylene en millieu industriel: observations
cliniques et measures physio-chemiques des atmosphere
pollues. Toxicol Eur Res 1:43-48.
Ballou B, Lagerholm BC, Ernst LA, Bruchez MP, Waggoner
AS. 2004. Non-invasive imaging of quantum dots in mice.
Bioconjug Chem 15:79-86.
Bazile DV, Ropert C, Huve P, Verrecchia T, Marlard
M, Frydman A, et al. 1992. Body distribution of fully
biodegradable [14C]-poly(lactic acid) nanoparticles coated
with albumin after parenteral administration to rats.
Biomaterials 13(15):1093-1102.
Bergeson LL, Auerbach B. 2004. The Environmental Regulatory
Implications of Nanotechnology. Daily Environment Report
No. 71. Washington, DC:Bureau of National Affairs, Inc.
Available: http://www.lawbc.com/other_pdfs/The%20Environmental%20Regulatory%20Implications%20of%20Nanotechnology.pdf [accessed 26 April 2005].
Berry JP, Arnoux B, Stanislas G, Galle P, Chretien
J. 1977. A microanalytic study of particles transport
across the alveoli: role of blood platelets. Biomedicine
27: 354-357.
Blakemore R. 1975. Magnetotactic bacteria. Science
190:377-379.
Blundell G, Henderson WJ, Price EW. 1989. Soil particles
in the tissues of the foot in endemic elephantiasis of
the lower legs. Ann Trop Med Parasitol 83 (4): 381-385.
Böckmann J, Lahl H, Eckert T,
Unterhalt B. 2000. Titan-Blutspiegel vor und nach Belastungsversuchen
mit
Titandioxid [in German]. Pharmazie 55: 140-143.
Bodian D, Howe HA. 1941a. Experimental studies on intraneural
spread of poliomyelitis virus. Bull Johns Hopkins Hosp
69:248-267.
Bodian D, Howe HA. 1941b. The rate of progression of
poliomyelitis virus in nerves. Bull Johns Hopkins Hosp
69:79-85.
Brand P, Gebhart J, Below M, Georgi B, Heyder J. 1991.
Characterization of environmental aerosols on Helgoland
Island. Atmos Environ 25A(3/4):581-585.
Brown DM, Stone V, Findlay P, MacNee W, Donaldson K.
2000. Increased inflammation and intracellular calcium
caused by ultrafine carbon black is independent of transition
metals or other soluble components. Occup Environ Med
57:685-691.
Brown DM, Wilson MR, MacNee W, Stone V, Donaldson K.
2001. Size-dependent proinflammatory effects of ultrafine
polystyrene particles: a role for surface area and oxidative
stress in the enhanced activity of ultrafines. Toxicol
Appl Pharmacol 175:191-199.
Brown JS, Zeman KL, Bennett WD. 2002. Ultrafine particle
deposition and clearance in the healthy and obstructed
lung. Am J Respir Crit Care Med 166:1240-1247.
Cagle DW, Kenmnel SJ, Mirzadeh S, Alford JM, Wilson
LJ. 1999. In vivo studies of fullerene-based materials
using endohedral metallofullerene radiotracers. Proc
Natl Acad Sci USA 96:5182-5187.
Calderon-Garcidueñas L, Azzarelli
B, Acuna H, Garcia R, Gambling TM, Osnaya N, et al.
2002. Air pollution
and brain damage. Toxicol Pathol 30(3):373-389.
Campbell A, Oldham M, Becaria A, Bondy SC, Meacher
D, Sioutas C, et al. 2005. Particulate matter in polluted
air may increase biomarkers of inflammation in mouse
brain. Neurotoxicology 26:133-140.
Cavagna G, Finulli M, Vigliani EC. 1961. Studio sperimentale
sulla patogenesi della febre da inalazione di fumi di
Teflon (politetrafluoroetilene) [in Italian]. Med Lavoro
52:251-261.
Chalupa DC, Morrow PE, Oberdörster
G, Utell MJ, Frampton MW. 2004. Ultrafine particle
deposition in subjects
with asthma. Environ Health Perspect 112: 879-882.
Chen B, Wilson S, Das M, Coughlin D, Erlanger B. 1998.
Antigenicity of fullerenes: antibodies specific for fullerenes
and their characteristics. Proc Natl Acad Sci USA 95:10809-10813.
Cheng X, Kan A, Tomson M. 2004. Napthalene adsorption
and desorption from aqueous C60 fullerene. J Chem Engineer
Data 49:675-678.
Chitose N, Ueta S, Seino S, Yamamoto T. 2003. Radiolysis
of aqueous phenol solutions with nanoparticles. 1. Phenol
degradation and TOC removal in solutions containing TiO2 induced
by UV, gamma-ray and electron beams. Chemosphere 50:1007-1013.
Cohen A, Hnasko R, Schubert W, Lisanti M. 2004. Role
of caveolae and caveolins in health and disease. Physiol
Rev 84:1341-1379.
Coleman WE, Scheel LD, Gorski CH. 1968. The particle
resulting from polytetrafluorethylene (PTFE) pyrolysis
in air. AIHA J 29:54-60.
Corachan M, Tura JM, Campo E, Soley M, Traveria A.
1988. Poedoconiosis in Aequatorial Guinea. Report of
two cases from different geological environments. Trop
Geogr Med 40:359-364.
Cyrys J, Stolzel M, Heinrich J, Kreyling WG, Menzel
N, Wittmaack K, et al. 2003. Elemental composition and
sources of fine and ultrafine ambient particles in Erfurt,
Germany. Sci Tot Environ 305:143-156.
Daughton C, Ternes T. 1999. Pharmaceuticals and personal
care products in the environment: agents of subtle change?
Environ Health Perspect 107(suppl 6): 907-938.
de Lorenzo AJ. 1957. Electron microscopic observations
of the olfactory mucosa and olfactory nerve. J Biophys
Biochem Cytol 3:839-850.
de Lorenzo AJ. 1970. The olfactory neuron and the blood-brain
barrier. In: Taste and Smell in Vertebrates (Wolstenholme
G, Knight J, eds). London:Churchill, 151-176.
Derfus AM, Chan WCW, Bhatia SN. 2004. Probing the cytotoxicity
of semiconductor quantum dots. Nano Lett 4(1):11-18.
Dieckmann G, Dalton A, Johnson P, Razal J, Chen J,
Giordano GM, et al. 2003. Controlled assembly of carbon
nanotubes by designed amphiphilic peptide helices. J
Am Chem Soc 125:1770-1777.
Dobson J. 2001. Nanoscale biogenic iron oxides and
neurodegenerative disease. FEBS Lett 496:1-5.
Donaldson K, Brown D, Clouter A, Duffin R, MacNee W,
Renwick L, et al. 2002. The pulmonary toxicology of ultrafine
particles. J Aerosol Med 15:213-220.
Donaldson K, Li XY, MacNee W. 1998. Ultrafine (nanometre)
particle mediated lung injury. J Aerosol Sci 29:553-560.
Donaldson K, Stone V. 2003. Current hypotheses on the
mechanisms of toxicity of ultrafine particles. Ann Ist
Super Sanita 39:405-410.
Donaldson K, Tran C-L. 2002. Inflammation caused by
particles and fibers. Inhal Toxicol 14:5-27.
Donlin M, Frey R, Putnam C, Proctor J, Bashkin J. 1998.
Analysis of iron in ferritin, the iron-storage protein.
J Chem Educ 75:437-441.
Driscoll KE. 1996. Role of inflammation in the development
of rat lung tumors in response to chronic particle exposure.
Inhal Toxicol 8(suppl):139-153.
Dunn JR, Fuller M, Zoeger J, Dobson J, Heller F, Hamnmann
J, et al. 1995. Magnetic material in the human hippocampus.
Brain Res Bull 36:149-153.
Elder ACP, Gelein R, Azadniv M, Frampton M, Finkelstein
J, Oberdorster G. 2002. Systemic interactions between
inhaled ultrafine particles and endotoxin. Ann Occup
Hyg 46(suppl 1):231-234.
Elder ACP, Gelein R, Azadniv M, Frampton M, Finkelstein
J, Oberdorster G. 2004. Systemic effects of inhaled ultrafine
particles in two compromised, aged rat strains. Inhal
Toxicol 16(6/7):461-471.
Elder ACP, Gelein R, Finkelstein
JN, Cox C, Oberdörster
G. 2000. Pulmonary inflammatory response to inhaled ultrafine
particles is modified by age, ozone exposure, and bacterial
toxin. Inhal Toxicol 12(suppl 4):227-246.
Essential Day Spa. 2005. Skin Anatomy and Physiology.
Available: http://www.essentialdayspa.com/Skin_Anathomy_and_Physiology.htm [accessed 26 April 2005].
Evelyn A, Mannick S, Sermon PA. 2003. Unusual carbon-based
nanofibers and chains among diesel-emitted particles.
Nano Lett 3:63-64.
Fechter LD, Johnson DL, Lynch RA. 2002. The relationship
of particle size to olfactory nerve uptake of a non-soluble
form of manganese into brain. Neurotoxicology 23: 177-183.
Feikert T, Mercer P, Corson N, Gelein R, Opanashuk
L, Elder A, et al. 2004. Inhaled solid ultrafine particles
(UFP) are efficiently translocated via neuronal naso-olfactory
pathways [Abstract]. Toxicologist 78(suppl 1): 435-436.
Ferin J, Oberdörster G. 1992.
Translocation of particles from pulmonary alveoli into
the interstitium.
J Aerosol Med 5:179-187.
Ferin J, Oberdörster G, Penney
DP. 1992. Pulmonary retention of ultrafine and fine
particles in rats. Am
J Resp Cell Mol Biol 6:535-542.
Ferin J, Oberdörster G, Penney
DP, Soderholm SC, Gelein R, Piper HC. 1990. Increased
pulmonary toxicity
of ultrafine particles? I. Particle clearance, translocation,
morphology. J Aerosol Sci 21:381-384.
Ferin J, Oberdörster G, Soderholm
SC, Gelein R. 1991. Pulmonary tissue access of ultrafine
particles.
J Aerosol Med 4(1):57-68.
Foley S, Crowley C, Smaihi M, Bonfils C, Erlanger BF,
Seta P, et al. 2002. Cellular localisation of a water-soluble
fullerene derivative. Biochem Biophys Res Commun 294:116-119.
Foresight and Governance Project.
2003. Nanotechnology & Regulation:
A Case Study using the Toxic Substance Control Act (TSCA):
A Discussion Paper. Publication 2003-6. Washington, DC:Woodrow
Wilson International Center for Scholars. Available: http://www.nanotechcongress.com/Nanotech-Regulation.pdf [accessed 26 April 2005].
Fox JE, Starcevic M, Kow KY, Burow ME, McLachlan JA.
2001. Nitrogen fixation: endocrine disrupters and flavonoid
signalling. Nature 413:128-129.
Freitas RA Jr. 1999. Nanomedicine, Volume I: Basic
Capabilities. Georgetown, TX:Landes Bioscience.
Gianutsos G, Morrow GR, Morris JB. 1997. Accumulation
of manganese in rat brain following intranasal administration.
Fundam Appl Toxicol 37:102-105.
Gibaud S, Andreux JP, Weingarten C, Renard M, Couvreur
P. 1994. Increased bone marrow toxicity of doxorubicin
bound to nanoparticles. Eur J Cancer 30A: 820-826.
Gibaud S, Demoy M, Andreux JP, Weingarten C, Gouritin
B, Couvreur P. 1996. Cells involved in the capture of
nanoparticles in hematopoietic organs. J Pharm Sci 85:
944-950.
Gibaud S, Rousseau C, Weingarten C, Favier R, Douay
L, Andreux JP, et al. 1998. Polyalkylcyanoacrylate nanoparticles
as carriers for granulocyte-colony stimulating factor
(G-CSF). J Control Release 52:131-139.
Goldstein M, Weiss H, Wade K, Penek J, Andrews L, Brandt-Rauf
P. 1987. An outbreak of fume fever in an electronics
instrument testing laboratory. J Occup Med 29: 746-749.
Gopinath PG, Gopinath G, Kumar A. 1978. Target site
of intranasally sprayed substances and their transport
across the nasal mucosa: a new insight into the intranasal
route of drug delivery. Curr Ther Res 23 (5): 596-607.
Greim H, Borm P, Schins R, Donaldson K, Driscoll K,
Hartwig A, et al. 2001. Toxicity of fibers and particles--report
of the workshop held in Munich, Germany, 26-27
October 2000. Inhal Toxicol 13:737-754.
Griffith FD, Stephens SS, Tayfun FO. 1973. Exposure
of Japanese quail and parakeets to the pyrolysis products
of fry pans coated with teflon and common cooking oils.
Am Ind Hyg Assoc J 34:176-178.
Gumbleton M. 2001. Caveolae as potential macromolecule
trafficking compartments within alveolar epithelium.
Adv Drug Deliv Rev 49:281-300.
Hautot D, Pankhurst QA, Khan N, Dobson
J. 2003. Preliminary evaluation of nanoscale biogenic
magnetite in Alzheimer’s
disease brain tissue. Proc R Soc Lon B 270(suppl 1):S62-S64.
Heckel K, Kiefmann R, Dorger M, Stoeckelhuber M, Goetz
AE. 2004. Colloidal gold particles as a new in vivo marker
of early acute lung injury. Am J Physiol Lung Cell Mol
Physiol 287:L867-L878.
Henneberger A, Zareba W, Ibald-Mulli A, Ruckerl R,
Cyrys J, Couderc JP, et al. 2005. Repolarization changes
induced by air pollution in ischemic heart disease patients.
Environ Health Perspect 113:440-446.
Howe HA, Bodian D. 1940. Portals of entry of poliomyelitis
virus in the chimpanzee. Proc Soc Exp Biol Med 43:718-721.
Huczko A, Lange H, Calko E, Grubek-Jaworska H, Droszcz
P. 2001. Physiological testing of carbon nanotubes: are
they asbestos-like? Fullerene Sci Technol 9:251-254.
Hughes LS, Cass GR, Gone J, Ames M, Olmez I. 1998.
Physical and chemical characterization of atmospheric
ultrafine particles in the Los Angeles area. Environ
Sci Technol 32:1153-1161.
Hunter DD, Dey RD. 1998. Identification and neuropeptide
content of trigeminal neurons innervating the rat nasal
epithelium. Neuroscience 83:591-599.
Hunter DD, Undem BJ. 1999. Identification and substance
P content of vagal afferent neurons innervating the epithelium
of the guinea pig trachea. Am J Respir Crit Care Med
159:1943-1948.
IARC (International Agency for Research on Cancer).
2002. Man-made Vitreous Fibres. IARC Monogr Eval Carcinog
Risks Hum 81:1-418.
ICON. 2004. International Council on Nanotechnology
Home page. Available: http://icon.rice.edu [accessed
16 March 2005].
International Commission on Radiological Protection.
1994. Human respiratory model for radiological protection.
Ann ICRP 24:1-300.
Jani P, Halbert GW, Langridge J, Florence
AT. 1990. Nanoparticle uptake by the rat gastrointestinal
mucosa: quantitation and particle size dependency. J
Pharm Pharmacol 42:821-826
Jani PU, McCarthy DE, Florence AT. 1994. Titanium dioxide
(rutile) particle uptake from the rat GI tract and translocation
to systemic organs after oral administration. Int J Pharm
105:157-168.
Jaques PA, Kim CS. 2000. Measurement of total lung
deposition of inhaled ultrafine particles in healthy
men and women. Inhal Toxicol 12:715-731.
Johnston CJ, Finkelstein JN, Mercer P, Corson N, Gelein
R, Oberdorster G. 2000. Pulmonary effects induced by
ultrafine PTFE particles. Toxicol Appl Pharmacol 168:
208-215.
Joo SH, Feitz AJ, Waite TD. 2004. Oxidative degradation
of the carbothioate herbicide, molinate, using nanoscale
zero-valent iron. Environ Sci Technol 38: 2242-2247.
Kato T, Yashiro T, Murata Y, Herbert DC, Oshikawa K,
Bando M, et al. 2003. Evidence that exogenous substances
can be phagocytized by alveolar epithelial cells and
transported into blood capillaries. Cell Tiss Res 311:
47-51.
Kennedy P, Chaudhuri A. 2002. Herpes simplex encephalitis.
J Neurol Neurosurg Psychiatry 73: 237-238.
Keyhani K, Scherer PW, Mozell MM. 1997. A numerical
model of nasal odorant transport for the analysis of
human olfaction. J Theor Biol 186:279-301.
Kim S, Lim YS, Soltesz EG, De Grand AM, Lee J, Nakayama
A, et al. 2004. Near infrared fluorescent type II quantum
dots for sentinel lymph node mapping. Nat Biotechnol
22(1):93-97.
Kimbell JS, Godo MN, Gross EA, Joyner DR, Richardson
RB, Morgan KT. 1997. Computer simulation of inspiratory
airflow in all regions of the F344 rat nasal passages.
Toxicol Appl Pharmacol 145:388-398.
Kirschvink JL, Kobayashi-Kirschvink A, Woodford BJ.
1992. Magnetite biomineralization in the human brain.
Proc Natl Acad Sci USA 89:7683-7687.
Kirschvink J, Walker M, Diebel C. 2001. Magnetite-based
magneto-reception. Curr Opin Neurobiol 11: 462-468.
König MF, Lucocq JM, Webel ER.
1993. Demonstration of pulmonary vascular perfusion
by electron and light
microscopy. J Appl Physiol 75(4):1877-1883.
Kreuter J. 2001. Nanoparticulate systems for brain
delivery of drugs. Adv Drug Deliv Rev 47:65-81.
Kreuter J. 2004. Influence of the surface properties
on nanoparticle-mediated transport of drugs to the brain.
J Nanosci Nanotech 4:484-488.
Kreuter J, Shamenkov D, Petrov V, Ramge P, Cychutek
K, Koch-Brandt C, et al. 2002. Apolipoprotein-mediated
transport of nanoparticle-bound drugs across the blood-brain
barrier. J Drug Target 10:317-325.
Kreyling WG, Scheuch G. 2000. Clearance of particles
deposited in the lungs. In: Particle-Lung Interactions
(Gehr P, Heyder J, eds). New York:Marcel Dekker Inc.,
323-376.
Kreyling W, Semmler M, Erbe F, Mayer P, Takenaka S,
Schulz H, et al. 2002. Translocation of ultrafine insoluble
iridium particles from lung epithelium to extrapulmonary
organs is size dependent but very low. J Toxicol Environ
Health 65A:1513-1530.
Kulmala M. 2004. Formation and growth rates of ultrafine
atmospheric particles: a review of observations. J Aerosol
Sci 35:143-176.
Lam CW, James JT, McCluskey R, Hunter RL. 2004. Pulmonary
toxicity of single-wall carbon nanotubes in mice 7 and
90 days after intratracheal instillation. Toxicol Sci
77:126-134.
Lecoanet H, Bottero J, Wiesner M. 2004. Laboratory
assessment of the mobility of nanomaterials in porous
media. Environ Sci Technol 38:5164-5169.
Lecoanet H, Wiesner M. 2004. Velocity effects on fullerene
and oxide nanoparticle deposition in porous media. Environ
Sci Technol 38(16):4377-4382.
Lee CH, Guo YL, Tsai PJ, Chang HY, Chen CR, Chen CW,
et al. 1997. Fatal acute pulmonary oedema after inhalation
of fumes from polytetrafluoroethylene (PTFE). Eur Res
J 10:1408-1411.
Li N, Sioutas C, Cho A, Schmitz D, Misra C, Sempf J,
et al. 2003. Ultrafine particulate pollutants induce
oxidative stress and mitochondrial damage. Environ Health
Perspect 111:455-460.
Li X, Brown D, Smith S, MacNee W, Donaldson K. 1999.
Short-term inflammatory responses following intratracheal
instillation of fine and ultrafine carbon black in rats.
Inhal Toxicol 11:709-731.
Lu Q, Moore JM, Huang G, Mount AS, Rao AM, Larcom LL,
et al. 2004. RNA polymer translocation with single-walled
carbon nanotubes. Nano Lett 4:2473-2477.
Mach R. 2004. Nanoscale Particle Treatment
of Groundwater. Federal Remedial Technology Roundtable:
Naval Facilities
Engineering Command. Available http://www.frtr.gov/pdf/meetings/l--mach_09jun04.pdf [accessed 16 March 2005].
Malmgren L, Olsson Y, Olsson T, Kristensson K. 1978.
Uptake and retrograde axonal transport of various exogenous
macromolecules in normal and crushed hypoglossal nerves.
Brain Res 153:477-493.
Maynard AD, Baron PA, Foley M, Shvedova AA, Kisin ER,
Castranova V. 2004. Exposure to carbon nanotube material:
aerosol release during the handling of unrefined single-walled
carbon nanotube material. J Toxicol Environ Health A
67:87-107.
McMurry PH, Woo KS. 2002. Size distributions of 3 to
100 nm urban Atlanta aerosols: measurement and observations.
J Aerosol Med 15(2): 169-178.
Mehta D, Bhattacharya J, Matthay MA, Malik AB. 2004.
Integrated control of lung fluid balance. Am J Physiol
Lung Cell Mol Physiol 287:L1081-L1090.
Monteiro-Riviere N, Nemanich R, Inman A, Wang Y, Riviere
J. 2005. Multi-walled carbon nanotube interactions with
human epidermal keratinocytes. Toxicol Lett 155:377-384.
Nagaveni K, Sivalingam G, Hegde M, Madras G. 2004.
Photocatalytic degradation of organic compounds over
combustion-sized nano-TiO2. Environ Sci Technol
38:1600-1604.
Nemmar A, Delaunois A, Nemery B, Dessy-Doize C, Beckers
JF, Sulon J, et al. 1999. Inflammatory effect of intratracheal
instillation of ultrafine particles in the rabbit: role
of C-fiber and mast cells. Toxicol Appl Pharmacol 160:250-261.
Nemmar A, Hoet PHM, Vanquickenborne B, Dinsdale D,
Thomeer M, Hoylaerts MF, et al. 2002a. Passage of inhaled
particles into the blood circulation in humans. Circulation
105:411-414.
Nemmar A, Hoylaerts MF, Hoet PHM, Dinsdale D, Smith
T, Xu H, et al. 2002b. Ultrafine particles affect experimental
thrombosis in an in vivo hamster model. Am J Respir Crit
Care Med 166:998-1004.
Nemmar A, Hoylaerts MF, Hoet PHM, Vermylen J, Nemery
B. 2003. Size effect of intratracheally instilled particles
on pulmonary inflammation and vascular thrombosis. Toxicol
Appl Pharmacol 186:38-45.
Nghiem LD, Schäfer AI, Elimelech
M. 2004. Removal of natural hormones by nanofiltration
membranes: measurement,
modeling, and mechanisms. Environ Sci Technol 38: 1888-1896.
Nikula KJ, Avila KJ, Griffith WC, Mauderly JL. 1997.
Lung tissue responses and sites of particle retention
differ between rats and cynomolgus monkeys exposed chronically
to diesel exhaust and coal dust. Fundam Appl Toxicol
37: 37-53.
NNI (National Nanotechnology Initiative). 2004. What
Is Nanotechnology? Available: http://www.nano.gov/html/facts/whatIsNano.html [accessed
16 March 2005].
NRC (National Research Council). 1983. Risk Assessment
in the Federal Government: Managing the Process. Washington,
DC:National Academy Press.
Nuttall JB, Kelly RJ, Smith BS, Whiteside CK Jr. 1964.
Inflight toxic reactions resulting from fluorocarbon
resin pyrolysis. Aerospace Med 35:676-683.
Oberdörster E. 2004a. Manufactured
nanomaterials (fullerenes, C60) induce oxidative stress
in brain of
juvenile largemouth bass. Environ Health Perspect 112:
1058-1062.
Oberdörster E. 2004b. Toxicity of nC60 fullerenes
to two aquatic species: Daphnia and largemouth bass [Abstract].
In: 227th American Chemical Society National Meeting,
27 March-1 April 2004, Anaheim, CA. Washington,
DC:American Chemical Society, IEC 21.
Oberdörster G. 2000. Toxicology
of ultrafine particles: in vivo studies. Philos Trans
R Soc Lond A 358:2719-2740.
Oberdörster G, Ferin J, Finkelstein
J, Wade P, Corson N. 1990. Increased pulmonary toxicity
of ultrafine
particles? II. Lung lavage studies. J Aerosol Sci 21:
384-387.
Oberdörster G, Ferin J, Gelein
R, Soderholm SC, Finkelstein J. 1992a. Role of the
alveolar macrophage
in lung injury: studies with ultrafine particles. Environ
Health Perspect 97:193-197.
Oberdörster G, Ferin J, Morrow
PE. 1992b. Volumetric loading of alveolar macrophages
(AM): a possible basis
for diminished AM-mediated particle clearance. Exp Lung
Res 18:87-104.
Oberdörster G, Finkelstein JN,
Johnston C, Gelein R, Cox C, Baggs R, et al. 2000.
Acute pulmonary effects
of ultrafine particles in rats and mice. Res Rep Health
Eff Inst 96:5-74.
Oberdörster G, Gelein RM, Ferin
J, Weiss B. 1995. Association of particulate air pollution
and acute mortality:
involvement of ultrafine particles? Inhal Toxicol 7:111-124.
Oberdörster G, Morrow PE, Spurny
K. 1988. Size dependent lymphatic short term clearance
of amosite fibers
in the lung. Ann Occup Hyg 32(suppl 4):149-156.
Oberdörster G, Sharp Z, Atudorei
V, Elder A, Gelein R, Kreyling W, et al. 2004. Translocation
of inhaled
ultrafine particles to the brain. Inhal Toxicol 16(6/7):
437-445.
Oberdörster G, Sharp Z, Atudorei
V, Elder A, Gelein R, Lunts A, et al. 2002. Extrapulmonary
transloction
of ultrafine carbon particles following whole-body inhalation
exposure of rats. J Toxicol Environ Health 65A: 1531-1543.
Oberdörster G, Yu CP. 1990.
The carcinogenic potential of inhaled diesel exhaust:
a particle effect? J Aerosol
Sci 21(suppl 1):S397-S401.
Ohl L, Mohaupt M, Czeloth N, Hintzen G, Kiafard Z,
Zwirner J, et al. 2004. CCR7 governs skin dendritic cell
migration under inflammatory and steady-state conditions.
Immunity 21:279-288.
Oldfors A, Fardeau M. 1983. The permeability of the
basal lamina at the neuromuscular junction. An ultrastructural
study of rat skeletal muscle using particulate tracers.
Neuropathol Appl Neurobiol 9: 419-432.
Olsson T, Kristensson K. 1981. Neuronal uptake of iron:
somatopetal axonal transport and fate of cationized and
native ferretin, and iron-dextran after intramuscular
injections. Neuropath Appl Neurobiol 7: 87-95.
Patton JS. 1996. Review--mechanisms of macromolecule
absorption by the lungs. Adv Drug Deliv Rev 19: 3-36.
Pekkanen J, Peters A, Hoek G, Tiittanen P, Brunekreef
B, de Hartog J, et al. 2002. Particulate air pollution
and risk of ST-segment depression during repeated submaximal
exercise tests among subjects with coronary heart disease.
The Exposure and Risk Assessment for Fine and Ultrafine
Particles in Ambient Air [ULTRA] study. Circulation 106:933-938.
Pekkanen J, Timonen KL, Ruuskanen J, Reponen A, Mirme
A. 1997. Effects of ultrafine and fine particles in urban
air on peak expiratory flow among children with asthmatic
symptoms. Environ Res 74:24-33.
Penttinen P, Timonen KL, Tiittanen P, Mirme A, Ruuskanen
J, Pekkanen J. 2001. Ultrafine particles in urban air
and respiratory health among adult asthmatics. Eur Resp
J 17:428-435.
Peters A, Doring A, Wichmann H-E, Koenig W. 1997a.
Increased plasma viscosity during an air pollution episode:
a link to mortality? Lancet 349:1582-1587.
Peters A, Wichmann HE, Tuch T, Heinrich J, Heyder J.
1997b. Respiratory effects are associated with the number
of ultrafine particles. Am Respir Crit Care Med 155:
1376-1383.
Pietropaoli AP, Frampton MW, Oberdörster
G, Cox C, Huang L-S, Marder V, et al. Blood markers
of coagulation
and inflammation in healthy human subjects exposed to
carbon ultrafine particles. In: Effects of Air Contaminants
on the Respiratory Tract - Interpretations from Molecular
to Meta Ananlysis (Heinrich U, ed). INIS Monographs.
Stuttgart, Germany:Fraunhofer IRB Verlag, 181-194.
Plattig K-H. 1989. Electrophysiology of taste and smell.
Clin Phys Physiol Meas 10:91-126.
Rancan F, Rosan S, Boehm F, Cantrell A, Brellreich
M, Schoenberger H, et al. 2002. Cytotoxicity and photocytotoxicity
of a dendritic C(60) mono-adduct and a malonic acid C(60)
tris-adduct on Jurkat cells. J Photochem Photobiol B
67(3):157-162.
Rejman J, Oberle V, Zuhorn IS, Hoekstra D. 2004. Size-dependent
internalization of particles via the pathways of clathrin-
and caveolae-mediated endocytosis. Biochem J 377:159-169.
Rodoslav S, Laibin L, Eisenberg A, Dusica M. 2003.
Micellar nanocontainers distribute to defined cytoplasmic
organelles. Science 300:615-618.
Sato K, Imai Y, Irimura RT. 1998. Contribution of dermal
macrophage trafficking in the sensitization phase of
contact hypersensitivity. J Immunol 161: 6835-6844.
Sayes C, Fortner J, Guo W, Lyon D, Boyd AM, Ausman
KD, et al. 2004. The differential cytotoxicity of water-soluble
fullerenes. Nano Lett 4:1881-1887.
Schlesinger RB, Ben-Jebria A, Dahl AR, Snipes MB, Ultman
J. 1997. Disposition of inhaled toxicants. In: Handbook
of Human Toxicology (Massaro EJ, ed). New York:CRC Press,
493-550.
Schultheiss-Grassi PP, Wessiken R, Dobson J. 1999.
TEM investigations of biogenic magnetite extracted from
the human hippocampus. Biochim Biophys Acta 1426: 212-216.
Seaton A, MacNee W, Donaldson K, Godden D. 1995. Particulate
air pollution and acute health effects. Lancet 345:176-178.
Semmler M, Seitz J, Erbe F, Mayer
P, Heyder J, Oberdörster
G, et al. 2004. Long-term clearance kinetics of inhaled
ultrafine insoluble iridium particles from the rat lung,
including transient translocation into secondary organs.
Inhal Toxicol 16:453-459.
Shi JP, Evans DE, Khan AA, Harrison
RM. 2001. Sources and concentration of nanoparticles
(< 10 nm diameer)
in the urban atmosphere. Atmos Environ 35: 1193-1202.
Shvedova AA, Kisin E, Keshava N, Murray AR, Gorelik
O, Arepalli S, et al. 2004a. Cytotoxic and genotoxic
effects of single wall carbon nanotube exposure on human
keratinocytes and bronchial epithelial cells [Abstract].
In: 227th American Chemical Society National Meeting,
27 March-1 April 2004, Anaheim, CA. Washington,
DC:American Chemical Society, IEC 20.
Shvedova AA, Kisin E, Murray A, Schwegler-Berry
D, Gandelsman V, Baron P, et al. 2004b. Exposure of
human
bronchial cells to carbon nanotubes caused oxidative
stress and cytotoxicity. In: Proceedings of the Meeting
of the SFRR Europe 2004, Ioannina, Greece. Philadelphia:
Taylor & Francis Group, 91-103.
Silva VM, Corson N, Elder A, Oberdörster
G. In press. The rat ear vein model for investigating in
vivo thrombogenicity of ultrafine particles
(UFP). Toxicol Sci.
Simionescu N, Simionescu M, Palade GE. 1975. Permeability
of muscle capillaries to small heme-peptides. J Cell
Biol 64:586-607.
Smith AE, Helenius A. 2004. How viruses enter animal
cells. Science 304:237-242
Terasaki S, Kameyama T, Yamamoto S. 1997. A case of
zoster in the 2nd and 3rd branches of the trigeminal
nerve associated with simultaneous herpes labialis infection--a
case report. Kurume Med J 44(1):61-66.
Tiittanen P, Timonen KL, Ruuskanen J, Mirme A, Pekkanen
J. 1999. Fine particulate air pollution, resuspended
road dust and respiratory health among symptomatic children.
Eur Resp J 13(2):266-273.
Tinkle SS, Antonini JM, Rich BA, Roberts JR, Salmen
R, DePree K, et al. 2003. Skin as a route of exposure
and sensitization in chronic beryllium disease. Environ
Health Perspect 111:1202-1208.
Tran CL, Buchanan D, Cullen RT, Searl A, Jones AD,
Donaldson K. 2000. Inhalation of poorly soluble particles.
II. Influence of particle surface area on inflammation
and clearance. Inhal Toxicol 12:1113-1126.
Tran CL, Jones AD, Cullen RT, Donaldson K. 1998. Influence
of particle characteristics on the clearance of low toxicity
dusts from lungs. J Aerosol Sci 29(suppl 1): S1269-S1270.
Tungittiplakorn W, Lion LW, Cohen C, Kim J-Y. 2004.
Engineered polymeric nanoparticles for soil remediation.
Environ Sci Technol 38(5):1605-1610.
Turetsky BI, Moberg PJ, Arnold SE, Doty RL, Gur RE.
2003. Low olfactory bulb volume in first-degree relatives
of patients with schizophrenia. Am J Psychiatry 160(4):703-708.
U.S. EPA. 1994. 10-Day Chronic Daphnia magna or Daphnia
pulex. SOP 2028. Washington DC:U.S. Environmental
Protection Agency. Available: http://www.ert.org/products/2028.pdf [accessed 16 March 2005].
U.S. EPA. 2004. Air Quality Criteria for Particulate
Matter. Vol 3. 600/P-95-001cF. Washington DC:U.S. Environmental
Protection Agency, Office of Research and Development.
Utell M, Frampton M, Zareba W, Devlin R, Cascio W.
2002. Cardiovascular effects associated with air pollution:
potential mechanisms and methods of testing. Inhal Toxicol
14:1231-1247.
Veronesi B, Makwana O, Pooler M, Chen LC. In press.
Effects of subchronic exposure to CAPs in ApoE-/- mice:
VII. Degeneration of dopaminergic neurons. Inhal Toxicol.
von Klot S, Wolke G, Tuch T, Heinrich J, Dockery DW,
Schwartz J, et al. 2002. Increased asthma medication
use in association with ambient fine and ultrafine particles.
Eur Respir J 20:691-702.
Warheit DB, Hartsky MA. 1993. Role of alveolar macrophage
chemotaxis and phagocytosis in pulmonary clearance responses
to inhaled particles: comparisons among rodent species.
Microsc Res Tech 26:412-422.
Warheit DB, Hill LH, George G, Brody AR. 1986. Time
course of chemotactic factor generation and the corresponding
macrophage response to asbestos inhalation. Am Rev Respir
Dis 134:128-133.
Warheit DB, Laurence BR, Reed KL, Roach DH, Reynolds
GAM, Webb TR. 2004. Comparative pulmonary toxicity assessment
of single-wall carbon nanotubes in rats. Toxicol Sci
77:117-125.
Warheit DB, Overby LH, George G, Brody AR. 1988. Pulmonary
macrophages are attracted to inhaled particles on alveolar
surfaces. Exp Lung Res 14:51-66.
Waritz RS, Kwon BK. 1968. The inhalation toxicity of
pyrolysis products of polytetrafluoroethylene heated
below 500 degrees centigrade. Am Ind Hyg Assoc J 29:
19-26.
WHO. 1985. Reference Methods for Measuring Airborne
Man-Made Mineral Fibers. Environmental Health Series
4. Copenhagen:World Health Organization.
Wichmann H-E, Cyrys J, Stölzel M, Spix C, Wittmaack
K, Tuch T, et al. 2002. Sources and elemental composition
of ambient particles in Erfurt, Germany. In: Fortschritte
in der Umweltmedizin (Wichmann HE, Schlipköter HW,
Fülgraff G, eds). Erfurt, Germany: Ecomed Publishers.
Wichmann H-E, Spix C, Tuch T, Wolke G, Peters A, Heinrich
J, et al. 2000. Daily Mortality and Fine and Ultrafine
Particles in Erfurt, Germany. Part I: Role of Particle
Number and Particle Mass. Res Rep Health Eff Inst 98:
5-86.
Widmaier EP, Raff H, Strang KT. 2004.
Vander, Sherman & Luciano's
Human Physiology: The Mechanisms of Body Functions. 9th
ed. New York:McGraw Hill.
Williams N, Atkinson G, Patchefsky A. 1974. Polymer
fume fever: not so benign. J Occup Med 16: 519-522.
Wilson M, Lightbody J, Donaldson K, Sales J, Stone
V. 2002. Interactions between ultrafine particles and
transition metals in vivo and in vitro. Toxicol Appl
Pharmacol 184(3):172-179.
Woo KS, Chen D-R, Pui D, McMurry P. 2001. Measurement
of Atlanta aerosol size distributions: observations of
ultrafine particle events. Aerosol Sci Technol 34: 75-87.
Yamago S, Tokuyama H, Nakamura E, Kikuchi K, Kananishi
S, Sueki K, et al. 1995. In vivo biological behavior
of a water-miscible fullerene: 14C labeling,
absorption, distribution, excretion and acute toxicity.
Chem Biol 2:385-389.
Yamakoshi Y, Umezawa N, Ryu A, Arakane K, Miyata N,
Goda Y, et al. 2003. Active oxygen species generated
from photoexcited fullerene (C60) as potential
medicines: O2-* versus 1O2.
J Am Chem Soc 125:12803-12809.
Zhou Y-M, Zhong C-Y, Kennedy IM, Leppert VJ, Pinkerton
KE. 2003. Oxidative stress and NF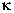 B
activation in the lungs of rats: a synergistic interaction
between soot and iron particles. Toxicol Appl Pharmacol
190:157-169. B
activation in the lungs of rats: a synergistic interaction
between soot and iron particles. Toxicol Appl Pharmacol
190:157-169.
Zhu Y, Hinds WC, Kim S, Shen SK, Sioutas C. 2002. Study
of ultrafine particles near a major highway with heavy-duty
diesel traffic. Atmos Environ 36:4323-4335.
Last Updated: June 14, 2005 |
|
 |
|
| |

