Title: Joseph Tart
Around the turn of the century, the German bacteriologist Paul Ehrlich predicted that drugs would be invented that would perform like "magic bullets"--locating and destroying diseased tissue, without harming healthy tissue. The well-aimed drugs that Ehrlich imagined would provide unheard-of effectiveness with virtually no side effects. Unfortunately, such drugs remained largely in Ehrlich's fantasies until last year when modern medical gunslingers developed the equivalent of a magic bullet--drugs incorporated into fats.
The advent of pharmaceuticals packaged in fats was in the early 1960s, when Alec D. Bangham, then head of the biophysics unit at the Institute of Animal Physiology in Babraham, England, discovered that mixing phospholipids with water produces microscopic bubbles, ranging from 0.25 micrometers (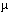 m) to more than 20
m) to more than 20 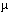 m across. These bubbles, called liposomes, consist of a bilayer of lipids that surrounds a watery interior. Almost immediately, liposomes appeared promising as drug-delivery devices because of three characteristics. First, liposomes can carry water-soluble drugs in their hollow center or fat-soluble drugs in their lipid bilayer. Second, the lipids in liposomes also serve as building blocks for cells in our bodies, suggesting that liposomes should be safe for medical use. Third, it appeared possible to design liposomes that would accumulate in specific tissues, where the encapsulated drug would leak out slowly.
m across. These bubbles, called liposomes, consist of a bilayer of lipids that surrounds a watery interior. Almost immediately, liposomes appeared promising as drug-delivery devices because of three characteristics. First, liposomes can carry water-soluble drugs in their hollow center or fat-soluble drugs in their lipid bilayer. Second, the lipids in liposomes also serve as building blocks for cells in our bodies, suggesting that liposomes should be safe for medical use. Third, it appeared possible to design liposomes that would accumulate in specific tissues, where the encapsulated drug would leak out slowly.
"In the nearly 30 years since their rather serendipitous discovery," Bangham wrote in the 15 December 1992 issue of Hospital Practice, "liposomes have demonstrated a versatility and utility that have exceeded expectations. . . . A stream of pharmaceutical agents may be expected in the years to come." Several new drugs now promise to swell that lipid-based stream.
Fighting Fungus
Some fungus-fighting pharmaceutical benefits arise from how our body handles liposomes. When a liposome enters the body, plasma proteins bind to it. In some cases, the proteins then tag the structure for destruction by the immune system, particularly by macrophages located largely in the liver and spleen. When macrophages gobble up the intruding liposomes, the drug is released into the macrophages, which are common hiding places for systemic fungal infections. This process spurred investigators to explore liposome-based antifungals.
Physicians need new tools for fighting fungal infections. According to Mitchell Cohen, director of the Division of Bacterial and Mycotic Diseases at the Centers for Disease Control, "Many fungal diseases are becoming more common, primarily because of an increasing frequency of susceptible hosts. We have more patients with diseases that suppress the immune system, such as malignancies and HIV infections. In addition, patients are becoming more susceptible because of intentional treatments, such as organ transplantation, chemotherapy, or high-dose steroids. All of these conditions and treatments make people susceptible to a variety of fungal agents, many of which are ubiquitous in the environment. Some of these agents, such as aspergillosis, are very, very difficult to treat." Aspergillosis, which is caused by a mold, is often devastating to people who are immunocompromised.
If a patient contracts a systemic fungal infection, a physician may prescribe amphotericin B--a powerful, broad-spectrum antifungal. Unfortunately, a dose large enough to kill the fungus might also kill the patient, because the drug can damage a patient's bone marrow, central nervous system, and kidneys. Some of these side effects can be reduced by enveloping amphotericin B in a liposome, or by simply mixing the drug with lipids.
Last November, the FDA approved treating some cases of aspergillosis with an antifungal mixed with lipids. The Liposome Company in Princeton, New Jersey, produces the antifungal, Abelcet, which consists of amphotericin B mixed with two phospholipids. A precise ratio of these ingredients generates structures that resemble ribbons, which, in cross section, resemble a daisy with petals composed of alternating phospholipids and amphotericin B.
According to Thomas Walsh, head of the mycology unit in the Pediatric Branch of the National Cancer Institute, "Abelcet, also known as amphotericin B lipid complex [ABLC], is indicated for the treatment of children and adults who have invasive aspergillosis and are refractory to or intolerant of conventional amphotericin B. Abelcet has been shown in open-label and controlled trials to be significantly less nephrotoxic than conventional amphotericin B, while retaining antifungal activity that is at least equivalent to that of the parent compound." Walsh added, "The cost of Abelcet, which is higher than that of conventional amphotericin B, must be balanced against the cost of the dose-limiting nephrotoxicity of amphotericin B. Prudent use of Abelcet in appropriate patients will also contribute to controlling institutional costs." In conclusion, Walsh called Abelcet "an important and long-awaited addition to our therapeutic armamentarium against invasive fungal infections."
At least three other lipid-based antifungals should reach the U.S. market over the next couple of years. Sequus Pharmaceuticals, Inc. in Menlo Park, California, is awaiting FDA approval for Amphotec, a nonliposome mixture of amphotericin B and lipids. Likewise, NeXstar Pharmaceuticals, Inc. in Boulder, Colorado, hopes to bring AmBisome--a liposomal form of amphotericin B--to the U.S. market. Finally, Aronex Pharmaceuticals, Inc. in The Woodlands, Texas, is performing clinical tests on NystatinLF--a liposomal form of the antifungal nystatin. Nystatin alone proves too toxic for intravenous use, but the liposomal form can be injected and appears effective in fighting fungal infections.
Targeting Tumors
Although being consumed by macrophages may improve the effectiveness of some lipid-based drugs, avoiding that destruction presents other pharmaceutical opportunities. In fact, liposomes that escape detection by macrophages can end up in cancerous tumors. Loading such tumor-targeting liposomes with anticancer drugs produces a concentrated attack on cancer, while sparing healthy tissue from the side effects of traditional chemotherapy. Liposomes are now engineered to avoid detection by macrophages, largely by altering the liposome's surface molecules.
Liposomes must also be designed appropriately to concentrate in tumors. These microscopic bubbles must be manufactured to a precise size and must possess long-term stability in blood for a cancerous tumor to reel in the drug-filled liposomes. A growing tumor releases growth factors that stimulate blood-vessel formation, (angiogenesis). These tumor-triggered vessels include gaps in their walls and even open-ended tubes in some cases. So a liposome of the right size is contained in regular blood vessels but slips through the gaps in tumor-feeding vessels, concentrating drug-filled liposomes within the tumor.
In 1989, investigators at Sequus discovered that adding a polyethylene glycol coating to a liposome gives it a long, stable life in circulation. Frank Martin, vice president and chief scientific officer at Sequus, said, "The fact that these liposomes circulate for so long means that they're passing through a tumor's . . . capillary bed many times. Over a period of days after injection, the liposomes move from the lumen of a tumor blood vessel through the gaps and into the tumor, where they get stuck. The liposomes sit there and, as my mother would say, 'stew in their own juice.' Eventually, the liposomes break down over a period of days to weeks and the drug comes out. So it's like delivering a little time-released pill to the tumor."
Liposome-based chemotherapy appears particularly suited to Kaposi's sarcoma, a form of skin cancer that appears most often in HIV-infected men. This cancer generates reddish or purplish skin lesions, or tumors, which are colored by red blood cells that escape defective capillaries and leave deposits of hemoglobin. At an advanced stage, conventional treatment for this cancer involves a combination of the drugs adriamycin, bleomycin, and vincristine (ABV). Unfortunately, this trio of drugs produces serious side effects, including damage to a patient's heart and bone marrow. These side effects and the fact that Kaposi's sarcoma produces a highly vascular tumor encouraged trials with liposome-based chemotherapy.
Kaposi's sarcoma can be treated with Sequus's Doxil, the first liposomal drug approved by the FDA. Doxil consists of doxorubicin--a conventional anticancer drug--encapsulated in liposomes that are coated with polyethylene glycol. The polymer coating gives this liposomal drug a circulating half-life (the time it takes for half the dose to be eliminated or disintegrated) of 50 hours compared with a 10-minute half-life for doxorubicin alone. In a Sequus-sponsored study of therapy for Kaposi's sarcoma, Doxil produced a clinical response in 43.2% of the patients, compared with ABV's 24.5% response rate. Moreover, only 32.3% of the patients using Doxil could not complete the study because of adverse effects, whereas 65.6% of the patients receiving ABV could not complete the treatment. In other words, Doxil proved more effective and less toxic in comparison with ABV. Doxil is now being tested against breast, ovarian, and prostate cancers.
Donald W. Northfelt, a hematologist and oncologist with the Pacific Oaks Medical Group who served as a principal investigator in studies of Doxil as a treatment for Kaposi's sarcoma, said, "It was especially gratifying that patients who'd received regular doxorubicin as part of their previous treatment for Kaposi's sarcoma, but were not getting the response that they needed, would start responding when switched to Doxil." When asked about the overall value of liposomal chemotherapy, he said, "I think we're just on the verge of a new era in chemotherapy with the use of this technology."
The added benefits of Doxil do impose a cost, however. According to Tony Goosmann, director of marketing at Sequus, Doxil costs $485 per 20 milligrams. When being used against Kaposi's sarcoma, that unit price translates to about $1,000 per treatment cycle, with a patient usually receiving a treatment every three weeks. Goosmann says that, in comparison to using ABV, Doxil costs roughly twice as much.
Several other companies hope to market additional liposomal forms of chemotherapy. For instance, NeXstar is awaiting FDA approval for a liposomal drug, DaunoXome, which delivers a conventional anticancer drug called daunorubicin. DaunoXome will also be used against Kaposi's sarcoma. In addition, The Liposome Company--in a joint effort with Pfizer, Inc.--is producing another liposomal form of doxorubicin called TLC D-99, which remains under development. Results from clinical trials indicate that this drug may battle metastatic breast cancer, particularly when combined with other forms of chemotherapy. Finally, Aronex's TretinoinLF, a liposomal form of all-trans-retinoic acid (vitamin A), is being tested against Kaposi's sarcoma and leukemia.
Expanding Applications
Today's world of lipid-associated pharmaceuticals revolves largely around antifungal and chemotherapeutic drugs for two reasons. First, many other types of drugs cannot be combined with liposomes. Second, other drugs may not benefit from being delivered with lipids, or going where lipids take them. Despite these obstacles, researchers expect to see liposomal drugs approved for other diseases. For example, The Liposome Company's TLC C-53, a liposomal form of prostaglandin E1, is being developed to treat a variety of conditions, including adult respiratory distress syndrome and heart attacks.
Moreover, the natural circulation of liposomes in a human body suggests other potential applications. Instead of fighting cancer cells in a tumor, for instance, liposomes might be formulated with drugs that interfere with angiogenesis, thereby starving a growing tumor. And, because liposomes collect at sites of inflammation and wounds, they might be filled with anti-inflammatory or wound-healing drugs. Projects are underway in these areas.
 |
|
Blood bubbles. Liposomes filled with hemoglobin may some day provide an alternative to natural red blood cells.Alan Rudolph/Naval Research Lab |
Nevertheless, liposomes are not limited to delivering drugs. For example, liposomes may be used to carry oxygen. Alan Rudolph, a program manager at the Naval Research Laboratory's Center for Biomolecular Science and Engineering, and his colleagues pack liposomes with hemoglobin, and the combined structure performs much like a natural red blood cell, carrying oxygen. Rudolph hopes that these "artificial red blood cells" will be employed when physicians need units of packed red blood cells. So far, this product has performed well in the early stages of animal testing, but it remains to be seen how a human will react to the high doses of liposomes that are involved in this application. Rudolph added that some modifications might be required to make the product cost-competitive, but it may provide a safe red-cell substitute--one with no risk of viral or antigen transmission.
Although fat is often considered a public-health enemy, that characterization may change as lipid-based pharmaceuticals become more widely available. In the end, Bangham's liposomal "stream of pharmaceutical agents" may overrun its banks, generating a flood of tiny fat bubbles that make us well.
Mike May
Suggested Reading
Lasic D. Liposomes. Am Sci 80(1):20-31 (1992).
Lasic D, Martin F. Stealth liposomes. Boca Raton, FL: CRC Press, 1995.
Rudolph AS. Biomaterial biotechnology using self-assembled lipid microstructures.
J Cell Biochem 56:183-187 (1994).
Last Update: May 23, 1997





