 |
|
|
April
2006
|
 |
|
|
|
|

|
|
|
| |
 |
|

H5N1 Avian Flu Virus Vaccine Induces Immune
Responses in Healthy Adults, March 29, 2006
NIH Researchers Identify OCD Risk Gene, March 29,
2006
New Federally Funded Research Program Aims to
Improve Survival from Cardiac Arrest and Severe
Trauma, March 24, 2006
New Strategies Help Depressed Patients Become
Symptom-Free, March 22, 2006
Male Fat Distribution Pattern and Coronary Risk
Profile Linked to X Chromosome & Women Lacking
Ovarian Function Shy, Anxious, March 21, 2006
Defective Immune System Response to Smallpox Vaccine
Detailed, March 21, 2006
 Back
to top Back
to top
|
|
|
|
 |
|
FREQUENTLY ASKED QUESTIONS:
NIH PUBLIC ACCESS POLICY
Q: What
is the NIH Public Access Policy?
A: The Public Access Policy requests
NIH-funded investigators to submit an electronic
version of the author’s final manuscript upon
acceptance for publication to the NIH National
Library of Medicine’s PubMed Central (PMC). This
includes all manuscripts resulting from research
supported in whole or in part, with direct funds
from NIH.
Q:
Why should there be a public resource of published
peer-reviewed research findings of NIH-funded
research?
A: The Public Access Policy has three goals:
develop a central archive of NIH-funded research
publications—for now and in the future, preserving
vital medical research results and information for
years to come; advance science by creating an
information resource that will make it easier for
scientists to mine medical research publications,
and for NIH to manage better its entire research
investment; and, provide electronic access to
NIH-funded research publications for patients,
families, health professionals, scientists,
teachers, and students.
Q: What is PubMed
Central (PMC)?
A: PubMed Central (PMC) is the NIH digital
repository of full-text, peer-reviewed biomedical,
behavioral, and clinical research manuscripts. It is
a publicly accessible, stable, permanent, and
searchable electronic archive that is fully
integrated with other National Library of Medicine
databases. Anyone with entry to the Internet can
access
PMC.
Q: When do I submit my
manuscript?
A: NIH-funded investigators are requested to
submit an electronic version of the author’s final
manuscript to NIH upon acceptance for publication.
The policy gives authors the flexibility to
designate a specific timeframe for public release of
the document—ranging from immediately after final
publication to 12 months.
Q: How do I submit a
manuscript?
A: Manuscripts can be submitted through the
NIH Manuscript Submission (NIHMS) system. This
password-protected, Web-based system allows for easy
identification of NIH grant numbers (past and
present) and NIH intramural project numbers by
associating them with the corresponding Principle
Investigator (PI). For detailed instructions on how
to submit a manuscript, refer to the
Authors Manual.
Q: Can authors
and journals continue to assert copyright in
scientific publications resulting from NIH funding?
A: Yes. The Public Access Policy does
not affect the ability to assert copyright. Funding
recipients can continue to assert copyright in works
arising from NIH-funded research, and they can
assign these copyrights to journals, as is the
current practice. Copyright holders can enforce
these copyrights as before. A member of the public
viewing or downloading a copyrighted document from
PubMed Central (PMC) is subject to the same rights
and restrictions as when copying an article from the
library.
Q: How can I be sure
that my journal publication contract allows me to
also publish in PubMed?
A: Individual publication arrangements are
made between authors and publishers, and can take
many forms. NIH encourages investigators to sign
agreements that specifically allow the manuscript to
be deposited with NIH for public posting on PubMed
Central as soon as possible after journal
publication. Institutions and investigators may want
to develop particular contract terms in consultation
with their own legal counsel, as appropriate. As an
example, the kind of language that an author or
institution might add to a copyright agreement
includes the following: "Journal acknowledges that
Author retains the right to provide a copy of the
final manuscript to NIH upon acceptance for Journal
publication or thereafter, for public archiving in
PubMed Central as soon as possible after publication
by Journal."
Q: Will the NIH Public
Access Policy apply to NIH-supported investigators
in foreign countries?
A: Yes. The Policy applies to all NIH-funded
investigators, including those in foreign countries.
The PMC archive will be available through the
Internet, so all investigators will have access to
it, provided they have a computer with an Internet
connection.
Q: Where can I send
questions or comments about the NIH Public Access
Policy?
A: Send
email
questions or comments, or visit the
NIH Public
Access Web site.
 Back
to top Back
to top
|
|
|
 |
|
COMMUNICATE
WITH THE
NIH EXTRAMURAL NEXUS — WE WANT TO HEAR FROM
YOU
Feedback
(to the Editor) from recipients and
subscribers of the NIH Extramural Nexus is
vital. Your comments, questions, and suggestions for
topics will enable Nexus editorial staff to
deliver appropriate content to the extramural
community.
 Back
to top Back
to top
|
|
|
|
|
Printer
Friendly Version
(Adobe
Acrobat Reader Required) |
|
|
|
NEWS FROM THE DIRECTOR OF OER:
R01 Electronic Timeline Adjusted and Multiple-PI
Initiative Piloted |
|
 Dear
Extramural Community, Dear
Extramural Community,
Immediately below, you will find updates on two
major NIH initiatives—Electronic Submission
and Multiple Principal Investigators—which
will have a significant effect on how NIH and
extramural institutions do business. The success
of these complex initiatives, which at NIH and
likely at your institutions touch on many aspects
of the conduct of biomedical research, depend
heavily on our continuing to work together. I very
much appreciate your feedback and hope that you
will find these updates and the other items in
this Nexus update both informative and
helpful.
Recently, the Office of Extramural Research (OER)
issued notices in the NIH Guide announcing
adjustments to electronic submission and the
rollout of the multiple Principal Investigator
(PI) option on research grants. To summarize these
changes:
Electronic Submission: The
NIH has made considerable progress in
transitioning to electronic applications. We have
already received electronic SBIR/STTR, R13/U13,
R36 and R15 applications and we will receive R03,
R21/R33 and R34 applications beginning in June.
Our commitment and enthusiasm for electronic
submission remains unchanged. However, after
considering input from the scientific community we
have
adjusted the implementation timeline for the
electronic R01s to provide an additional four
months to address business processes here and at
applicant organizations. We will begin receiving
R01 applications via Grants.gov and the SF424
(R&R) with the February 1, 2007, receipt date. We
also have
changed the submission deadline from 8:00 p.m.
Eastern Time to 5:00 p.m. local time at the
applicant organization. Additional information is
available at the
Electronic Submission Web site.
Multiple Principal
Investigators: A pilot program, beginning in May
with a few select RFAs and PAs, will permit the
identification of more than one PI on applications
for NIH research grants. This change is being
taken to catalyze multidisciplinary research and
team science in ways that have been difficult with
a single-PI model. The database changes and the
necessary modifications to the computer interfaces
required to process grant applications are
expected to be completed by May 1. We hope to make
the multiple-PI option available for most
investigator-initiated research grant mechanisms
submitted for February 2007 and later application
receipt dates. Initial features will include the
ability to record the names of all PIs in NIH
databases, display those names on the notice of
grant award, and list all PIs in
CRISP. In the future, we will consider ways to
make linked awards, apportion funds to individual
PIs and recognize all key personnel. We will
continue to work with the scientific community to
identify features that will stimulate
collaborative research approaches. Additional
information on the pilots and the project timeline
is available in the
NIH Guide and on the
Multiple-PI Web site.
If you have questions about
any aspect of the Multiple PI Project, send them
by email to
multi_PI@mail.nih.gov.
— Norka Ruiz Bravo, Ph.D. -
Director, OER and NIH Deputy Director for
Extramural Research
 Back
to top Back
to top
|
|
NIH OFFERS NEW SCIENTISTS
A “PATHWAY TO INDEPENDENCE” |
|

One of the most challenging
transitions in any research career is the
transition from postdoctoral trainee training to
independence as a scientist. New investigators who
successfully cross the bridge from research
dependence to research independence have a new
opportunity to act on fresh ideas and bring
innovative perspectives to the research
enterprise. These are critical to sustaining NIH’s
ability to push forward and advance the frontiers
of medical research. In today’s challenging budget
environment, supporting a healthy cohort of
NIH-supported investigators is the number one
priority of NIH Director Elias A. Zerhouni, M.D.
Dr. Zerhouni formed the NIH
New Investigators Committee, chaired by Dr. Norka
Ruiz Bravo, the NIH deputy director for extramural
research and Dr. Story Landis, director of the
National Institute of Neurological Disorders and
Stroke. The new
NIH Pathway to Independence Award (K99/R00)
program is an initial result of the committee’s
intensive efforts to develop and implement
programs that facilitate an investigator’s ability
to receive their first R01 award earlier in their
research career. All NIH Institutes and Centers
are participating.
The NIH Pathway to
Independence Award (K99/R00) program is a new and
exciting opportunity for promising postdoctoral
scientists. The award includes both mentored and
independent research support. Features of the
award, including eligibility, citizenship, review
issues, fiscal matters, etc., are described in
detail in the
NIH Guide and on the
New Investigators Web site. NIH will issue
between 150 and 200 awards for this program in its
initial year, beginning in fall 2006, and
anticipates issuing the same number of awards each
of the following five years. During this time, the
NIH will provide almost $400 million in support of
this program.
Since the program’s
announcement in the NIH Guide on January 27,
2006, there has been a high level of interest from
the grantee community. To answer some of the most
common questions regarding eligibility,
citizenship, review issues, fiscal matters, etc.,
the NIH has posted several
Questions & Answers.
Each candidate is encouraged
to discuss their potential competitiveness for
this funding opportunity with his/her advisor,
department chair, and
relevant NIH program staff prior to preparing
an application. Only the most competitive
candidates are encouraged to apply for this award.
 Back
to top Back
to top
|
|
DO YOU BELIEVE THIS EXTRAMURAL URBAN LEGEND?
“Preparing preliminary data for applications requires Investigators to ‘fudge’ their payroll certifications (effort reports)” |
|
In the world of Principal
Investigators (PIs), tracking the time and effort
devoted to scientific, administrative, and
institutional responsibilities for the purpose of
payroll certification can be complex and more than a
little confusing. It is commonly heard that the PI’s
time spent preparing a grant or contract proposal,
including the collection of preliminary data, must
be tracked and reported separately when certifying
payroll. Many believe that these activities must be
completed after-hours and ‘off the books,’ therefore
not in compliance with Federal rules.
Is this an urban legend or
is it true?...It is an urban legend! For
additional information, read on...
Reporting requirements
differ with institution category because
while different types of institutions have similar
Federal costing requirements, each is required to
address distinct requirements under Federal cost
principles. The Office of Management and Budget
(OMB) Circular A-21 (Cost Principles for Educational
Institutions) applies to colleges and universities,
and OMB Circular A-122 (Cost Principles for
Non-Profit Organizations) applies to non-profits.
College or university
Investigators collecting preliminary data or
preparing a competing application do not have to
track this time separately from their other
activities. There is no Federal requirement to
consider ‘Bid and Proposal’ effort, including the
preparation of preliminary data, in payroll
certifications (effort reporting). It simply is not
necessary.
Non-profit organization
Investigators are required to track and
certify time associated with collecting preliminary
data or preparing bids and proposals. Institutions
that fall in this category have developed procedures
to do this.
For non profit institutions OMB Circular A-122 (Cost
Principles for Non-Profit Organizations) does not
specifically address the treatment of these costs.
However, the Code of Federal Regulations,
45 CFR Part 74.27(b) states that bid and proposal costs of
the current accounting period are allowable as F&A.
Accordingly, preparing preliminary data to support a
competing application should be certified as part of
the F&A costs, in accordance with the established
policy and procedure of the non-profit organization.
Finally, it is important to
remember that while these Federal rules are
constant, each grantee institution implements
Federal requirements locally, with their own policy and
procedures. Therefore, it is always important to
understand and be fully responsive to local policies
and procedures.
For further reference, links
are provided for the following documents in full:
 Back
to top Back
to top
|
|
NIH CREATES THE OFFICE OF PORTFOLIO ANALYSIS AND STRATEGIC INITIATIVES |
|
NIH recently launched the
Office of Portfolio Analysis and Strategic
Initiatives (OPASI) in the Office of the NIH
Director to transform the way NIH finds and funds
cutting-edge research, improve our ability to
identify public health challenges, and increase
trans-NIH dialogue, decision-making and
priority-setting.
“This process has essentially
two goals,” said NIH director Dr. Elias A.
Zerhouni. “To allow new science that falls through
the cracks to be funded quickly, and to make sure
new ideas have a chance.”
OPASI has three branches that
will work together to accomplish its goals: the
Division of Resource Development and Analysis
(DRDA), the Division of Strategic Coordination
(DSC), and the Division of Evaluation and
Systematic Assessments (DESA).
The OPASI Process
(image opens in new browser window in Acrobat format)
OPASI will build upon the
model of the NIH Roadmap for Medical Research and
will coordinate with NIH Institute and Centers
(ICs) and external stakeholders to identify
research priorities that will ultimately improve
NIH’s ability to be nimble, dynamic, and
responsive to emerging scientific opportunities
and public health needs. OPASI will solicit
regular input from the biomedical and behavioral
research community including proposals from
individual scientists, stakeholders and
organizations outside NIH; a regular, Roadmap-like
planning process; data about the burden of
illness; as well as considerable input from IC
directors and such NIH Office of the Director
components as women’s health, behavioral science,
and AIDS research. Although OPASI will not have
grant-making authority, it will provide an
“incubator space” to jump-start trans-NIH
initiatives and support ICs that will take the
lead on priority projects on a time-limited basis
(5 to 10 years). These initiatives will be managed
primarily by the ICs, and the turnover of projects
will ensure that sufficient funds are available
for continuous development of new, trans-NIH
efforts.
These OPASI initiatives will
be supported by the “Common Fund for Shared
Needs,” a central funding source built upon the
Roadmap budget model. The Common Fund is derived
from the ICs—and the Office of the Director—which
will contribute a percentage of their annual
budgets to the Fund. By pooling resources in this
formal, structured way, NIH will eliminate the
need to collect money in an ad hoc manner from ICs
every time a trans-NIH opportunity presents
itself. Building from current Roadmap funds, which
amount to about 1.6 percent of NIH’s total budget
in fiscal year 2007, the Fund will increase to up
to 5 percent of the total NIH budget depending on
NIH budget growth, scientific opportunities and
public health needs.
OPASI was formally introduced
on September 28 with an announcement in the
Federal Register. More information will be
available on the OPASI Web site, which will be
launched soon. Additionally, OPASI has been
described in detail in the
NIH Record.
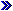 Back
to top Back
to top
|
|
PUBLIC PRIVATE PARTNERSHIPS: A
Component of the NIH Roadmap for Medical Research
|
|
 As researchers tackle ever
more complex biomedical problems, strategic
partnerships between NIH, private industry and
nonprofit organizations will become more
important. Promoting and facilitating new and
ongoing partnerships is the mission of the Program
on Public-Private Partnerships, located in the
Office of Science Policy (OSP) within the NIH's
Office of the Director. As researchers tackle ever
more complex biomedical problems, strategic
partnerships between NIH, private industry and
nonprofit organizations will become more
important. Promoting and facilitating new and
ongoing partnerships is the mission of the Program
on Public-Private Partnerships, located in the
Office of Science Policy (OSP) within the NIH's
Office of the Director.
The NIH Program on
Public-Private Partnerships (PPP) represents an
important aspect of the
NIH Roadmap. The PPP Program is designed to
promote and facilitate the formation of
partnerships that will foster excellent science in
the public interest and leverage NIH and non-NIH
resources. The NIH takes a broad view of
partnering and will work cooperatively with other
federal agencies, academic scientists and
institutions, charitable foundations, patient
advocacy groups and individuals, industry, and
others. Partnerships may take many forms and range
widely in size and scope. Partnerships provide an
opportunity for NIH to participate in the greater
scientific community and for non-NIH entities to
participate in science that is in the interest of
the public health.
With an eye toward keeping
pace with changes in the business sector and
dealing with concerns about intellectual property,
patents, and licensing rights, the PPP office will
work with relevant NIH Institutes, Centers and
Offices to review existing partnership mechanisms
and provide recommendations for policies or legal
authorities needed to achieve NIH's objectives.
The PPP also will work closely with the
Foundation for the National Institutes of Health, a private charitable
foundation chartered to help support NIH
activities. The PPP also will serve as a point of
contact for entities wanting to partner with NIH,
and can provide contact information and advice
regarding the initiation, establishment and
implementation of new partnerships. This effort
will complement, not replace, the role of the
Institutes and Centers as they work with the
private sector to serve their missions.
There are a number
of successful public-private partnerships that are ongoing within NIH, such as
the Osteoarthritis Initiative (OA),
The Alzheimer’s Disease Neuroimaging Initiative (ADNI), and the recently
launched Genetic Association Information Network (GAIN). Additionally, there are
several others in development. For information about specific partnership
opportunities, contact the relevant NIH Institute or Center. For additional
information or advice, visit the
Public-Private Partnerships Web site or contact the
Program on Public-Private Partnerships.
 Back
to top Back
to top
|
|
DO I REALLY NEED TO READ THE APPLICATION GUIDE? |
|
Whether you’ve been writing
grant applications for 20 years or 20 minutes, you
will benefit from reading the
Department of Health and Human Services Grants.gov
Application Guide SF424 (R&R). The National
Institutes of Health (NIH) and the Agency for
Healthcare Research and Quality (AHRQ) are
converting to the electronic SF424 Research and
Related (R&R) grant application forms. The
Application Guide is a comprehensive resource for
applicants that covers the new electronic
submission process, the new SF424 (R&R)
application form set and current policy
information.
The NIH/AHRQ electronic
submission of grant applications process and the
SF424 (R&R) form set are new to everyone. The
Application Guide, together with the funding
opportunity announcement instructions, provides
all the information you need to submit your grant
application successfully.
Additional benefits of
reading the Application Guide are as follows:
 |
The new process
automatically checks the application to
ensure it follows the instructions in the
Application Guide and funding opportunity
announcement. These automated checks may
catch and flag as errors things that
customarily passed unnoticed through the
paper application process. Errors can halt
an application in its tracks. You cannot
complete the submission process without
correcting all errors in the application and
submitting a corrected application through
Grants.gov. Following along in the
Application Guide as you fill out your
application will help you avoid errors and
save you the time and frustration of
submitting a corrected application.
|
 |
The SF424 (R&R) forms
are used by all Federal agencies that
provide Research and Related grant funding.
Unless a form field is required by multiple
agencies, it will not be marked as required
on the federal-wide form. For this reason,
not all fields that are required by NIH/AHRQ
are marked as required on the form. For
example, the “Credential, e.g., agency
login” field in the PD/PI section of the
Senior/Key Person Profile component is
required by NIH/AHRQ, but not marked as such
on the federal-wide form. If you pay special
attention to the agency-specific
instructions included in the Application
Guide and denoted by the HHS logo, you will
know exactly what NIH/AHRQ is looking for in
each form field.
|
 |
Although Grants.gov
will accept attachments in multiple formats,
NIH accepts attachments only in PureEdge™ or
Portable Document Format (PDF) format. The
Application Guide provides tips for creating
acceptable attachments.
|
Details relating to all
aspects of electronic grant applications are
available on the
Electronic Submission of Grant Applications Web
site.
 Back
to top Back
to top
|
|
CONTINUING SERIES ON PROGRAMS IN THE OER
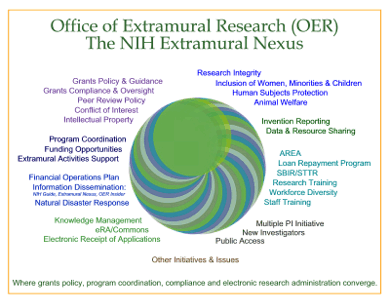
Click on
graphic to expand (opens in new window)
|
|
 OLAW, or the Office of
Laboratory Animal Welfare, is an office in the
Office of Extramural Research that supports the
NIH research enterprise by administering the PHS
Policy on Humane Care and Use of Laboratory
Animals (Animal Policy). Appropriate animal care
and use are integral components of good research
because they affect research findings,
reproducibility of results, and reliability of
data. OLAW promotes compliance with the PHS Animal
Policy through educational outreach,
communication, negotiation of Animal Welfare
Assurances, and evaluation of institutional
self-reports of noncompliance. OLAW, or the Office of
Laboratory Animal Welfare, is an office in the
Office of Extramural Research that supports the
NIH research enterprise by administering the PHS
Policy on Humane Care and Use of Laboratory
Animals (Animal Policy). Appropriate animal care
and use are integral components of good research
because they affect research findings,
reproducibility of results, and reliability of
data. OLAW promotes compliance with the PHS Animal
Policy through educational outreach,
communication, negotiation of Animal Welfare
Assurances, and evaluation of institutional
self-reports of noncompliance.
Congress delegated authority
and responsibility for Animal Policy to the NIH
Director in the 1985 Health Research Extension Act,
which established a system of oversight by local
Institutional Animal Care and Use Committees
(IACUCs). The system is based on institutional
self-evaluation, self-monitoring,
self-identification and self-correction of
noncompliance. Within the scope of the Policy,
IACUCs exercise autonomy in reviewing and approving
research and the use of performance-based standards
found in the
Guide for the Care and Use of Laboratory Animals.
Institutions are able to identify and report
deficiencies and non-compliance as well as
correction of deficiencies and non-compliance
without fear of regulatory reprisal due to the
emphasis in the law on providing grantees reasonable
opportunity to take corrective actions. Refer to the
Guidance on Prompt Reporting to OLAW under the PHS
Policy on Humane Care and Use of Laboratory Animals
for complete details.
OLAW is divided into a Division of Assurances, a
Division of Compliance Oversight, and the Office of the Director. Educational
outreach activities are a function of the Office of the Director. Many
educational conferences and workshops are co-sponsored each year, and
educational resources developed in conjunction with outside scientific experts are
available. The OLAW Web
site is a rich source of information and guidance. You can send questions
to OLAW Help or find information for the grantee
community on the OLAW
listserv.

Left to Right:
Axel Wolff, Director, Division of Compliance Oversight;
Carol Wigglesworth, Acting Director, Office the Director;
Denis Doyle, Director, Division of Assurances
 Back
to top Back
to top
|
|
THE
AMERICAN PHYSIOLOGICAL SOCIETY ISSUES A RESOURCE
BOOK FOR THE DESIGN OF ANIMAL EXERCISE PROTOCOLS
|
|
With sponsorship from the NIH
Office of Laboratory Animal Welfare (OLAW),
the American Physiological Society (APS) has
published the
Resource Book for the Design of Animal Exercise
Protocols. Experts in the fields of exercise
physiology and animal research models developed
the book, intended for researchers, Institutional
Animal Care and Use Committees (IACUCs), and those
involved with research oversight. The authoring
committee, which comprised exercise physiologists
and laboratory animal veterinarians, reviewed
reference material and drew upon their own
experience to compile suggestions about how to
design, review, and implement experimental
paradigms involving animals and exercise. The APS
Resource Book was peer reviewed by other exercise
physiologists and laboratory animal veterinarians.
The opening chapter of the
Resource Book outlines the scope of the document
and addresses the relevance of studying exercise
in general as well as the specific question, why
study exercise in animals? It explains how the use
of animals in exercise protocols contained in the
APS Resource Book fit into the context of U.S.
animal welfare requirements, including the Animal
Welfare Act, the Public Health Service Policy on
Humane Care and Use of Animals, and the Institute
of Laboratory Animal Resources Guide for the Care
and Use of Laboratory Animals. Specifically, the
APS Resource Book is intended to promote an
informed dialogue that can help researchers and
their IACUCs arrive at satisfactory answers to
questions about how to assure the welfare of
animals in exercise research protocols.
Single copies of the APS
Resource Book are available free of charge from
OLAW while supplies last;
contact OLAW to request a free copy. Copies
also may be purchased for $9.50 from the
APS store.
 Back
to top Back
to top
|
|
NIH BALANCED SCORECARD GRANTEE SATISFACTION SURVEY: COMING IN
MAY 2006
|
|
Starting in May 2006, you may
be selected to participate in a Grantee
Satisfaction Survey to asses the performance of
NIH Grants Management Offices and Program Offices.
Your feedback as a grantee is important to helping
us improve the grants management function at NIH.
In 2000, NIH began
undertaking periodic self-assessments based on
Balanced Scorecard (BSC) methodology, a strategic
management tool developed in recent years. BSC
methodology attempts to evaluate a system's
operation from multiple perspectives to provide an
overview of the effectiveness of the operational
process under review. In doing so, it identifies
areas needing improvement and can further serve as
tool for effective strategic management within an
organization. All HHS Operating Divisions are now
conducting assessments via BSC surveys. An
NIH-wide Grantee Survey was last conducted in
2002.
The Grantee Satisfaction
Survey is Web-based and by invitation only. The
survey will be sent to a random sample of
grantees, including Principal Investigators,
Business Offices, and Program Directors, so more
than one individual from an organization may be
selected. Selected participants will receive an
e-mail invitation which will include a link to the
survey.
Survey respondents will be
asked to evaluate both the Grants Management
Office and Program Offices they interact with on
all phases of the grants process from the
pre-award phase to reporting and post-award
administration, and overall satisfaction.
The contractor for this effort is LMI Government
Consulting, located in McLean, VA. They will host
the Grantee Survey on their servers and will
contact those selected to participate directly.
The Office of Extramural Research’s Office of
Policy for Extramural Research Administration is
assisting LMI in coordinating the survey effort.
 Back
to top Back
to top
|
|
NATIONAL LEADERSHIP FORUM ANNOUNCES WORKSHOP ON
NIH ROADMAP—INVENTORY AND EVALUATION OF CLINICAL
RESEARCH NETWORKS |
|
The National Leadership Forum (NLF) will present
findings from the NIH-sponsored Inventory and
Evaluation of Clinical Research Networks (IECRN)
on May 31 and June 1, 2006, at the DoubleTree
Hotel in Rockville, MD.
The NLF will discuss ways to improve the
infrastructure of clinical research networks and
make recommendations for increasing the efficiency
and effectiveness of research networks. The target
audience includes principal investigators,
epidemiologists, behavior scientists, clinical
study coordinators, pharmacists, biostatisticians,
clinicians, data managers, bioinformatics and
information technologists. Registration will be
limited to the first 500 participants due to room
capacity. The Forum is open to the public. The
plenary sessions will be simultaneously Web cast.
Details are available through the
NIH Roadmap Web site.
 Back
to top Back
to top
|
|
OHRP
ANNOUNCES NEW WEB RESOURCE |
|
 The
HHS Office for Human Research Protections
(OHRP) invites you to visit the
new OHRP human subject protections Web resource
page. It provides links to reference
documents, historical materials and Common Rule
departments and agencies. Additional materials
will be added as they become available. The
HHS Office for Human Research Protections
(OHRP) invites you to visit the
new OHRP human subject protections Web resource
page. It provides links to reference
documents, historical materials and Common Rule
departments and agencies. Additional materials
will be added as they become available.
OHRP encourages all in the
research community to use this reference page.
 Back
to top Back
to top
|
|
|
|
|
|

|
|
|
The NIH Extramural Nexus is a bimonthly update
from the NIH Office of Extramural Research. Articles,
comments, questions and suggestions may be addressed to
the Editor.
The NIH Extramural Nexus reserves the right to
select and edit items submitted for inclusion.
To subscribe to the NIH Extramural Nexus, send a
plain text email to Listserv@list.nih.gov
including only the words Subscribe EXTRAMURALNEXUS
in the body of the message. To unsubscribe, follow the
same procedure, using the words Unsubscribe
EXTRAMURALNEXUS in the message body.
|
|
|
|
|
|
