Appendix E: Stem Cell Markers
Appendix E.i. How Do Researchers Use Markers to Identify Stem Cells?
In recent years, scientists have discovered a wide array of stem cells that have unique capabilities to self-renew, grow indefinitely, and differentiate or develop into multiple types of cells and tissues. Researchers now know that many different types of stem cells exist but they all are found in very small populations in the human body, in some cases 1 stem cell in 100,000 cells in circulating blood. And, when scientists examine these cells under a microscope, they look just like any other cell in the tissue where they are found. So, how do scientists identify these rare type of cells found in many different cells and tissues—a process that is much akin to finding a needle in a haystack? The answer is rather simple thanks to stem cell "markers." This feature describes stem cell marker technology and how it is used in the research laboratory. Following this is a listing of some of the commonly used stem cell markers (see Appendix E.ii. Markers Commonly Used to Identify Stem Cells and to Characterize Differentiated Cell Types).
What are stem cell markers? Coating the surface of every cell in the body are specialized proteins, called receptors, that have the capability of selectively binding or adhering to other "signaling" molecules. There are many different types of receptors that differ in their structure and affinity for the signaling molecules. Normally, cells use these receptors and the molecules that bind to them as a way of communicating with other cells and to carry out their proper functions in the body. These same cell surface receptors are the stem cell markers. Each cell type, for example a liver cell, has a certain combination of receptors on their surface that makes them distinguishable from other kinds of cells. Scientists have taken advantage of the biological uniqueness of stem cell receptors and chemical properties of certain compounds to tag or "mark" cells. Researchers owe much of the past success in finding and characterizing stem cells to the use of markers.
Stem cell markers are given short-hand names based on the molecules that bind to the stem cell surface receptors. For example, a cell that has the receptor stem cell antigen -1, on its surface, is identified as Sca-1. In many cases, a combination of multiple markers is used to identify a particular stem cell type. So now, researchers often identify stem cells in shorthand by a combination of marker names reflecting the presence (+) or absence (-) of them. For example, a special type of hematopoietic stem cell from blood and bone marrow called "side population" or "SP" is described as (CD34-/low, c-Kit+, Sca-1+) [4].
Researchers use the signaling molecules that selectively adhere to the receptors on the surface of the cell as a tool that allows them to identify stem cells. Many years ago, a technique was developed to attach to the signaling molecule another molecule (or the tag) that has the ability to fluoresce or emit light energy when activated by an energy source such as an ultraviolet light or laser beam (see Figure E.i.1. Identifying Cell Surface Markers Using Fluorescent Tags). At the researchers' disposal are multiple fluorescent tags with emitted light that differ in color and intensity.
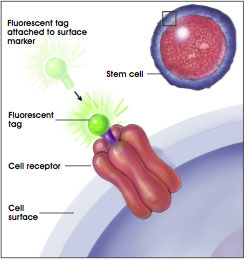
Figure E.i.1. Identifying Cell Surface Markers Using Fluorescent Tags.
(© 2001 Terese Winslow)
Described here are two approaches of how researchers use the combination of the chemical properties of fluorescence and unique receptor patterns on cell surfaces to identify specific populations of stem cells. One approach for using markers as a research tool is with a technique known as fluorescence-activated cell sorting (FACS) (see Figure E.i.2. Looking for a Needle in a Haystack: How Researchers Find Stem Cells) [1, 3, 5]. Researchers often use a FACS instrument to sort out the rare stem cells from the millions of other cells. With this technique, a suspension of tagged cells (i.e., bound to the cell surface markers are fluorescent tags) is sent under pressure through a very narrow nozzle—so narrow that cells must pass through one at a time. Upon exiting the nozzle, cells then pass, one-by-one, through a light source, usually a laser, and then through an electric field. The fluorescent cells become negatively charged, while nonfluorescent cells become positively charged. The charge difference allows stem cells to be separated from other cells. The researchers now have a population of cells that have all of the same marker characteristics, and with these cells they can conduct their research.
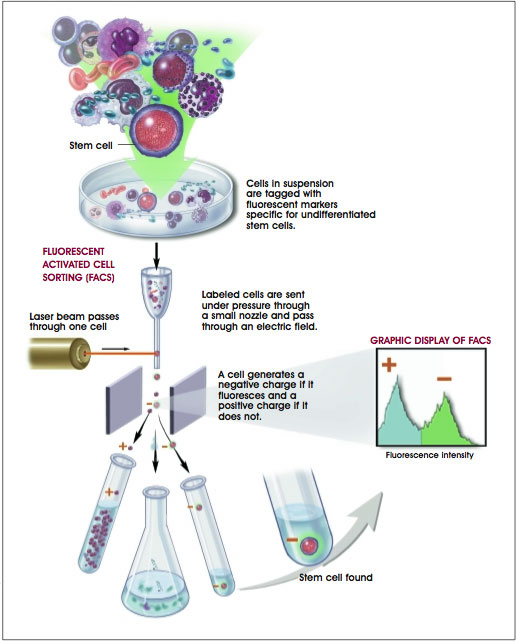
Figure E.i.2. Looking for a Needle in a Haystack: How Researchers Find Stem Cells.
(© 2001 Terese Winslow, Lydia Kibiuk, Caitlin Duckwall)
A second method uses stem cell markers and their fluorescent tags to visually assess cells as they exist in tissues. Often researchers want to assess how stem cells appear in tissues and in doing so they use a microscope to evaluate them rather than the FACS instrument. In this case, a thin slice of tissue is prepared, and the stem cell markers are tagged by the signaling molecule that has the fluorescent tag attached. The fluorescent tags are then activated either by special light energy or a chemical reaction. The stem cells will emit a fluorescent light that can easily be seen under the microscope.
Genetic and molecular biology techniques are extensively used to study how cells become specialized in the organism's development. In doing so, researchers have identified genes and transcription factors (proteins found within cells that regulate a gene's activity) that are unique in stem cells. Scientists use techniques such as polymerase chain reaction (PCR) to detect the presence of genes that are "active" and play a role guiding the specialization of a cell. This technique has is helpful to researchers to identify "genetic markers" that are characteristic of stem cells. For example, a gene marker called PDX-1 is specific for a transcription factor protein that initiates activation of the insulin gene. Researchers use this marker to identify cells that are able to develop islet cells in the pancreas.
Recently, researchers have applied a genetic engineering approach that uses fluorescence, but isn't dependent on cell surface markers. The importance of this new technique is that it allows the tracking of stem cells as they differentiate or become specialized. Scientists have inserted into a stem cell a "reporter gene" called green fluorescent protein or GFP [2]. The gene is only activated or "reports" when cells are undifferentiated and is turned off once they become specialized. Once activated, the gene directs the stem cells to produce a protein that fluoresces in a brilliant green color (see Figure E.i.3. Microscopic Image of Fluorescent-Labeled Stem Cell). Researchers are now coupling this reporting method with the FACS and microscopic methods described earlier to sort cells, identify them in tissues, and now, track them as they differentiate or become specialized.
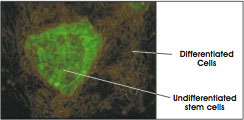
Figure E.i.3. Microscopic Image of Fluorescent-Labeled Stem Cell.
(© 2001 Terese Winslow)
These discovery tools are commonly used in research laboratories and clinics today, and will likely play important roles in advancing stem cell research. There are limitations, however. One of them is that a single marker identifying pluripotent stem cells, those stem cells that can make any other cell, has yet to be found. As new types of stem cells are identified and research applications of them become increasingly complex, more sophisticated tools will be developed to meet investigators' needs. For the foreseeable future, markers will continue to play a major role in the rapidly evolving world of stem cell biology.
References
- Bonner, W.A., Hulett, H.R., Sweet, R.G., and Herzenberg, L.A. (1972). Fluorescence activated cell sorting. Rev. Sci. Instrum. 43, 404–409.
- Eiges, R., Schuldiner, M., Drukker, M., Yanuka, O., Itskovitz-Eldor, J., and Benvenisty, N. (2001). Establishment of human embryonic sten cell-transduced clones carrying a marker of undifferentiated cells. Curr. Biol. 11, 514–518.
- Herzenberg, L.A. and De Rosa, S.C. (2000). Monoclonal antibodies and the FACS: complementary tools for immunobiology and medicine. Immunol. Today. 21, 383390.
- Jackson, K., Majka SM, Wang H, Pocius J, Hartley CJ, Majesky MW, Entman ML, Michael LH, Hirschi KK, and Goodell MA (2001). Regeneration of ischemic cardiac muscle and vascular endothelium by adult stem cells. J. Clin. Invest. 107, 1–8.
- Julius, M.H., Masuda, T., and Herzenberg, L.A. (1972). Demonstration that antigen-binding cells are precursors of antibody-producing cells after purification with a fluorescence-activated cell sorter. Proc. Natl. Acad. Sci. U. S. A. 69, 1934–1938.
Appendix E.ii. Markers Commonly Used to Identify Stem Cells and to Characterize Differentiated Cell Types
| Marker Name |
Cell Type |
Significance |
| Blood Vessel |
| Fetal liver kinase-1 (Flk1) |
Endothelial |
Cell-surface receptor protein that identifies endothelial cell progenitor; marker of cell-cell contacts |
| Smooth muscle cell-specific myosin heavy chain |
Smooth muscle |
Identifies smooth muscle cells in the wall of blood vessels |
| Vascular endothelial cell cadherin |
Smooth muscle |
Identifies smooth muscle cells in the wall of blood vessels |
| Bone |
| Bone-specific alkaline phosphatase (BAP) |
Osteoblast |
Enzyme expressed in osteoblast; activity indicates bone formation |
| Hydroxyapatite |
Osteoblast |
Minerlized bone matrix that provides structural integrity; marker of bone formation |
| Osteocalcin (OC) |
Osteoblast |
Mineral-binding protein uniquely synthesized by osteoblast; marker of bone formation |
| Bone Marrow and Blood |
| Bone morphogenetic protein receptor (BMPR) |
Mesenchymal stem and progenitor cells |
Important for the differentiation of committed mesenchymal cell types from mesenchymal stem and progenitor cells; BMPR identifies early mesenchymal lineages (stem and progenitor cells) |
| CD4 and CD8 |
White blood cell (WBC) |
Cell-surface protein markers specific for mature T lymphocyte (WBC subtype) |
| CD34 |
Hematopoietic stem cell (HSC), satellite, endothelial progenitor |
Cell-surface protein on bone marrow cell, indicative of a HSC and endothelial progenitor; CD34 also identifies muscle satellite, a muscle stem cell |
| CD34+Sca1+ Lin- profile |
Mesencyhmal stem cell (MSC) |
Identifies MSCs, which can differentiate into adipocyte, osteocyte, chondrocyte, and myocyte |
| CD38 |
Absent on HSC
Present on WBC lineages |
Cell-surface molecule that identifies WBC lineages. Selection of CD34+/CD38- cells allows for purification of HSC populations |
| CD44 |
Mesenchymal |
A type of cell-adhesion molecule used to identify specific types of mesenchymal cells |
| c-Kit |
HSC, MSC |
Cell-surface receptor on BM cell types that identifies HSC and MSC; binding by fetal calf serum (FCS) enhances proliferation of ES cells, HSCs, MSCs, and hematopoietic progenitor cells |
| Colony-forming unit (CFU) |
HSC, MSC progenitor |
CFU assay detects the ability of a single stem cell or progenitor cell to give rise to one or more cell lineages, such as red blood cell (RBC) and/or white blood cell (WBC) lineages |
| Fibroblast colony-forming unit (CFU-F) |
Bone marrow fibroblast |
An individual bone marrow cell that has given rise to a colony of multipotent fibroblastic cells; such identified cells are precursors of differentiated mesenchymal lineages |
| Hoechst dye |
Absent on HSC |
Fluorescent dye that binds DNA; HSC extrudes the dye and stains lightly compared with other cell types |
| Leukocyte common antigen (CD45) |
WBC |
Cell-surface protein on WBC progenitor |
| Lineage surface antigen (Lin) |
HSC, MSC
Differentiated RBC and WBC lineages |
Thirteen to 14 different cell-surface proteins that are markers of mature blood cell lineages; detection of Lin-negative cells assists in the purification of HSC and hematopoietic progenitor populations |
| Mac-1 |
WBC |
Cell-surface protein specific for mature granulocyte and macrophage (WBC subtypes) |
| Muc-18 (CD146) |
Bone marrow fibroblasts, endothelial |
Cell-surface protein (immunoglobulin superfamily) found on bone marrow fibroblasts, which may be important in hematopoiesis; a subpopulation of Muc-18+ cells are mesenchymal precursors |
| Stem cell antigen (Sca-1) |
HSC, MSC |
Cell-surface protein on bone marrow (BM) cell, indicative of HSC and MSC Bone Marrow and Blood cont. |
| Stro-1 antigen |
Stromal (mesenchymal) precursor cells, hematopoietic cells |
Cell-surface glycoprotein on subsets of bone marrow stromal (mesenchymal) cells; selection of Stro-1+ cells assists in isolating mesenchymal precursor cells, which are multipotent cells that give rise to adipocytes, osteocytes, smooth myocytes, fibroblasts, chondrocytes, and blood cells |
| Thy-1 |
HSC, MSC |
Cell-surface protein; negative or low detection is suggestive of HSC |
| Cartilage |
| Collagen types II and IV |
Chondrocyte |
Structural proteins produced specifically by chondrocyte |
| Keratin |
Keratinocyte |
Principal protein of skin; identifies differentiated keratinocyte |
| Sulfated proteoglycan |
Chondrocyte |
Molecule found in connective tissues; synthesized by chondrocyte |
| Fat |
| Adipocyte lipid-binding protein (ALBP) |
Adipocyte |
Lipid-binding protein located specifically in adipocyte |
| Fatty acid transporter (FAT) |
Adipocyte |
Transport molecule located specifically in adipocyte |
| Adipocyte lipid-binding protein (ALBP) |
Adipocyte |
Lipid-binding protein located specifically in adipocyte |
| General |
| Y chromosome |
Male cells |
Male-specific chromosome used in labeling and detecting donor cells in female transplant recipients |
| Karyotype |
Most cell types |
Analysis of chromosome structure and number in a cell |
| Liver |
| Albumin |
Hepatocyte |
Principal protein produced by the liver; indicates functioning of maturing and fully differentiated hepatocytes |
| B-1 integrin |
Hepatocyte |
Cell-adhesion molecule important in cell-cell interactions; marker expressed during development of liver |
| Nervous System |
| CD133 |
Neural stem cell, HSC |
Cell-surface protein that identifies neural stem cells, which give rise to neurons and glial cells |
| Glial fibrillary acidic protein (GFAP) |
Astrocyte |
Protein specifically produced by astrocyte |
| Microtubule-associated protein-2 (MAP-2) |
Neuron |
Dendrite-specific MAP; protein found specifically in dendritic branching of neuron |
| Myelin basic protein (MPB) |
Oligodendrocyte |
Protein produced by mature oligodendrocytes; located in the myelin sheath surrounding neuronal structures |
| Nestin |
Neural progenitor |
Intermediate filament structural protein expressed in primitive neural tissue |
| Neural tubulin |
Neuron |
Important structural protein for neuron; identifies differentiated neuron |
| Neurofilament (NF) |
Neuron |
Important structural protein for neuron; identifies differentiated neuron |
| Neurosphere |
Embryoid body (EB), ES |
Cluster of primitive neural cells in culture of differentiating ES cells; indicates presence of early neurons and glia |
| Noggin |
Neuron |
A neuron-specific gene expressed during the development of neurons |
| O4 |
Oligodendrocyte |
Cell-surface marker on immature, developing oligodendrocyte |
| O1 |
Oligodendrocyte |
Cell-surface marker that characterizes mature oligodendrocyte |
| Synaptophysin |
Neuron |
Neuronal protein located in synapses; indicates connections between neurons |
| Tau |
Neuron |
Type of MAP; helps maintain structure of the axon |
| Pancreas |
| Cytokeratin 19 (CK19) |
Pancreatic epithelium |
CK19 identifies specific pancreatic epithelial cells that are progenitors for islet cells and ductal cells |
| Glucagon |
Pancreatic islet |
Expressed by alpha-islet cell of pancreas |
| Insulin |
Pancreatic islet |
Expressed by beta-islet cell of pancreas Pancreas |
| Insulin-promoting factor-1 (PDX-1) |
Pancreatic islet |
Transcription factor expressed by beta-islet cell of pancreas |
| Nestin |
Pancreatic progenitor |
Structural filament protein indicative of progenitor cell lines including pancreatic |
| Pancreatic polypeptide |
Pancreatic islet |
Expressed by gamma-islet cell of pancreas |
| Somatostatin |
Pancreatic islet |
Expressed by delta-islet cell of pancreas |
| Pluripotent Stem Cells |
| Alkaline phosphatase |
Embryonic stem (ES), embryonal carcinoma (EC) |
Elevated expression of this enzyme is associated with undifferentiated pluripotent stem cell (PSC) |
| Alpha-fetoprotein (AFP) |
Endoderm |
Protein expressed during development of primitive endoderm; reflects endodermal differentiation Pluripotent Stem Cells |
| Bone morphogenetic protein-4 |
Mesoderm |
Growth and differentiation factor expressed during early mesoderm formation and differentiation |
| Brachyury |
Mesoderm |
Transcription factor important in the earliest phases of mesoderm formation and differentiation; used as the earliest indicator of mesoderm formation |
| Cluster designation 30 (CD30) |
ES, EC |
Surface receptor molecule found specifically on PSC |
| Cripto (TDGF-1) |
ES, cardiomyocyte |
Gene for growth factor expressed by ES cells, primitive ectoderm, and developing cardiomyocyte |
| GATA-4 gene |
Endoderm |
Expression increases as ES differentiates into endoderm |
| GCTM-2 |
ES, EC |
Antibody to a specific extracellular-matrix molecule that is synthesized by undifferentiated PSCs |
| Genesis |
ES, EC |
Transcription factor uniquely expressed by ES cells either in or during the undifferentiated state of PSCs |
| Germ cell nuclear factor |
ES, EC |
Transcription factor expressed by PSCs |
| Hepatocyte nuclear factor-4 (HNF-4) |
Endoderm |
Transcription factor expressed early in endoderm formation |
| Nestin |
Ectoderm, neural and pancreatic progenitor |
Intermediate filaments within cells; characteristic of primitive neuroectoderm formation |
| Neuronal cell-adhesion molecule (N-CAM) |
Ectoderm |
Cell-surface molecule that promotes cell-cell interaction; indicates primitive neuroectoderm formation |
| Oct-4 |
ES, EC |
Transcription factor unique to PSCs; essential for establishment and maintenance of undifferentiated PSCs |
| Pax6 |
Ectoderm |
Transcription factor expressed as ES cell differentiates into neuroepithelium |
| Stage-specific embryonic antigen-3 (SSEA-3) |
ES, EC |
Glycoprotein specifically expressed in early embryonic development and by undifferentiated PSCs |
| Stage-specific embryonic antigen-4 (SSEA-4) |
ES, EC |
Glycoprotein specifically expressed in early embryonic development and by undifferentiated PSCs |
| Stem cell factor (SCF or c-Kit ligand) |
ES, EC, HSC, MSC |
Membrane protein that enhances proliferation of ES and EC cells, hematopoietic stem cell (HSCs), and mesenchymal stem cells (MSCs); binds the receptor c-Kit |
| Telomerase |
ES, EC |
An enzyme uniquely associated with immortal cell lines; useful for identifying undifferentiated PSCs |
| TRA-1-60 |
ES, EC |
Antibody to a specific extracellular matrix molecule is synthesized by undifferentiated PSCs |
| TRA-1-81 |
ES, EC |
Antibody to a specific extracellular matrix molecule normally synthesized by undifferentiated PSCs |
| Vimentin |
Ectoderm, neural and pancreatic progenitor |
Intermediate filaments within cells; characteristic of primitive neuroectoderm formation |
| Skeletal Muscle/Cardiac/Smooth Muscle |
| MyoD and Pax7 |
Myoblast, myocyte |
Transcription factors that direct differentiation of myoblasts into mature myocytes |
| Myogenin and MR4 |
Skeletal myocyte |
Secondary transcription factors required for differentiation of myoblasts from muscle stem cells |
| Myosin heavy chain |
Cardiomyocyte |
A component of structural and contractile protein found in cardiomyocyte |
| Myosin light chain |
Skeletal myocyte |
A component of structural and contractile protein found in skeletal myocyte |
 Appendix D | Table of Contents | Appendix F
Appendix D | Table of Contents | Appendix F 
Historical content: June 17, 2001