
|
 |
Dispatch
Enteropathogenic Escherichia
coli O157 Strains from Brazil
T. Eric Blank,* David W. Lacher,† Isabel C.A. Scaletsky,‡ Hailang
Zhong,* Thomas S. Whittam,† and Michael S. Donnenberg*
*University of Maryland School of Medicine, Baltimore, Maryland, USA;
†Michigan State University, East Lansing, Michigan, USA; and ‡Universidade
Federal de São Paulo, São Paulo, Brazil
Suggested citation for this article: Blank TE,
Lacher DW, Scaletsky ICA, Zhong H, Whittam TS, Donnenberg MS. Enteropathogenic
Escherichia coli O157 strains from Brazil. Emerg Infect Dis [serial
online] 2003 Jan [date cited]. Available from: URL: http://www.cdc.gov/ncidod/EID/vol9no1/02-0072.htm
We describe two
serogroup O157 Escherichia coli strains from Brazilian infants
with diarrhea. A variety of assays indicate that these strains belong
to the enteropathogenic, not the enterohemorrhagic, pathotype. These
strains possess a novel bfpA allele encoding the type IV pilin
characteristic of typical enteropathogenic E. coli strains. Our
results emphasize the pitfalls of classifying pathogenic E. coli
by serogroup.
Enterohemorrhagic Escherichia coli (EHEC) O157:H7 is the serotype
most commonly associated with hemorrhagic colitis and the hemolytic uremic
syndrome (1). EHEC strains share with enteropathogenic
E. coli (EPEC), a leading cause of infant diarrhea in developing
countries, the ability to induce the attaching and effacing effect on
host cells. This property is specified by a pathogenicity island that
includes the eae gene encoding the outer membrane adhesin intimin.
EPEC are defined by this attaching and effacing phenotype or, at a molecular
level, by the presence of the eae gene and the absence of the genes
for Shiga toxins (2). Typical EPEC strains have a large
plasmid that encodes bundle-forming pili and the localized adherence phenotype,
while atypical strains lack these properties. EHEC differ from EPEC in
that they produce Shiga toxins but not bundle-forming pili.
Here we describe two strains of the O157 serogroup identified as part
of an ongoing epidemiologic survey of pathogenic E. coli in Brazil
(3). Strain SC373/2 was isolated from a 9-month-old infant
in Joinville, Santa Catarina, in 1997, and RN587/1, from a 7-month-old
infant in Natal, Rio Grande do Norte, in 1998. Both patients had acute
diarrhea of 7 days’ duration, accompanied by vomiting, fever, and moderate
dehydration. These strains were the only enteropathogenic bacteria isolated
from the patients’ stools. Rotavirus and cryptosporidia were not detected.
We used a combination of phenotypic assays and DNA sequencing to further
characterize these isolates. DNA probe testing was performed by established
methods (4). A fragment of the eae gene was amplified
by polymerase chain reaction (PCR) and sequenced as described (5).
Serotyping was performed at the E. coli Reference Laboratory (University
Park, PA). Tests of localized adherence, autoaggregation, and attaching
and effacing were performed as described (6,7). The bfpA
gene was amplified by PCR and sequenced as described (8).
Multilocus sequence typing was performed as described (9).
We found a number of indications that these O157 strains are not EHEC
but unexpectedly fall within the EPEC pathotype. First, both strains tested
probe negative for the Shiga toxin (stx) genes characteristic of
EHEC. Second, both strains carried an eae (intimin) gene, identified
by sequencing as an a allele characteristic of the group of strains known
as EPEC 1, rather than the γ allele characteristic of O157:H7 EHEC
(C.L. Tarr and T.S. Whittam, unpub. data). As expected of both EPEC and
EHEC, both strains are capable of attaching and effacing as demonstrated
by using the fluorescence actin staining test. E. coli strains
that are eae+ and stx– are by definition, EPEC
(2). Third, both strains lacked the H7 flagellar antigen,
although they were motile. The H antigens were difficult to type; RN587/1
reacts weakly with H8 antiserum. Fourth, both strains rapidly fermented
sorbitol, a phenotype lacking in most O157:H7 strains. Fifth, the nucleotide
sequences of part of the coding regions of 14 chromosomal loci encoding
proteins with housekeeping functions indicated a close relationship of
these strains with the EPEC 1 clonal group. The DNA sequences of the two
O157 strains were identical over a total of 2,240 codons. Comparison of
the multilocus sequence data to the reference strains K-12, EDL-933 (O157:H7;
EHEC 1), and E2348/69 (O127:H6; EPEC 1) indicated that the EPEC O157 isolates
were most closely related to E2348/69. The percentage of nucleotide difference
in the total of 6,720 bp was 0.72 ± 0.10 (E2348/69), 2.66 ± 0.20 (K-12),
and 2.95 ± 0.21 (EDL-933). Phylogenetic analysis with these sequences
from a diverse collection of strains, including EPEC, EHEC, and other
pathotypes, likewise shows that RN587/1 and SC373/2 are much more closely
related to EPEC 1 than to EHEC O157 strains (Figure
1). Both strains also tested probe positive for the EPEC adherence
factor plasmid, which is associated with localized adherence.
Finally, both strains carried the bfpA gene encoding bundlin,
the structural subunit of the bundle-forming pilus expressed by typical
EPEC strains but not by EHEC. Both strains are also capable of autoaggregation
and localized adherence, the two phenotypes associated with expression
of bundle-forming pili. The bfpA gene isolated from these
strains has a unique sequence (GenBank accession number AF474407). We
call this version of bfpA the β6 allele. When compared to
the a and β bfpA alleles described previously (8),
the β6 bfpA allele maps between the β1 allele and a cluster
of the β2–β5 alleles (Figure 2). The
predicted β6 bundlin amino acid sequence maps between the β1
and β2 and the β3–5 pilin proteins.
Our findings emphasize the fact that E. coli with the O157 O antigen
are not always EHEC but may belong to other pathotypes. Scotland et al.
described Shiga toxin–negative O157:H8 E. coli strains isolated
from children with diarrhea (10) . These strains were
positive for attaching and effacing and localized adherence phenotypes
and are therefore EPEC, although, unlike the strains we describe, they
were negative for the adherence factor probe. EPEC strains of the O157:H45
serotype have been noted as agents of diarrhea in a large outbreak in
Japan and isolated cases in Germany, Japan, and Thailand (11–13).
These previously described O157 EPEC strains were not tested for bundlin
alleles, and their phylogenetic relationships to other pathogenic E.
coli have not been reported. However, the results of several studies
have indicated that O157 strains lacking the H7 flagellar antigen and
stx genes, which were not necessarily EPEC, are clonally distinct
from O157:H7 strains and often from each other (14-16).
Such findings suggest that recombination events causing the O antigen
gene cluster to specify the O157 antigen have occurred in multiple E.
coli lineages, including an EPEC 1 strain of an unknown serotype that
is the progenitor of the strains described here. The results described
in this study also highlight the pitfalls of classifying pathogenic E.
coli by serogroup.
Acknowledgments
We are grateful to Wensheng Luo and Xiaolin Wang for technical assistance.
This work was supported by
Public Health Service grant (R01 AI-37606) to M.S.D., a National Research
Service Award postdoctoral training fellowship (F32 AI-10191) to T.E.B.,
and in part by the National Institutes of Health through the Enteric
Pathogens Research Unit, University of Maryland Medical School (T.S.W.).
Dr. Blank is a microbiologist
with GEO-CENTERS, Inc. and conducts research in the laboratory of Arthur
Friedlander at the U.S. Army Medical Research Institute of Infectious
Diseases in Frederick, Maryland. Dr. Blank’s research focuses on genetic
variations that attenuate the virulence of bacterial pathogens.
References
- Donnenberg MS, Whittam TS. Pathogenesis
and evolution of virulence in enteropathogenic and enterohemorrhagic
Escherichia coli. J Clin Invest 2001;107:539–48.
- Kaper JB. Defining EPEC. Revista de Microbiologia, Sao Paulo 1996;27(Suppl1):130–3.
- Scaletsky ICA, Fabbricotti SH, Aranda KR, Morais MB, Fagundes-Neto
U. Comparison
of DNA hybridization and PCR assays for detection of putative pathogenic
enteroadherent Escherichia coli. J Clin Microbiol 2002;40:1254–8.
- Scotland SM, Smith HR, Cheasty T, Said B, Willshaw GA, Stokes N, et
al. Use
of gene probes and adhesion tests to characterise Escherichia coli
belonging to enteropathogenic serogroups isolated in the United Kingdom.
J Med Microbiol 1996;44:438–43.
- McGraw EA, Li J, Selander RK, Whittam TS. Molecular
evolution and mosaic structure of
 , ,
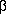 , and , and  intimins of pathogenic Escherichia coli. Mol Biol Evol 1999;16:12–22.
intimins of pathogenic Escherichia coli. Mol Biol Evol 1999;16:12–22.
- Donnenberg MS, Nataro JP. Methods
for studying adhesion of diarrheagenic Escherichia coli.
Methods Enzymol 1995;253:324–36.
- Anantha RP, Stone KD, Donnenberg MS. Effects
of bfp mutations on biogenesis of functional enteropathogenic
Escherichia coli type IV pili. J Bacteriol 2000;182:2498–506.
- Blank TE, Zhong H, Bell AL, Whittam TS, Donnenberg MS. Molecular
variation among type IV pilin (bfpA) genes from diverse enteropathogenic
Escherichia coli strains. Infect Immun 2000;68:7028–38.
- Reid SD, Herbelin CJ, Bumbaugh AC, Selander RK, Whittam TS. Parallel
evolution of virulence in pathogenic Escherichia coli. Nature
2000;406:64–7.
- Scotland SM, Willshaw GA, Cheasty T, Rowe B. Strains
of Escherichia coli O157:H8 from human diarrhoea belong to attaching
and effacing class of E. coli. J Clin Pathol 1992;45:1075–8.
- Schmidt H, Rüssmann H, Karch H. Virulence
determinants in nontoxinogenic Escherichia coli O157 strains
that cause infantile diarrhea. Infect Immun 1993;61:4894–8.
- Makino S, Asakura H, Shirahata T, Ikeda T, Takeshi K, Arai K, et al.
Molecular
epidemiological study of a mass outbreak caused by enteropathogenic
Escherichia coli O157:H45. Microbiol Immunol 1999;43:381–4.
- Echeverria P, Orskov F, Orskov I, Knutton S, Scheutz F, Brown JE,
et al. Attaching
and effacing enteropathogenic Escherichia coli as a cause of
infantile diarrhea in Bangkok. J Infect Dis 1991;164:550–4.
- Whittam TS, Wilson RA. Genetic
relationships among pathogenic Escherichia coli of serogroup
O157. Infect Immun 1988;56:2467–73.
- Feng P, Lampel KA, Karch H, Whittam TS. Genotypic
and phenotypic changes in the emergence of Escherichia coli O157:H7.
J Infect Dis 1998;177:1750–3.
- Tarr PI, Schoening LM, Yea YL, Ward TR, Jelacic S, Whittam TS. Acquisition
of the rfb-gnd cluster in evolution of Escherichia coli
O55 and O157. J Bacteriol 2000;182:6183–91.
|
