4.1. Detection limit of the analytical procedure
The detection limit of the analytical procedure was determined by injecting 0.40 µL of an analytical standard of 0.40 µg/mL (0.40 µg/mL × 0.40 µL = 0.16 ng/injection). This
gave a peak whose height was approximately five times the baseline noise (Figure 4.1.).
4.2. Detection limit of the overall procedure
The detection limit of the overall procedure is 0.45 µg/sample (0.045 mg/m3 or 0.02 ppm). This is the amount of DMF which, when spiked on the sampling device, will allow recovery of an amount approximately equivalent to the detection limit of the analytical procedure (0.16 ng/injection or 0.40 µg/sample).
Table 4.2.
Data for Detection Limit of the Overall Procedure
|
sample
number |
theoretical amount
(µg) |
amount recovered
(µg) |
|
| 1 |
0.4528 |
0.4528 |
| 2 |
0.4528 |
0.4057 |
| 3 |
0.4528 |
0.4333 |
| 4 |
0.4528 |
0.4664 |
| 5 |
0.4528 |
0.4338 |
| 6 |
0.4528 |
0.4247 |
|
4.3. Reliable quantitation limit
The reliable quantitation limit is that amount of DMF which, when spiked onto the sampling device, will allow recovery of at least 75% and have a precision (1.96 SD) of ±25% or better. The reliable quantitation limit is the same as the detection limit of the overall procedure in this evaluation (using the recommended injection size of 0.40 µL). The front sections of six SKC Lot 120 charcoal tubes were spiked with 8 µL of a 56.6 µg/mL solution of DMF in acetone, and stored in a laboratory refrigerator overnight. The tubes were desorbed with 1.0 mL of acetone containing 1 µL/mL propionitrile as an internal standard and analyzed.
Table 4.3.
Data for Reliable Quantitation Limit
(Based on samples and data of Table 4.2.)
|
sample
number |
percent
recovered |
statistics |
|
1 |
100. |
 = = |
96.3% |
| 2 |
89.6 |
| 3 |
95.7 |
| 4 |
103. |
SD = |
4.7% |
| 5 |
95.8 |
Precision = |
(1.96)(±4.7%) |
| 6 |
93.8 |
= |
±9.2% |
|
4.4. Instrument response to the analyte
The instrument response to the analyte over the range of 0.5 to 2 times the PEL is linear with a sensitivity of 3800 area counts per (µg/mL). (Table 4.4., Figure. 4.4.)
Table 4.4.
DMF Sensitivity Data
|
× target conc.
µg/mL |
0.5×
142 |
1×
283 |
2×
566 |
|
| area |
458500 |
1104000 |
2114000 |
| counts |
469400 |
1105000 |
2130000 |
|
482500 |
1105000 |
2140000 |
|
497300 |
1100000 |
2150000 |
|
507800 |
1103000 |
2159000 |
|
514200 |
|
2159000 |
|
 |
488283 |
1103400 |
2142000 |
|
4.5. Storage Data
Thirty-three charcoal tubes were liquid spiked with a loading equivalent to the PEL based on a 10-L air volume. Three tubes were desorbed and analyzed immediately. Of the remaining thirty tubes, fifteen were refrigerated at -5°C and fifteen were stored at ambient temperature in a laboratory drawer. Approximately every three days, three tubes from the refrigerated group and three from the ambient group were desorbed and analyzed. The recovery of DMF from charcoal, as determined from a best fit curve, remained above 91% during the sixteen day storage test. No appreciable difference was observed between
the refrigerated and the ambient samples (Table 4.5.1., Figures
4.5.1. and 4.5.2.).
Table 4.5.1.
Storage Study of Liquid Spiked Samples
|
| storage time |
|
% recovery |
| (days) |
|
(refrigerated) |
|
(ambient) |
|
| 0 |
|
93.2 |
90.9 |
96.4 |
|
93.2 |
90.9 |
96.4 |
| 4 |
91.0 |
95.1 |
93.6 |
92.4 |
92.4 |
93.0 |
| 6 |
92.2 |
92.2 |
85.0 |
91.2 |
91.9 |
91.2 |
| 9 |
89.3 |
99.7 |
94.7 |
89.2 |
91.1 |
93.3 |
| 13 |
89.0 |
94.7 |
92.4 |
89.6 |
90.9 |
90.9 |
| 16 |
84.0 |
89.7 |
86.1 |
92.7 |
91.6 |
92.7 |
|
Storage samples were also collected from a dynamically generated atmosphere of DMF at 71% relative humidity and 23°C. Thirty-six charcoal tubes were used to sample this atmosphere. Ten-liter air samples were taken at a rate of 0.2 L/min. Six samples were
desorbed and analyzed immediately; these represent day-zero storage. The remaining thirty were divided into two groups of fifteen; one group was stored under refrigeration at -5°C and the other at ambient temperature in a laboratory drawer. The samples from
each group were analyzed so as to provide storage data at
3-day intervals for a period of fifteen days. Front and back sections of each tube were analyzed separately.
Analysis of six vapor-generated samples on day zero yielded a recovery of 65%, rather than 92% which would have been expected if the only bias present was the desorption efficiency. The additional bias was attributed to DMF loss in the vapor generating system; NIOSH reported a similar finding (Ref. 5.8.). Therefore the theoretical concentration of
the DMF test atmosphere was considered to be approximately 70% of the PEL. Since the day-zero samples would show only the effects of desorption efficiency and not storage, the normalized results were adjusted to reflect only the loss due to desorption on day zero. This was accomplished by multiplying the actual storage recoveries by 1.41 (91.7/65). These storage results, which are comparable to the storage results obtained with liquid-spiked samples, indicate no significant bias that could be attributed to storage instability
(Table 4.5.2., Figures 4.5.3. and
4.5.4.).
The precision of the overall procedure must be ±25% or better at the 95% confidence level for samples collected at the target concentration. The precision at the 95% confidence level for the 15-day storage test at ambient temperature (Figure 4.5.4.) is ±14.5%. This includes an additional ±5% for sampling error.
Table 4.5.2.
Storage Study - Vapor Generated Samples
|
| storage time |
% recovery |
| (days) |
(refrigerated) |
|
(ambient) |
|
| 0 |
94 |
95 |
93 |
|
94 |
95 |
93 |
|
90 |
93 |
91 |
90 |
93 |
91 |
| 3 |
99 |
108 |
-- |
95 |
-- |
93 |
| 6 |
99 |
99 |
98 |
97 |
97 |
97 |
| 9 |
94 |
94 |
91 |
85 |
77 |
90 |
| 12 |
101 |
104 |
101 |
97 |
94 |
95 |
| 15 |
116 |
99 |
99 |
95 |
-- |
95 |
|
4.6. Precision (analytical method only)
The precision of the analytical procedure is defined as the pooled coefficient of variation determined from replicate injections of analytical standards at 0.5, 1 and 2 times the target concentration. Based on the data of Table 4.4., coefficients of variation (CV) for the three
levels and the pooled coefficient of variation (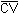 ) are given in Table 4.6.
) are given in Table 4.6.
Table 4.6.
Precision of the analytical method
(Based on the data of Table 4.4)
|
× target conc.
µg/sample |
0.5×
142 |
1×
283 |
2×
566 |
|
| SD (area counts) |
2.196 × 104 |
2.074 × 103 |
1.774 × 104 |
| CV |
0.0450 |
0.0019 |
0.0083 |
|
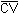 = 0.027 = 0.027 |
|
4.7. Precision (overall procedure)
The precision of the overall procedure is determined from the storage data. The determination of the standard error of estimate for a regression line plotted through the graphed storage data, as in
Figure 4.5.4., allows the inclusion of storage time as one of the factors affecting overall precision. The standard error of estimate (SEE) is similar to the standard deviation, except it is a measure of dispersion of data about a regression line
instead of about a mean. It is determined with the following equation:
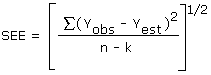
| where |
n =
k =
k = |
total no. of data points
2 for linear regression
3 for quadratic regression |
| Yobs = |
observed % recovery at a given time |
| Yest = |
estimated % recovery from the regression line at the same given time |
An additional 5% for pump error is added to the standard error of estimate by the addition of variances. The precision at the 95% confidence level is obtained by multiplying the standard error of estimate (with pump error included) by 1.96 (the z-statistic from the standard normal distribution at the 95% confidence level). The 95% confidence intervals are drawn about their respective regression lines in the storage graphs.
4.8. Reproducibility
Six liquid-spiked samples and a draft copy of this procedure were given to a chemist unassociated with this evaluation. The samples were analyzed after 1 day of storage at ambient temperature.
Table 4.8.
DMF Reproducibility
|
sample
no. |
theoretical
amount (µg) |
amount
found (µg) |
% found
|
% deviation
|
|
| 1 |
302.2 |
294.0 |
97.3 |
-2.7 |
| 2 |
234.2 |
235.3 |
100.5 |
+0.5 |
| 3 |
136.0 |
134.7 |
99.0 |
-1.0 |
| 4 |
302.2 |
295.5 |
97.8 |
-2.2 |
| 5 |
136.0 |
127.6 |
93.8 |
-6.2 |
| 6 |
234.2 |
225.6 |
96.3 |
-3.7 |
Precision of the overall procedure (from Section 1.2.7.) is ±14.5%.
DE for charcoal = 91.7%
|
4.9. Sampler Capacity
Analyte breakthrough in this evaluation was determined by sampling a dynamically generated test atmosphere at approximately twice the target level using only the front section of the sorbent tube.
Breakthrough is defined as the point at which the concentration of DMF in the atmosphere which has passed through the sorbent tube is 5% of the upstream concentration of the test atmosphere.
This initial study using the vapor generator indicated a discrepancy between the theoretical concentration of DMF and the experimentally determined concentration. This discrepancy was found when a charcoal tube that had been used to sample the atmosphere was analyzed. The experimentally determined concentration was only 70% of the theoretical
concentration (Table 4.9.1.). As previously stated in Section 4.5., NIOSH mentions this type of problem in the backup data report for their dimethylformamide method (Ref. 5.8.). A further discussion of this is given in Section 4.11.
Before proceeding with the evaluation on charcoal, the effect of humidity on the capacity of the standard size silica gel tube was determined. The NIOSH method, which uses the silica gel tube, did not address the effects of humidity. The concentration of the test atmosphere was 46.22 mg/m3 as determined by the analysis of the front portion of a silica gel tube. Breakthrough was monitored downstream from the sampling tube with a gas chromatograph equipped with a gas sampling valve. DMF was introduced into the vapor generation system through a heated inlet at a rate of 9.36 mg/min. The test atmosphere was sampled at a rate of 1 L/min and an average relative humidity of 67% at 23°C.
The results of two capacity studies on silica gel yielded an average 5% breakthrough volume of 159 L. The capacity of silica gel for DMF is 7.36 mg (Table 4.9.2., Figure 4.9.2.). Examination of the capacity data indicates that silica gel is an adequate solid sorbent for collection of DMF even at the studied humidity.
The capacity of SKC, Inc. Lot 120 charcoal was tested using the same vapor generation system and delivery rate as in the silica gel studies. The concentration of the test atmosphere, generated at 68% relative humidity and 25°C, was found to be 46.56 mg/m3
as determined by the analysis of the front portion of a charcoal tube. At a sampling rate of 1 L/min the 5% breakthrough volume was found to be 349 L and the capacity of the charcoal for DMF 16.46 mg (Table 4.9.3.,
Figure 4.9.3.). Only one capacity study was performed since the breakthrough volume greatly exceeded the commonly used air volume of 10 L for charcoal. The capacity study indicates that charcoal is an effective collection
medium for DMF.
Table 4.9.1.
Analysis of Charcoal Tube
|
theor. conc.
(mg/m3) |
air vol.
(L) |
expected
(mg) |
recovered
(mg) |
DE
corr. |
%
recovered |
|
| 61.05 |
255.9 |
15.62 |
10.10 |
11.00 |
70 |
DE for charcoal = 91.7% |
Table 4.9.2.
DMF Breakthrough with Silica Gel
|
| test |
5% breakthrough
volume (L) |
capacity
(mg) |
|
| 1 |
164.4 |
7.46 |
| 2 |
154.2 |
7.25 |
|
 |
159.3 |
7.36 |
|
Table 4.9.3.
DMF Breakthrough with Charcoal
|
| test |
5% breakthrough
volume (L) |
capacity
(mg) |
|
| 1 |
349.0 |
16.46 |
|
4.10. Desorption efficiency
The desorption efficiency was determined at 0.5, 1 and 2 times the target concentration by injecting known amounts of solutions containing DMF in acetone onto the 100-mg portion of charcoal tubes. Six tubes at both the 0.5 and 2 times the PEL level were prepared. Seven tubes were prepared at the PEL level. All tubes were capped and allowed to equilibrate overnight under refrigeration. The tubes were desorbed with 1.0 mL of acetone and analyzed.
Desorption efficiencies at 0.1, 0.05 and 0.02 times the target concentration were also determined to ensure that the desorption was constant at lower analyte concentrations. Charcoal tubes at each of the three levels were prepared by injecting known amounts of DMF in acetone onto the 100-mg portion of the tubes. All tubes were capped and allowed to equilibrate overnight under refrigeration. The tubes were then desorbed and analyzed.
Table 4.10.
The Desorption Efficiency of DMF from Charcoal
|
0.5× target conc.
142 µg spiked
|
|
1× target conc.
283 µg spiked
|
|
2× target conc.
566 µg spiked
|
| µg found |
DE (%) |
|
µg found |
DE (%) |
|
µg found |
DE (%) |
|
| 133 |
93.7 |
|
249 |
88.0 |
|
530 |
93.6 |
| 133 |
93.7 |
249 |
88.0 |
528 |
93.3 |
| 133 |
93.7 |
261 |
92.2 |
526 |
92.9 |
| 131 |
92.2 |
254 |
89.7 |
524 |
92.6 |
| 128 |
90.1 |
260 |
91.9 |
510 |
90.1 |
| 132 |
93.0 |
252 |
89.0 |
514 |
90.8 |
|
|
260 |
91.9 |
|
|
| average |
| 132 |
92.7 |
255 |
90.1 |
522 |
92.2 |
| Average DE over the range of 0.5× to 2× target conc. is 91.7%. |
|
0.1× target conc.
28.3 µg spiked
|
|
0.05× target conc.
14.2 µg spiked
|
|
0.02× target conc.
5.66 µg spiked
|
| µg found |
DE (%) |
|
µg found |
DE (%) |
|
µg found |
DE (%) |
|
| 24.4 |
86.2 |
|
12.4 |
87.3 |
|
5.49 |
96.9 |
| 24.6 |
86.9 |
12.5 |
88.0 |
5.48 |
96.8 |
| 24.6 |
86.9 |
12.7 |
89.4 |
5.21 |
92.0 |
| 24.8 |
87.6 |
12.4 |
87.3 |
5.57 |
98.4 |
| 24.8 |
87.6 |
12.8 |
90.1 |
5.37 |
94.9 |
| 28.0 |
98.9 |
11.4 |
80.3 |
5.12 |
90.4 |
| 25.0 |
88.3 |
12.5 |
88.0 |
4.90 |
86.6 |
| 27.4 |
96.8 |
11.8 |
83.1 |
4.53 |
80.0 |
| 26.0 |
91.9 |
11.7 |
82.4 |
4.59 |
81.1 |
|
|
11.4 |
80.3 |
5.98 |
106 |
| average |
| 25.5 |
90.1 |
12.2 |
85.6 |
5.22 |
92.3 |
| Average DE over the range of 0.1× to 0.02× target conc. is 89.3%. |
|
4.11. Loss of DMF in the vapor generating system
A number of tests were performed to determine if the loss of DMF in the vapor generating system was due to sampling technique.
The syringe pump which was used to meter DMF into the vapor generating system was recalibrated. Initial and final calibrations were within 1%.
The calibration of the dilution air rotameters was checked and found to be accurate.
The glass-wool plug and the foam plug separating the front and back sections of a vapor-spiked charcoal tube were each desorbed and analyzed to check for DMF adsorption. DMF was found in neither.
In order to determine if collection on charcoal was the problem, two sets of side-by-side samples utilizing silica gel sorbent tubes and methanol bubblers, in addition to charcoal, were collected. The test atmosphere theoretically contained 30 mg/m3 DMF in dry air. Samples were collected at approximately 1 L/min for 50 min. Results from the analysis of the side-by-side samples indicated the concentration of the test atmosphere to be
12-23% lower than the theoretical value (Table 4.11.4.). The results show that the discrepancy between the theoretical concentration and the observed concentration is not unique to sampling with charcoal.
In order to determine if DMF losses in the vapor generating system were constant or concentration dependent, DMF test atmospheres at theoretical test concentrations of twice and ten times the PEL were studied. Three charcoal tubes were collected at a rate of 0.2 L/min for 25 min from a dry test atmosphere which was theoretically 10 times the PEL. These tubes were analyzed and the results compared with the data of Table 4.9.1. which
was collected at twice the PEL. This comparison is shown in Table 4.11.5. and indicates that at both twice and ten times the PEL the recovery is about 70%. Therefore the loss does not appear to be concentration dependent. This work also indicated that pacification of the vapor generating system with high DMF concentrations prior to use would not
alleviate the loss of DMF.
Humidity was not the cause of low results. Samples collected under both dry-air conditions (represented by the charcoal tubes in side-by-side sampling, Table 4.11.4.) and humid air conditions (represented by the vapor-spiked storage samples, Section 4.5.) gave results lower than the theoretical concentrations.
In conclusion, the reason for the discrepancy between the theoretical concentrations of DMF in the vapor generating system and experimentally determined concentrations could not be attributed to any of the parameters tested, including the sampling technique.
Table 4.11.4.
Three Collection Media at 30.21 mg/m3
|
| media |
test |
air
vol.(L) |
expected
(µg) |
recovered
(µg) |
DE corr.
(µg) |
%
recovery |
|
| charcoal |
1 |
48.8 |
1473 |
1046 |
1139 |
77 |
| bubbler |
1 |
44.4 |
1341 |
1076 |
1076 |
80 |
| silica gel |
1 |
52.0 |
1571 |
874 |
1053 |
67 |
| charcoal |
2 |
48.4 |
1462 |
1187 |
1293 |
88 |
| bubbler |
2 |
46.5 |
1405 |
1212 |
1212 |
86 |
| silica gel |
2 |
51.2 |
1547 |
973 |
1172 |
76 |
DE for charcoal = 91.7%. DE for silica gel = 83.0% |
Table 4.11.5.
Concentration Effects
|
theor. conc.
(mg/m3) |
air
vol.(L) |
expected
(mg) |
recovered
(mg) |
DE corr.
(mg) |
%
recovery |
|
| 61.05 |
255.9 |
15.62 |
10.10 |
10.98 |
70 |
| 318.0 |
5.26 |
1.673 |
1.060 |
1.152 |
69 |
| 318.0 |
4.96 |
1.577 |
0.993 |
1.079 |
68 |
| 318.0 |
5.21 |
1.657 |
1.121 |
1.218 |
74 |
DE for charcoal = 91.7% |

