|
VOLATILE NITROSAMINE MIXTURE II |
|
N-nitrosomethylethylamine (NMEA)
N-nitrosodiisopropylamine (NDiPA)
N-Nitrosomethyl-n-butylamine (NMBA)
N-Nitrosoethyl-n-butylamine (NEBA)
N-Nitroso-n-propyl-n-butylamine (NPBA)
N-nitrosodiamylamine (NDAmA) |
| Method no.: |
38 |
|
| Matrix: |
Air |
|
| Procedure: |
Samples are collected using ThermoSorb/N air samplers. The samples are desorbed with a dichloromethane/methanol (75:25)(v/v) solution. Analysis is by gas chromatography and detection with a Thermal Energy Analyzer. |
|
Recommended air volume
and sampling rate: |
75 L at 1 L/min (0.2 to 2 L/min is permissible) |
|
| Special requirements: |
It is recommended that collected samples be stored in a freezer. |
|
| Status of method: |
A sampling and analytical method that has been subjected to the established procedures of the Organic Methods Evaluation Branch. |
|
| Date: June 1982 |
Chemist: Warren Hendricks |
Organic Methods Evaluation Branch
OSHA Analytical Laboratory
Salt Lake City, Utah
Volatile Nitrosamine Mixture II
|
| analyte |
NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
target concentration1
µg/m3
ppb
reliable quantitation limit
µg/m3
ppb
standard error of estimate
at the target concentration,
% (Section 4.7.) |
2.7
0.74
0.13
0.036
7.83 |
2.9
0.55
0.15
0.028
6.48 |
2.5
0.53
0.13
0.027
6.68 |
2.9
0.55
0.15
0.028
7.01 |
2.7
0.45
0.13
0.022
6.7 |
4.0
0.52
0.20
0.026
6.67 |
|
| 1 Air concentrations are based on a 75-L air sample volume. |
1. General Discussion
1.1. Background
1.1.1. History
The purpose of this work was to extend previously developed volatile N-nitrosamine air sampling and analytical techniques (Ref. 5.1.) to another set of N-nitrosamine analytes. This air sampling procedure has now been validated for 13 N-nitrosamines.
Volatile N-nitrosamines have been collected using cold traps (Ref. 5.2.), bubblers (Ref. 5.2.) and various solid adsorbents (Ref. 5.4.). Most of these air sampling techniques have serious deficiencies. The cold traps are difficult to maintain and are reported to enable the artifactual (in situ) formation of the analytes from precursor amines and nitrogen oxides (Ref. 5.5.). Bubblers are inconvenient and analyte retention efficiency decreases with increased sampling time and temperature (Ref. 5.4.). Solid adsorbents, in general, may concentrate precursor amines and nitrosating agents resulting in artifactual formation of the analytes (Ref. 5.4.).
The commercially available ThermoSorb/N air sampling system is reported to be free of the many problems associated with sampling air for N-nitrosamines (Ref. 5.4.). The device is an opaque plastic cartridge containing a solid adsorbent. The manufacturer states that the device has adequate capacity to collect most N-nitrosamines and also to prevent artifactual formation of the analytes because of a proprietary amine trap and nitrosation inhibitor (Ref. 5.4.). Since the device has been successfully used by OSHA industrial hygienists to collect similar analytes, it was decided to evaluate ThermoSorb/N cartridges as sampling media for this method.
Previous analytical methods for N-nitrosamines include polarographic (Ref. 5.6.), spectrophotometric (Ref. 5.6.), and thin layer chromatographic (Ref. 5.6.) techniques. The analytes have been determined both directly and following derivatization. The main disadvantages of these methods are that they have limited application to low level analysis of complex samples, they are only semi-quantitative, they have insufficient resolution and lack selective detection systems (Ref. 5.6).
Gas and liquid chromatographic analytical techniques have greater application to trace level analysis of N-nitrosamines than the methods previously mentioned because they provide better analyte resolution and also because these methods utilize more selective detectors (Ref. 5.6.).
The Thermal Energy Analyzer (TEA) is a highly selective detector for N-nitroso compounds that has been successfully interfaced to gas and liquid chromatographs. In the gas chromatography (GC) mode of operation, the chromatographed N-nitroso compound exits the GC and enters the TEA pyrolyzer through a heated transfer line. The chemical bond between the two nitrogen atoms (N-NO) is thermally broken, resulting in an organic fragment and a nitrosyl radical (·NO). Organic compounds, solvents and fragmentation products are collected in a cold trap. The nitrosyl radical is a stable gas which passes through the cold trap to react with ozone under vacuum to form electronically excited nitrogen dioxide. The excited nitrogen dioxide quickly decays to its ground state and emits light, at a characteristic wavelength, which is measured by a photomultiplier tube. When the TEA is used as a high performance liquid chromatograph (HPLC) detector, the sequence of events is similar to those of the GC mode. The chromatographed analyte exits the HPLC column and enters the TEA pyrolyzer. A higher temperature pyrolyzer is used because the HPLC mobile phase is flash vaporized at the same time the N-nitroso bond is thermally broken. The vaporized HPLC mobile phase, organic compounds and fragmentation products are condensed in a cold trap. The nitrosyl radical is swept through the cold trap with an inert gas, usually helium or argon, and is detected as before (Ref. 5.7.).
This method uses a GC/TEA procedure for the analysis of the component analytes. A HPLC/TEA procedure is also presented for use as a confirmatory technique.
1.1.2. Toxic effects (This section is for information and should not be taken as the basis of OSHA policy).
The effects of acute exposure to each of the analytes were similar. The administration of a lethal dose to rats led to a progressively weakened and emaciated condition frequently with the appearance of jaundice. Death usually occurred within seven days. The autopsy revealed severe centrilobular liver damage with hemorrhaging into the lungs in most cases. The response to a lethal dose was the same for male and female rats (Ref. 5.8.).
Each of the analytes evaluated in this method has a carcinogenic effect on the rat. There is no evidence that exposure to the analytes has led to cancer in humans and extrapolation of animal data to humans is controversial, but experience with similar N-nitroso compounds suggests that human exposure should be kept to a minimum (Ref. 5.28.).
A high correlation between carcinogenicity and mutagenicity has been reported (Ref. 5.10.). NDAmA, NMBA, and NPBA were found to be mutagenic on Salmonella typhimurium when incubated with a rat liver activating system (Ref. 5.11.). No information regarding the mutagenicity of NMEA, NDiPA, and NEBA was obtained through a literature search.
A generally accepted theory of N-nitrosamine carcinogenesis proposes that the agents require biological activation in order to exert their carcinogenic effects. If a compound is directly active, local sarcomas should be observed at the site of subcutaneous injection because of the high concentration of the agent and sensitivity of the subcutaneous tissue. If, however, local sarcomas do not develop and tumors in distant organs are observed, the agent is an intermediate from which the actual carcinogen is produced by metabolic processes. Subcutaneous injection of N-nitrosodialkylamines did not result in local sarcomas and the carcinogenic effects were evident in specific organs (lungs, liver, bladder, etc.). The N-nitrosodialkylamines are, therefore, not considered to be the ultimate carcinogens (Ref. 5.8.).
The metabolic activation of a N-nitrosodialkylamine initially involves the enzymatic hydroxylation of the carbon atom immediately adjacent (in the alpha position) to the N-nitroso group. This is followed by a series of other steps, one of which is the formation of a diazoalkane which ultimately leads to a carbonium ion (Figure 1.). A carbonium ion is believed to be the carcinogenic species. This mechanism for the formation of the carbonium ion is supported by the fact that ethyl-tert-butylnitrosamine, which is structurally unable to form the intermediate diazoalkane species, was found not to cause cancer in rats, even at high doses. Exposure to a similar compound, ethyl-n-butylnitrosamine, which structurally can form a diazoalkane did lead to cancer in rats (Ref. 5.8.).
Detailed information regarding chronic exposure to the analytes is presented in Section 4.9.
1.1.3. Potential workplace exposure
A literature search resulted in no citations regarding occupational exposure to any of the analytes.
N-nitrosamines have been detected in various industries (Refs. 5.12. - 5.14.) and industrial products (Refs. 5.12. and 5.15. - 5.22.).
Occupational exposure to the analytes is probably the result of the formation of the agent from precursor amines and suitable nitrosating species. The amino group can be primary, secondary or tertiary (Ref. 5.25.). The amine can be free or a portion of a more complex molecule such as a drug or a herbicide. Amines can be nitrosated in air (Ref. 5.41.) or in solution under acidic, neutral or alkaline conditions (Refs. 5.23. and 5.25.). The nitrosation reaction is catalyzed by thiocyanate, halide ions, metal ions, formaldehyde and ozone (Refs. 5.23., 5.25., and 5.31.). Suitable nitrosating species include nitrogen oxides (NO, NO2, N2O4, N2O3), nitrite and nitrous acid (Refs. 5.23. and 5.25.).
Nitrosation can occur as a result of transnitrosation. This is a chemical reaction in which a N-nitrosamine transfers its nitroso group to another amine (Refs. 5.24. and 5.25.).
Exposure to the analytes includes the endogenous formation of the agents in the human gastrointestinal tract. Precursor amines have been shown to react with nitrite to form the corresponding N-nitrosamines under conditions found in the mammalian stomach (Ref. 5.30.). N-nitrosomorpholine has been produced in vivo by mice gavaged with morpholine and later exposed to nitrogen dioxide in inhalation chambers (Ref. 5.32.). The source of volatile N-nitrosamines found in normal human feces, urine and saliva has been suggested to be in vivo nitrosation of ingested secondary and tertiary amines (Ref. 5.29 and 5.33. - 5.34.).
The size of the work population that is exposed is unknown. Since amines and suitable nitrosating species are ubiquitous, the number of potential exposures could be large (Refs. 5.3. and 5.25.).
1.1.4. Physical properties
The following data were taken from Refs. 5.8. and 5.26.
|
| analyte |
NMEA |
NDiPA |
NEBA |
NDAmA |
|
CAS no.
mol wt
bp, °C (mm Hg)
solubility in
water, g/100 mL
UV absorption data
l max, nm
log e
l max, nm
log e |
10595-95-6
88.13
70 (35)
30
230
3.85
335
1.90 |
601-77-4
130.22
96 (28)
194.5 (760)
1.3
232
3.86
343
1.84 |
4549-44-4
130.22
94-96 (14)
1.2
231
3.88
337
1.94 |
13256-06-9
186.34
136 (14)
0.014
234
3.86
349
1.94 |
|
The following data were taken from Refs. 5.27. and 5.28.
|
| analyte |
NMEA |
NDiPA |
NEBA |
NDAmA |
|
physical
appearance
density
mp, °C |
yellow
liquid
0.9448 (18/4)
|
yellow1
solid
0.9442 (20/4)
48 |
yellow1
liquid
|
yellow1
liquid
|
|
| 1 personal observation |
The following data were taken from Refs. 5.26. and 5.37.
|
| analyte |
NMBA |
NPBA |
|
CAS no.
mol wt
bp, °C
physical
appearance |
7068-83-9
116.19
56 (1.5 mm Hg)
yellow1
liquid |
25413-64-3
144.25
yellow1
liquid |
|
| 1 personal observation |
synonyms (Ref. 5.26.)
N-nitrosomethylethylamine
ethylamine, N-methyl-N-nitroso; ethylmethylnitrosamine; methylaethyl-nitrosamin (German); N,N-methylethylnitrosamine; N-methyl-N-nitrosoethylamine; N-nitrosoethylmethylamine; NEMA; NMEA.
N-nitrosodiisopropylamine
diisopropylamine, N-nitroso; diisopropylnitrosamin (German); NDiPA.
N-nitrosomethyl-n-butylamine
butylamine, N-methyl-N-nitroso-; butanamine, N-methyl-N-nitroso-; methyl-butyl-nitrosamin (German); methylbutyl-nitrosamine; methyl-n-butylnitrosamine; N-methyl-N-nitrosobutylamine; N-nitroso-n-butylmethylamine; nitrosomethyl-n-butylamine; MBNA; NMBA.
N-nitrosoethyl-n-butylamine
aethyl-n-butyl-nitrosamin (German); N-ethyl-N-nitrosobutylamine; butylamine, N-ethyl-N-nitroso-; butanamine, N-ethyl-N-nitroso-; ethyl-n-butylnitrosamine; N-ethyl-N nitrosobutylamine; N-nitroso-n-butylethylamine; NEBA.
N-nitroso-n-propyl-n-butylamine
1-butanamine,N-nitroso-N-propyl-; butylamine, N-nitroso-N-propyl-; N-propyl-N-butylnitrosoamine; NPBA.
N-nitrosodiamylamine
dipentylamine, N-nitroso-; diamylnitrosamin (German); di-n-amylnitrosamine; di-n-pentylnitrosamine; NDAmA.
structure and molecular formula (Ref. 5.26.).
| NMEA |
 |
C3H8N2O |
|
| NDiPA |
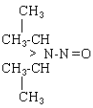 |
C6H14N2O |
|
| NMBA |
 |
C5H12N2O |
|
| NEBA |
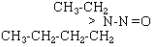 |
C6H14N2O |
|
| NPBA |
 |
C7H16N2O |
|
| NDAmA |
 |
C10H22N2O |
1.2. Limit defining parameters (All air concentrations presented are based on a 75 L air sample volume and a 1-mL desorption volume.)
1.2.1. Detection limits of the analytical procedure
The detection limit of the analytical procedure is the mass of analyte per injection which will result in a peak whose height is about five times the amplitude of the baseline noise. (Section 4.1.)
Detection Limits of the
Analytical Procedure (pg/injection)
|
| NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
| 50 |
55 |
47 |
55 |
50 |
75 |
|
1.2.2. Detection limits of the overall procedure
The detection limit of the overall procedure is the amount of analyte spiked on the sampling device which allows recovery of an amount of analyte equivalent to the detection limit of the analytical procedure. (Section 4.2.)
Detection Limits of the Overall Procedure
|
| analyte |
NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
ng/sample
µg/m3
ppb |
10
0.13
0.036 |
11
0.15
0.028 |
9.4
0.13
0.027 |
11
0.15
0.028 |
10
0.13
0.022 |
15
0.20
0.026 |
|
1.2.3. Reliable quantitation limits
The reliable quantitation limit is the smallest amount of analyte which can be quantitated within the requirements of at least 75% recovery and a precision (±1.96 SD) of ±25% or better, based on six samples.
The reliable quantitation limits were the same as the detection limits of the overall procedure since the desorption efficiencies were above 75% and the precisions were better than ±25%. (Section 4.2.)
Reliable Quantitation Limits
|
| analyte |
NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
ng/sample
µg/m3
ppb |
10
0.13
0.036 |
11
0.15
0.028 |
9.4
0.13
0.027 |
11
0.15
0.028 |
10
0.13
0.022 |
15
0.20
0.026 |
|
The reliable quantitation limit and detection limits reported in the method are based upon optimization of the instrument for the smallest possible amount of analyte. When the target concentration of an analyte is exceptionally higher than these limits, they may not be attainable at the routine operating parameters.
1.2.4. Sensitivity
The sensitivity of the analytical procedure is determined by the slope of the calibration curve over a concentration range from 0.5 to 2 times the target concentration. The sensitivity will vary somewhat with the particular instrument used in the analysis. (Section 4.4.)
Sensitivity of the Analytical Procedure
(area units per µg/mL)
|
| NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
| 279441 |
204894 |
192333 |
231792 |
174773 |
114093 |
|
1.2.5. Recovery
The recovery of analyte from the collection medium during storage must be 75% or greater. The minimum recoveries which are presented below were determined from the regression line of the plotted ambient temperature storage data. (Section 4.7.)
Recovery (%)
|
| NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
| 93.6 |
97.6 |
93.6 |
93.3 |
90.6 |
93.4 |
|
1.2.6. Precision (analytical method only)
The pooled coefficients of variation obtained from replicate determinations of analytical standards at 0.5, 1 and 2 times the target concentrations are presented below. (Section 4.3.)
The Pooled Coefficients of Variation
|
| NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
| 0.013 |
0.018 |
0.020 |
0.026 |
0.017 |
0.025 |
|
1.2.7. Precision (overall procedure)
The overall procedure must provide results at the target concentrations that are ±25% or better at the 95% confidence level. The precisions at the 95% confidence level for the 16-day storage tests are presented below (Section 4.7., Figures 4.7.1. - 4.7.6.). The reported values each include an additional ±5% for sampling error.
Precision (%) of the Overall Procedure
|
| NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
| 15.3 |
12.7 |
13.0 |
13.7 |
13.1 |
13.0 |
|
1.2.8. Reproducibility
Six vapor-spiked samples and a draft copy of this procedure were given to a chemist unassociated with this evaluation. The samples were analyzed after one day of storage at ambient temperature. The average recoveries and standard deviations are presented below (Section 4.10.). The samples were spiked at the target concentrations.
Reproducibility Study
|
| analyte |
NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
amount vapor
spiked, µg
 , % , %
SD, % |
0.20
100.8
6.65 |
0.22
99.2
5.29 |
0.19
100.9
3.99 |
0.22
100.0
2.85 |
0.20
103.4
4.01 |
0.30
104.4
3.45 |
|
1.3. Advantages
1.3.1. The sampling and analytical procedures permit the simultaneous determination of a mixture of analytes.
1.3.2. The sampling and analytical procedures are precise, reliable, safe and convenient.
1.3.3. The sampling procedure is artifact free under the tested conditions. The capacity of the sampling device to prevent artifacts is probably limited.
1.3.4. The air sampling device is commercially available and is constructed of opaque plastic to prevent photo-degradation of the collected N-nitrosamines.
1.3.5. The samples are stable when stored at ambient temperatures for at least 16 days.
1.4. Disadvantages
1.4.1. Smaller laboratories may not be able to support the cost of the recommended sampling and analytical instruments.
1.4.2. The ability of the sampling device to collect and retain the analytes is limited.
2. Sampling Procedure
2.1. Apparatus
2.1.1. An air sampling pump, the flow of which can be determined to within ±5% at the recommended air flow rate with the air sampler in line.
2.1.2. ThermoSorb/N air sampling cartridges, available from Thermo Electron Corporation, Waltham, Mass.
2.1.3. Equipment to calibrate the air flow rate through the sampling device.
2.2. Reagents
None required
2.3. Technique
2.3.1. Prior to sampling, remove the ThermoSorb/N air sampling device from the foil container. Save the container for reuse.
2.3.2. Prior to sampling, remove the red end caps from the inlet and outlet ports. Store the caps on the air sampler in the places that are provided for this use.
2.3.3. Label the air sampler and attach the device to the air sampling pump with flexible tubing. Adjust the pump to obtain the proper air flow rate. The recommended rate is 1 L/min but flow rates of from 0.2 to 2 L/min may be used. If air volumes larger than the recommended 75 L are required or if large amounts of N-nitrosamines are suspected, two air samplers may be connected in series. Always calibrate the pump with the sampling device in line.
2.3.4. Attach the sampling device in the breathing zone of the employee to be monitored. The molded clip is convenient for this purpose.
2.3.5. After sampling for the appropriate time, remove the device and replace the red end caps on the inlet and outlet ports of sampler.
2.3.6. Wrap each sample end to end with official OSHA seals (Form 21). Place the sealed air sampler inside the foil container from Section 2.3.1.
2.3.7. With each set of samples, submit at least one blank sample. The blank should be subjected to the same handling as the sample except that no air is drawn through it.
2.3.8. Place the samples in a freezer if they are to be stored before shipping to the laboratory.
2.3.9. List possible interferences on the sample data sheet.
2.4. Breakthrough
Breakthrough studies were conducted by connecting two samplers in series. The first sampler was vapor spiked at twice the target concentrations with a mixture of the analytes and then air at about 80% relative humidity and 22°C was drawn through the sampling train. The rear sampler was removed and analyzed after an appropriate air volume had been sampled. The rear sampler was replaced with a new cartridge and sampling was continued. The first tube was also analyzed when the study was terminated.
NMEA was the only component of the mixture to be lost from the first sampling device. Two breakthrough studies were conducted and the average 5% breakthrough air volume was 169 L (Section 4.5.). The 5% breakthrough air volume was defined as the air volume at which 5% of the amount of the component vapor spiked on the first tube was found on the second tube. None of the other analytes moved from the front tube after 365 L of air was drawn through the sampling train.
In the interests of a general N-nitrosamine air sampling procedure, 75 L was selected as the recommended air volume for this method because previously evaluated N-nitrosamines have been shown to have a lower 5% breakthrough air volume than NMEA. (Ref. 5.1.)
2.5. Desorption efficiency
The average desorption efficiency for each of the analytes vapor spiked at 0.5, 1 and 2 times the target concentration on ThermoSorb/N air samplers is presented below (Section 4.6.).
Average Desorption Efficiencies, %
|
| NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
| 101.1 |
100.7 |
100.7 |
99.5 |
99.5 |
95.7 |
|
2.6. Recommended air volume and sampling rate
2.6.1. The recommended air volume is 75 L.
2.6.2. The recommended air sampling rate is 1 L/min. Studies indicate that flow rates of from 0.2 to 2 L/min may be used if required.
2.7. Interferences (sampling)
2.7.1. The commercial ThermoSorb/N N-nitrosamine air sampler is constructed to prevent the in situ formation of N-nitrosamines from airborne precursors. The manufacturer has tested the sampling device for the artifactual formation of N-nitrosodimethylamine (NDMA), N-nitrosopiperidine (NPIP), N-nitrosopyrrolidine (NPYR), N-nitrosomorpholine (NMOR) and NDiPA using 50 µg each of the respective precursor secondary amines and nitrogen oxides (NOx). The amines were pre-loaded on the ThermoSorb/N tubes and then 100 L of air containing 0.2, 0.5, 1, 2 or 4 ppm NO/NO2 at a 1/1 ratio were sampled. Various untreated solid adsorbents formed the N-nitrosamines quite easily but at these levels of amines and NOx, the ThermoSorb/N cartridges were shown to be artifact free (Ref. 5.4.).
The process by which the ThermoSorb/N tubes prevent artifact formation is proprietary. The manufacturer states that the device has an amine trap that removes incoming amines and holds them unavailable for nitrosation. The sampled air, after passing through the amine trap, enters a solid sorbent bed where the N-nitrosamines are quantitatively removed and retained. The sampled air then passes through fiber pads containing a nitrosation inhibitor and finally exits the sampler. When the air sampling device is backflushed with solvent, the nitrosation inhibitor is put into solution and is washed together with the collected N-nitrosamines and amines into a collection vial. The nitrosation inhibitor successfully competes with the amine and chemically removes any nitrosating species (Ref. 5.35.).
The resistance of the ThermoSorb/N air sampler to artifact formation was verified using morpholine (a precursor amine for NMOR) and nitrogen dioxide. Morpholine was selected as the test amine because it has been shown to be easily nitrosated. Nitrogen dioxide in a gas bag, mixed with humid air, has been shown to be an effective nitrosating species. A Teflon gas bag was prepared containing 33 µg/L morpholine in nitrogen and another containing 4.8 ppm v/v NO2 in air at about 75% relative humidity. The morpholine bag was sampled for 50 min at 1 L/min and then the nitrogen dioxide bag was sampled for 50 min at 1 L/min. The total morpholine loaded on the tube was 1650 µg. There was no NMOR formed on the ThermoSorb/N cartridge. The experiment was repeated using a standard Florisil tube. The result showed that 7 µg of NMOR could be formed, as an artifact on the tube, after sampling each bag for 7 min.
2.7.2. It is unknown if there are other interferences with the collection of N-nitrosamines using ThermoSorb/N cartridges.
2.8. Safety precautions (sampling)
2.8.1. Attach the sampling equipment to the worker so it will not interfere with work performance or safety.
2.8.2. Follow all safety practices that apply to the work area to be monitored.
3. Analytical Procedure
3.1. Apparatus
3.1.1. A temperature programmable gas chromatograph (GC).
3.1.2. A high performance liquid chromatographic (HPLC) pump.
3.1.3. A HPLC sample injector.
3.1.4. A Thermal Energy Analyzer (TEA), Thermo Electron Corporation, Waltham, Mass.
3.1.5. A GC column capable of resolving the analytes from each other and potential interferences. The column used in this work was 10-ft × 1/8-in. stainless steel containing 10% Carbowax 20M with 2% KOH on 80/100 mesh Chromosorb W AW.
3.1.6. A HPLC analytical column capable of resolving the analytes from each other and potential interferences. The column used in this work was a DuPont Zorbax CN (4.6 mm × 25-cm).
3.1.7. The necessary hardware to interface the TEA to the GC and the HPLC apparatus.
3.1.8. An electronic integrator or other suitable means to measure peak area and record chromatograms. A Hewlett-Packard 3354 B/C data system was used for this evaluation. The A/D converter was set for 2 readings per second.
3.1.9. Vials, 2-mL with Teflon-lined caps.
3.1.10. Syringes, of convenient sizes for sample and standard preparations and injections.
3.1.11. Hypodermic needles, 23 gauge × 1 in., Luer hub.
3.1.12. Volumetric flasks, 1-mL and other convenient sizes.
3.1.13. Dewar flasks, of convenient sizes for liquid nitrogen.
3.1.14. Microanalytical balance.
3.2. Reagents
3.2.1. Analytical standards: NMEA, NDiPA, NMBA, NEBA, NPBA and NDAmA.
3.2.2. Dichloromethane and methanol, HPLC grade. The sample desorption solution is composed of 75% dichloromethane and 25% methanol by volume.
3.2.3. Isopropanol and 2,2,4-trimethylpentane, HPLC grade.
3.2.4. n-Propanol, technical grade.
3.2.5. Nitrogen, liquid.
3.2.6. Helium, GC grade.
3.2.7. Oxygen and air, medical grade.
3.3. Standard preparation
3.3.1. Keep the exposure of the standards to light at a minimum because light will decompose each of the analytes.
3.3.2. Prepare individual stock standards of by diluting known quantities of each component with isopropanol.
3.3.3. Prepare an intermediate standard mixture using known volumes of each stock standard and diluting the mixture with 2,2,4-trimethylpentane. The following intermediate standard mixture was used for this evaluation.
Concentration of the Standard Mixture, µg/mL
|
| NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
| 10 |
11 |
9.4 |
11 |
10 |
15 |
|
3.3.4. Prepare fresh working range standards daily by diluting the standard mixture with desorbing solution. Standards at the target concentration were obtained by diluting the above standard mixture 1 to 50 with desorbing solution.
3.3.5. Additional standards at other than the target concentration should be prepared in order to generate the calibration curve.
3.3.6. Store the standards in a freezer using well-sealed, dark containers.
3.4. Sample preparation
3.4.1. Store the samples in a freezer until analysis.
3.4.2. The sample should be received in a foil container. Remove the sample from the container.
3.4.3. Insure that the official OSHA seal (Form 21) is intact and complete.
3.4.4. Check the laboratory sample number against the field identification number to be sure that the sample has been properly identified.
3.4.5. Prepare the desorption solution described in Section 3.2.2. Store the solution in a well sealed, dark bottle.
3.4.6. Label two 1-mL volumetric flasks with the sample number. Further label one flask "A" and the other "B".
3.4.7. Remove the OSHA seal and the red end caps from the sample.
3.4.8. Attach a syringe needle to the male Luer fitting at the inlet port of the air sampler.
3.4.9. Fill a syringe with about 4 mL of the desorbing solution. Attach the syringe to the female Luer fitting located at the outlet end of the air sampler.
3.4.10. Elute the sample by gently forcing the desorption solvent through the air sampler at approximately 0.5 mL/min. Collect the first 1-mL portion of solvent in the volumetric flask labeled "A" in Section 3.4.6. and the second 1 mL of eluant in the flask labeled "B".
3.4.11. Because light will decompose N-nitrosamines, it is necessary to prevent exposure of the eluted samples to light.
3.4.12. If the eluted samples are not to be analyzed immediately, transfer the contents of each flask to a separate vial which can be sealed with a Teflon-lined cap. Protect the samples from light and store them in a freezer.
3.5. Analysis
3.5.1. Instrument conditions
3.5.1.1. GC conditions
| injector temperature: |
150°C |
| column temperature: |
150 to 220°C at 4°C/min |
| carrier gas flow rate: |
30 mL/min (helium) |
| injection volume: |
5 µL |
The recommended GC column is 10-ft × 1/8-in. stainless steel packed with 10% Carbowax 20M with 2% KOH on 80/100 mesh Chromosorb W AW.
3.5.1.2. TEA Conditions
The following conditions apply to the Model TEA-502 with the retrofit Explosives Analysis Package (EAP).
| GC transfer temperature: |
220°C |
| GC pyrolyzer temperature: |
500°C |
| oxygen: |
5 mL/min |
| attenuation: |
4 |
| cold trap temperature: |
-130°C |
| (n-propanol and liquid nitrogen) |
3.5.2. Chromatogram Section 4.8.
3.5.3. Detector response is measured with an electronic integrator or other suitable means.
3.5.4. An external standard procedure is used to prepare a calibration curve using at least 3 standard solutions of different concentrations. The calibration curve is prepared daily. The integrator is calibrated to report results in µg/mL.
3.5.5. Bracket the samples with analytical standards.
3.6. Interferences (analytical)
3.6.1. N-nitrosodipropylamine has the same general GC retention time as NMBA (Figure 4.8.2.). These analytes may be resolved on a 1/8-in. × 10-ft stainless steel GC column packed with 20% Versamid 900 on 80/100 Chromosorb W AW (Figure 4.8.3.). If either of these analytes are detected in samples, the identity of the peak in question must be confirmed.
3.6.2. Because the TEA has been shown to respond to compounds other than N-nitrosamines, it is strongly recommended that positive GC/TEA results be confirmed by HPLC/TEA analysis. Since GC and HPLC separation techniques operate using different principles, the component elution order is not the same. It is unlikely that two different compounds will have a coincidence of retention time on both GC and HPLC columns.
3.6.2.1. HPLC conditions
| column: |
DuPont Zorbax CN (4.6 mm × 25 cm) |
| mobile phase: |
94.5% 2,2,4-trimethylpentane/5% dichloromethane/0.5% isopropanol (v/v/v) |
| flow rate: |
1.3 mL/min |
| injection volume: |
5 to 25 µL |
The TEA conditions are the same as for the GC/TEA analysis except that the HPLC pyrolyzer is used at 550°C and the cold trap is maintained at -80°C with a mixture of water, n-propanol and liquid nitrogen.
3.6.2.2. Chromatogram Figure 4.8.4.
3.6.3. GC and HPLC parameters may be changed to circumvent interferences. Possible interferences are listed on the sample data sheets.
3.6.4. The only unequivocal means of structure designation is by gas chromatography/mass spectrometry with continuous peak matching. It is recommended this procedure be used to confirm samples whenever possible.
3.7. Calculations
3.7.1. The integrator value in µg/mL is used for reference only. More reliable results are obtained by use of a calibration curve. The detector response, for each standard, is compared to its equivalent concentration in µg/mL and the best straight line through the data points is determined by linear regression.
3.7.2. The concentration, in µg/mL, for a particular determination is obtained by comparing its detector response to the calibration curve.
3.7.3. The result obtained from the analysis of each vial or flask is corrected by the appropriate desorption efficiency, and then the corrected results from the "A" and "B" determinations that compose a particular air sample are added together.
3.7.4. The analyte air concentrations for a sample are calculated by the following equation:
concentration in µg/m3 = (C)(D)(1000)/E
| where |
C |
= |
µg/mL from Section 3.7.3. |
|
D |
= |
desorption volume in milliliters (1 mL) |
|
E |
= |
air volume in liters |
3.7.5. To convert the results from Section 3.7.4. to parts per billion (at 760 mm and 25°C) the following relationship is used:
concentration in ppb = (µg/m3)(24.46)/MW
| where |
µg/m3 |
= |
result from Section 3.7.4. |
|
24.46 |
= |
molar volume of an ideal gas at 25°C and 760 mm Hg. |
|
MW |
= |
molecular weight of the analyte, obtained below. |
Molecular Weights
|
| NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
| 88.13 |
130.22 |
116.19 |
130.22 |
144.25 |
186.34 |
|
3.8. Safety precautions (analytical)
3.8.1. The analytes are extremely potent animal carcinogens and utmost care must be exercised when working with these compounds.
3.8.2. Avoid skin contact with liquid nitrogen and the solvents.
3.8.3. Confine the use of solvents to a fume hood.
3.8.4. Wear safety glasses in all laboratory areas.
3.8.5. Check to be sure that the TEA exhaust is connected to a fume hood.
4. Backup Data
4.1. Detection limit of the analytical procedure
Figure 4.1. is a chromatogram obtained from a 5-µL injection of a standard solution at the following concentrations:
Concentrations of the Detection Limit Standard in µg/mL
|
| NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
| 0.010 |
0.011 |
0.0094 |
0.011 |
0.010 |
0.015 |
|
Procedure used to Vapor Spike ThermoSorb/N Tubes
Each ThermoSorb/N tube was vapor spiked with the analytes by first injecting a liquid mixture of the analytes on Polar Partition resin. The spiked resin tube was then placed in front of the ThermoSorb/N cartridge and 50 L (1 L/min) of air, at about 80% relative humidity and 22°C, were drawn through the sampling train. The analytes were vaporized from the resin and collected on the ThermoSorb/N tube. The vaporization process was determined to be complete after 25 L of air had passed through the resin.
4.2. The detection limit of the overall procedure and the reliable quantitation limit
The following data were obtained by vapor spiking the analytes on air samplers. The injection size recommended in the analytical procedure (5-µL) was used to determine the detection limit of the overall procedure and the reliable quantitation limit.
Table 4.2.
Desorption Efficiency at the Detection Limit
|
analyte
ng/sample |
NMEA
10 |
NDiPA
11 |
NMBA
9.4 |
NEBA
11 |
NPBA
10 |
NDAmA
15 |
|
recovery,
%

SD
1.96 SD |
95.6
92.7
102.0
91.5
86.9
99.1
94.6
5.45
10.7 |
94.8
88.2
85.0
103.9
97.2
97.5
94.4
6.85
13.4 |
95.2
87.3
83.9
100.0
95.4
85.1
91.2
6.58
12.9 |
94.9
99.9
95.2
98.8
102.1
105.9
99.5
4.20
8.22 |
83.2
95.1
104.5
94.4
102.7
102.6
97.1
8.01
15.7 |
107.2
107.0
98.5
105.1
101.9
100.6
103.4
3.59
7.03 |
|
Since the desorption efficiencies were near 100% and also the precisions were better than ±25%, the detection limits of the overall procedure and the reliable quantitation limits were the same.
4.3. Precision data
The following data were obtained from multiple injections of analytical standards:
Table 4.3.1.
Precision at 0.5× Target Concentration
|
analyte
µg/mL |
NMEA
0.10 |
NDiPA
0.11 |
NMBA
0.094 |
NEBA
0.11 |
NPBA
0.10 |
NDAmA
0.15 |
|
area counts

SD
CV |
27065
27193
27690
27758
27851
27511.4
356.612
0.0130 |
22586
22420
22124
22999
22323
22490.4
329.788
0.0147 |
18524
19648
19363
19412
19727
19334.8
478.557
0.0248 |
25698
23684
25931
25189
24922
25084.8
879.157
0.0350 |
18686
17699
18217
18418
18052
18214.4
372.786
0.0205 |
19604
19083
18838
18479
19805
19161.8
544.637
0.0284 |
|
Table 4.3.2.
Precision at 1× Target Concentration
|
analyte
µg/mL |
NMEA
0.20 |
NDiPA
0.22 |
NMBA
0.19 |
NEBA
0.22 |
NPBA
0.20 |
NDAmA
0.30 |
|
area counts

SD
CV |
52963
53081
54020
54539
53835
53687.6
661.458
0.0123 |
44183
44391
43683
42981
42851
43617.8
691.924
0.0159 |
38450
37254
36628
38216
37771
37663.8
737.453
0.0196 |
47220
49112
47969
48028
46244
47714.6
1063.36
0.0223 |
34422
34512
33540
34727
34285
34297.2
452.753
0.0132 |
35067
34259
35449
35120
36502
35279.4
811.673
0.0230 |
|
Table 4.3.3.
Precision at 2× Target Concentration
|
analyte
µg/mL |
NMEA
0.40 |
NDiPA
0.44 |
NMBA
0.38 |
NEBA
0.44 |
NPBA
0.40 |
NDAmA
0.60 |
|
area counts

SD
CV |
111830
108836
110374
112323
111588
110990.2
1401.72
0.0126 |
87667
89592
87938
92364
91555
89823.2
2104.25
0.0234 |
73717
75536
73669
73607
75048
74315.4
908.889
0.0122 |
1011134
100335
98393
102487
102664
101002.6
1749.05
0.0173 |
70263
69945
68823
71697
71112
70368.0
1106.43
0.0157 |
69269
69635
72803
71145
68670
70304.4
1668.91
0.0237 |
|
Table 4.3.4.
The Pooled Coefficients of Variation
|
| NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
| 0.013 |
0.018 |
0.020 |
0.026 |
0.017 |
0.025 |
|
4.4. Sensitivity
The data in Tables 4.3.1. - 4.3.3. are presented graphically in Figures 4.4.1. - 4.4.6. The sensitivity of the analytical procedure is determined by the slope of the calibration curve over a concentration range of from 0.5 to 2 times the target concentrations.
4.5. Breakthrough
Two breakthrough studies were conducted at about 80% relative humidity and 22°C. Two ThermoSorb/N tubes were connected in series and the front tube was vapor spiked at 2 times the target concentration with a mixture of the analytes. The rear tube was removed and replaced at intervals and analyzed. NMEA was the only analyte to move from the front to the rear tube. The data is presented in Tables 4.5.1. and 4.5.2. and also in Figures 4.5.1. and 4.5.2.
Table 4.5.1.
Breakthrough Study One
|
air sampled
L |
NMEA recovery,
% |
cumulative NMEA
recovery, % |
|
100
150
200
250
300
front tube |
ND
2.8
24.3
36.4
26.0
10.5 |
0.0
2.8
27.1
63.5
89.5 |
|
Table 4.5.2.
Breakthrough Study Two
|
air sampled
L |
NMEA recovery,
% |
cumulative NMEA
recovery, % |
|
123
174
229
275
365
front tube |
ND
2.0
19.1
35.5
43.4
ND |
0.0
2.0
21.1
56.6
100.0 |
|
4.6. Desorption efficiency
The following data represent the analysis of ThermoSorb/N tubes vapor spiked with the analytes at 0.5, 1, and 2 times the target concentrations.
Table 4.6.1.
Desorption Efficiency at 0.5× the Target Concentration
|
analyte
ng/sample |
NMEA
100 |
NDiPA
110 |
NMBA
94 |
NEBA
110 |
NPBA
100 |
NDAmA
150 |
|
% recovery
 |
98.4
104.2
97.9
103.6
105.2
98.6
101.3 |
94.1
93.9
102.8
102.2
111.0
94.2
99.7 |
99.0
88.6
103.2
96.6
112.1
97.1
99.4 |
99.4
89.2
97.9
91.6
105.5
96.2
96.6 |
92.8
84.5
98.5
97.9
109.1
100.5
97.2 |
102.3
99.6
104.2
91.0
94.7
101.0
98.8 |
|
Table 4.6.2.
Desorption Efficiency at 1× the Target Concentration
|
analyte
ng/sample |
NMEA
200 |
NDiPA
220 |
NMBA
188 |
NEBA
220 |
NPBA
200 |
NDAmA
300 |
|
% recovery
 |
92.0
105.3
105.1
100.0
98.7
106.7
101.3 |
92.4
105.4
106.8
100.6
97.5
104.0
101.1 |
90.8
106.2
106.0
104.4
95.4
104.5
101.2 |
92.9
106.9
105.7
102.1
94.4
104.5
101.1 |
96.6
104.0
105.1
100.0
95.4
101.8
100.5 |
95.4
96.3
93.7
90.2
99.6
96.0
95.2 |
|
Table 4.6.3.
Desorption Efficiency at 2× the Target Concentration
|
analyte
ng/sample |
NMEA
400 |
NDiPA
440 |
NMBA
376 |
NEBA
440 |
NPBA
400 |
NDAmA
600 |
|
% recovery
 |
101.3
102.1
103.2
103.8
98.0
96.5
100.8 |
101.5
103.7
100.8
100.0
104.9
97.8
101.4 |
102.6
102.6
99.8
101.4
103.9
99.3
101.6 |
101.6
103.0
97.8
101.2
102.4
99.5
100.9 |
100.9
102.2
100.1
102.0
104.4
95.2
100.8 |
87.0
92.5
93.6
95.5
94.6
94.8
93.0 |
|
Table 4.6.4.
Average Desorption Efficiency at
0.5, 1, and 2 Times the Target Concentration
|
| NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
| 101.1 |
100.7 |
100.7 |
99.5 |
99.5 |
95.7 |
|
4.7. Storage data
The data in Tables 4.7.1. and 4.7.2. represent the effects of storage at ambient (21 to 26°C) and reduced (-20°C) temperatures on vapor spiked ThermoSorb/N cartridges. The tubes were vapor spiked at the following levels. The results are not corrected for desorption efficiency. The data are also presented graphically in Figures 4.7.1. to 4.7.12.
Amount Vapor spiked, ug/Cartridge
|
| NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
| 0.20 |
0.22 |
0.19 |
0.22 |
0.20 |
0.30 |
|
Table 4.7.1.
Ambient Temperature Storage, % Recovery
|
| day |
sample |
NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
0
3
6
9
13
16 |
1
2
3
1
2
3
1
2
3
1
2
3
1
2
3
1
2
3 |
105.8
102.8
105.1
96.1
99.9
104.2
85.9
86.4
92.8
91.8
96.9
91.0
103.8
95.4
92.0
95.5
100.6
93.2 |
105.4
101.5
104.8
102.8
108.5
104.7
94.0
90.2
98.1
100.2
101.9
98.5
101.9
98.3
100.9
98.1
100.8
95.1 |
105.8
100.2
104.0
98.2
105.0
97.1
92.4
87.5
95.9
94.8
95.2
92.9
100.8
98.4
96.0
95.0
97.0
90.2 |
104.2
98.6
104.5
100.4
107.2
104.0
89.7
86.2
95.4
99.3
98.8
98.2
94.2
92.2
97.1
93.4
99.0
91.8 |
104.2
101.0
105.1
96.1
105.0
102.0
89.8
88.2
98.1
97.1
96.9
95.0
91.6
88.4
89.5
93.6
98.5
89.6 |
92.0
97.3
98.4
91.3
102.7
99.4
88.8
85.4
94.4
90.7
93.4
89.7
93.0
93.3
94.3
94.0
100.0
94.4 |
|
Table 4.7.2.
Reduced Temperature Storage, % Recovery
|
| day |
sample |
NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
0
3
6
9
13
16 |
1
2
3
1
2
3
1
2
3
1
2
3
1
2
3
1
2
3 |
99.0
94.4
91.6
98.0
93.8
99.3
94.2
100.2
97.3
100.6
101.6
100.6
103.1
92.8
97.0
91.1
92.1
93.2 |
100.8
94.9
101.9
100.4
99.8
99.7
100.4
101.7
99.9
103.9
99.5
101.1
104.8
95.2
98.8
95.1
99.1
97.4 |
102.6
94.3
98.4
97.6
98.0
97.7
98.7
97.6
97.1
99.1
97.2
95.8
98.6
91.9
89.8
90.2
94.2
96.9 |
103.5
94.5
98.5
99.5
100.0
98.3
99.3
100.1
98.2
104.4
99.0
97.4
98.7
94.8
94.1
88.8
92.6
94.2 |
103.3
93.6
95.0
103.5
101.5
97.0
97.5
93.6
89.6
101.2
91.4
92.7
101.1
94.5
99.4
91.0
94.2
93.3 |
101.1
91.3
86.8
87.7
98.1
97.8
94.4
96.8
98.3
88.4
91.4
95.1
96.4
88.2
103.4
93.4
93.4
93.2 |
|
4.8. Chromatograms
4.8.1. GC/TEA Chromatogram
Figure 4.8.1. is a typical GC/TEA chromatogram obtained by the injection of a standard mixture containing the analytes. The column was 1/8-in. × 10-ft stainless steel packed with 10% Carbowax 20M with 2% KOH on 80/100 Chromosorb W AW.
4.8.2. GC/TEA Chromatogram
Figure 4.8.2. is a GC/TEA chromatogram obtained from the analysis of a mixture of N-nitrosamines. The analytes are those of Volatile Mixtures I and II. Note that NMBA and N-nitrosodipropylamine are not resolved. The analytical column was 1/8-in. × 10-ft stainless steel packed with 10% Carbowax 20M with 2% KOH on 80/100 Chromosorb W AW.
N-nitrosamines of Volatile Mixture I shown in Figures 4.8.2.-4.8.4. are: N-nitrosodimethylamine (NDMA); N-nitrosodiethylamine (NDEA); N-nitrosodipropylamine (NDPA); N-nitrosodibutylamine (NDBA); N-nitrosopiperidine (NPIP); N-nitrosopyrrolidine (NPYR); N-nitrosomorpholine (NMOR).
4.8.3. GC/TEA Chromatogram
Figure 4.8.3. is a GC/TEA chromatogram obtained by the analysis of the same N-nitrosamine mixture as used in Section 4.8.2. on a different GC column. Note that while NMBA and NDPA are resolved, NPIP and NPYR are not resolved. The column was 1/8-in. × 10-ft stainless steel packed with 20% Versamid 900 on 80/100 Chromosorb W AW.
4.8.4. HPLC/TEA Chromatogram
Figure 4.8.4. is a HPLC/TEA chromatogram obtained by the injection of the same standard mixture used in 4.8.2. The column was 4.6-mm i.d. × 25-cm DuPont Zorbax CN. The mobile phase was 94.5% 2,2,4-trimethylpentane, 5% dichloromethane and 0.5% isopropanol. The flow rate was 1.3 mL/min.
4.9. Toxic effects (This data is for information only and should not be taken as the basis of OSHA policy.)
NMEA: The LD50 for NMEA is 90 mg/kg following administration by esophageal probe to the rat. In a chronic effects study, 4 rats received 2 mg/kg and 11 rats 1 mg/kg daily in the drinking water. Five of the rats in the second group died without tumors and all the other rats had extensive liver cancers. One animal had a tumor of the vagina. The mean carcinogenic doses were 750 and 420 mg/kg and the mean induction periods were 360 and 500 days respectively. The substance was designated a weaker carcinogen than N-nitrosodiethylamine (Ref. 5.8.).
NDiPA: The LD50 for NDiPA was 850 mg/kg following oral administration to the rat. In a chronic effects study, 25 and 50 mg/kg of NDiPA were administered to 2 groups of rats daily in the drinking water. The experiment was discontinued after a total dose of 11 and 14 g/kg had been administered. These rather large doses produced liver cancer in only 9 of a total of 20 test animals. The mean induction periods were 770 and 430 days for each group respectively. The weak carcinogenic effect of NDiPA was attributed to steric hindrance of the enzymatic hydroxylation of the alpha-carbon atom due to the branched alkyl groups (Ref. 5.8.). In another chronic effects study, 450 and 2,400 mg of NDiPA were administered to 2 groups of rats over 50 and 40 weeks. Twenty-two of thirty rats had tumors primarily of the nasal turbinate (Ref. 5.36.).
NMBA: The LD50 for NMBA is 130 mg/kg following oral administration to the rat. The lowest dose which resulted in liver tumors for 50% of the test rats was 600 mg/kg NMBA. The substance was administered by intraperitoneal injection. (Ref. 5.37.). Inhalation of NMBA has been reported to result in incidences of 100% for esophageal tumors and 23% for nasal tumors in rats (Ref. 5.38. and 5.39.).
NEBA: The LD50 for NEBA was 380 mg/kg, following oral administration to the rat. In a chronic effects study, five rats received 10 mg/kg and 25 rats 5 mg/kg NEBA daily in the drinking water. All of the test animals died between the 175th and the 290th day with extensive cancer of the esophagus. Three animals also had liver cancer and one a pulmonary cancer. The mean carcinogenic doses were 1.6 and 0.97 g/kg and the mean induction periods were 200 and 240 days respectively. In another experiment, 15 rats received 25 mg/kg once per week by intravenous injections. Five animals developed papillomas and nine cancer of the esophagus. Six rats had liver tumors, one cancer of the ethmoturbinalia and another cancer of the pancreas. Seven animals had tumors in multiple organs. The mean carcinogenic dose was 1.04 g/kg and mean induction period was 363 days. Bladder cancer which is often the result of exposure to N-nitrosodibutylamine was not observed in these experiments (Ref. 5.8.).
NPBA: The daily oral administration of 415 mg NPBA per animal, in the drinking water, for 16 to 19 weeks led to a 100% incidence of liver cancer in the rat. Six of 10 rats also developed esophageal papillomas (Ref. 5.40.).
NDAmA: The LD50 for NDAmA was 3000 mg/kg following subcutaneous administration to the rat. The oral administration of 100 mg/kg NDAmA, daily, in the diet led to liver cancer in 17 of 18 animals; in 5 animals the liver tumors were accompanied by pulmonary metastases. One animal also had lung cancer. The carcinogenic mean dose was 48 g/kg and the mean induction period was 360 days. In another chronic experiment, 500 mg/kg of undiluted NDAmA were administered to rats once per week by subcutaneous injection. Only one animal developed liver cancer but six of ten rats died with lung cancer. The mean carcinogenic dose was 12 g/kg and the mean induction period was 340 days. This experiment was seen as fundamentally important because it demonstrated that pulmonary cancer is not only produced by locally acting inhaled agents but also that it can result from absorbed carcinogens. These experiments also show that the affected organs are not only a function of the chemical and physical properties of the substance but also by the size of the dose and mode of administration (Ref. 5.8.).
Molecular Structure and carcinogenicity: A few generalities regarding the carcinogenic properties of nitrosodialkylamines have been made. Branching of the alkyl group at the alpha carbon tends to decrease carcinogenicity. Increasing molecular weight and polarity seems also to reduce carcinogenic activity. The carcinogenicity of unsymmetrical nitrosamines is often selective toward the esophagus (Ref. 5.9.).
4.10. Reproducibility
Six vapor spiked ThermoSorb/N tubes and a draft copy of this procedure were given to a chemist unassociated with this evaluation. The samples were analyzed after one day of storage at ambient temperature. The recoveries and standard deviations are presented in Table 4.10. The samples were spiked at the target concentrations.
Table 4.10.
Reproducibility Study
|
|
NMEA |
NDiPA |
NMBA |
NEBA |
NPBA |
NDAmA |
|
amount vapor
spiked, µg
recovery, %
 , % , %
SD, % |
0.20
100.0
100.0
110.0
100.0
105.0
90.0
100.8
6.65 |
0.22
100.0
95.5
104.5
100.0
104.5
90.9
99.2
5.29 |
0.19
100.0
94.7
105.3
100.0
105.3
100.0
100.9
3.99 |
0.22
100.0
95.5
104.5
100.0
100.0
100.0
100.0
2.85 |
0.20
100.0
110.0
105.0
105.0
100.5
100.0
103.4
4.01 |
0.30
106.7
100.0
110.0
103.3
103.3
103.3
104.4
3.45 |
|
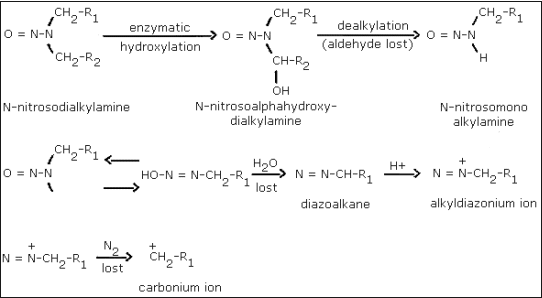
Figure 1. The metabolic activation of N-nitrosodialkylamines.
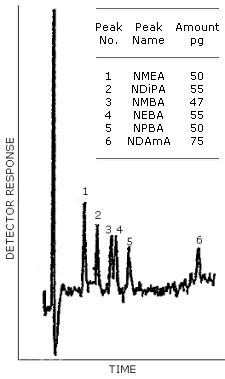
Figure 4.1. The detection limits of the analytical procedure.
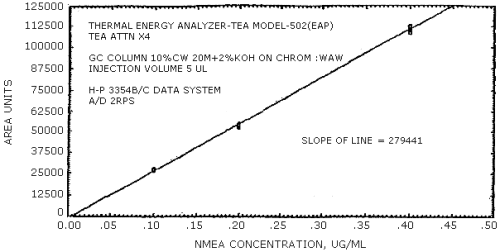
Figure 4.4.1. N-nitrosomethylethylamine calibration curve.

Figure 4.4.2. N-nitrosodiisopropylamine calibration curve.
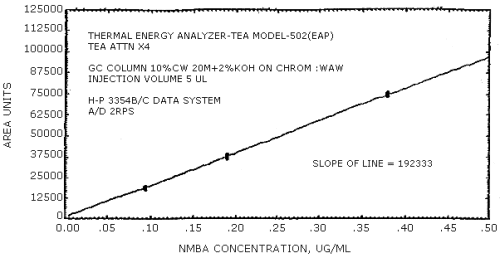
Figure 4.4.3. N-nitrosomethylbutylamine calibration curve.

Figure 4.4.4. N-nitrosoethylbutylamine calibration curve.

Figure 4.4.5. N-nitrosopropylbutylamine calibration curve.

Figure 4.4.6. N-nitrosodiamylamine calibration curve.

Figure 4.5.1. Breakthrough study one.

Figure 4.5.2. Breakthrough study two.

Figure 4.7.1. Ambient temperature storage test for nitrosomethylethylamine.

Figure 4.7.2. Ambient temperature storage test for N-nitrosodiisopropylamine.

Figure 4.7.3. Ambient temperature storage test for N-nitrosomethylbutylamine.

Figure 4.7.4. Ambient temperature storage test for N-nitrosoethylbutylamine.
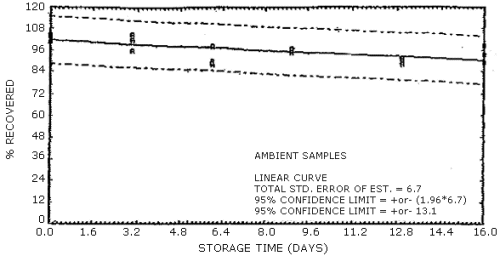
Figure 4.7.5. Ambient temperature storage test for N-nitrosopropylbutylamine.
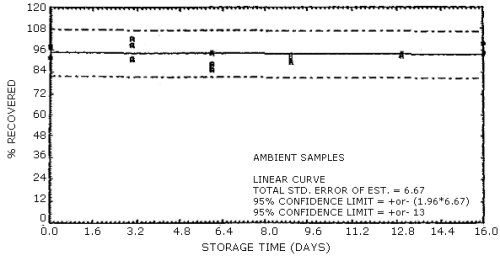
Figure 4.7.6. Ambient temperature storage test for N-nitrosodiamylamine.

Figure 4.7.7. Reduced temperature storage test for N-nitrosomethylethylamine.

Figure 4.7.8. Reduced temperature storage test for N-nitrosodiisopropylamine.

Figure 4.7.9. Reduced temperature storage test for N-nitrosomethylbutylamine.

Figure 4.7.10. Reduced temperature storage test for N-nitrosoethylbutylamine.

Figure 4.7.11. Reduced temperature storage test for N-nitrosopropylbutylamine.
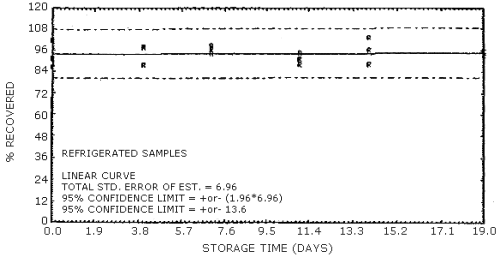
Figure 4.7.12. Reduced temperature storage test for N-nitrosodiamylamine.
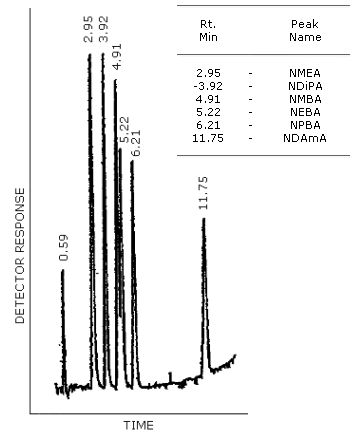
Figure 4.8.1. GC/TRA chromatogram of the analytes with the Carbowax 20M column. The column temperature was programmed from 150 to 220°C at 4°C/min. The injector was set at 150°C and the carrier gas flow rate was 30 mL/min.
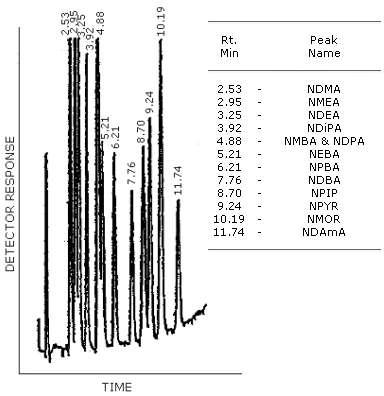
Figure 4.8.2. GC/TEA chromatogram of a mixture of N-nitrosamines with the Carbowax 20 M column. The column temperature was programmed from 150 to 220°C at 4°C/min. The injector was set at 150°C and the carrier gas flow rate was 30 mL/min.

Figure 4.8.3. GG/TEA chromatogram of a mixture of N-nitrosamines with the Versamid 900 column. The column temperature was programmed from 150 to 220°C at 4°C/min. The injector was set at 150°C and the carrier gas flow rate was 30 mL/min.
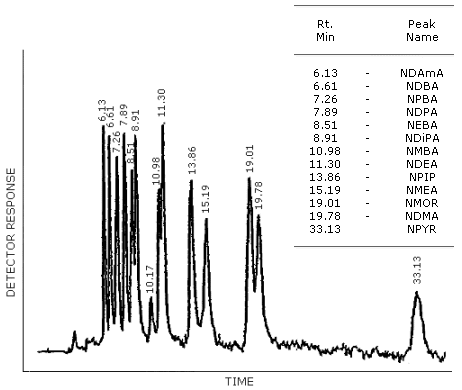
Figure 4.8.4. HPLC/TEA chromatogram of a mixture of N-nitrosamines with the CN column. The mobile phase was 94.5% 2,2,4-trimethylpentane, 5% dichloromethane and 0.5% isopropanol. The flowrate was 1.3 mL/min.
5. References
5.1. Hendricks, W. Volatile Nitrosamine Mixture I (Method 27, Organic Methods Evaluation Branch, OSHA Analytical Laboratory, Salt Lake City, Utah) Unpublished (2-81).
5.2. Fine, D.H.; Rounbehler, D.P.; Sawicki, E. and Krost, K. Environ. Science Technol., 11, 577-580, (1977).
5.3. Fine, D.H. Oncology, 37, 199-202, (1980).
5.4. Rounbehler, D.P.; Reisch, J.W.; Coombs, J.R. and Fine, D.H. Anal. Chem., 52, 273-276, (1980).
5.5. Fisher, R.L.; Rieser, R.W., and Lasoski, B.A. Anal. Chem., 49, 1821-1823, (1977).
5.6. Sen, N.P. "Environmental Carcinogens Selected Methods of Analysis, Volume 1-Analysis of Volatile Nitrosamines in Food", IARC Scientific Publications No. 18, Egan, H., Editor-in-Chief, International Agency for Research on Cancer: Lyon (1978).
5.7. Instruction Manual - Thermal Energy Analyzer, Model 502/LC, Thermal Electron Corporation, Waltham, Mass. 02145, 3-1, (12-77).
5.8. Druckrey, D.; Preussmann, R.; Ivankovic, S. and Schmahl, D. Z. Krebsforsch, 69, 103-201, (1967).
5.9. Edelman, A.S.; Kraft, P.L.; Rand, W.M., and Wishnok, J.S. Chem. Biol. Interactions, 31, 81-92, (1980).
5.10. McCann, J.; Choi, E.; Yamasaki, E., and Ames, B.N. Proc. Nat. Acad. Sci. USA, 72, 5135-5139, (1975).
5.11. Yahagi, T.; Nagao, M.; Seino, Y.; Matsushima, T.; Sugimura, T., and Okada, M. Mutation Res., 48, 121-130, (1977).
5.12. Ember, L.R. Chem. Eng. News, 58, 20-26, (1980).
5.13. Rounbehler, D.P.; Krull, I.S.; Goff, E.U.; Mills, K.M.; Edwards, G.S.; Fine, D.H.; Rheinhold, V.; Morrison, J.; Fagen, J.M. and Carson, G.A. Fd. Cosmet. Toxicol. 17, 487-491, (1979).
5.14. Fajen, J.M.; Carson, G.A.; Rounbehler, D.P.; Fan, T.Y.; Vita, R.; Goff, V.E.; Wolf, M.H.; Edwards, G.S.; Fine, D.H.; Reinhold, V. and Biemann, K. Science, 205, 1262-1264, (1979).
5.15. Chem. Eng. News, 57, 6, (1979).
5.16. Chem. Eng. News, 57, 18, (1979).
5.17. Brunnemann, K.D.; Yu, L. and Hoffmann, D. Cancer Research, 37, 3218-3222, (1977).
5.18. Day, E.W.; West, S.D.; Koenig, D.K. and Powers, F.L. J. Agric. Food Chem., 27, 1081-1085, (1979).
5.19. Fiddler, W.; Pensabene, J.W.; Doerr, R.C. and Dooley, C.J. Fd. Cosmet. Toxicol., 15, 441-443, (1977).
5.20. Spiegelhalder, B.; Eisenbrand, G. and Preussmann, R. Angew. Chem. Int. Ed. Engl., 17, 367-368, (1978).
5.21. Archer, M.C. and Wishnok, J.S., J. Environ. Sci. Health, 10 & 11, 583-590, (1976).
5.22. Goff, E.U.; Coombs, J.R. and Fine, D.H. Anal. Chem., 52, 1833-1836, (1980).
5.23. Challis, B.C.; Edwards, A.; Hunna, R.R.; Kyrtopoulos, S.A. and Outram, J.P. "Environmental Aspects of N-Nitroso Compounds, IARC Scientific Publications No. 19", Walker, E.A., Lyle, R.E., Castegnaro M. and Griciute, L. Eds., International Agency for Research on Cancer: Lyon, 127-139, (1978).
5.24. March, J. "Advanced Organic Chemistry: Reactions, Mechanisms and Structure", McGraw-Hill, Inc.: New York, 430-431, (1968).
5.25. Fine, D.H. "Monitoring Toxic Substances", Schuetzle, D. Ed., American Chemical Society Symposium, Series 94: Washington, D.C., 246-254, (1979).
5.26. Registry of Toxic Effects of Chemical Substances, 1979 Edition. (Lewis, R.J. and Tatken, R.L., Eds.) U.S. Department of Health, Education and Welfare, Public Health Service, Center for Disease Control, National Institute for Occupational Safety and Health, U.S. Government Printing Office, Washington, D.C. (1980).
5.27. "CRC Handbook of Chemistry and Physics", CRC Press: Boca Raton, FL (1979).
5.28. "IARC Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans, Some N-nitroso Compounds, Volume 17", International Agency for Research on Cancer: Lyon (1978).
5.29. Wishnok, J.S. and Tannenbaum, S.R. Anal. Chem., 49, 715A-718A, (1977).
5.30. Lijinsky, W. and Epstein, 5.5. Nature, 225, 21-23, (1970).
5.31. Sander, J.; Schweinsberg, F.; La Bar, J. and Burkle, G. "GANN Monograph on Cancer Research 17", 145-160, (1975).
5.32. Iqbal, Z.M.; Dahl, K. and Epstein, 5.5. Science, 207, 1475-1476, (1980).
5.33. Wang, T.; Kakizoe, T.; Dion, P.; Furrer, R.; Varghese, A.J. and Bruce, W.R. Nature, 276, 280-281, (1978).
5.34. Kakizoe, T.; Wang, T.; Eng, V.W.S.; Furrer, R.; Dion, P. and Bruce, W.R. Cancer Research, 39, 829-832, (1979).
5.35. "ThermoSorb/N Air Sampler-Instructions for Monitoring", 5-25 Thermo Electron Corporation (1980).
5.36. Lijinsky, W. and Taylor, W. J. Natl. Cancer Inst., 62, 407-410, (1979).
5.37. Heath, D.F. and Magee, P.N. Brit. J. Indur. Med., 19, 276-282, (1962).
5.38. Druckrey, H.; Landschutz, C. and Preussmann, R. Z. Krebsforsch, 75, 221-224, (1971).
5.39. Druckrey, H. and Landschutz, C. Z. Krebsforsch, 75, 221-224, (1971).
5.40. Okada, M.; Suzuki, E. and Hashimoto, Y. Gann, 67, 825-834, (1976).
5.41. Pitts, J.N.; Grosjean, D.; Cauwenberghe, K.V.; Schmid, J.P. and Fitz, D.R. Environ. Science Technol., 12, 946-953, (1978).
|

