|
HYDRAZINE
| Method number: | 108 |
| |
| Matrix: | Air |
| |
| Target concentration: |
10 ppb (13 µg/m3) and 1 ppm (1.3 mg/m3) |
| OSHA PEL: | 1 ppm (1.3 mg/m3) |
| ACGIH TLV: | 10 ppb (13 µg/m3) |
| |
| Procedure: | Samples are collected closed-face by drawing known volumes of air
through sampling devices consisting of three-piece cassettes, each containing two sulfuric acid treated 37-mm glass fiber filters
separated by the ring section. The filters are extracted with a buffered EDTA disodium solution. An aliquot of the extract is derivatized with a benzaldehyde solution to form benzalazine from any hydrazine in the samples. The benzalazine is quantitated by LC
using a UV detector. |
| |
Recommended air volume
and sampling rate: | 240 L at 1.0 L/min |
| |
| Reliable quantitation limit: | 0.058 ppb (0.076 µg/m3) |
| |
Standard error of estimate at
the target concentration:
|
7.5% at 10 ppb Target Concentration
5.2% at 1 ppm Target Concentration |
| |
| Status of method: |
Evaluated method. This method has been subjected to the established evaluation
procedures of the Organic Methods Evaluation Branch. |
| |
| Date: February 1997 | Chemist: Carl J. Elskamp |
Organic Methods Evaluation Branch
OSHA Salt Lake Technical Center
Salt Lake City, UT 84165-0200
1. General Discussion
1.1 Background
1.1.1 History
In 1980, an air sampling and analytical procedure to determine hydrazine was validated
by the OSHA Analytical Laboratory. (Ref. 5.1) The method
(OSHA Method 20) is based on a field procedure developed by the U.S. Air Force that involves collection of samples using sulfuric acid coated Gas Chrom R and colorimetric analysis using
r-dimethylaminobenzaldehyde (Ref. 5.2) Because colorimetric procedures are more susceptible to interferences, an LC analysis procedure was also developed by OSHA in Method 20 where benzalazine, the reaction product between hydrazine and benzaldehyde, is quantitated using a UV detector.
After Method 20 was completed, sulfuric acid treated Gas Chrom R adsorbent tubes
became commercially available. Since that time, the OSHA SLTC (Salt Lake Technical
Center) has received comments from other laboratories reporting low extraction
efficiencies and sample recoveries when using the commercial tubes. It was decided that
these concerns would be investigated. Also, because ACGIH lowered the TLV from 100
ppb to 10 ppb (Ref. 5.3) since Method 20 was validated and because OSHA may consider lowering the PEL from 1 ppm in the future, the methodology was evaluated at
lower levels with test atmospheres. It was not possible for OSHA to generate hydrazine
test atmospheres when Method 20 was validated. Since that time, a controlled test
atmosphere generation system was constructed at the OSHA SLTC that can be used to
safely generate atmospheres from toxic compounds such as hydrazine.
A 2.1-ppm atmosphere was generated and samples were collected using both
commercial sulfuric acid treated Gas Chrom R sampling tubes and sampling tubes that
were prepared in-house. Both samplers gave similar recoveries, but most of the
hydrazine was found on the front glass wool plug of the commercial tubes. This indicated
that as prepared samplers are stored for an extended period of time before use, some
of the sulfuric acid apparently migrates to the glass wool plugs, turning the plug into an
effective sampler for hydrazine. It is not known how old the commercial tubes were for
this study, but they were probably several years old. It was felt that the commercial tubes
would be suitable, as long as the glass wool plug was extracted and analyzed along with
the treated Gas Chrom R. But further tests at ppb levels of hydrazine showed poor
recoveries and poor sample extract stability from acid treated Gas Chrom R, especially
for the commercial tubes. The poor recoveries and stability may be attributed to the
possible presence of trace metals in the adsorbent, which was indicated by the yellow
color of EDTA extracts of the acid treated Gas Chrom R. The EDTA extracts from the
commercial tubes were deeper yellow than that from the in-house prepared tubes,
suggesting the presence of more metals. It was also found that the commercially
prepared tubes had a higher acid content than the in-house tubes, which may be
important for recovery yields because the reaction between hydrazine and benzaldehyde
is somewhat pH dependent. An alternate sampling device that would avoid the problems
associated with acid treated Gas Chrom R was desirable. A promising candidate was a
sampler consisting of sulfuric acid treated glass fiber filters that had been validated by
the OSHA SLTC for a number of aromatic amines (Refs. 5.4-5.11).
Initial tests using sulfuric acid treated Gelman A/E filters at ppm levels of hydrazine
showed collection efficiencies, recoveries, and extraction efficiencies to be essentially
100%. The recovery of hydrazine collected from test atmospheres is not quite as good
at ppb levels. In an attempt to enhance recoveries, filters treated with either EDTA or
Vitamin C alone or with sulfuric acid were tested with no improvement. The filters were
also treated with different mineral acids, including hydrochloric, nitric and phosphoric
acid, but again recoveries were no better than when just sulfuric acid was used. Gelman
A/E filters were used in the methods for aromatic amines and may be suitable for
hydrazine, but it was found that the thicker A/B filters formed tighter seals in the filter
cassettes and they also provided slightly better collection efficiencies. The A/B filters can also be used for aromatic amines with no other change in the methods.
Improvements were also made to the analytical procedure. It was found that the room
temperature formation of benzalazine from hydrazine and benzaldehyde proceeded to
completion in less than 30 minutes at about pH 3.5. Also, the use of EDTA disodium
enhanced the stability of extracted samples, so an aqueous EDTA disodium solution
buffered to pH 3.5 is used to extract the filter samples. Acetonitrile is now used instead
of methyl alcohol as the solvent for benzaldehyde. The recommended air volume was
20 L in Method 20, but in order to obtain a lower reliable quantitation limit and to have a
more convenient sampling time of 4 hours, 240 L is the new recommended air volume.
The amount of benzalazine formed from 240-L samples at ppm levels was found to be
somewhat insoluble in methyl alcohol, but was found to be much more soluble in acetonitrile.
1.1.2 Toxic effects (This section is for information only and should not be taken as the basis
of OSHA policy.)
Hydrazine is carcinogenic in experimental animals and is a suspected human carcinogen.
(Ref. 5.3) It is corrosive to the eyes, skin, and mucous membranes. The liver, kidney, and hematopoietic system are the main target organs following repeated exposures. (Ref. 5.12)
1.1.3 Workplace exposure
Hydrazine is chiefly used as a chemical intermediate in the production of agricultural
chemicals, spandex fibers and antioxidants. It is also used as a rocket fuel, oxygen
scavenger in boiler water treatment, polymerization catalyst, blowing agent, and
scavenger for gases. (Ref. 5.3)
1.1.4 Physical properties (Ref. 5.13 unless otherwise denoted)
| CAS number: | 302-01-2 |
| molecular weight: | 32.06 |
| boiling point: | 113.5°C |
| melting point: | 1.4°C |
| appearance: | colorless, oily, fuming liquid or white crystals |
| density: | 1.1011 at 15°C |
| molecular formula: | H4N2 |
| vapor pressure: | 1.3 kPa at 20°C (Ref. 5.3) |
| flash point: | 100°F (37.8°C) (open cup) |
| odor: | penetrating, resembling ammonia |
| lower explosive limit: | 4.7% (Ref. 5.3) |
| synonyms: | Diamide; diamine; hydrazine, anhydrous (DOT); hydrazine, aqueous solution (DOT); hydrazine base; hydrazyna (Polish); RCRA waste number U133 |
| structural formula: | H2NNH2 |
The analyte air concentrations throughout this method are based on the recommended
sampling and analytical parameters. Air concentrations listed in ppb and ppm are
referenced to 25°C and 101.3 kPa (760 mmHg). Although benzalazine is the actual
species analyzed, all masses presented are in terms of hydrazine.
1.2 Limit defining parameters
1.2.1 Detection limit of the analytical procedure
The detection limit of the analytical procedure is 10.6 pg. This is the amount of analyte
that will give a response that is significantly different from the background response of
a reagent blank. (Sections 4.1 and 4.2)
1.2.2 Detection limit of the overall procedure
The detection limit of the overall procedure is 5.48 ng per sample (0.017 ppb or 0.023
µg/m3). This is the amount of analyte spiked on the sampler that will give a response that is significantly different from the background response of a sampler blank. (Sections 4.1 and 4.3)
1.2.3 Reliable quantitation limit
The reliable quantitation limit is 18.3 ng per sample (0.058 ppb or 0.076 µg/m3). This is the amount of analyte spiked on a sampler that will give a signal that is considered the lower limit for precise quantitative measurements. (Section 4.4)
1.2.4 Precision (analytical procedure)
The precisions of the analytical procedure, measured as the pooled relative standard
deviation over concentration ranges equivalent to 0.5 to 2 times the target concentration,
are 0.26% and 0.09% based on the 10-ppb and the 1-ppm target concentrations respectively. (Section 4.5)
1.2.5 Precision (overall procedure)
The precisions of the overall procedure at the 95% confidence level for the ambient
temperature storage tests are ±14.8% and ±10.1% at 9.4 ppb and 1.06 ppm respectively.
These include an additional 5% for sampling error. (Section 4.6)
1.2.6 Recovery
The recovery of hydrazine from samples used in a 19-day storage test remained above
78% at the 10-ppb target concentration when the samples were stored at ambient
temperatures. The recovery of hydrazine from samples used in a 20-day storage test remained above 96% at the 1-ppm target concentration when the samples were stored at ambient temperatures. (Section 4.7)
1.2.7 Reproducibility
Six samples at each target concentration that were collected from controlled test
atmospheres, along with a draft copy of this procedure, were submitted to an SLTC
service branch for analysis. The samples were analyzed after 63 and 58 days of storage
at 0°C for the 10-ppb and 1-ppm target concentrations respectively. One of the 10-ppb target concentration samples was an outlier, while none of the remaining sample results deviated from its theoretical value by more than the precisions reported in Section 1.2.5. (Section 4.8)
2. Sampling Procedure
2.1 Apparatus
2.1.1 Samples are collected using a personal sampling pump calibrated, with a sampling
device attached, to within ±5% at the recommended flow rate.
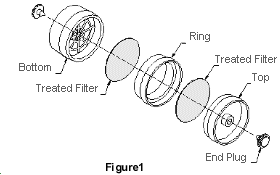 2.1.2 Samples are collected closed-face using a sampling device consisting of
two sulfuric acid treated 37-mm Gelman Sciences type A/B glass fiber extra thick filters (part no. 66208) contained in a three-piece SAN (styrene acrylonitrile) plastic cassette (Gelman part no. 4339 or Millipore Corp., Bedford, MA, part no.
M000037A0). The filters are prepared by soaking each filter with 1.0 mL of 0.26 N sulfuric acid. The 0.26 N sulfuric acid can be prepared by diluting 1.5 mL of 36 N sulfuric
acid to 200 mL with either methyl alcohol or deionized water. The filters are dried in an
exhaust hood when a methanolic solution is used and in an oven at 100°C when an
aqueous solution is used and then assembled into three-piece 37-mm cassettes without support pads. The front filter is separated from the back filter by the ring section. The cassettes are sealed with shrink bands and the ends are plugged with plastic plugs.
2.1.2 Samples are collected closed-face using a sampling device consisting of
two sulfuric acid treated 37-mm Gelman Sciences type A/B glass fiber extra thick filters (part no. 66208) contained in a three-piece SAN (styrene acrylonitrile) plastic cassette (Gelman part no. 4339 or Millipore Corp., Bedford, MA, part no.
M000037A0). The filters are prepared by soaking each filter with 1.0 mL of 0.26 N sulfuric acid. The 0.26 N sulfuric acid can be prepared by diluting 1.5 mL of 36 N sulfuric
acid to 200 mL with either methyl alcohol or deionized water. The filters are dried in an
exhaust hood when a methanolic solution is used and in an oven at 100°C when an
aqueous solution is used and then assembled into three-piece 37-mm cassettes without support pads. The front filter is separated from the back filter by the ring section. The cassettes are sealed with shrink bands and the ends are plugged with plastic plugs.
2.2 Reagents
None required
2.3 Technique
2.3.1 Remove the plastic end plugs from the sampling device immediately before sampling.
Samples are collected closed-face.
2.3.2 Attach the sampling device to the sampling pump with flexible tubing and place the
device in the employee's breathing zone. Position the sampler so it does not impede
work performance or safety.
2.3.3 Do not pass the sampled air through any hose or tubing before it enters the sampling
device.
2.3.4 Immediately after sampling, seal the sampling device with plastic end plugs and seal and
identify with a Form OSHA-21 seal.
2.3.5 Submit at least one blank with each sample set. Blanks should be handled in the same
manner as samples, except no air is drawn through them.
2.3.6 Record sample volumes (in liters of air) for each sample. Also list any compounds
considered potential interferences that could be present in the sampling area.
2.3.7 If any bulk samples are submitted for analysis, ship them in separate containers from the air samples.
2.3.8 Submit the samples to the laboratory for analysis as soon as possible after sampling. If
delay is unavoidable, store the samples at reduced temperature.
2.4 Sampler capacity
Sampler capacity is determined by measuring how much air can be sampled before breakthrough
of analyte through the sampler occurs, i.e., the sampler capacity is exceeded. Breakthrough is
considered to occur when the effluent from the sampler contains a concentration of analyte that is 5% of the upstream concentration (5% breakthrough). Tests for breakthrough were performed by simultaneously sampling at 1 L/min from a hydrazine atmosphere (81.5% RH, 23.6°C) at 2.1 ppm using four samplers. The sampler flows were momentarily interrupted and the back filters were removed and replaced with fresh filters every hour after sampling began over a period of 8 hours. The back filters were analyzed to determine the hydrazine concentration in the effluents. At no time during the 8-hour tests did the effluent concentrations exceed 5% of the upstream concentration for any of the four samplers. The filters are coated with an amount of sulfuric acid that could theoretically collect about 4.5 mg of hydrazine. For a 240-L air sample, this would be equal to 14.3 ppm (18.8 mg/m3).
2.5 Extraction efficiency
2.5.1 The average extraction efficiencies over the range of 0.5 to 2 times the target
concentrations are 98.7% and 98.9% based on the 10-ppb and 1-ppm levels respectively. (Section 4.9.1)
2.5.2 The extraction efficiency at 0.05, 0.1, and 0.2 times the target concentration was found
to be 97.7%, 97.5%, and 96.3% respectively based on the 10-ppb level and 98.2%,
98.0%, and 98.6% respectively based on the 1-ppm level. (Section 4.9.1)
2.5.3 Both extracted and extracted/derivatized samples at each target concentration remain
stable for at least 24 h. (Section 4.9.2)
2.6 Recommended air volume and sampling rate
2.6.1 For long-term samples, sample 240 L of air at 1 L/min (4-h samples).
2.6.2 For short-term samples, sample 15 L of air at 1 L/min (15-min samples).
2.6.3 When short-term samples are collected, the air concentration equivalent to the reliable
quantitation limit becomes larger. For example, the reliable quantitation limit is 0.93 ppb
(1.22 µg/m3) when 15 L of air is sampled.
2.7 Interferences (sampling)
2.7.1 It is not known if any compounds will severely interfere with the collection of hydrazine on sulfuric acid treated filters. In general, any compound in the sampled air that would react with sulfuric acid to decrease the amount on the filter will decrease the
breakthrough volume. Also, any compound that will react with hydrazine or hydrazine
sulfate is a potential interference.
2.7.2 Suspected interferences should be reported to the laboratory with submitted samples.
2.8 Safety precautions (sampling)
2.8.1 Attach the sampling equipment to the employee so that it will not interfere with work
performance or safety.
2.8.2 Follow all safety procedures that apply to the work area being sampled.
3. Analytical Procedure
3.1 Apparatus
3.1.1 An LC system equipped with an ultraviolet detector. A Hewlett-Packard 1050 Series LC
consisting of a pumping system, a programmable variable wavelength detector and an autosampler was used in this evaluation.
3.1.2 An LC column capable of separating the analyte of interest from any interferences. A
12.5-cm × 4-mm i.d. LiChrospher 100 RP-18 column (Hewlett-Packard Company, part no. 79925OD-564) was used in this evaluation.
3.1.3 An electronic integrator or some other suitable means of measuring peak areas or
heights. A Waters Millennium Networking Computer System was used in this evaluation.
3.1.4 Glass vials with Teflon®-lined caps. Kimble Glass Co. 7-mL disposable scintillation vials (part no. 74502-7) and Thomas Scientific caps (cat. no.
2390-B16) were used in the preparation of standards and extraction of sample filters. National Scientific Co. 2-mL Target DP™ vials and caps (part no.
C4000-1 and C4000-54Y respectively) were used in the derivatization of standards and filter extracts.
3.1.5 Hand held dispensers (repeating pipettors) capable of delivering 0.5, 1.0 and 5.0 mL for
preparing and transferring standards and sample extracts and for dispensing the
derivatizing solution. If dispensers are not available, volumetric pipettes may be used
instead.
3.1.6 A test tube rotator to mix the samples during the extraction step.
3.1.7 A laboratory centrifuge.
3.2 Reagents
3.2.1 Hydrazine or hydrazine solution, reagent grade. Aldrich Chemical (Milwaukee, WI) Lot
21131EN anhydrous hydrazine was used in this evaluation. Hydrazine is a highly toxic
sensitizer, mutagen, and cancer suspect agent that is readily absorbed through skin. It
is also a corrosive and combustible liquid and a strong reducing agent that should be
stored under nitrogen.
Standards may alternatively be prepared from reagent grade benzalazine or from a
reagent grade hydrazine salt, such as hydrazine dihydrochloride or hydrazine sulfate.
3.2.2 Acetonitrile, methanol, and water, LC grade. The acetonitrile and methanol used in this
evaluation were "Optima" brand from Fisher Chemical (Fair Lawn, NJ) and the water was
from a Millipore Milli-Q water purification system.
3.2.3 Benzaldehyde, reagent grade. Aldrich Lot 03628KN was used in this evaluation.
Benzaldehyde is an irritant, mutagen, sensitizer, and cancer suspect agent. It is air and
light sensitive and should be store under nitrogen.
3.2.4 Sodium phosphate, monobasic monohydrate (NaH2PO4· H2O), reagent grade. Fisher Lot 704979 was used in this evaluation.
3.2.5 EDTA disodium, from ethylenediaminetetraacetic acid, disodium salt dihydrate, reagent
grade. Aldrich Lot 14428LZ was used in this evaluation.
3.2.6 Phosphoric acid, reagent grade.
3.2.7 Extraction solution, consisting of an aqueous solution of 0.1 M NaH2PO4· H2O/0.05 M EDTA disodium adjusted to pH 3.5 with phosphoric acid.
3.2.8 Derivatizing solution, prepared by diluting 1.0 mL of benzaldehyde to 100 mL with
acetonitrile. The derivatizing solution should be prepared fresh daily.
3.3 Standard preparation
3.3.1 Prepare concentrated stock solutions by accurately diluting a known amount of hydrazine
with methanol. Stock solutions are stable for at least 3 days when refrigerated.
Standards can alternatively and more conveniently be prepared from hydrazine
dihydrochloride, hydrazine sulfate or benzalazine. Stock standards from hydrazine salts
should be prepared in water, while standards from benzalazine should be prepared in
acetonitrile. If standards are preprared from one of the salts or benzalazine, the
appropriate conversion factor to determine the equivalent mass of hydrazine must be
used. For example, if hydrazine sulfate (MW = 130.12) is used to make standards, the
mass of hydrazine sulfate weighed out must be multiplied by 0.2464 (32.06 ÷ 130.12) to
obtain the equivalent mass of hydrazine.
3.3.2 Prepare analytical standards by injecting microliter amounts of stock standards into
7-mL vials containing 5.0 mL of extraction solution delivered from the same dispenser or pipet used to extract samples.
3.3.3 Derivatize the analytical standards by transferring 1.0 mL of the standards to separate
autosampler vials and adding 0.5 mL of derivatizing solution to each vial. Cap each vial
and shake it for a few seconds to obtain thorough mixing. Allow the vials to sit at room
temperature for at least 30 min before analysis. Analyze the derivatized standards by LC.
3.3.4 Bracket sample concentrations with analytical standard concentrations. If samples fall
outside of the concentration range of prepared standards, prepare and analyze additional
standards at the appropriate concentrations to ascertain the linearity of response.
Theoretically, there is enough benzaldehyde added to the vials to prepare standards as
high as 2.5 ppm for 240-L air samples (789 µg/sample). Alternatively, if sample concentrations are higher than the highest standard, the extracts can be diluted with
extraction solution, and 1.0 mL aliquots of the diluted extracts can be derivatized and
analyzed.
3.4 Sample preparation
3.4.1 Transfer front and back filters to individual 7-mL vials.
3.4.2 Add 5.0 mL of extraction solution to each vial using the same dispenser or pipet as used
for preparation of standards.
3.4.3 Cap the vials and mix them on a rotator for 30 min.
3.4.4 Centrifuge the sample vials for 10 min at 2000 rpm.
3.4.5 Derivatize the samples by transferring 1.0 mL of the centrifuged extracts to separate
autosampler vials and adding 0.5 mL of derivatizing solution to each vial. Cap each vial
and shake them for a few seconds to obtain thorough mixing. Allow the vials to sit at
room temperature for at least 30 min before analysis. Analyze the derivatized samples
by LC. As a precautionary step, the sample extracts should be derivatized as soon as
possible after they are extracted and centrifuged.
3.5 Analysis
3.5.1 LC conditions
| mobile phase: | 67/33, acetonitrile/water |
| flow rate: | 1.0 mL/min |
| UV detector wavelength: | 300 nm |
| detector range: | 2 AUFS for 1-ppm target concentration 0.02 AUFS for 10-ppb target concentration |
| injection volume: | 4.5 µL |
| column: | 12.5-cm × 4-mm i.d. LiChrospher 100 RP-18 |
| retention time: | 5.8 min |
3.5.2 Peak areas or heights are measured by an integrator or other suitable means.
3.5.3 An external standard (ESTD) calibration method is used. Calibration curves are prepared
by plotting the amount of analyte per sample versus peak heights or area counts of the
standards. Sample concentrations must be bracketed by standards.
3.6 Interferences (analytical)
3.6.1 Any compound that produces a response on a UV detector at 300 nm and has the same
general retention time of benzalazine is a potential interference. Possible interferences
should be reported to the laboratory with submitted samples by the industrial hygienist.
These interferences should be considered before samples are extracted.
3.6.2 LC parameters may be changed to possibly circumvent interferences.
3.6.3 When necessary, the identity or purity of an analyte peak may be confirmed with
additional analytical data, such as wavelength ratioing. As an aid in choosing appropriate
wavelengths to ratio, the UV spectrum of benzalazine is given in Section 4.10.
3.7 Calculations
The hydrazine concentration for samples is obtained from the calibration curve in terms of
micrograms of hydrazine per sample. The back filter of each sampler is analyzed primarily to
determine if there was any breakthrough from the front filter during sampling. If a significant amount of analyte is found on the back filter, this fact should be reported with sample results. If any analyte is found on the back filter, it is added to the amount found on the front filter. This total amount is then corrected by subtracting the total amount (if any) found on the blank. The air concentration is calculated using the following formula.
mg/m³ = (µg of hydrazine per sample) / [(L of air sampled)(extraction efficiency)]
ppm = [(mg/m³)(24.46)] / 32.06 = (mg/m³)(0.7629)
3.8 Safety precautions (analytical)
3.8.1 Adhere to the rules set down in your Chemical Hygiene Plan.
3.8.2 Avoid skin contact and inhalation of all chemicals.
3.8.3 Wear safety glasses and a lab coat at all times while in the lab area.
4. Backup Data
4.1 Determination of detection limits
Detection limits (DL), in general, are defined as the amount (or concentration) of analyte that gives a response (YDL) that is significantly different (three standard deviations (SDBR)) from the background response (YBR).
YDL - YBR = 3(SDBR)
The direct measurement of YBR and SDBR in chromatographic methods is typically inconvenient and difficult because YBR is usually extremely low. Estimates of these parameters can be made with data obtained from the analysis of a series of analytical standards or samples whose
responses are in the vicinity of the background response. The regression curve obtained for a plot of instrument response versus concentration of analyte will usually be linear. Assuming SDBR and the precision of data about the curve are similar, the standard error of estimate (SEE) for the regression curve can be substituted for SDBR in the above equation. The following calculations derive a formula for DL:
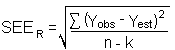
| Yobs = |
observed response |
| Yest = |
estimated response from regression curve |
| n = |
total no. of data points |
| k = |
2 for linear regression curve |
At point YDL on the regression curve
YDL = A(DL) + YBR
A = analytical sensitivity (slope)
therefore
Substituting 3(SEE) + YBR for YDL gives
4.2 Detection limit of the analytical procedure (DLAP)
The DLAP is measured as the mass of analyte introduced into the chromatographic column. Ten
analytical standards were prepared in equal descending increments with the highest standard
containing 31.88 ng of hydrazine per sample. This standard produces a peak approximately 10
times the baseline noise of a reagent blank. Standards, plus a reagent blank, were analyzed and the data obtained were used to determine the required parameters (A and SEE) for the calculation of the DLAP. Values of 2222 and 7881 were obtained for A and SEE respectively. The DLAP was calculated to be 10.6 pg.
4.3 Detection limit of the overall procedure (DLOP)
The DLOP is measured as mass per sample and expressed as equivalent air concentrations,
based on the recommended sampling parameters. Ten samplers were spiked with equal
descending increments of hydrazine such that the highest sampler loading was 31.88 ng/sample.
This is the amount, when spiked on a sampler, that would produce a peak approximately 10 times
the baseline noise for a sample blank. These spiked samplers, plus a sample blank, were
analyzed with the recommended analytical parameters, and the data obtained used to calculate
the required parameters (A and SEE) for the calculation of the DLOP. Values of 1553 and 2839
were obtained for A and SEE respectively. The DLOP was calculated to be 5.48 ng/sample (0.017
ppb or 0.023 µg/m3).
4.4 Reliable quantitation limit (RQL)
The RQL was calculated to be 18.3 ng/sample (0.058 ppb or 0.076 µg/m3). The recovery at this level is 90.2%.
4.5 Precision (analytical method)
The precisions of the analytical procedure are defined as the pooled relative standard deviations (RSDP). Relative standard deviations were determined from six replicate injections of standards at 0.5, 0.75, 1, 1.5, and 2 times the target concentrations. After assuring that the RSDs satisfy the Cochran test for homogeneity at the 95% confidence level, the RSDPs were calculated to be 0.26% and 0.09% based on the 10-ppb and 1-ppm target concentrations respectively.
Table 4.5.1
Instrument Response Based on a 10-ppb Target Concentration
|
× target concn
(ng/sample) |
0.5×
1594 |
0.75×
2390 |
1.0×
3187 |
1.5×
4781 |
2.0×
6374 |
|
peak areas
(µV· s) |
3187300
3176400
3164800
3175300
3173000
3176900 |
5022900
5002500
5006200
5001200
4991800
4996400 |
6648300
6601100
6615000
6595100
6620900
6627700 |
10115000
10032000
10035000
10043000
10017000
10026000 |
13192000
13161000
13144000
13184000
13167000
13130000 |
| |
mean
SD
RSD (%) |
3175600
7248.6
0.228 |
5003500
10745
0.215 |
6618000
19181
0.290 |
10045000
35545
0.354 |
13163000
23461
0.178 |
|
The Cochran test for homogeneity:
|
g =
|
largest RSD2
RSD20.5x +
RSD20.75x +
RSD21x +
RSD21.5x +
RSD22x
|
= 0.369
|
The critical value of the g statistic at the 95% confidence level for five variances, each associated with six observations, is 0.5065. Because the g statistic does not exceed this value, the RSDs can be considered equal and they can be pooled (RSDP) to give an estimated RSD for the concentration range studied.
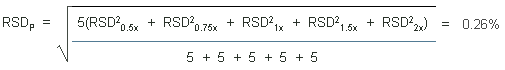
Table 4.5.2
Instrument Response Based on a 1-ppm Target Concentration
|
× target concn
(µg/sample) |
0.5×
159.4 |
0.75×
239.0 |
1.0×
318.7 |
1.5×
478.1 |
2.0×
637.4 |
|
peak areas
(µV· s) |
3338300
3331000
3334800
3336100
3336200
3336800 |
5030400
5035000
5036400
5039600
5040700
5046400 |
6668100
6672700
6674500
6682700
6684100
6685400 |
9869800
9877700
9877900
9878900
9879000
9890700 |
12721000
12741000
12744000
12745000
12746000
12752000 |
| |
mean
SD
RSD (%) |
3335500
2494.5
0.075 |
5038100
5471.5
0.109 |
6677900
7104.5
0.106 |
9879000
6699.9
0.068 |
12742000
10672
0.084 |
|
The Cochran test for homogeneity:
|
g =
|
largest RSD2
RSD20.5x +
RSD20.75x +
RSD21x +
RSD21.5x +
RSD22x
|
= 0.294
|
The critical value of the g statistic at the 95% confidence level for five variances, each associated with six observations, is 0.5065. Because the g statistic does not exceed this value, the RSDs can be considered equal and they can be pooled (RSDP) to give an estimated RSD for the concentration range studied.
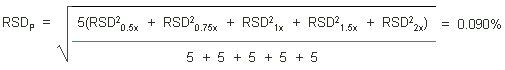
4.6 Precision (overall procedure)
The precision of the overall procedure is determined from the storage data in
Section 4.7. The determination of the standard error of estimate (SEER) for a regression line plotted through the graphed storage data allows the inclusion of storage time as one of the factors affecting overall precision. The SEER is similar to the standard deviation, except it is a measure of dispersion of data about a regression line instead of about a mean. It is determined with the following equation:
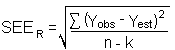
n =
k =
k = |
total no. of data points
2 for linear regression
3 for quadratic regression |
| Yobs = |
observed % recovery at a given time |
| Yest = |
estimated % recovery from the regression line at the same given time |
An additional 5% for pump error (SP) is added to the SEER by the addition of variances to obtain the total standard error of estimate.

The precision at the 95% confidence level is obtained by multiplying the standard error of estimate (with pump error included) by 1.96 (the z statistic from the standard normal distribution at the 95% confidence level). The 95% confidence intervals are drawn about their respective regression lines in the storage graphs, as shown in Figures
4.7.1.1, 4.7.1.2, 4.7.2.1 and 4.7.2.2. The precisions of the overall procedure of ±14.8% and ±10.1% were obtained from Figures 4.7.1.2 and 4.7.2.2 for 10-ppb and
1-ppm target concentrations respectively.
4.7 Storage test
Storage samples were prepared by sampling at 1 L/min from two different controlled test
atmospheres, one at 9.40 ppb and the other at 1.06 ppm. Both atmospheres were at
approximately 80% RH and at room temperatures ranging from 22-26°C. Six samples for each level were analyzed immediately after generation, fifteen were stored in a refrigerator at 0°C, and fifteen were stored in a closed drawer at ambient temperatures of 20-25°C. At approximately three-day intervals, three samples were selected from each of the two storage sets for each level and analyzed.
Table 4.7.1
Storage Test at 9.40 ppb
|
time
(days) |
refrigerated storage
recovery (%) |
|
ambient storage
recovery (%) |
|
0
0
3
6
11
14
19 |
90.7
90.9
82.0
90.0
92.8
91.3
86.8 |
72.4
94.9
90.1
92.6
89.3
94.6
98.2 |
84.5
85.7
81.4
86.8
83.6
82.6
89.4 |
|
90.7
90.9
83.8
78.9
75.8
83.3
77.9 |
72.4
94.9
87.0
90.0
77.8
79.6
74.5 |
84.5
85.7
86.6
86.9
80.3
75.2
88.7 |
|
Table 4.7.2
Storage Test at 1.06 ppm
|
time
(days) |
refrigerated storage
recovery (%) |
|
ambient storage
recovery (%) |
|
0
0
3
7
10
15
20 |
96.2
95.5
94.8
102.4
98.9
97.0
99.6 |
97.8
95.8
92.7
99.2
97.6
97.8
98.4 |
95.8
97.2
93.5
100.7
97.2
99.1
99.6 |
|
96.2
95.5
95.3
98.7
96.3
99.5
97.6 |
97.8
95.8
93.2
97.6
96.9
98.1
98.4 |
95.8
97.2
94.2
98.1
97.1
99.3
98.1 |
|
4.8 Reproducibility
Reproducibility samples were prepared by sampling at 1 L/min from two different controlled test atmospheres, one at 9.40 ppb and the other at 1.06 ppm. Both atmospheres were at
approximately 80% RH and at room temperatures ranging from 22-26°C. Six samples for each target concentration were submitted to an SLTC service branch for analysis. The samples were stored for 63 and 58 days at 0°C before they were analyzed for the
10-ppb and 1-ppm target concentration samples respectively. One sample at the 9.40-ppb level was much lower than the other samples and is considered to be an outlier. It is suspected that there was a possible leak on the seal of the sample cassette and the manifold adapter used in the controlled test atmosphere generator. The rest of the samples did not deviate greater than the precisions of the overall procedure determined in Section 4.7, which are ±14.8% and ±10.1% for the
10-ppb and 1-ppm target concentrations respectively.
Table 4.8.1
Reproducibility Data for 10-ppb Samples
|
| sample |
ng reported |
ng expected |
percent |
deviation |
|
1
2
3
4
5
6 |
2497
2319
2406
2492
1880
2434 |
2657
2524
2732
2641
2780
2611 |
94.0
91.9
88.1
94.4
67.6
93.2 |
-6.0
-8.1
-11.9
-5.6
-32.4
-6.8 |
|
Table 4.8.2
Reproducibility Data for 1-ppm Samples
|
| sample |
µg reported |
µg expected |
percent |
deviation |
|
1
2
3
4
5
6 |
340.5
331.8
293.4
340.2
286.4
330.6 |
335.5
328.8
308.9
343.2
290.7
332.8 |
101.5
100.9
95.0
99.1
98.5
99.3 |
+1.5
+0.9
-5.0
-0.9
-1.5
-0.7 |
|
4.9 Extraction efficiency and stability of extracted samples
4.9.1 Extraction efficiency
The extraction efficiencies (EE) for the two levels were determined by injecting standards
onto sulfuric acid treated filters with amounts equivalent to 0.05 to 2 times the target
concentrations. The average extraction efficiencies over the working range of 0.5 to 2
times the target concentrations are 98.7% and 98.9% based on the 10-ppb and 1-ppm levels respectively.
Table 4.9.1.1
Extraction Efficiency Based on a 10-ppb Target Concentration
|
× target concn
mass spiked (ng) |
0.05×
157.4 |
0.1×
314.7 |
0.2×
629.5 |
0.5×
1574 |
1.0×
3147 |
2.0×
6295 |
|
| EE (%) |
98.2
96.1
96.7
96.4
101.5
97.2 |
98.4
99.0
97.7
97.0
96.4
96.3 |
96.0
96.4
97.0
95.3
96.1
97.1 |
98.4
98.6
98.8
99.0
99.0
98.9 |
98.2
98.3
98.1
98.3
98.0
97.9 |
98.6
98.7
100.4
99.3
99.0
99.0 |
| |
| mean |
97.7 |
97.5 |
96.3 |
98.8 |
98.1 |
99.2 |
|
Table 4.9.1.2
Extraction Efficiency Based on a 1-ppm Target Concentration
|
× target concn
mass spiked (µg) |
0.05×
15.74 |
0.1×
31.47 |
0.2×
62.95 |
0.5×
157.4 |
1.0×
314.7 |
2.0×
629.5 |
|
| EE (%) |
98.3
98.2
98.3
98.1
98.3
98.2 |
98.5
97.9
97.4
98.2
98.1
97.9 |
98.5
98.6
98.6
98.6
98.7
98.4 |
98.9
99.0
99.2
99.4
99.5
99.2 |
99.3
99.0
99.2
99.0
99.0
98.5 |
98.7
98.3
98.3
98.3
98.6
98.5 |
| |
| mean |
98.2 |
98.0 |
98.6 |
99.2 |
99.0 |
98.4 |
|
4.9.2 Stability of extracted and extracted/derivatized samples
The stability of extracted samples at the two target concentrations was investigated by
analyzing subsequent aliquots of the extracted samples 24 h after the samples were
initially extracted. Three of the extracted samples (the filters were still in the vials) were
refrigerated, and the other three were allowed to stand at room temperature on a
laboratory bench top for each target concentration. Aliquots of the stored extracts were
analyzed with fresh standards. The average percent change was +0.8% and +0.7% for
sample extracts that were refrigerated, and +0.3% and +0.3% for those stored at room
temperature at the 10-ppb and 1-ppm target concentrations respectively.
Table 4.9.2.1
Stability of Extracted Samples
at the 10-ppb Target Concentration
| | |
|
| | |
|
| extracts refrigerated |
|
extracts at room temperature |
initial
EE
(%) |
EE after
one day
(%) |
difference |
|
initial
EE
(%) |
EE after
one day
(%) |
difference |
|
98.2
98.3
98.1
98.2 |
98.8
99.0
99.3
(averages)
99.0 |
+0.6
+0.7
+1.2
+0.8 |
|
98.3
98.0
97.9
98.1 |
98.4
98.4
98.3
(averages)
98.4 |
+0.1
+0.4
+0.4
+0.3 |
|
Table 4.9.2.2
Stability of Extracted Samples
at the 1-ppm Target Concentration
| | |
|
| | |
|
| extracts refrigerated |
|
extracts at room temperature |
initial
EE
(%) |
EE after
one day
(%) |
difference |
|
initial
EE
(%) |
EE after
one day
(%) |
difference |
|
99.3
99.0
99.2
99.2 |
101.0
99.2
99.6
(averages)
99.9 |
+1.7
+0.2
+0.4
+0.7 |
|
99.0
99.0
98.5
98.8 |
99.4
98.7
99.3
(averages)
99.1 |
+0.4
-0.3
+0.8
+0.3 |
|
The stability of extracted and derivatized samples was investigated by reanalyzing the
target concentration samples 24 h after derivatization and initial analyses. After the
original analyses were performed, three vials were recapped with new septa while the
remaining three retained their punctured septa for each target concentration set. The
samples were reanalyzed with fresh standards. The average percent change was -0.4%
and -0.2% for samples that were resealed with new septa, and -0.1% and 0.0% for those
that retained their punctured septa for samples at the 1-ppb and
10-ppm target concentrations respectively.
Table 4.9.2.3
Stability of Extracted and Derivatized Samples
at the 10-ppb Target Concentration
| | |
|
| | |
|
| punctured septa replaced |
|
punctured septa retained |
initial
EE
(%) |
EE after
one day
(%) |
difference |
|
initial
EE
(%) |
EE after
one day
(%) |
difference |
|
98.2
98.3
98.1
98.2 |
98.2
97.7
97.6
(averages)
97.8 |
0.0
-0.6
-0.5
-0.4 |
|
98.3
98.0
97.9
98.1 |
98.0
98.0
98.1
(averages)
98.0 |
-0.3
0.0
+0.2
-0.1 |
|
Table 4.9.2.4
Stability of Extracted and Derivatized Samples
at the 1-ppm Target Concentration
| | |
|
| | |
|
| punctured septa replaced |
|
punctured septa retained |
initial
EE
(%) |
EE after
one day
(%) |
difference |
|
initial
EE
(%) |
EE after
one day
(%) |
difference |
|
99.3
99.0
99.2
99.2 |
98.7
99.1
99.1
(averages)
99.0 |
-0.6
+0.1
-0.1
-0.2 |
|
99.0
99.0
98.5
98.8 |
98.8
98.8
98.7
(averages)
98.8 |
-0.2
-0.2
+0.2
0.0 |
|
4.10 Qualitative analysis
A UV spectrum for benzalazine was obtained from a Hewlett-Packard 1050 Series programmable
variable wavelength detector by injecting a standard using the same conditions given in Section 3.5.1.
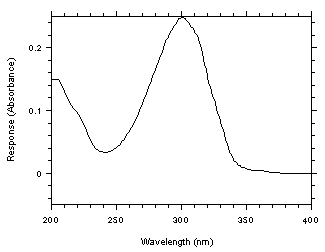
Figure 4.10. UV spectrum of benzalazine in 67/33, acetonitrile/water.
5. References
5.1. OSHA Analytical Methods Manual; Vol. 1, Publ. #4542, U.S. Department of Labor, Occupational Safety and Health Administration; OSHA Salt Lake Technical Center: Salt Lake City, UT, 1990; Method 20: Hydrazine; American Conference of Governmental Industrial Hygienists (ACGIH): Cincinnati, OH.
5.2. Suggs, H.J., L.J. Luskus and J.K. Herman, Am. Ind. Hyg. Assoc. J., 1980, 41, 879-883.
5.3. Documentation of the Threshold Limit Values and Biological Exposure Indices, 6th ed., on TLVs® and Other Occupational Exposure Values - 1995- CD-Rom, Publ. #9521W, American Conference
of Governmental Industrial Hygienists (ACGIH): Cincinnati, OH, 1995.
5.4. OSHA Analytical Methods Manual; Vol. 3, Publ. #4542, U.S. Department of Labor, Occupational Safety and Health Administration; OSHA Salt Lake Technical Center: Salt Lake City, UT, 1990; Method 57: 4,4'-Methylenedianiline; American Conference of Governmental Industrial Hygienists (ACGIH): Cincinnati, OH.
5.5. ibid. Method 65: Benzidine, 3,3'-Dichlorobenzidine,
2,4- and 2,6-Toluenediamine.
5.6. ibid. Method 71: o-Dianisidine,
4,4'-Methylenebis(o-chloroaniline), o-Tolidine.
5.7. ibid. Method 73: o-, m-, and r-Toluidine.
5.8. ibid. Method 78: Diphenylamine, N-Isopropylaniline.
5.9. ibid., Vol. 4, Method 87: m-, o-, and
r-Phenylenediamine.
5.10. ibid. Method 93: 4-Aminobiphenyl, 1-Naphthylamine and
2-Naphthylamine.
5.11. Elskamp, C.J. "OSHA Method 105: m-Xylylenediamine and
r-Xylylenediamine", OSHA Salt Lake Technical Center, unpublished, Salt Lake City, UT 84165-0200, December 1994.
5.12. Clayton, G.D. and F.E. Clayton, Eds. "Patty's Industrial Hygiene and Toxicology", 4th ed., Vol. 2, Part E; John Wiley & Sons, Inc.: New York, NY, 1994.
5.13. Lewis, R.J., Sr., Ed. "Sax's Dangerous Properties of Industrial Materials", 8th ed., Vol. 3; Van Nostrand Reinhold Co.: New York, NY, 1992.
|

