This article is part of the monograph on Children's Environmental Health and Disease Prevention Research.
Address correspondence to B. Eskenazi, School of Public Health, University of California, Berkeley, 140 Warren Hall, Berkeley, CA 94720-7360. Telephone: (510) 642-3496. Fax: (510) 642-5815. E-mail: eskenazi@uclink4.berkeley.edu
The authors thank M. O'Rourke for preliminary information on pesticide levels in Arizona children. We also thank G. Solomon for review of early drafts. Finally, we thank our collaborators in the Center who have contributed to parts of this review, including I. Tager, J. Leckie, V. Zartarian, T. Boyce, and M. Lipsett.
This research is jointly funded by the U.S. Environmental Protection Agency and the National Institute of Environmental Health Sciences award R826709-01-0.
Received 2 February 1999; accepted 7 April 1999.
Recent studies have demonstrated that home environments throughout the United States are commonly contaminated with pesticides, including organophosphate (OP), carbamate, organochlorine, pyrethroid, and herbicide compounds (1-12). Young children may be highly exposed to these pesticides because of their normal tendency to explore their environment orally, combined with their proximity to potentially contaminated floors, surfaces, and air. Physiologic characteristics of young children, such as high intake of food, water, and air per unit of body weight, may also increase their exposures (13). Because children are developmentally immature, they may also be at higher risk for adverse health effects (13). This paper reviews information on children's exposure to OP pesticides and potential adverse health effects. We also outline our planned research in Monterey County, California, to investigate exposures to children living in an agricultural area and possible effects on growth, neurobehavioral development, and respiratory disease.
Nationally, approximately 750-800 million pounds of conventional pesticides are used annually in agriculture, excluding sulfur, oils and repellants (14). Total conventional pesticide use, including home, structural, and other applications, averages about 1 billion pounds in the United States. During the mid-1990s, national pesticide use levels have been stable (15), although trends vary by region. In California, which has the largest agricultural output of all 50 states, approximately 200 million pounds of pesticidal active ingredient are used annually in agriculture (16). Pesticide use data for California suggest a trend of increasing use between 1991 and 1995 for production agriculture, postharvest treatment, structural fumigation, and landscape maintenance (16). These changes may be due, in part, to unique meteorologic and economic factors, including heavy rains, shifts to lower toxicity compounds that require higher volumes, and changes in planted acreage (16-18). Agricultural use of neurotoxic pesticides, including the OPs chlorpyrifos and diazinon, was also higher in 1995 than in 1991, most likely due to increased use on cotton, and to a lesser extent on oranges, alfalfa, apples, and broccoli (16,17). Overall, pesticide use in California appears to be stable or increasing, with annual fluctuations making it difficult to identify long-term trends.
Pesticide residue in food may also contribute to children's exposures. In response to concern about low-level exposure, the Food Quality Protection Act of 1996 (P.L. 104-170) (19) was unanimously passed by the U.S. Congress to address pesticide food safety issues raised by the seminal 1993 National Academy of Sciences report Pesticides in the Diets of Infants and Children (13). This report drew the public's attention to the specific vulnerability of children to many pesticides. The National Academy of Sciences committee found that current tolerances for pesticide levels in food are not health based and may not adequately protect children. Congress specifically directed the U.S. Environmental Protection Agency (U.S. EPA) to reevaluate food tolerances and establish health-based standards that account for children's unique sensitivity to environmental toxicants. The law requires the U.S. EPA to consider all nonoccupational sources of pesticide exposure, especially exposure to compounds with similar mechanisms of toxicity. The National Academy of Sciences recommended in 1993 that the U.S. EPA modify its decision-making process for setting pesticide tolerances to reflect the unique characteristics of the diets of infants and children and account also for all nondietary intake of pesticides (13). Findings from several small, cross-sectional studies (3-6,8,12) indicate that nondietary exposures to young children from residential contamination may be an important component of total pesticide exposure. Unfortunately, our knowledge about the actual levels of pesticide exposures of children from food and environmental exposures and their potential health effects is extremely limited.
National population-based surveys of pesticide urinary metabolites in adults indicate widespread exposure to pesticides, including organophosphates, carbamates, wood preservatives, and fungicides (20,21). For example, Hill et al. (20) detected chlorpyrifos, an OP pesticide, in 82% of 993 adults tested through the National Health and Nutrition Examination Survey, and found a 5-fold increase in the proportion of adults with levels over 5 µg/L compared to earlier surveys, suggesting increasing exposure in the general population. OPs are eliminated from the body after 3-6 days (22), so the widespread detection of these compounds indicates continuing exposure.
Biologic information on children's pesticide exposure is very limited. Hill et al. (23) reported detections of dichlorobenzene and wood preservatives in 96% and 100%, respectively, of 197 Arkansas children, whereas phenoxy herbicide metabolites were found in 20% of all samples. Preliminary results from the federal Agricultural Health Study indicate detectable pesticide residues in children's urine (8). Loewenherz et al. (24), working in Washington state, found that 44% of children of pesticide applicators and 27% of nonfarm, rural children had detectable OP residues. In preliminary data from Arizona, chlorpyrifos was detected in 100% of about 40 children > 6 years of age sampled in a population-based survey, and approximately 25% of 150 children 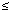 6 years of age sampled in an agricultural area. Detection limits in the second survey were higher (25). Comparison of these studies to each other and to data reported by Hill et al. (20) is difficult because of differences in detection limits, sample type (spot samples vs first morning void), and ages of participants. Overall, these studies suggest the potential for widespread low-level pesticide exposure in children and the need for population-based studies to establish norms.
6 years of age sampled in an agricultural area. Detection limits in the second survey were higher (25). Comparison of these studies to each other and to data reported by Hill et al. (20) is difficult because of differences in detection limits, sample type (spot samples vs first morning void), and ages of participants. Overall, these studies suggest the potential for widespread low-level pesticide exposure in children and the need for population-based studies to establish norms.
Pesticides enter children's bodies via dermal absorption, ingestion, and inhalation. Exposure in the home depends on the frequency, duration, and nature (i.e., dermal contact, hand-to-mouth behavior) of the child's interaction with contaminated media such as house dust. Children may have higher exposure to pesticides than other residents living in the same contaminated environment, in part because young children spend more of their time indoors at home (26,27). Thus, they are likely to spend more time in proximity to any pesticides present in their immediate environment. The importance of specific exposure-related behaviors, such as hand-to-mouth activity, will be age dependent, suggesting that consequent exposure levels and pathways will vary with age, as has been observed for lead exposure (28). For example, children younger than 6 months of age may receive their greatest exposures through breast milk or inhalation, but dermal absorption and ingestion may be the major pathway of exposure when children begin crawling and placing their hands on dusty surfaces and increasing their hand-to-mouth behavior. The level of exposure may continue to increase, given that the normal tendency of young children to explore their environment orally increases through 2 years of age. The actual dose to the child will depend on environmental concentrations and the efficiency of pesticide uptake for the different types of exposure routes, i.e., dermal contact versus ingestion. To date, direct observation and quantification of children's exposure-related activity patterns and their interaction with their environment is very limited. Time-activity analysis thus could provide information about age-specific exposure pathways.
To assess time-activity patterns, most researchers have preferred self-administered time diaries and interviews (29,30). However, these diaries are subject to inaccurate recall and thus have limited validity (31-33). Moreover, they fail to document microactivities such as dermal and hand-to-mouth contacts, which are important pathways of exposure in young children. Observational techniques are more detailed and accurate than conventional methods of questionnaires and diaries for recording such microscopic data (34-37). Leckie and Zartarian and co-workers (36-39) have successfully developed videotaping methodologies and video translation software to quantify children's activity patterns for dermal and nondietary contacts, and have piloted these techniques on 2- to 4-year-old children of Mexican American farmworkers in California (36). More extensive data collection is needed on children of various ages to assess the changing pathways and routes of exposure as children develop.
Nationally, an estimated 5 million farmworkers work on America's farms, including approximately 1 million California residents (40). A growing body of literature indicates that resident farm families, hired farmworkers, and their children are among those most highly exposed to pesticides (5,6,8,10,12,24,41-49). These studies suggest that farm children can be exposed by the same pathways as other children, namely through consumption of contaminated food, by household use of pesticides, as a result of drift from nearby agricultural applications, by contaminated breast milk from their farmworker mothers, by playing in the fields, and through pesticides tracked into their homes by their parents or other household members working in fields (5,6,8,10,12,24,41-43,50,51). For example, preliminary data from pilot studies conducted for the Agricultural Health Study in North Carolina and Iowa indicate elevated levels of recently applied pesticides in the food, homes, and bodies of farm families and their children (8,10,12,43,44). In a study of 88 children in the Yakima Valley, Washington, Loewenherz et al. (24) reported more frequent detection of OP metabolites in children of pesticide applicators compared to nonapplicators, particularly those living less than 200 ft from orchards. Trends of increasing exposure with decreasing age also suggested that child activity is an important exposure variable.
Simcox et al. (5) studied 59 families in the Yakima Valley, and compared levels of four organophosphate (OP) pesticides in the homes of hired farmworkers, families residing on farms, and nonagricultural families. Chlorpyrifos was detected in 95% of the homes. House dust concentrations were consistently higher for agricultural families than for nonagricultural families, and pesticide applicators tended to have higher house dust concentrations compared to nonapplicators. There was a 3-fold difference in median chlorpyrifos house dust concentration between farmworkers who did not directly handle pesticides and reference families of nonfarmworkers living in agricultural communities (median =172 ng/g for farmworker vs 53 ng/g for nonagricultural families).
Bradman et al. (6) conducted a pilot study of pesticide exposures to children of migrant farmworkers and nonfarmworkers living in a largely Latino community in California's Central Valley. Floor dust samples and child hand wipes were collected from the homes of 10 families, 5 of which had at least one resident farmworker. Higher levels of the OPs diazinon (maximum = 160 ppm), chlorpyrifos (maximum = 33 ppm), and malathion (maximum = 1.6 ppm) were found in house dust in farmworker homes (6). Residues of diazinon and chlorpyrifos were detected on the hands of two and three farmworker toddlers, respectively, who also lived in the homes with the highest dust concentrations. A preliminary risk assessment suggested that diazinon exposures in children could exceed the U.S. EPA Office of Pesticide Program's oral reference dose of 9E-5 mg/kg/day.
Overall, the findings from these studies suggest that inadvertent carry-homes of occupational pesticides are occurring and that contamination in the homes of farm families are likely to be higher than in other homes. Further, a significant source of exposure to farmworker families may derive from their residential proximity to fields.
Studies of the effects of pesticide exposure on children's health have been limited to those of birth defects, childhood cancer, and acute poisonings following ingestion. Several case-control studies have associated parental exposure to pesticides or pesticide use in the home with childhood brain tumors, leukemias and lymphomas, testicular cancers, and other cancers (52-55). Other studies have reported that parental exposure to pesticides or application of pesticides in the home is associated with certain birth defects including neural tube and other birth defects (56,57).
To date, only one small ecologic study has examined whether low-level chronic exposure of children to pesticides can lead to adverse health consequences (58). This study of Yaqui children in Mexico found that children 4-5 years of age (n = 33) living in an agricultural valley with presumably higher pesticide exposure had deficits in tests of stamina, coordination, recall, and ability to draw a person, compared to children (n = 17) living in the foothills where there was mostly ranching. This study, although suggestive of an effect of pesticides, is limited by small sample size, utilization of a convenience sample, the lack of individual exposure data, and no statistical control of potential confounders. At present, the only prospective study investigating pesticides and adverse health effects is the National Cancer Institute/U.S. EPA Agricultural Health Study, which is a large cohort study of cancer in midwestern and eastern farmers and their families (49). In spite of the paucity of information concerning the potential health effects in children of chronic low-level exposure to organophosphates, there is substantial evidence in developing rodents and limited evidence in adult humans who have been chronically exposed to OPs that low-level chronic exposure to organophosphates may affect neurologic functioning, neurodevelopment, and growth. Because OP exposure may cause dysregulation of the autonomic control of airways, it is biologically plausible that exposure may be related to the occurrence of asthma in children.
The primary effects of OP and carbamate acute exposure are on the parasympathetic, sympathetic, and central nervous system. These pesticides interfere with the metabolism of acetylcholine (ACh) by inhibiting the enzyme that hydrolyzes it, acetylcholinesterase (AChE) (59). ACh accumulates at the neuronal junctions, resulting in the continued stimulation and then suppression of neurotransmission to organs. ACh is the chemical transmitter of somatic motor neurons to skeletal muscle, postganglionic parasympathetic nerve fibers, preganglionic fibers of both sympathetic and parasympathetic nerves, and some fibers in the central nervous system. The accumulation of ACh at the motor nerves results in weakness, fatigue, muscle cramps, fasciculations, and muscular weakness of respiratory muscles. Accumulation at the autonomic ganglia results in increased heartbeat and blood pressure, pallor, and hypoglycemia. Accumulation of ACh at muscarinic receptors results in visual disturbances, tightness in the chest and wheezing due to bronchoconstriction and increased bronchial secretions, and increased salivation, lacrimation, sweating, peristalsis (resulting in nausea, vomiting, cramps, diarrhea), and urination. Central nervous system effects from ACh accumulation include anxiety, headache, confusion, convulsions, ataxia, depression of respiration and circulation, slurred speech, tremor, and generalized weakness (59,60). Carbamates, unlike OPs, do not irreversibly inhibit ACHE. Thus, their activity is quickly reversed after excretion of the pesticide (61). Pregnancy may pose a time of increased risk because plasma AChE activity is already reduced, at least during the first two trimesters (62,63).
The most frequent acute symptoms of OP poisoning in children include miosis, excessive salivation, nausea and vomiting, lethargy, muscle weakness, tachycardia, hyporeflexia, and hypertonia, and respiratory distress (60,64). Duration of symptoms depends on the dose, with the highest doses resulting in death. In one study, pneumonitis developed in about one-third of poisoned children (64). OP-induced delayed onset peripheral neuropathy (OPIDN), reported for adults, has not been reported in children.
Although no studies have examined the long-term sequelae of acute pesticide poisoning in children, some studies in adults suggest that there are residual effects. Neuropsychologic investigations of poisoned farmworkers, pest control workers, and industrial workers tested a number of months to years after acute exposure to various OP pesticides have revealed deficits in overall abstraction, verbal and visual attention, visual memory, visuomotor speed, sequencing, visuomotor problem solving, motor steadiness, motor dexterity, and fine motor speed (65-68). These workers report anxiety, depression, irritability, confusion, and impaired concentration and memory. On neurologic exam, lower vibrotactile sensitivity has been reported (68,69). High acute or subchronic exposures to OPs may also result in delayed neurotoxicity or OPIDN (59,70,71). OPIDN usually manifests 1 to 6 weeks after exposure and may result in moderate to severe peripheral neuropathies lasting months, years, or indefinitely (59).
Although there are no studies in children on the neuropsychologic effects of chronic pesticide exposure, small studies of chronic low-level exposures of farmers or pest control workers who had levels of AChE within normal limits found no differences in tests of their neurobehavioral functioning compared to those of unexposed controls (72) or in a pre/post exposure comparison (73). Results of other studies in adults indicate that there may be mild peripheral effects of chronic lower level exposure as indicated by slower reaction times (74), impaired proprioception (postural sway) (75), decreased conduction velocities in motor (median and peroneal) and sensory (median and sural) nerves (76), wider two-point discrimination (77), as well as some neurobehavioral effects such as increased anxiety (78), decreased visuomotor speed (65,79,80), and short-term verbal memory (79). Daily exposure to OPs that are insufficient to cause signs and symptoms of acute poisoning may also produce an influenza-type illness characterized by weakness, anorexia, and malaise (81). In chronic lower level exposures, depression of cholinesterase activity may be cumulative, and there is no predictable correlation between the severity of symptoms and the degree of cholinesterase inhibition.
There is a strong and growing body of evidence linking exposure to OP pesticides during gestation or the early postnatal period and neurodevelopmental effects in animals. These effects may be due to the direct impact of OPs on the cholinergic system of the fetus, although effects on cellular intermediates such as adenylyl cyclase (82) and altered DNA synthesis in the brain through noncholinergic mechanisms (83-85) have been hypothesized. Table 1 summarizes animal studies investigating different organophosphate pesticides, dosing regimes, and exposure routes and their impacts on the developing nervous system.



Chlorpyrifos
Neurobehavioral tests given postnatally found that animals exposed in utero demonstrated decreased balance (86), increased righting reflex time, and poorer cliff avoidance (87,88). When exposure occurred in the early postnatal period, there was a lowered threshold for convulsions (89) as well as increased gait abnormalities and tremors (90) and deficits in delayed alternation on mazes (91).
Some studies suggest that early gestation may be a critical period for the neurodevelopmental effects of certain pesticides. Muto et al. (86) studied the effects of exposure to chlorpyrifos in rats occurring both during gestation (gestation days 0-7 and 7-21) and the postnatal period. They reported that early prenatal exposures were more likely to result in poorer performance on the rotorod test than exposures during later gestation, which was in turn, more likely to result in deficits than those occurring postnatally.
A number of mechanisms have been proposed to explain the observed neurobehavioral effects in animals. Chanda and Pope (88) found that repeated exposure of rodents to low levels of chlorpyrifos during gestation was related to inhibited levels of AChE and downregulated muscarinic receptors in the fetal brain. Transient brain AChE inhibition also has been consistently reported in neonates postnatally exposed to chlorpyrifos (90-94). Other effects of chlorpyrifos that, in part, could explain the neurobehavioral impairment include decreased muscarinic receptor binding (90,94,95), altered brain RNA concentrations (96), and inhibition of brain DNA synthesis (84,85,97). For example, after treating rats on postnatal days 1-4 with a dosage that produced minimal AChE inhibition (1 mg/kg), Dam et al. (85) reported large deficits in DNA synthesis in the brain stem and forebrain, with lesser effects on the cerebellum. Similar deficits in DNA synthesis were observed after a single early postnatal exposure but at a slightly higher dose (97). Early postnatal exposure to chlorpyrifos (postnatal days 1-4 or 11-45) also altered RNA concentrations in the brain stem and forebrain of rats (96). By targeting RNA, the macromolecule that controls postmitotic processes of cell differentiation and growth, the chemical may evoke alterations in cell function and number in developing organisms (96).
The results of these studies indicate that OP pesticides could contribute to behavioral abnormalities in young animals by producing cellular deficits in the developing brain. Recently, Campbell et al. (84) concluded that regions rich in cholinergic projections, such as the brain stem and forebrain may be more affected than the less cholinergic regions such as the cerebellum. However, the maturational timetable of each region (brain stem then forebrain then cerebellum) may be an important factor in determining relative vulnerability. Nevertheless, there is reasonable evidence that even subtoxic exposure to chlorpyrifos during the critical period of brain development could produce cellular, synaptic, and neurobehavioral aberrations in animals (97).
Other OP Pesticides
OPs other than chlorpyrifos have been associated with lowered AChE in the brain of rodents exposed prenatally [bromophos (98), dichlorvos (99), dimethoate (100), methyl parathion (101), quinalphos (102)]. Studies in which animals were exposed early in the postnatal period to these other organophosphate pesticides have also reported inhibition of brain AChE [dichlorvos (103), diisopropylfluorophosphate (104), quinalphos (105), parathion (92,93,95,106)] and downregulation of muscarinic receptors [diisopropylfluorophosphate (104,107), parathion (95,106)]. In addition, evidence from a single in vitro study suggests that prenatal exposure to organophosphates could alter human fetal brain AChE levels. For example, Banerjee et al. (108) reported a dose-dependent inhibition of cerebellar AChE activity in human fetal brain cells (8-10 weeks gestation) treated with 5  10-11 - 5
10-11 - 5  10-8 M diisopropylfluorophosphate. Further research in rodents has found reductions in brain weight, most pronounced in the cerebellum and brainstem, following OP exposure during gestation [dichlorvos (109), trichlorfon (109-111)].
10-8 M diisopropylfluorophosphate. Further research in rodents has found reductions in brain weight, most pronounced in the cerebellum and brainstem, following OP exposure during gestation [dichlorvos (109), trichlorfon (109-111)].
Neurobehavioral deficits such as impaired maze performance [dichlorvos (99), diazinon (112)], decreased open-field activity [sumithion (113)], impaired locomotion or swimming [trichlofon (110), diazinon (112)], and reduced time on the rotorod test [diazinon (112), sumithion (113)] have also been associated with prenatal organophosphate exposure. In addition, permanent alterations in spontaneous motor behavior (i.e., locomotion, rearing, and total activity) have been observed in mice exposed to a single subsymptomal dose of diisopropylfluorophosphate early in the postnatal period (107).
Decreased Birth Weight and Altered Growth
A number of the animal studies reported above have demonstrated a decrease in birth weight or body weight in developing animals exposed to OPs. Anticholinesterase agents such as OPs may have a nonspecific regulatory effect on growth, perhaps by an influence on placental transport of nutrients (112,114) or by altering the activity and reactivity of the adenylyl cyclase signaling cascade, which would disrupt cell development in all areas of the body, not only those cholinergically regulated (82).
Muto et al. (86) reported lower body weight in rats exposed during the first 7 days of gestation to chlorpyrifos (0.03 mg/kg); with higher doses they found a decrease in the length of the limbs (0.1 and 0.3 mg/kg) and head circumference (0.3 mg/kg). Other studies have reported a decrease in pup weight (115) and a decrease in weight gain postnatally following exposure to chlorpyrifos (88) or diazinon (112) during gestation. Spyker and Avery (112) also reported lower birth weight and a slower rate of postnatal weight gain in mice exposed to diazinon (9 mg/kg) throughout gestation. In rats, low levels of two carbamates and a triazine herbicide administered postnatally interacted to increase thyroxine levels and alter levels of somatotropin, hormones that regulate growth (116).
Potential Respiratory Health Effects
Much of the animal literature reviewed here has focused on the central nervous system effects of organophosphate exposure. Because OP pesticides exert their pharmacologic effects through inhibition of AChE, both short- and long-term effects on autonomic regulation are prominent features in the toxicology of this class of pesticides (76,117). No previous work has addressed the autonomic sequelae of pesticide exposure per se, yet disorders of autonomic regulation may be one of the earliest and most sensitive measures of long-term physiologic effects of exposures in infants and young children.
Similarly, the parasympathetic nervous system provides the principal neural control of lung airway tone. There are considerable data indicating that dysregulation of both parasympathetic (cholinergic) and sympathetic autonomic control of airways, such as by pesticide exposure, may be important in the occurrence of asthma and its severity (118). Dysregulation of parasympathetic function, as measured by respiratory sinus arrhythmia, predicts the onset of wheezing in adults (119). Although there are few direct studies of the effects of OP and carbamate pesticide exposure on asthma risk, farmworkers' exposure to carbamate pesticides has been associated with the occurrence of asthma after adjustment for other relevant factors (120). Professional fumigators reportedly have an increased occurrence of allergy and asthma in parallel with a higher risk of a > 20% decrease in red blood cell AchE (121). Exposure to chlorpyrifos has also been associated with an increase in the occurrence of atopic conditions (122). Although none of these studies involved children, they raise the prospect that pesticide exposure could be important etiologic and morbidity-modifying factors in the occurrence of childhood asthma.
Tests on young rodents demonstrate a progressive decrease in susceptibility to OP pesticides with increasing age (13,123-125). In some cases, the lethal dose in immature animals is only 1% of the adult lethal dose (92,93,97). A study of rats found that animals 1 and 7 days of age tolerated only 4% and 17% of the adult dose, respectively (92,93,97). Seven-day-old rats were 2.3, 8.6, and 6.2 times more sensitive than adults to the acute toxicity of methyl parathion, parathion, and chlorpyrifos, respectively (92). In humans, children have had higher fatality rates than adults in several cases of OP poisoning (13).
Young animals may be more susceptible to the toxic effects of organophosphates due to lower activity of detoxifying enzymes such as paraoxonase that deactivate active OP metabolites (e.g., paraoxon, chlorpyrifos-oxon) (123,126-131). For example, Mortensen et al. (126) reported markedly lower plasma and liver chlorpyrifos-oxonase levels in neonate compared to adult rat tissue. They concluded that the higher sensitivity of young rats to acute chlorpyrifos toxicity may not be explained by increased sensitivity of the target enzyme, brain AChE, but it may be partially explained by a deficiency of chlorpyrifos-oxonase activity (126).
Although young animals are more sensitive than adults to the acute toxic effects of chlorpyrifos, some researchers have suggested that lower level chlorpyrifos exposures may produce more extensive neurobehavioral effects in the adult rat than in the neonate (94). In addition, more extensive changes have been found in cholinergic parameters in the maternal brain compared to the fetus or neonate (88). Developing animals also appear to recover more quickly from cholinesterase inhibition than the adult (92,98), and may be less susceptible to developing OPIDN (71). However, repeated low-dose chlorpyrifos exposure during gestation has been associated with persistent neurochemical and neurobehavioral changes in developing rodents (88).
In summary, young children may be especially vulnerable to pesticides because of the sensitivity of their developing organ systems combined with a limited ability to enzymatically detoxify these chemicals (13,123,126-131). According to the National Academy of Sciences (13), children's OP exposures are of special concern because "exposure to neurotoxic compounds at levels believed to be safe for adults could result in permanent loss of brain function if it occurred during the prenatal and early childhood period of brain development" (13). Because there is so little information available on the levels and routes of children's pesticide exposure, it is not feasible to conduct a risk assessment predicting the likelihood of adverse effects based on animal studies. Thus far, there are no data in children to support or refute the hypothesized health effects of chronic low-level pesticide exposure.
There is clearly a lack of information on the sources, pathways, and levels of pesticide exposures of children, and in particular, of those children at highest risk because they live in agricultural communities. Similarly, there is a dearth of information on whether low-level chronic exposure to pesticides is associated with adverse health effects. The goal of the Center for Children's Environmental Health Research at the University of California, Berkeley is to address these questions by conducting a longitudinal cohort study of approximately 500 pregnant women and their children who live in a rural agricultural community in the Salinas Valley of Monterey County, California. The specific aims of this Center for the Health Assessment of Mothers and Children of Salinas, or CHAMACOS (which means "little child" in Chicano Spanish), are to a) characterize OP exposure levels and pathways in pregnant women and their children; b) determine the predictors of OP levels in the body and home; c) describe the exposure-prone behavior of children at different developmental stages using time-activity analysis; and d) follow up the children to 3 years of age to determine whether exposure in utero and/or during the postnatal period is associated with poor neurodevelopment (assessed by tests of the central and autonomic nervous system), slower and stunted growth, and increased prevalence of respiratory symptoms and disease. Our ultimate goal is to involve community partners in planning, coordinating, and conducting an intervention to reduce pesticide exposures to young children in this agricultural community and to evaluate the efficacy and sustainability of the intervention. To accomplish our goals, we have established a multidisciplinary partnership comprised of farmworkers, health care providers, growers, journalists, scientists, educators, and representatives of community groups and state and county health and agricultural departments. We are hopeful that the results of this study will benefit this community and agricultural communities, in general, and will directly contribute to the information necessary for the implementation of the Food Quality Protection Act.
REFERENCES AND NOTES
1. Starr HG Jr, Aldrich FD, MacDougall WD, Mounce LM. Contribution of household dust to the human exposure to pesticides. Pestic Monit J 8:209-212 (1974).
2. Roinestad KS, Louis JB, Rosen JD. Determination of pesticides in indoor air and dust. J AOAC Int 76:1121-1126 (1993).
3. Lewis RG, Fortmann RC, Camann DE. Evaluation of methods for monitoring the potential exposure of small children to pesticides in the residential environment. Arch Environ Contam Toxicol 26:37-46 (1994).
4. Whitmore RW, Immerman FW, Camann DE, Bond AE, Lewis RG, Schaum JL. Non-occupational exposures to pesticides for residents of two U.S. cities. Arch Environ Contam Toxicol 26:47-59 (1994).
5. Simcox NJ, Fenske RA, Wolz SA, Lee IC, Kalman DA. Pesticides in household dust and soil: exposure pathways for children of agricultural families. Environ Health Perspect 103:1126-1134 (1995).
6. Bradman MA, Harnly ME, Draper W, Seidel S, Teran S, Wakeham D, Neutra R. Pesticide exposures to children from California's Central Valley: results of a pilot study. J Expo Anal Environ Epidemiol 7:217-234 (1997).
7. Buckley T, Liddle J, Ashley D, Paschal D, Burse V, Needham L, Akland G. Environmental and biomarker measurements in nine homes in the lower Rio Grande Valley: multimedia results for pesticides, metals, PAHs, and VOCs. Environmental International 23:705-732 (1997).
8. Steen W, Bond A, Mage D. Agricultural Health Study-Exposure Pilot Study Report. Research Triangle Park, NC:U.S. Environmental Protection Agency, 1997.
9. Colt JZ, SH. Camann, DE. Hartge, P. Comparison of pesticides and other compounds in carpet dust samples collected from used vacuum cleaner bags and from a high-volume surface sampler. Environ Health Perspect 106:721-724 (1998).
10. Camann D, Harding H, Clothier J, Kuchibhatla R, Bond A. Dermal and in-home exposure of the farm family to agricultural pesticides. In: Measurement of Toxic and Related Air Pollutants. Pittsburgh, PA:Air & Waste Management Association, 1995;548-554.
11. Camann D, MacIntosh D, Weker R, Hammerstrom K, Ryan P. Associations among pesticide and PAH concentrations in residential environmental measurements. In: Unpublished presentation at ISEA Annual Conference, 5-7 November 1997, Research Triangle Park, North Carolina.
12. Camann D, Akland G, Buckley J, Bond A, Mage D. Carpet dust and pesticide exposure of farm children. In: Unpublished presentation at International Society of Exposure Analysis Annual Meeting, 5 November 1997, Research Triangle Park, North Carolina.
13. National Research Council. Pesticides in the Diets of Infants and Children. Washington:National Academy Press, 1993.
14. Aspelin A. Pesticides Industry Sales and Usage, 1994 and 1995 Market Estimates. Washington: U.S. Environmental Protection Agency, 1997.
15. Aspelin A. Personal communication.
16. DPR. Pesticide Use Report, Annual 1995, Indexed by Chemical and by Crop. Sacramento, CA:Department of Pesticide Regulation, California Environmental Protection Agency, 1996.
17. Wilhoit L, Supkoff D, Steggall J, Braun A, Goodman C, Hobza B, Todd B, Lee M. An Analysis of Pesticide Use in California. Sacramento, CA:Department of Pesticide Regulation, California Environmental Protection Agency, 1998.
18. Liebman J, Brewer C, Bourque M, Katten A, Rice C, Campbell K. Rising Toxic Tide: Pesticide use in California, 1991-1995. San Francisco: Pesticide Action Network, 1997.
19. Food Quality Protection Act of 1996. U.S. Congress. Pub L No 104-170, 110 Stat 1489.
20. Hill RH Jr, Head SL, Baker S, Gregg M, Shealy DB, Bailey SL, Williams CC, Sampson EJ, Needham LL. Pesticide residues in urine of adults living in the United States: reference range concentrations. Environ Res 71:99-108 (1995).
21. Murphy RS, Kutz FW, Strassman SC. Selected pesticide residues or metabolites in blood and urine specimens from a general population survey. Environ Health Perspect 48:81-86 (1983).
22. Bradway D, Shafik T, Lores E. Comparison of cholinesterase activity, residue levels, and urinary metabolite excretion of rats exposed to organophosphorus pesticides. J Agric Food Chem 25:1353-1358 (1977).
23. Hill R, To T, Holler J, Fast D, Smith S, Needham L, Binder S. Residues of chlorinated phenols and phenoxy acid herbicides in the urine of Arkansas children. Arch Environ Contam Toxicol 18:469-474 (1989).
24. Loewenherz C, Fenske RA, Simcox NJ, Bellamy G, Kalman D. Biological monitoring of organophosphorus pesticide exposure among children of agricultural workers in central Washington State. Environ Health Perspect 105:1344-1353 (1997).
25. O'Rourke M. Personal Communication, 1998.
26. Silvers A, Florence BT, Rourke DL, Lorimor RJ. How children spend their time: a sample survey for use in exposure and risk assessments. Risk Anal 14:931-944 (1994).
27. CARB. Studies of Children's Activity Patterns. (Final Report) A733-149. Sacramento, CA: California Air Resources Board, 1991.
28. Clark C, Bornschein R, Succop P, Que Hee S, Hammond P, Peace B. Condition and type of housing as an indicator of potential environmental lead exposure and pediatric blood lead levels. Environ Res 38:46-53 (1985).
29. As D. Observing environmental behavior: the behavior setting. In: Behavioral Research in Environmental Design, Vol 8 (Michelson W, ed). Stroudsburg, PA:Dowden, Hutchinson, and Ross, 1975;280-300.
30. Michelson W. From Sun to Sun: Daily Obligations and Community Structure in the Lives of Employed Women and Their Families. Totowa, NJ:Rowman & Allanheld, 1985.
31. Ott W. Exposure estimates based on computer generated activity patterns. J Toxicol Clin Toxicol 21:97-128 (1983).
32. Ott MG, Teta MJ, Greenberg HL. Assessment of exposure to chemicals in a complex work environment. Am J Ind Med 16:617-630 (1989).
33. Stock T, Morandi M. A comparative evaluation of self-reported and independently-observed activity patterns in an air pollution health effects study. In: Unpublished presentation at Research Planning Conference on Human Activity Patterns, Environmental Research Center, Las Vegas, Nevada, 1989.
34. Wright HF. Recording and Analyzing Child Behavior with Ecological Data from an American Town. New York:Harper & Row, 1967.
35. Michelson W, Reed P. The time budget--behavioral research in environmental design. In: Behavioral Research Methods in Environmental Design (Michelson W, ed). Stroudsburg, PA:Dowden, Hutchinson & Ross, 1975;185-234.
36. Zartarian VG, Streicker J, Rivera A, Cornejo CS, Molina S, Valadez OF, Leckie JO. A pilot study to collect micro-activity data of two- to four-year-old farm labor children in Salinas Valley, California. J Expo Anal Environ Epidemiol 5:21-34 (1995).
37. Zartarian V, Ferguson A, Leckie J. Quantified dermal activity data from a four-child pilot field study. J Expo Anal Environ Epidemiol 7:543-552 (1997).
38. Zartarian V. DERM (Dermal Exposure Reduction Model): A Physical-Stochastic Model for Understanding Dermal Exposure to Chemicals. Palo Alto, CA:Stanford University, 1996.
39. Zartarian V, Ferguson A, Ong C, Leckie J. Quantifying videotaped activity patterns: video translation software and training methodologies. J Expo Anal Environ Epidemiol 7:535-542 (1997).
40. Mobed K, Gold EB, Schenker MB. Occupational health problems among migrant and seasonal farm workers. West J Med 157:367-373 (1992).
41. Grieshop J, Villanueva N, Stiles M. Wash day blues: secondhand exposure to agricultural chemicals. J Rural Health 10:247-257 (1994).
42. NIOSH. Report to Congress on Workers' Home Contamination Study Conducted Under the Workers' Family Protection Act. Cincinnati, OH: National Institute for Occupational Safety and Health, 1995.
43. Melnyk LJ, Berry MR, Sheldon LS. Dietary exposure from pesticide application on farms in the Agricultural Health Pilot Study. J Expo Anal Environ Epidemiol 7:61-80 (1997).
44. Shealy DB, Barr JR, Ashley DL, Patterson DG Jr, Camann DE, Bond AE. Correlation of environmental carbaryl measurements with serum and urinary 1-naphthol measurements in a farmer applicator and his family. Environ Health Perspect 105:510-513 (1997).
45. Musicco M, Sant M, Molinari S, Filippini G, Gatta G, Berrino F. A case-control study of brain gliomas and occupational exposure to chemical carcinogens: the risk to farmers. Am J Epidemiol 128:778-785 (1988).
46. Brown LM, Blair A, Gibson R, Everett GD, Cantor KP, Schuman LM, Burmeister LF, Van Lier SF, Dick F. Pesticide exposures and other agricultural risk factors for leukemia among men in Iowa and Minnesota. Cancer Res 50:6585-6591 (1990).
47. Richter ED, Chuwers P, Levy Y, Gordon M, Grauer F, Marzouk J, Levy S, Barron S, Gruener N. Health effects from exposure to organophosphate pesticides in workers and residents in Israel. Isr J Med Sci 28:584-598 (1992).
48. Moses M, Johnson ES, Anger WK, Burse VW, Horstman SW, Jackson RJ, Lewis RG, Maddy KT, McConnell R, Meggs WJ, et al. Environmental equity and pesticide exposure. Toxicol Ind Health 9:913-959 (1993).
49. Alavanja MC, Sandler DP, McMaster SB, Zahm SH, McDonnell CJ, Lynch CF, Pennybacker M, Rothman N, Dosemeci M, Bond AE, et al. The Agricultural Health Study. Environ Health Perspect 104:362-369 (1996).
50. Sonawane B. Chemical contaminants in human milk: an overview. Environ Health Perspect 103:197-205 (1995).
51. USDA. United States Department of Agriculture, Pesticide Data Program: Annual Summary Calendar Year 1997. Agricultural Marketing Service, 1997.
52. Buckley JD, Robison LL, Swotinsky R, Garabrant DH, LeBeau M, Manchester P, Nesbit ME, Odom L, Peters JM, Woods WG. Occupational exposures of parents of children with acute nonlymphocytic leukemia: a report from the Childrens Cancer Study Group. Cancer Res 49:4030-4037 (1989).
53. Blair A, Zahm SH, Pearce NE, Heineman EF, Fraumeni JF Jr. Clues to cancer etiology from studies of farmers. Scand J Work Environ Health 18:209-215 (1992).
54. Leiss JK, Savitz DA. Home pesticide use and childhood cancer: a case-control study [see comments]. Am J Public Health 85:249-252 (1995).
55. Kristensen P, Andersen A, Irgens LM, Bye AS, Vagstad N. Testicular cancer and parental use of fertilizers in agriculture. Cancer Epidemiol Biomarkers Prev 5:3-9 (1996).
56. Shaw GM, Wasserman CR, O'Malley CD. Maternal pesticide exposures as risk factors for orofacial clefts and neural tube defects. Am J Epidemiol 141(suppl 11):S3 (1995).
57. Blatter BM, Hermens R, Bakker M, Roeleveld N, Verbeek AL, Zielhuis GA. Paternal occupational exposure around conception and spina bifida in offspring. Am J Ind Med 32:283-291 (1997).
58. Guillette EA, Meza MM, Aquilar MG, Soto AD,Garcia IE. An anthropological approach to the evaluation of preschool children exposed to pesticides in Mexico. Environ Health Perspect 106:347-353 (1998).
59. Ecobichon D. Organophosphorus ester insecticides. In: Pesticides and Neurological Diseases (Ecobichon DJ, Joy RM, eds). Boca Raton, FL:CRC Press, 1994;171-250.
60. Sherman JD. Organophosphate pesticides--neurological and respiratory toxicity. Toxicol Ind Health 11:33-39 (1995).
61. Keifer MC, Mahurin RK. Chronic neurologic effects of pesticide overexposure. Occup Med 12:291-304 (1997).
62. Howard JK, East NJ, Chaney JL. Plasma cholinesterase activity in early pregnancy. Arch Environ Health 33:277-279 (1978).
63. Evans RT, O'Callahan J, Norman A. A longitudinal study of cholinesterase changes in pregnancy. Clin Chem 34:2249-2252 (1988).
64. Zwiener RJ, Ginsburg CM. Organophosphate and carbamate poisoning in infants and children Pediatrics 81:121-126 (1988). [published erratum appears in Pediatrics 81(5):683 (1988)].
65. Eskenazi B, Maizlish NA. Effects of occupational exposure to chemicals on neurobehavioral functioning. In: Medical Neuropsychology: The Impact of Disease on Behavior (Tarter RE, Thiel DHV, Edwards KL, eds). New YorkCity:Plenum Press, 1988;223-264.
66. Savage EP, Keefe TJ, Mounce LM, Heaton RK, Lewis JA, Burcar PJ. Chronic neurological sequelae of acute organophosphate pesticide poisoning. Arch Environ Health 43:38-45 (1988).
67. Rosenstock L, Keifer M, Daniell WE, McConnell R, Claypoole K. Chronic central nervous system effects of acute organophosphate pesticide intoxication. The Pesticide Health Effects Study Group [see comments]. Lancet 338:223-227 (1991).
68. Steenland K, Jenkins B, Ames RG, O'Malley M, Chrislip D, Russo J. Chronic neurological sequelae to organophosphate pesticide poisoning. Am J Public Health 84:731-736 (1994).
69. McConnell R, Keifer M, Rosenstock L. Elevated quantitative vibrotactile threshold among workers previously poisoned with methamidophos and other organophosphate pesticides. Am J Ind Med 25:325-334 (1994).
70. Abou-Donia MB. Organophosphorus ester-induced delayed neurotoxicity. Annu Rev Pharmacol Toxicol 21:511-548 (1981).
71. Abou-Donia MB, Lapodula DM. Mechanisms of organophosphorus ester-induced delayed neurotoxicity: type I and type II. Annu Rev Pharmacol Toxicol 30:405-440 (1990).
72. Rodnitzky RL. Occupational exposure to organophosphate pesticides: a neurobehavioral study. Arch Environ Health 30:98-103 (1975).
73. Maizlish N, Schenker M, Weisskopf C, Seiber J, Samuels S. A behavioral evaluation of pest control workers with short-term, low-level exposure to the organophosphate diazinon. Am J Ind Med 12:153-172 (1987).
74. Fiedler N, Kipen H. Chemical sensitivity: the scientific literature. Environ Health Perspect 105 (suppl 2):409-415 (1997).
75. Sack D, Linz D, Shukla R, Rice C, Bhattacharya A, Suskind R. Health status of pesticide applicators: postural stability assessments. J Occup Med 35:1196-1202 (1993).
76. Ruijten MW, Salle HJ, Verberk MM, Smink M. Effect of chronic mixed pesticide exposure on peripheral and autonomic nerve function. Arch Environ Health 49:188-195 (1994).
77. Beach JR, Spurgeon A, Stephens R, Heafield T, Calvert IA, Levy LS, Harrington JM. Abnormalities on neurological examination among sheep farmers exposed to organophosphorous pesticides. Occup Environ Med 53:520-525 (1996).
78. Levin HS, Rodnitzky RL. Behavioral effects of organophosphate in man. Clin Toxicol 9:391-403 (1976).
79. Chuwers P, Levy Y, Levy S, Grauer F, Barron S, Melnik Y, Richter E. Neurobehavioral effects in workers and residents exposed to organophosphates [conference abstract]. Public Health Rev 17:346 (1989/90).
80. Ames RG, Steenland K, Jenkins B, Chrislip D, Russo J. Chronic neurologic sequelae to cholinesterase inhibition among agricultural pesticide applicators. Arch Environ Health 50:440-444 (1995).
81. Morgan WK. The effects of particles, vapours, fumes and gases. Eur J Respir Dis Suppl 123:7-12 (1982).
82. Song X, Seidler F, Saleh J, Zhang J, Padilla S, Slotkin T. Cellular mechanisms for developmental toxicity of chlorpyrifos: targeting the adenylyl cyclase signaling cascade. Toxicol Appl Pharmacol 145:158-174 (1997).
83. Song X, Violin JD, Seidler FJ, Slotkin TA. Modeling the developmental neurotoxicity of chlorpyrifos in vitro: macromolecule synthesis in PC12 cells. Toxicol Appl Pharmacol 151:182-191 (1998).
84. Campbell C, Seidler FJ, Slotkin TA. Chlorpyrifos interferes with cell development in rat brain regions. Brain Res Bull 43:179-189 (1997).
85. Dam K, Seidler FJ, Slotkin TA. Developmental neurotoxicity of chlorpyrifos: delayed targeting of DNA synthesis after repeated administration. Brain Res Dev Brain Res 108:39-45 (1998).
86. Muto MA, Lobelle F Jr, Bidanset JH, Wurpel JN. Embryotoxicity and neurotoxicity in rats associated with prenatal exposure to DURSBAN. Vet Hum Toxicol 34:498-501 (1992).
87. Chanda SM, Harp P, Liu J, Pope CN. Comparative developmental and maternal neurotoxicity following acute gestational exposure to chlorpyrifos in rats. J Toxicol Environ Health 44:189-202 (1995).
88. Chanda SM, Pope CN. Neurochemical and neurobehavioral effects of repeated gestational exposure to chlorpyrifos in maternal and developing rats. Pharmacol Biochem Behav 53:771-776 (1996).
89. Wurpel JN, Hirt PC, Bidanset JH. Amygdala kindling in immature rats: proconvulsant effect of the organophosphate insecticide-chlorpyrifos. Neurotoxicology 14:429-436 (1993).
90. Moser VC, Padilla S. Age- and gender-related differences in the time course of behavioral and biochemical effects produced by oral chlorpyrifos in rats. Toxicol Appl Pharmacol 149:107-119 (1998).
91. Stanton M, Mundy W, Ward T, Dulchinos V, Barry C. Time-dependent effects of acute chlorpyrifos administration on spatial delayed alternation and cholinergic neurochemistry in weanling rats. Neurotoxicology 15:201-208 (1994).
92. Pope C, Chakraborti T, Chapman M, Farrar J, Arthun D. Comparison of in vivo cholinesterase inhibition in neonatal and adult rats by three organophosphorothioate insecticides. Toxicology 68:51-61 (1991).
93. Pope CN, Chakraborti TK. Dose-related inhibition of brain and plasma cholinesterase in neonatal and adult rats following sublethal organophosphate exposures. Toxicology 73:35-43 (1992).
94. Chakraborti TK, Farrar JD, Pope CN. Comparative neurochemical and neurobehavioral effects of repeated chlorpyrifos exposures in young and adult rats. Pharmacol Biochem Behav 46:219-224 (1993).
95. Stamper CR, Balduini W, Murphy SD, Costa LG. Behavioral and biochemical effects of postnatal parathion exposure in the rat. Neurotoxicol Teratol 10:261-266 (1988).
96. Johnson DE, Seidler FJ, Slotkin TA. Early biochemical detection of delayed neurotoxicity resulting from developmental exposure to chloropyrifos. Brain Res Bull 45:143-147 (1998).
97. Whitney KD, Seidler FJ, Slotkin TA. Developmental neurotoxicity of chlorpyrifos: cellular mechanisms. Toxicol Appl Pharmacol 134:53-62 (1995).
98. Santhoshkumar P, Shivanandappa T. Differential in vivo inhibition of the foetal and maternal brain acetylcholinesterase by bromophos in the rat. Neurotoxicol Teratol 16:227-232 (1994).
99. Schulz H, Nagymajtenyi L, Desi I. Life-time exposure to dichlorvos affects behaviour of mature rats. Hum Exp Toxicol 14:721-726 (1995).
100. Srivastava MK, Raizada RB. Development effect of technical dimethoate in rats: maternal and fetal toxicity evaluation. Indian J Exp Biol 34:329-333 (1996).
101. Gupta RC, Rech RH, Lovell KL, Welsch F, Thornburg JE. Brain cholinergic, behavioral, and morphological development in rats exposed in utero to methylparathion. Toxicol Appl Pharmacol 77:405-413 (1985).
102. Srivastava MK, Raizada RB, Dikshith TS. Fetotoxic response of technical quinalphos in rats. Vet Hum Toxicol 34:131-133 (1992).
103. Maslinska D, Lewandowska I, Prokopczyk J. Effect of prolonged acetylcholinesterase inhibition on postnatal brain development in rabbit. I:Level of serotonin in different brain regions. Acta Neuropathol Suppl 7:52-55 (1981).
104. Balduini W, Cimino M, Reno F, Marini P, Princivalle A, Cattabeni F. Effects of postnatal or adult chronic acetylcholinesterase inhibition on muscarinic receptors, phosphoinositide turnover and m1 mRNA expression. Eur J Pharmacol 248:281-288 (1993).
105. Gupta A, Gupta A, Shukla GS. Effects of neonatal quinalphos exposure and subsequent withdrawal on free radical generation and antioxidative defenses in developing rat brain. J Appl Toxicol 18:71-77 (1998).
106. Veronesi B, Pope C. The neurotoxicity of parathion-induced acetylcholinesterase inhibition in neonatal rats. Neurotoxicology 11:609-626 (1990).
107. Ahlbom J, Fredriksson A, Eriksson P. Exposure to an organophosphate (DFP) during a defined period in neonatal life induces permanent changes in brain muscarinic receptors and behaviour in adult mice. Brain Res 677:13-19 (1995).
108. Banerjee J, Ghosh P, Mitra S, Ghosh N, Bhattacharya S. Inhibition of human fetal brain acetylcholinesterase: marker effect of neurotoxicity. J Toxicol Environ Health 33:283-290 (1991).
109. Mehl A, Schanke TM, Johnsen BA, Fonnum F. The effect of trichlorfon and other organophosphates on prenatal brain development in the guinea pig. Neurochem Res 19:569-74 (1994).
110. Berge GN, Nafstad I, Fonnum F. Prenatal effects of trichlorfon on the guinea pig brain. Arch Toxicol 59:30-35 (1986).
111. Hjelde T, Mehl A, Schanke TM, Fonnum F. Teratogenic effects of trichlorfon (Metrifonate) on the guinea-pig brain. Determination of the effective dose and the sensitive period. Neurochem Int 32:469-477 (1998).
112. Spyker JM, Avery DL. Neurobehavioral effects of prenatal exposure to the organophosphate Diazinon in mice. J Toxicol Environ Health 3:989-1002 (1977).
113. Lehotzky K, Szeberenyi MJ, Kiss A. Behavioral consequences of prenatal exposure to the organophosphate insecticide sumithion. Neurotoxicol Teratol 11:321-324 (1989).
114. Fant ME, Harbison RD, Harrison RW. Glucocorticoid uptake into human placental membrane vesicles. J Biol Chem 254:6218-6221 (1979).
115. Breslin W, Liberacki A, Dittenber D, Quast J. Evaluation of the developmental and reproductive toxicity of chlorpyrifos in the rat. Fundam Appl Toxicol 29:119-130 (1996).
116. Porter WP, Green SM, Debbink NL, Carlson I. Groundwater pesticides: interactive effects of low concentrations of carbamates aldicarb and methomyl and the triazine metribuzin on thyroxine and somatotropin levels in white rats. J Toxicol Environ Health 40:15-34 (1993).
117. Eyer P. Neuropsychopathological changes by organophosphorus compounds--a review. Hum Exp Toxicol 14:857-864 (1995).
118. Barnes PJ. Is asthma a nervous disease? The Parker B. Francis Lectureship. Chest 107:119S-125S (1995).
119. Sparrow D, GT OC, Basner RC, Rosner B, Weiss ST. Predictors of the new onset of wheezing among middle-aged and older men. The Normative Aging Study. Am Rev Respir Dis 147:367-371 (1993).
120. Senthilselvan A, McDuffie HH, Dosman JA. Association of asthma with use of pesticides. Results of a cross-sectional survey of farmers. Am Rev Respir Dis 146:884-887 (1992).
121. Garry V, Kelly J, Sprafka J, Edwards S, Griffith J. Survey of health and use characterization of pesticide appliers in Minnesota. Arch Environ Health 49:337-343 (1994).
122. Thrasher JD, Madison R, Broughton A. Immunologic abnormalities in humans exposed to chlorpyrifos: preliminary observations. Arch Environ Health 48:89-93 (1993).
123. Benke GM, Murphy SD. The influence of age on the toxicity and metabolism of methyl parathion and parathion in male and female rats. Toxicol Appl Pharmacol 31:254-269 (1975).
124. Brodeur J, DuBois K. Comparison of acute toxicity of anticholinesterase insecticides to weanling and adult male rats. Proc Soc Exp Biol Med 114:509-511 (1963).
125. Lu F, Jessup D, Lavallee A. Toxicity of pesticides in young versus adult rats. Food Cosmet Toxicol 3:591-596 (1965).
126. Mortensen SR, Chanda SM, Hooper MJ, Padilla S. Maturational differences in chlorpyrifos-oxonase activity may contribute to age-related sensitivity to chlorpyrifos. J Biochem Toxicol 11:279-287 (1996).
127. Gagne J, Brodeur J. Metabolic studies on the mechanisms of increased susceptibility of weanling rats to parathion. Can J Physiol Pharmacol 50:902-915 (1972).
128. Murphy S. Toxicity and hepatic metabolism of organophosphate insecticides in developing rats. Banbury Report 11:125-136 (1982).
129. Atterberry TT, Burnett WT, Chambers JE. Age-related differences in parathion and chlorpyrifos toxicity in male rats: target and nontarget esterase sensitivity and cytochrome P450-mediated metabolism. Toxicol Appl Pharmacol 147:411-418 (1997).
130. Davies HG, Richter RJ, Keifer M, Broomfield CA, Sowalla J, Furlong CE. The effect of the human serum paraoxonase polymorphism is reversed with diazoxon, soman and sarin. Nat Genet 14:334-336 (1996).
131. Grossman J. What's hiding under the sink: dangers of household pesticides [News]. Environ Health Perspect 103:550-554 (1995).
132. Crowder LA, Lanzaro GC, Whitson RS. Behavioral effects of methyl parathion and toxaphene exposure in rats. J Environ Sci Health [B] 15:365-378 (1980).
133. Pope AM, Heavner JE, Guarnieri JA, Knobloch CP. Trichlorfon-induced congenital cerebellar hypoplasia in neonatal pigs. J Am Vet Med Assoc 189:781-783 (1986).
134. Clemens GR, Hartnagel RE, Bare JJ, Thyssen JH. Teratological, neurochemical, and postnatal neurobehavioral assessment of METASYSTOX-R, an organophosphate pesticide in the rat. Fundam Appl Toxicol 14:131-143 (1990).
135. Nagymajtenyi L, Schulz H, Desi I. Changes in EEG of freely-moving rats caused by three-generation organophosphate treatment. Arch Toxicol Suppl 17:288-294 (1995).
Last Updated: May 17, 1999







