Address correspondence to A. Nel, Department of Medicine, UCLA School of Medicine, 52-175 CHS, 10833 Le Conte Avenue, Los Angeles, CA 90095 USA. Telephone: (310) 825-6620. E-mail: anel@mednet.ucla.edu
This study was supported by the National Institute of Environmental Health Sciences (grant RO1-ES10553) and the Southern California Particle Center and Supersite, funded by the U.S. Environmental Protection Agency (STAR award R82735201) and the California Air Resources Board (grant 98-316).
This manuscript has not been subjected to the U.S. EPA peer and policy review.
Received 18 September 2002; accepted 16 December 2002.
Epidemiologic studies have shown associations between ambient air particulate matter (PM) and adverse health outcomes, including increased mortality, emergency room visits, and time lost from work and school [Dockery et al. 1993; Health Effects Institute (HEI) 2002; Samet et al. 2000; Wichmann et al. 2000]. The underlying toxicologic mechanisms by which air pollutant particles induce adverse health effects are of intense scientific interest and have been earmarked as a key scientific priority by the National Academy of Sciences [National Research Council (NRC) 1998]. This includes a call for research on the physicochemical properties that promote particle toxicity (NRC 1998). PM with aerodynamic diameter < 2.5 µm (PM
2.5) is currently regulated by the U.S. Environmental Protection Agency. Within that spectrum of particle sizes, ultrafine particles (UFPs), defined as having an aerodynamic diameter < 0.1 µm, may have a central role in health effects of PM (Oberdörster and Utell 2002; Samet et al. 2000). Primary UFPs are formed during gas-to-particle conversion or during incomplete fuel combustion (HEI 2002). Due to their small size, high number concentration, and relatively large surface area per unit mass, UFPs have unique characteristics, including increased adsorption of organic molecules and enhanced ability to penetrate cellular targets in the lung and systemic circulation (Frampton 2001; HEI 2002; Nemmar et al. 2002; Oberdörster 1996; Utell and Frampton 2000).
Particle composition may also be critical in PM toxicity. We are interested in organic PM compounds because organic extracts made from diesel exhaust particles (DEPs) mimic intact particles in their ability to form reactive oxygen species (ROS) (Hiura et al. 1999, 2000; Kumagai et al. 1997; Nel et al. 1998). One of the major advances in PM research has been the recognition that the organic and metal PM components can induce proinflammatory effects in the lung due to their ability to cause oxidative stress (Kumagai et al. 1997; Nel et al. 1998, 2001; Saldiva et al. 2002). Quinones present in PM can act as catalysts to produce ROS directly and may be key compounds in PM-based oxidative stress (Monks et al. 1992; Penning et al. 1999). PAHs can induce oxidative stress indirectly, through biotransformation by cytochrome P450, expoxide hydrolase, and dihydrodiol dehydrogenase to generate redox active quinones (Penning et al. 1999). The involvement of quinones and PAHs was confirmed by demonstrating that compounds present in aromatic and polar fractions of DEP extracts mimic the pro-oxidative effects of intact particles in bronchial epithelial cells and macrophages (Li et al. 2000, 2002b). DEPs also induce cytochrome P450 1A1 induction in bronchial epithelial cells (Bonvallot et al. 2001). Animal and human experiments confirm that DEPs and PAHs derived from DEPs promote allergic airway inflammation and cytochrome P450 1A1 induction in the lungs of exposed mice (Miyabara et al. 1998; Nel et al. 1998; Takano et al. 2002; Tsien et al. 1997). Epidemiologic studies have also shown an association between PM exposure and asthma exacerbation (Nemmar et al. 2002; Penttinen et al. 2001; Utell and Frampton 2000).
The Versatile Aerosol Concentration Enrichment System (VACES), which uses three parallel sampling lines to collect concentrated ambient coarse, fine, and ultrafine particles for biological analysis, is now available for use in toxicologic studies aimed at identifying the relative toxicity of the different particle sizes (Kim et al. 2001a, 2001b). This technology enables us to probe the relationship between particle size, chemical composition, and toxicity (Li et al. 2002a). These concentrators are mobile and can be used to test hypotheses about particle toxicity in the Los Angeles basin in California. Concentrated air particulates (CAPs) of different sizes were collected to study their oxidative stress effects and subcellular localization in cultured macrophages and epithelial cells. We demonstrate that UFPs are more potent than fine (< 2.5 µm) or coarse (2.5-10 µm) particles toward inducing oxidative stress. This effect may be explained by adsorbed chemicals (organics and metals) capable of generating ROS and the ability of UFPs to localize in mitochondria.
Ambient particle collection. Ambient coarse particles (2.5-10 µm), fine plus ultrafine particles (< 2.5 µm), and UFPs (< 0.15 µm) were collected in the Los Angeles basin during November 2001-March 2002 using the VACES (Kim et al. 2001a, 2001b). Coarse particles were concentrated using a single nozzle virtual impactor, while fine and ultrafine particles were concentrated by drawing air samples through two parallel lines, using 2.5 µm and 0.15 µm cut-point preimpactors, respectively, to remove larger-sized particles. These particles are drawn through a saturation-condensation system that grows particles to 2-3 µm droplets, which are subsequently concentrated by virtual impaction. Highly concentrated particle suspensions were obtained by connecting the VACES output to a sterilized liquid impinger (BioSampler; SKC West Inc., Fullerton, CA) (Willeke et al. 1998). Aerosols were collected using ultrapure (Milli-Q; Millipore Corp., Bedford, MA) deionized water (resistivity 18.2 megaohm; total organic compounds < 10 ppb; particle-free; bacteria < 1 colony forming unit/mL) as the collection medium. The concentration enrichment process does not alter the physical, chemical, and morphologic properties of the particles (Kim et al. 2001a, 2001b). We determined the total amount of particulate loading in the collection medium by multiplying the ambient concentration of each PM mode by the total air sample volume collected by each VACES line. The particle concentration in the aqueous medium was then calculated by dividing the particle loading by the total volume collected in that time period. Five sample sets were collected, two at the University of Southern California (USC), and three at Claremont. USC is a typical urban site located 3 km south of downtown Los Angeles. This is a site in which aerosols are mostly generated from fresh vehicular emissions. Claremont is a receptor site approximately 45 km east (i.e., downwind) of downtown Los Angeles. In that location, ambient PM originates mostly from advection of polluted air parcels originally emitted in urban Los Angeles, after "aging" in the atmosphere for a few hours, as well as from secondary photochemical processes.
Particle chemical analysis. Samples were collected on Teflon and quartz filters with a Micro Orifice Uniform Deposit Impactor (MOUDI; MSP Corporation, Shoreview, MN) for chemical analysis (Li et al. 2002a). We used Teflon filters to determine the metal and trace element content by X-ray fluorescence and quartz filters to determine the organic carbon (MnO2-catalyzed CO2 formation), sulfate (ion chromatography), and nitrate (ion chromatography) contents (Li et al. 2002a). PAH content for each CAPs set was determined by an HPLC-fluorescence method that detects a signature group of 16 PAHs (Li et al. 2002a).
Cellular stimulation and heme oxygenase 1 (HO-1) immunoblotting. We used two cell lines in the study: RAW 264.7 and BEAS-2B. RAW 264.7 is a murine macrophage cell line that mimics the oxidative stress response of pulmonary alveolar macrophages in response to DEP exposure (Hiura et al. 1999, 2000; Li et al. 2002b). BEAS-2B is a transformed human bronchial epithelial cell line, which mimics the oxidative stress response of primary bronchial epithelial cells (Li et al. 2002b). For RAW 264.7 culture, particle suspensions were reconstituted with Dulbecco's Modified Eagle's Medium powder, a culture medium component that rapidly dissolves in deionized water. This culture medium was further replenished with 10% fetal calf serum and a 1:200 dilution of penicillin/streptomycin/amphotericin B (Li et al. 2002a). For BEAS-2B cells, particle suspensions were made up in hormonally defined F12 medium (Kawasaki et al. 2001). After incubating cells for 16 hr, we used 100 µg of lysate protein for HO-1 immunoblotting (Li et al. 2000, 2002a, 2002b). Densitometric analysis was performed on a laser Personal Densitometer SI using ImageQuant software (both from Amersham Biosciences, Piscataway, NJ).
GSH/GSSG assay. Total glutathione and oxidized glutathione (GSSG) were measured in a glutathione reductase recycling assay (Tietze 1969). We calculated the amount of total glutathione and GSSG in the samples from the standard curves. The amount of reduced glutathione (GSH) was calculated by subtracting the amount of GSSG from that of the total glutathione.
DTT assay. The dithiothreitol (DTT) assay quantitatively measures the formation of ROS by quinone catalysis (Kumagai et al. 2002). In the presence of quinones, 1 mol DTT + 2 mol O2 generate 1 mol DTT-disulfide + 2O2·-
a: Q + DTT 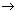 semi-Q + DTT-thiyl
semi-Q + DTT-thiyl
b: Q + DTT-thiyl 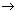 semi-Q
semi-Q
+ DTT-disulfide
c: 2 semi-Q + 2O2 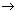 2Q ± 2O2·-
2Q ± 2O2·-
Net reaction: DTT + 2O2 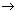 DTT-disulfide
DTT-disulfide
+ 2O2·-
The loss of DTT is followed by its reaction with 5,5´-dithiobis-(2-nitrobenzoic acid) (DTNB), which is converted to 5-mercapto-2-nitrobenzoic (Kumagai et al. 2002). We incubated the PM sample (5-50 µg/mL) with 10 µM DTT in a Tris buffer at pH 8.9 for 10-90 min. Aliquots of the incubation mixture were transfered to the DNTB solution and the optical density read at 412 nm.
Electron microscopy. We performed electron microscopy as previously described (Yang et al. 1987). Thin sections were cut with a Reichert-Jung ultracut and ultramicrotome (Leica, Stuttgart, Germany). Copper grids were stained with lead citrate and uranyl acetate and photographed in a Hitachi electron microscope (Hitachi Instrument Inc., Tokyo, Japan).
Particulate organic carbon and PAH content. To determine whether there is a link between UFP composition and biological effects, CAPs were collected at two outdoor sites in the Los Angeles basin with the VACES. Chemical analysis of the CAPs indicate that UFPs have a significantly higher organic (
p < 0.01) and elemental carbon ((
p < 0.001) content than fine plus ultrafine (designated "fine") or coarse particles (Table 1). Coarse and fine particles had a higher metal content than UFPs (Table 1). PAH content for each set of CAPs was determined and averaged for both collection sites; there was a statistically significant difference in total PAH content in UFPs compared to fine (
p = 0.04) and coarse (
p = 0.03) PM (Figure 1A). The PAH content of UFPs at USC was significantly higher than the PAH content at Claremont (Figure 1A), which reflects the fact that particles collected at the source site (USC) are derived from primary emissions that are far more abundant in the urban areas of Los Angeles than in receptor areas.

|
Figure 1. Correlation of PAH content with ROS formation. (A) PAH content for each set of CAPs determined by HPLC-fluorescence; values shown are mean ± SEM for Claremont (n = 3) and USC (n = 2). (B) In vitro electron transfer capacity of CAPs measured by a colorometric assay that distinguishes oxidized from reduced DTT (Kumagai et al. 2002). The mean was calculated for three separate measurements; SEM < 0.1. (C) Linear regression analysis demonstrating the correlation between PAH content and 15 DTT data points (5 sites  3 samples/site); r2 = 0.98. Inset: With the highest point removed, r2 remains significant at 0.86. 3 samples/site); r2 = 0.98. Inset: With the highest point removed, r2 remains significant at 0.86. |

The DTT assay of ROS formation by particles of varying size. The ability of PM to generate ROS was assessed with the DTT assay. Quinones with appropriate redox potentials can transfer electrons from DTT to oxygen (Kumagai et al. 2002). We used this reaction to determine the ability of PM to generate ROS in vitro. The DTT assay demonstrated that UFPs had significantly higher redox activity than fine and coarse PM (Figure 1B). Averaging of the data revealed that the redox cycling capacity of UFPs was 21.7- and 8.6-fold greater than coarse and fine PM, respectively (Figure 1B). Regression analysis of the DTT assay and PAH content showed a correlation coefficient (r2) of 0.98, suggesting the electron transfer capacity of CAPs is consistent with their organic chemical content (Figure 1C).
|

Figure 2. Induction of oxidative stress and HO-1 expression. (A) HO-1 expression in RAW 264.7 cells exposed to CAPs for 16 hr. (B) HO-1 expression in BEAS-2B cells treated with CAPs (Claremont Mar 02) for 16 hr. (C) Effects of CAPs (Claremont Jan 02) on the intracellular GSH/GSSG ratio in RAW 264.7 cells after 16 hr exposure; GSH/GSSG values shown are mean ± SEM from two separate experiments, with duplicate measurements per experiment (Tietze 1969). (D) Regression analysis demonstrating the correlation between in vitro redox activity of CAPs and HO-1 induction (15 data points); r2 = 0.97. Inset: After removal of the highest data point, r2 = 0.81.
|
|

Figure 3. Electron micrographs demonstrating effects of different sized particles in RAW 264.7 cells treated with USC-Jan 02 CAPs for 16 hr. (A) and (B) Untreated RAW 264.7 cells. (C) and (D) RAW 264.7 cells exposed to coarse particles. (E) and (F) RAW 264.7 cells exposed to fine particles. (G) and (H) RAW 264.7 cells exposed to UFPs. Notice damage to cristae as well as the presence of particles (P) inside mitochondria (M) in UFP- or fine + UFP-exposed cells.
|
Particle chemical composition and oxidative stress. Quinones and other redox-active compounds present in PM generate ROS and oxidative stress (Kumagai et al. 1997; Nel et al. 1998). We have demonstrated that DEP-induced oxidative stress generates hierarchical effects in pulmonary alveolar macrophages and bronchial epithelial cells (Li et al. 2002a, 2002b). Low levels of oxidative stress activate antioxidant defenses, whereas higher levels of oxidative stress lead to proinflammatory and cytotoxic effects (Li et al. 2002a, 2002b). An example of an antioxidant response is HO-1 expression via the antioxidant response element in its promoter (Choi and Alam 1996; Li et al. 2000). Utilizing an immunoblotting technique to assess HO-1 expression in RAW 264.7 cells, UFPs were more potent than fine or coarse particles (Figure 2A). Densitometric analysis demonstrated significantly higher HO-1 expression in ultrafine over fine (p = 0.001) and coarse (p = 0.001) particles, respectively. The increased potency of UFPs was seen for all CAPs collections (data not shown).
Sufficient CAPs were collected in March 2002 to study HO-1 expression in the bronchial epithelial cell line, BEAS-2B, in parallel with RAW 264.7. The BEAS-2B response mimics the DEP-induced oxidative stress response in human bronchial epithelial cells (Li et al. 2002b). Immunoblot analysis shows that UFPs but not coarse or fine particles induce HO-1 expression in BEAS-2B cells (Figure 2B). To demonstrate that these effects reflect differences in the level of oxidative stress, we compared HO-1 expression to changes in the reduced (GSH) to oxidized (GSSG) glutathione ratio. These data show abundant HO-1 expression by UFPs or 50 µg/mL fine PM (Figure 2A, Claremont Jan 02), which is accompanied by a sizable drop in glutathione ratios (Figure 2C). In contrast, coarse particles had no effect on either biological response (Figure 2C). Regression analysis showed a correlation coefficient (r2) of 0.97 between HO-1 expression and the DTT assay (Figure 2D). Overall, there is a strong correlation between particle size, chemical composition, ROS-generating capacity, and cellular oxidative stress.
UFP localization and mitochondrial damage. In defining the mechanistic features of PM toxicity, a key question is the subcellular localization of PM. This may determine ROS generation, as demonstrated by O2·- generation in lung microsomes during incubation with DEP extracts (Kumagai et al. 1997). Subcellular DEP targets include mitochondria, as demonstrated by the ability of organic DEP extracts to induce structural mitochondrial damage (Hiura et al. 1999, 2000; Li et al. 2002b). After exposure to CAPs, there were clear differences in the ultramicroscopic features of RAW 264.7 cells exposed to different particle sizes (Figure 3). Whereas coarse particles collected in large cytoplasmic vacuoles (Figure 3C and 3D), UFPs frequently lodged inside mitochondria (Figure 3G and H). Mitochondrial architecture remained intact in coarse PM incubations, but cells incubated with UFPs showed extensive disruption of mitochondrial cristae, resulting in a vacuolar cellular appearance (Figure 3H). These changes were time dependent, with fewer particles collecting inside mitochondria during shorter incubations (not shown). In cells exposed to fine particulates (which includes some UFPs), some particles lodged inside mitochondria but did not show the same degree of ultrastructural damage (Figure 3E and F).
Electron microscopy showed similar features in BEAS-2B cells--namely, considerable mitochondrial damage by UFPs, resulting in the formation of concentric structures, known as myelin figures (Figure 4). These structures result from the disassociation of lipoproteins, which facilitates water uptake and intercalation between lamellar membrane stacks (Figure 4C). Similar to RAW 264.7 cells, UFPs lodged inside damaged mitochondria (Figure 4C). Cells incubated with coarse or fine particles showed lesser mitochondrial damage (not shown). The extent of mitochondrial damage is in accordance with the redox cycling potential of the particles, as well as the HO-1 and glutathione results.

|
Figure 4. Electron micrographs demonstrating mitochondrial destruction in BEAS-2B cells treated with 8.4 µg/mL of USC-Jan 02 UFPs for 16 hr. (A) Untreated BEAS-2 cells; magnification  8,500. (B) UFP-treated cells; magnification 8,500. (B) UFP-treated cells; magnification  8,500. (C) UFP-treated cells; magnification 8,500. (C) UFP-treated cells; magnification  26,300. Notice the disappearance of cristae, formation of myelin figures (MF), and presence of particles (P) inside mitochondria (M). 26,300. Notice the disappearance of cristae, formation of myelin figures (MF), and presence of particles (P) inside mitochondria (M).
|
Our data demonstrate that the UFP mode in the Los Angeles basin is more potent than fine and coarse PM toward inducing oxidative stress as measured by the DTT, HO-1, and glutathione assays. Electron microscopy also indicates subcellular penetration and mitochondrial damage by UFPs and, to a lesser extent, fine particles. The findings correlate with PM organic carbon and PAH composition, suggesting a role of organic agents in generating redox activity.
The results from the DTT assay indicate UFPs are capable of producing greater ROS on a microgram basis than fine and coarse particles. This is the first time that a quantitative assay has been used to directly measure ROS generation by CAPs. Kumagai et al. (2002) focused the assay on assessment of quinones, whereas we have applied the assay to UFPs and fine and coarse particles. The DTT assay provides a quantitative measure of the relative redox activity of different PM sizes in the Los Angeles basin.
UFPs contain a higher percentage of organic carbon than fine and coarse particles, and this has relevance to the biologic potency of these particles. The enhanced biologic potency of UFPs is directly correlated with the PAH content. Although PAHs are capable of inducing ROS production in macrophages, it is also possible that these compounds may be a surrogate for other redox cycling chemicals in the DTT assay. We do not exclude a contribution by transition metals, which may interact with organic PM components in ROS generation (Saldiva et al. 2002).
These data are in accordance with the growing awareness that oxidative stress plays a key role in the induction of airway inflammation (HEI 2002; Nel et al. 1998). Recently, we demonstrated that macrophages and epithelial cells exhibit a stratified oxidative stress response to increasing concentrations of DEPs (Li et al. 2002a, 2002b). The stratified response commences with HO-1 expression when the GSH/GSSG ratio is minimally disturbed, proceeds to Jun kinase activation at intermediary levels of oxidative stress, and culminates in cellular toxicity at high oxidative stress levels. Ambient CAPs mimic the effects of organic DEP extracts (Li et al. 2002a), with UFPs showing increased potency in depressing the cellular GSH/GSSG ratio (Figure 2C). The significance of Jun kinase activation is the transcriptional activation of cytokine, chemokine, and adhesion receptor promoters (Nel et al. 1998). These products play a role in the proinflammatory effects of PM in the lung and possibly also the cardiovascular system (Nel et al. 1998). The finding of a significant correlation between heme oxygenase activity, GSH/GSSG ratio, and redox activity as measured by DTT production provides further evidence for the role of ROS generation in PM toxicity.
The biological significance of HO-1 expression in the lung is the antioxidant effect of its catabolic product, bilirubin (Choi and Alam 1996). In the process of heme catabolism, HO-1 also generates a gaseous substance, CO, which exerts anti-inflammatory effects in the lung and is exhaled in the expired air (Horvath et al. 1998; Maines 1997). It is interesting, therefore, that in a study in which normal human volunteers were exposed to DEPs, CO levels in the expired air was a more sensitive exposure marker than the presence of inflammatory products in the bronchoalveolar fluid (Nightingale et al. 2000). This is in agreement with the exquisite sensitivity of the HO-1 promoter to oxidative stress in vivo and in vitro (Choi and Alam 1996; Nightingale et al. 2000). HO-1 expression and CO generation are markers for airway inflammation in asthma (Horvath et al. 1998). Monitoring of CO levels in the expired air may be a useful marker for evaluating the pro-oxidative and proinflammatory effects of CAPs in the respiratory tract.
How exactly UFPs gain access to and induce mitochondrial damage is unknown. One possibility is that ROS generated outside of the mitochondrion may damage this organelle, allowing access to the particles. This is compatible with the ability of organic DEP extracts to induce ultrastructural mitochondrial damage in the absence of particles (Hiura et al. 2000; Li et al. 2002b). Our previous studies have demonstrated that organic DEP chemicals induced pro-apoptotic effects in macrophages and bronchial epithelial cells (Hiura et al. 1999, 2000; Li et al. 2002b). This effect may be mediated through the perturbation of mitochondrial permeability transition pore, which sets in motion cytochrome c release, caspase activation, and superoxide production in the mitochondrial inner membrane (Hiura et al. 2000). Ultramicroscopic visualization of human macrophages and BEAS-2B cells incubated with organic DEP extracts showed that the appearance of apoptotic bodies were accompanied by changes in mitochondrial morphology, including mitochondrial swelling and a loss of cristae (Li et al. 2002b). Another possibility is that UFPs gain access to mitochondria because of their small sizes. These particles might then release redox cycling chemicals that damage the inner membrane. All considered, we propose that enhanced tissue penetrance and ability to generate oxidative stress render UFPs more damaging at cellular level and consequently contribute to the adverse health effects of UFPs in the Los Angeles basin.
These findings may be of importance for PM regulation. Currently, the manufacture of cleaner combustion engines relies on mass output standards but do not consider the output of large numbers of UFPs, which have very low mass. Our data show that UFPs are more potent than PM2.5 and PM10 that contribute the majority of mass in the HO-1 and DTT assays. It may be necessary to consider standards based on particle number instead of mass if further studies confirm the differential toxicity of UFPs. Further research to more fully characterize the toxicity of UFPs in relation to particle number, surface area, and chemical composition is needed.
References
Bonvallot V, Baeza-Squiban A, Baulig A, Brulant S, Boland S, Muzea F, et al. 2001. Organic compounds from diesel exhaust particles elicit a proinflammatory response in human airway epithelial cells and induce cytochrome P450 1A1 expression. Am J Respir Cell Mol Biol 25:515-521.
Choi AM, Alam J. 1996. Heme oxygenase-1: function, regulation, and implication of a novel stress-inducible protein in oxidant-induced lung injury. Am J Respir Cell Mol Biol 15:9-19.
Dockery DW, Pope CA, Xu X, Spengler JD, Ware JH, Fay ME, et al. 1993. An association between air pollution and mortality in six U.S. cities. N Engl J Med 329:1753-1759.
Frampton MW. 2001. Systemic and cardiovascular effects of airway injury and inflammation: ultrafine particle exposure in humans. Environ Health Perspect 109(suppl 4):529-532.
HEI. 2002. Understanding the Health Effects of Components of the Particulate Matter Mix: Progress and Next Steps. Health Effects Institute Perspectives Series. Boston, MA:Health Effects Institute.
Hiura TS, Kaszubowski M, Li N, Nel AE. 1999. Chemicals in diesel exhaust particles generate reactive oxygen radicals and induce apoptosis in macrophages. J Immunol 163:5582-5590.
Hiura TS, Li N, Kaplan R, Horwitz M, Seagrave J, Nel AE. 2000. The role of a mitochondrial pathway in the induction of apoptosis by chemicals extracted from diesel exhaust particles. J Immunol 165:2703-2711.
Horvath I, Donnelly LE, Kiss A, Paredi P, Kharitonov SA, Barnes PJ. 1998. Raised levels of exhaled carbon monoxide are associated with an increased expression of heme oxygenase-1 in airway macrophages in asthma: a new marker of oxidative stress. Thorax 53:668-672.
Kawasaki S, Takizawa H, Takami K, Desaki M, Okazaki H, Kasama T, et al. 2001. Benzene-extracted components are important for the major activity of diesel exhaust particles. Am J Respir Cell Mol Biol 24:419-426.
Kim S, Jaques P, Chang MC, Froines JR, Sioutas C. 2001a. A versatile aerosol concentrator for simultaneous in vivo and in vitro evaluation of toxic effects of coarse, fine and ultrafine particles: Part I: Laboratory evaluation. J Aerosol Sci 11: 1281-1297.
Kim S, Jaques P, Chang MC, Sioutas C. 2001b. Versatile aerosol concentration enrichment system (VACES) for simultaneous in-vivo and in-vitro evaluation of toxic effects of ultrafine, fine and coarse ambient particles. Part II: Field evaluation. J Aerosol Sci 11:1299-1314.
Kumagai Y, Arimoto T, Shinyashiki M, Shimojo N, Nakai Y, Yoshikawa T, et al. 1997. Generation of reactive oxygen species during interaction of diesel exhaust particle components with NADPH-cytochrome P450 reductase and involvement of the bioactivation in the DNA damage. Free Radic Biol Med 22:479-487.
Kumagai Y, Koide S, Taguchi K, Endo A, Nakai Y, Yoshikawa T, et al. 2002. Oxidation of proximal protein sulfhydryls by phenanthraquinone, a component of diesel exhaust particles. Chem Res Toxicol 15:483-489.
Li N, Kim S, Wang M, Froines J, Sioutas C, Nel AE. 2002a. Use of a stratified oxidative stress model to study the biological effects of ambient concentrated and diesel exhaust particulate matter. Inhalation Toxicol 14:459-486.
Li N, Venkatesan MI, Miguel A, Kaplan R, Gujuluva C, Alam J, et al. 2000. Induction of heme oxygenase-1 expression in macrophages by diesel exhaust particle chemicals and quinones via the antioxidant-responsive element. J Immunol 165:3393-3401.
Li N, Wang M, Oberley TD, Sempf JM, Nel AE. 2002b. Comparison of the pro-oxidative and pro-inflammatory effects of organic DEP chemicals in bronchial epithelial cells and macrophages. J Immunol 169: 4531-4541.
Maines MD. 1997. The heme oxygenase system: a regulator of second messenger gases. Annu Rev Pharmacol Toxicol 37:517-554.
Miyabara Y, Ichinose T, Takano H, Lim HB, Sagai M. 1998. Effects of diesel exhaust on allergic airway inflammation in mice. J Allergy Clin Immunol 102:805-812.
Monks TJ, Hanzlik RP, Cohen GM, Ross D, Graham DG. 1992. Quinone chemistry and toxicity. Toxicol Appl Pharmacol 112:2-16.
National Research Council (NRC). 1998. Research Priorities for Airborne Particulate Matter: I-Immediate Priorities and a Long-Range Research Portfolio. Washington, DC:National Academy Press.
Nel AE, Diaz-Sanchez D, Li N. 2001. The role of particulate pollutants in pulmonary inflammation and asthma: evidence for the involvement of organic chemicals and oxidative stress. Curr Opinion Pulmonary Med 7:20-26.
Nel AE, Diaz-Sanchez D, Ng D, Hiura T, Saxon A. 1998. Enhancement of allergic inflammation by the interaction between diesel exhaust particles and the immune system. J Allergy Clin Immunol 102:539-559.
Nemmar A, Hoet PH, Vanquickenborne B, Dinsdale D, Thomeer M, Hoylaerts MF, et al. 2002. Passage of inhaled particles into the blood circulation in humans. Circulation 105:411-414.
Nightingale JA, Maggs R, Cullinan P, Donnelly LE, Rogers DF, Kinnersley R, et al. 2000. Airway inflammation after controlled exposure to diesel exhaust particulates. Am J Respir Crit Care Med 162:161-166.
Oberdörster G. 1996. Significance of particle parameters in the evaluation of exposure-dose-response relationships of inhaled particles. Inhal Toxicol 8(suppl):73-89.
Oberdörster G, Utell MJ. 2002. Ultrafine particles in the urban air: to the respiratory tract-and beyond [Editorial]. Environ Health Perspect 110:A440-A441.
Penning TM, Burczynski ME, Hung CF, McCoull KD, Palackal NT, Tsuruda LS. 1999. Dihydrodiol dehydrogenases and polycyclic aromatic hydrocarbon activation: generation of reactive and redox active o-quinones. Chem Res Toxicol 12:1-18.
Penttinen P, Timonen KL, Tiittanen P, Mirme A, Ruuskanen J, Pekkanen J. 2001. Number concentration and size of particles in urban air: effects on spirometric lung function in adult asthmatic subjects. Environ Health Perspect 109:319-323.
Saldiva PH, Clarke RW, Coull BA, Stearns RC, Lawrence J, Murthy GG, et al. 2002. Lung inflammation induced by concentrated ambient air particles is related to particle composition. Am J Respir Crit Care Med 165:1610-1617.
Samet JM, Dominici F, Curriero FC, Coursac I, Zeger SL. 2000. Fine particulate air pollution and mortality in 20 U.S. cities, 1987-1994. N Engl J Med 343:1742-1749.
Takano H, Yanagisawa R, Ichinose T, Sadakane K, Inoue K, Yoshida S, et al. 2002. Lung expression of cytochrome P450 1A1 as a possible biomarker of exposure to diesel exhaust particles. Arch Toxicol 76:146-151.
Tietze F. 1969. Enzymic method for quantitative determination of nanogram amounts of total and oxidized glutathione: applications to mammalian blood and other tissues. Anal Biochem 27:502-522.
Tsien A, Diaz-Sanchez D, Ma J, Saxon A. 1997. The organic component of diesel exhaust particles and phenanthrene, a major polyaromatic hydrocarbon constituent, enhances IgE production by IgE-secreting EBV-transformed human B cells in vitro. Toxicol Appl Pharmacol 142:256-263.
Utell MJ, Frampton MW. 2000. Acute health effects of ambient air pollution: the ultrafine particle hypothesis. J Aerosol Med 13:355-359.
Wichmann HE, Spix C, Tuch T, Wolke G, Peters A, Heinrich J, et al. 2000. Daily mortality and fine and ultrafine particles in Erfurt, Germany. Part I: role of particle number and particle mass. Res Rep Health Eff Inst 98:5-86.
Willeke K, Lin X, Grinshpun SA. 1998. Improved aerosol collection by combined impaction and centrifugal motion. Aerosol Sci Tech 28:439-456.
Yang AH, Gould-Kostka J, Oberley TD. 1987. In vitro growth and differentiation of human kidney tubular cells on a basement membrane substrate. In Vitro Cell Dev Biol 23:34-46.
Last Updated: March 15, 2002







