
| |  | |  |
Children's Health
|
| Vitamin D Receptor Fok1 Polymorphism and Blood Lead Concentration in Children Erin N. Haynes,1,2,3,4 Heidi J. Kalkwarf,1,2 Richard Hornung,1,5 Richard Wenstrup,6 Kim Dietrich,1,3 and Bruce P. Lanphear1,2,3 1Cincinnati Children's Environmental Health Center and 2General and Community Pediatrics, Cincinnati Children's Hospital Medical Center, Cincinnati, Ohio, USA; 3Department of Environmental Health, University of Cincinnati, Cincinnati, Ohio, USA; 4Department of Environmental Health Sciences, University of Michigan School of Public Health, Ann Arbor, Michigan, USA; 5Institute for Health Policy and Health Services Research Institute, University of Cincinnati, Cincinnati, Ohio, USA: 6Division of Human Genetics, Cincinnati Children's Hospital Medical Center, Cincinnati, Ohio, USA
Abstract
Variation in blood lead concentration is caused by a complex interaction of environmental, social, nutritional, and genetic factors. We evaluated the association between blood lead concentration and a vitamin D receptor (VDR) gene polymorphism. Environmental samples and blood were analyzed for lead, nutritional and behavioral factors were assessed, and VDR -Fok1 genotype was determined in 245 children. We found a significant interaction between floor dust lead and genotype on blood lead concentration. For every 1 µg/ft2 increase in floor dust, children with VDR -FF genotype had a 1.1% increase in blood lead [95% confidence interval (CI) , 0.69-1.5], VDR -Ff, 0.53% increase (95% CI, 0.1-0.92) , and VDR -ff, 3.8% increase (95% CI, 1.2-6.3) ; however, at floor dust levels < 10 µg/ft2, children with VDR -ff had the lowest blood lead concentrations. These data suggest that VDR -Fok1 is an effect modifier of the relationship of floor dust lead exposure and blood lead concentration. Key words: child, dust, environmental exposure, Fok1, gene-environment, lead, lead poisoning, VDR, vitamin D receptor. Environ Health Perspect 111:1665-1669 (2003) . doi:10.1289/ehp.6167 available via http://dx.doi.org/ [Online 7 July 2003] |
|
|
 |
Address correspondence to E.N. Haynes, University of Cincinnati, Department of Environmental Health, Division of Epidemiology and Biostatistics, PO Box 670056, Cincinnati, OH 45267-0056. Telephone: (513) 558-1986. Fax: (513) 558-6272. E-mail: erin.haynes@cchmc.org
This publication was funded in part by the National Institute of Environmental Health Sciences, National Institutes of Health (RO1-ES08338, ES06096, ES11261, and 5 T32 ES10957-02) and the Centers for Disease Control and Prevention (U67/CCU210773).
The authors declare they have no conflict of interest.
Received 18 December 2002; accepted 7 July 2003.
Despite a dramatic decline in blood lead concentration in U.S. children over the last three decades, lead toxicity remains a significant risk for some infants and children (Pirkle et al. 1998). Moreover, there is increasing evidence that blood lead concentrations < 10 µg/dL, the current level of concern [Centers for Disease Control and Prevention (CDC) 1997], are associated with deficits in cognition, hearing, and academic skills (Canfield et al. 2003; Lanphear et al. 2000a; Schwartz 1994; Schwartz and Otto 1991). Scientists have long recognized that children's blood lead concentrations vary widely by race, socioeconomic status, environmental exposure, mouthing behaviors, and unidentified biologic or genetic factors (CDC 1997; Lanphear et al. 2002; Lin-Fu 1973). But biologic factors that account for differences in blood lead concentrations are poorly understood.
Gastrointestinal lead absorption varies across individuals. Variation in lead absorption results from nutritional deficiencies (Fullmer 1992; Six and Goyer 1970), fasting conditions (Flanagan et al. 1982; Rabinowitz et al. 1980), and genetic factors that influence mineral metabolism (Bouton and Pevsner 2000; Schwartz et al. 2000). Observational and experimental studies have shown that dietary intake of calcium and iron are inversely associated with lead absorption (Fullmer 1992; Mahaffey et al. 1986; Six and Goyer 1970; Watson et al. 1980; Wright et al. 1999; Ziegler et al. 1978), indicating that variation in blood lead concentration may be caused partly by nutritional intake of calcium and iron or genetic factors that influence their absorption (Bouton and Pevsner 2000; Flanagan et al. 1982; Schwartz et al. 2000).
Lead follows the biologic pathway of other bivalent cations, such as calcium, competing for absorptive and protein-binding sites (Godwin 2001). Although the relationship between calcium and lead is complex, in vivo and in vitro studies have demonstrated that cellular lead uptake increases when calcium stores are reduced or depleted (Fullmer 1992; Kerper and Hinkle 1997). The high-affinity binding of lead to calcium-binding protein (Richardt et al. 1986) suggests that lead absorption would be increased in times of low calcium intake (Fullmer 1997). Indeed, children with elevated blood lead concentration have been reported to have significantly lower dietary calcium intake (Fullmer 1992; Johnson and Tenuta 1979; Mahaffey et al. 1986; Rosen et al. 1980). Calcium metabolism is governed, in part, by the vitamin D endocrine system and the vitamin D receptor (VDR) (DeLuca 1979; Henry 1982). Ames et al. (1999) reported that the VDR-Fok1 genotype FF was associated with increased bone mineral density (p = 0.02) and a 30-40% increase in calcium absorption (p = 0.04) in healthy children 7-12 years old.
The purpose of this study was to test whether the VDR-Fok1 polymorphism was associated with increased blood lead concentration in a cohort of 275 children. We hypothesized that the VDR-Fok1 polymorphism would modify blood lead concentrations. In particular, we predicted that children homozygous for the F allele--a marker for increased calcium absorption--would have higher blood lead concentrations than heterozygotes and children homozygous for the f allele, after adjusting for environmental lead exposure. We examined the interaction of floor dust lead loading, the major source of lead intake for children, and VDR-Fok1 to determine if VDR-Fok1 was an effect modifier of the relationship of floor dust lead exposure and blood lead concentration.
Study population. The study population is based on a cohort of 275 children enrolled in an ongoing longitudinal study (Lanphear et al. 1999). Families who lived in the city of Rochester, New York, were identified and recruited using sequential lists of live births from five urban hospitals in Rochester. Four home visits were made to families when the child was 6 (baseline), 12, 18, and 24 months old. A certified phlebotomist drew a blood sample (5-10 cc) from each infant at every visit, and a trained interviewer conducted a face-to-face survey with the primary caretaker to assess risk factors for lead exposure, including mouthing behaviors (e.g., soil ingestion, paint chip ingestion), time spent outdoors, and attainment of developmental milestones. The primary caretaker was questioned about the content of the child's diet using a food frequency checklist (Willett 1990) that was modified to reflect the dietary content of children in the study population. Nutritional contributions to infants' diets from breast milk were calculated based on published data (Lawrence 1994). The estimated breast milk intake of 6-month-old infants who were not exclusively breast-fed, as defined by Labbok and Krasovec (1990), was based on individual surveys and was 769 mL/day (Heinig et al. 1993). Contributions from formula were calculated by ounces of formula (brand and type) consumed daily at 6 and 12 months (Johnson 1993).
At each of the four visits, an environmental technician collected three or four composite interior dust wipe samples from surfaces that were accessible to a child (i.e., carpeted floors, noncarpeted floors, and window sills) or known to be heavily contaminated with lead (window troughs) in the child's bedroom, the kitchen, and the living room. A composite dust sample consisted of a maximum of three wipe samples collected from the same surface (i.e., carpeted floor, noncarpeted floor, and interior window sill or window wells). Because floor dust lead values were highly skewed and the most extreme floor dust values exerted a high degree of influence on regression coefficients, we truncated the extreme floor dust lead values (> 98.5th percentile) rather than log-transforming the data. Floor samples (carpeted and noncarpeted floor dust lead loading values) were combined to form a single floor dust lead variable. Dust samples were analyzed first by flame atomic absorption, followed by graphite furnace if levels were < 5 µg/sample. The detection limit of graphite furnace for the dust wipe was 0.5 µg/sample.
Soil and water samples were measured at baseline and when a child moved to a new residence. Three soil samples were taken from each side of the home along the perimeter of the foundation where bare soil was present. The samples were combined to form a single composite soil sample. Soil was analyzed separately with flame atomic absorption spectroscopy. The detection limit for lead in soil was 25 µg/g. Caregivers collected a water sample (250 cc) in the morning from the kitchen tap after the water flowed for 1 min. Water was analyzed by using atomic absorption, with a detection limit of 5 µg/L. Because only a small proportion of water samples had a lead concentration above the detection limit, water lead was dichotomized as at or above versus below the detection limit.
The study was approved by the institutional review boards at Cincinnati Children's Hospital Medical Center (Cincinnati, OH) and University of Michigan (Ann Arbor, MI).
Blood lead concentration. Children's venous blood samples were analyzed for lead using electrothermal atomization atomic absorption spectrometry at the New York State Department of Health (Wadsworth Laboratories, Albany, NY; Lanphear et al. 1999; Parsons and Slavin 1993). Briefly, all reported blood lead values were the means of six separate analyses performed on each blood sample. The routine within-run precision, expressed as standard deviation, was 0.23-0.26 µg/dL, and the between-run precision, based on duplicate measurements, ranged from 0.1 to 0.5 µg/dL for blood lead concentrations < 20 µg/dL. The detection limit for lead in blood was 1 µg/dL. Blood samples were stored in a -70°C freezer.
VDR-Fok1 genotyping. Whole blood samples were analyzed for VDR-Fok1 polymorphism by the Cincinnati Children's Hospital Medical Center, Division of Human Genetics. Genomic DNA was isolated from 3 mL of whole blood collected in EDTA-coated tubes by a standard phenol-chloroform extraction procedure. The 265-bp fragment of genomic DNA containing the polymorphic portion of exon 2 on the VDR gene was amplified by polymerase chain reaction (PCR), as described by Ames et al. (1999). Primers VDR2a: 5´-AGCTGGCCCTGGCACTGACTCTGCTCT-3´ and VDR2b: 5´-ATGGAAACACCTTGCTTCTTCTCCCTC-3´ were used to amplify the Fok1 polymorphic restriction site (Gibco BRL Custom Primers, Carlsbad, CA). In summary, 2 µL genomic DNA was added to 47.6 µL PCR nucleotide mix containing 1 µL dNTP, 2 µL MgCl2, and 5 µL 10% dimethyl sulfoxide. After 3 min of thermocycling at 95°C in the PCR machine, 0.4 µL Taq DNA polymerase was added to the reaction. Thermocycling conditions were 94°C for 30 sec, 60°C for 30 sec, and 72°C for 30 sec. A final elongation period of 5 min at 72°C was added after 30 cycles. PCR products were digested with 4 µL Fok1 and 5 µL New England Biolabs buffer (Beverly, MA) for 1.5 hr at 37°C and then electrophoresed through a 3% agarose gel for 2 hr at 80 V.
|

| |
Figure 1. Influence of floor dust lead loading and VDR-Fok1 genotype on blood lead concentration. The difference among the three VDR-Fok1 genotypes was statistically significant (p = 0.009). Slopes: FF, exp(1.584 + 0.011  floor dust), n = 117; Ff, exp(1.562 + 0.0053 floor dust), n = 117; Ff, exp(1.562 + 0.0053  floor dust), n = 96; ff, exp(1.405 + 0.038 floor dust), n = 96; ff, exp(1.405 + 0.038  floor dust), n = 17. floor dust), n = 17.
|
|
Table 1.

|
|
Table 2.

|
|
Table 3.

|
|

| |
Figure 2. Influence of floor dust lead loading and VDR-FF and VDR-Ff genotypes on blood lead concentration by race. The difference among the VDR-FF and VDR-Ff genotypes was statistically significant for non-African Americans (p = 0.021), but not statistically significant for African Americans (p = 0.272). Slopes: African-American FF, exp[1.69 + 0.011  floor dust), n = 83; African-American Ff, exp(1.61 + 0.0074 floor dust), n = 83; African-American Ff, exp(1.61 + 0.0074  floor dust), n = 56; non-African-American FF, exp(1.53+ 0.015 floor dust), n = 56; non-African-American FF, exp(1.53+ 0.015  floor dust), n = 38; non-African-American Ff: exp(1.61 + 0.0022 floor dust), n = 38; non-African-American Ff: exp(1.61 + 0.0022  floor dust), n = 44. floor dust), n = 44.
|
The f genotype was indicated by the presence of the restriction site that generates two fragments of 196 bp and 69 bp. The F genotype was indicated by a single uncleaved 265-bp fragment. Determination of VDR genotype FF, Ff, or ff was indicated based on the Fok1 cleavage pattern (Figure 1). The following safeguards were instituted to maintain consistency of the results: a) Genomic DNA, which was previously genotyped at the VDR locus, was repeated twice per month alongside new samples, to confirm previous results; b) "no DNA" controls were routinely run with samples to ensure that DNA contamination was prevented. The reproducibility of these methods is 100%.
Statistical analysis. We used descriptive statistics to examine the differences in characteristics of 24-month-old children by their VDR-Fok1 genotype. We used analysis of variance to make comparisons among the three genotypes for continuous variables, and chi square tests for categorical variables. For all statistical analyses, blood lead concentration was log transformed.
We developed multiple regression models to predict blood lead concentration as a function of environmental, nutritional, behavioral, and demographic variables. Data were analyzed using repeated measures analysis, a mixed model regression method that accounts for the correlation among outcomes measured on the same child over time. The subjects in this study were considered random effects, and the VDR-Fok1 genotype, environmental lead exposure variables, nutritional intake, and mouthing behaviors were considered fixed effects. The PROC MIXED procedure in SAS (version 8; SAS Institute, Cary, NC) was used to conduct the repeated measures analysis.
To examine effect modification of the VDR-Fok1 genotype, we tested numerous interactions, including calcium intake (lagged by one visit), vitamin D intake, race, environmental exposure to lead-contaminated floor dust, and the VDR-Fok1 genotype. Covariates were retained in the final model if they caused a meaningful change in the VDR coefficient or were significant predictors of blood lead, as determined by a two-tailed p-value of < 0.05. Final model covariates included age, African-American race, calcium intake, iron intake, renting a home, floor dust lead loading, window-well dust lead loading, soil ingestion, paint chip ingestion, and water lead concentration. Interactions included the VDR-Fok1 genotype by floor dust, age by total iron intake, age by African-American race, age by renting a home, age by chip ingestion, and age by window-well dust lead loading. After adjustment, we compared the geometric mean blood lead concentration for each age group by the VDR-Fok1 genotype.
Study population. Of the 275 children who were enrolled at 6 months of age, 245 (89%) were available for the 24-month follow-up blood test (Lanphear et al. 1999). The attrition rate was similar for African-American (4%) and non-African-American children (6%). Six children were removed from the analysis because their residences had floor dust lead loading values > 100 µg/ft2, which were considered to be outliers in this data set. There were adequate blood samples for 239 (97%) of the children (145 African American, 47 white, and 47 other, which included Asian, Latino, American Indian, and unknown). The overall prevalence of the VDR-Fok1 genotypes in the study population was 51% FF, 42% Ff, and 8% ff. Allele distributions in the study population were in Hardy-Weinberg equilibrium.
None of the lead exposure variables differed significantly by genotype except soil ingestion: 47% of children with VDR-ff reportedly ingested soil compared with approximately 20% in children with VDR-FF and VDR-Ff (p = 0.05; Table 1). The VDR-Fok1 polymorphisms also differed significantly by race (Table 2). The frequency of FF homozygotes in African-American children was significantly greater than that in non-African-American children (chi squared = 6.451, 1 df, p = 0.01). African-American children also had a higher percentage of the F allele than did non-African-American children (chi square = 9.068, 1 df, p = 0.003). Genotype frequencies for the FF, Ff, and ff genotypes were 57%, 39%, and 4%, respectively in African-American children, and 40%, 47%, and 13%, respectively in non-African-American children (Table 2). There was no significant difference among the frequencies of genotypes between white and other (Asian, Latino, American Indian, and unknown; chi square = 0.845, 2 df, p = 0.66).
Blood lead concentration increased during the first 24 months of life (p < 0.001), with the steepest increase occurring between 6 and 12 months. In an unadjusted repeated measures analysis, there was a significant difference in blood lead concentrations among the VDR-Fok1 genotypes (p = 0.04; Table 3). At 24 months, children with VDR-FF genotype had blood lead concentrations that were 1.2 µg/dL and 2.3 µg/dL higher than children with VDR-Ff and VDR-ff, respectively (Table 3).
Multivariable analysis. There were no changes in the determinants of children's blood lead concentrations from our previous publication (Lanphear et al. 2002), except for calcium intake. In this VDR model, calcium intake was inversely associated with children's blood lead (p = 0.03).
After adjusting for environmental lead exposure, there was no significant difference in blood lead concentrations among the VDR-Fok1 genotypes (p = 0.58; Table 3). At 24 months, children with VDR-FF genotype had blood lead concentrations that were 0.5 µg/dL and 0.1 µg/dL higher than in children with VDR-Ff and VDR-ff, respectively (Table 3).
To determine whether the VDR-Fok1 polymorphism was an effect modifier of the relationship between lead exposure and children's blood lead concentrations, we included interaction terms between environmental lead exposures and VDR-Fok1 categories. We examined all interactions of lead exposure and VDR-Fok1 categories. Only floor dust lead loading and paint chip ingestion were significant. Only 6% of children were reported to ingest paint chips, and the largest increase in blood lead compared with those with reportedly no paint chip ingestion was for those children with the VDR-ff genotype (n = 2, or 12%; Table 1). Therefore, we did not examine interactions of VDR with paint chip ingestion.
There was a significant interaction between floor dust lead loading and VDR-Fok1 genotypes on blood lead concentration (p = 0.009). With every 1 µg/ft2 increase in floor dust, children with the FF genotype had a 1.1% increase in blood lead [95% confidence interval (CI), 0.69-1.5], whereas children with the Ff genotype had a 0.53% increase in blood lead (95% CI, 0.1-0.92; Figure 1). Children with the ff genotype had a 3.8% increase in blood lead concentration (95% CI, 1.2-6.3) for each 1 µg/ft2 increase in floor dust lead loading (Figure 1). Mean blood lead concentrations among children with VDR-ff were lower than the other VDR-Fok1 genotypes at floor dust lead levels < 10 µg/ft2; however, when floor dust lead levels were > 10 µg/ft2, children with VDR-ff had the highest mean blood lead concentration. To address the issue of small sample size of VDR-ff group, we conducted secondary analyses without the VDR-ff group. The difference between the exposure-response functions for VDR-FF and VDR-Ff decreased in significance from p = 0.0009 to p = 0.03. The slope relating log of blood lead to floor dust loading for VDR-FF was 0.012; that for VDR-Ff was 0.005.
To examine the effect of race on the interaction between VDR genotype and floor dust lead loading, we ran the model for African Americans and non-African Americans separately. Because of the small sample size of the VDR-ff group, we removed it from the analysis. The floor dust lead loading interaction with VDR-FF and VDR-Ff was not significant for African-American children (p = 0.272), but was significant for non-African-American children (p = 0.021; Figure 2). However, the three-way interaction of race, VDR, and floor dust was not statistically significant (p = 0.48). The coefficients for the white and other categories comprising the non-African-American group were examined, but the results were similar.
There were also no interactions found with VDR and other environmental variables. In addition, we did not find a significant modification of the VDR-Fok1 effect on blood lead by calcium intake (p = 0.49).
The results of these analyses suggest that VDR-Fok1 may modify the relationship of lead exposure and blood lead levels during the first 2 years of life, when children are most susceptible to lead ingestion and absorption (Lanphear et al. 2002; Ziegler et al. 1978). As floor dust lead increased, children with FF genotype had a greater increase in blood lead concentration than did children with Ff genotype. Presumably, this difference is due to lead's metabolic mimicry of calcium and an increased efficiency of lead absorption and retention among children with VDR-FF. The dramatic rise in blood lead at floor dust lead > 10 µg/ft2 for children with VDR-ff could be attributed to small sample size (only 17 children had VDR-ff genotype, resulting in wide confidence intervals, and only two children with VDR-ff had floor dust lead loading > 10 µg/ft2). It is also likely that other genetic or biologic differences exist that we did not measure or could not measure precisely, such as calcium intake.
In secondary analysis, we examined whether VDR-Fok1 was an effect modifier after excluding children who had VDR-ff. Not wanting to assume allele dominance, we decided not to pool genotypes with the f allele, but rather removed the VDR-ff group from the analysis. When we ran the main model without the VDR-ff group, the difference between the VDR-FF and VDR-Ff groups was still significant.
To investigate the role of African-American race, we ran the full model for each race separately. Interestingly, the model without African-American children showed a significant interaction between VDR-Fok1 genotypes and floor dust (p = 0.015). The results were similar when we ran the model with the VDR-ff group removed. The interaction between floor dust lead loading and VDR genotypes was not significant for African-American children (p = 0.27), but for non-African-American children the interaction was significant (p = 0.02). This suggests a three-way interaction among VDR-Fok1, floor dust lead loading, and race; however, the three-way interaction in the model was not significant (p = 0.48). We interpret this with caution because of the small sample size of the groups.
Based on our model, calcium intake was a significant predictor of blood lead concentration (p = 0.03). We did not, however, find a significant effect modification of calcium intake on blood lead concentration by VDR-Fok1 genotype, perhaps because of the complexity of the lead-calcium relationship. Also, the mean calcium intake for study children was well above the dietary reference intakes (DRIs) for children. Similarly, in a study by Dawson-Hughes et al. (1995) the impact of the VDR-Bsm1 BB genotype was reduced at calcium intake of > 300 mg/day. The mean calcium intake for children in the present study was 679 mg/day (95% CI, 649-712) at 6 months, 987 mg/day at 12 months (95% CI, 940-1,034), and 968 mg/day (95% CI, 913-1023) at 18 months (Lanphear et al. 2002). The DRIs are 210 mg/day for children 0-6 months old, 270 mg/day for children 6-12 months old, and 500 mg/day for children 1-3 years old (Institute of Medicine 1997). Although the calcium intake for children in our study was greater than the DRI, the values are comparable with calcium intake levels in the third National Health and Nutrition Examination Survey (NHANES III). In NHANES III, children 5-7 months old had a mean calcium intake of 691 mg/day; 11-13 months, 933 mg/day; 17-19 months, 874 mg/day; and 23-25 months, 784 mg/day (Lanphear BP. Unpublished data).
The VDR gene has been cloned (Miyamoto et al. 1997) and several genetic variants have been described in humans (Morrison et al. 1992). Variation in the VDR gene has been reported, including sites cleaved by Bsm1 (Morrison et al. 1992), Apa1 (Faraco et al. 1989), Taq1 (Morrison et al. 1992), and Fok1 (Saijo et al. 1991), and a poly(A) site at the 5´ end of the gene (Ingles et al. 1997). Located within VDR translation initiation site on exon II, the Fok1 polymorphism, a C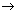 T transition, creates an upstream initiation codon, resulting in a VDR molecule elongated by three amino acids (f) compared with those initiating translation from the downstream site (F) (Arai et al. 1997; Gross et al. 1996). Unlike the Bsm1, Apa1, and Taq1 sites, the Fok1 polymorphism affects the amino acid sequence altering the VDR protein structure (Gennari et al. 1999). VDR-Fok1 is not genetically linked to the Bsm/Apa/Taq/poly(A) cluster (Whitfield et al. 2001). Very few studies have examined the association with the Apa/Bsm/Taq cluster and the Fok1 site. T transition, creates an upstream initiation codon, resulting in a VDR molecule elongated by three amino acids (f) compared with those initiating translation from the downstream site (F) (Arai et al. 1997; Gross et al. 1996). Unlike the Bsm1, Apa1, and Taq1 sites, the Fok1 polymorphism affects the amino acid sequence altering the VDR protein structure (Gennari et al. 1999). VDR-Fok1 is not genetically linked to the Bsm/Apa/Taq/poly(A) cluster (Whitfield et al. 2001). Very few studies have examined the association with the Apa/Bsm/Taq cluster and the Fok1 site.
Lead-contaminated house dust is a major source of childhood lead exposure (Adams 1991; Lanphear 1998; Lanphear et al. 1998a, 1998b; Manton et al. 2000; Sayre and Katzel 1979; Succop et al. 1998). Lead-contaminated house dust can be ingested during normal hand-to-mouth activity or on foods (Melnyk et al. 2000). Lead-contaminated floor dust was the primary source of exposure in our cohort. We ran the repeated measures model with each of the four lead exposure variables, floor dust, window-well dust, water lead, and soil lead, individually. Based on the likelihood ratio statistic, the floor dust model was the best fit for the data.
The prevalence of the VDR-Fok1 alleles has previously been determined for African-American women (Harris et al. 1997) and children (Ames et al. 1999). We found 55% of the African-American children in our study to be homozygous for the F allele, 40% to be heterozygous, and 4% to be homozygous for the f allele. The distributions reported by Harris et al. (1997) and Ames et al. (1999) were 65% FF, 31% Ff, 4% ff for African-American women and 67% FF, 33% Ff, and 0% ff for African-American children. Consistent with these other reports, we found that African-American children were significantly more likely to have VDR-FF than were non-African-American children (p = 0.02). Thus, although it is beyond the scope of this study, the ability of African-American individuals to have increased calcium absorption may partially explain the higher blood lead concentration seen in African-American children (Lanphear et al. 1996, 2000b, 2002; Pirkle et al. 1998) .
This study has some limitations. The semiquantitative food frequency survey required caregivers to recall a child's food intake over a 6-month period, which is subject to error. The food frequency surveys did not correlate with a 3-day diary for a random sample of 26 children 18 months old (Lanphear et al. 2002). However, the nutritional intake in our study population was similar for those surveyed in NHANES III (Lanphear BP. Unpublished data). Another limitation is the small number of children in our study homozygous for the f allele (n = 17). These low numbers may, in part, explain the higher blood lead concentrations among children who were exposed to floor dust lead loading > 10 µg/ft2. Only 2 of the 17 children with VDR-ff were exposed to floor dust lead loading > 10 µg/ft2. In contrast 44 and 43% of children with VDR-FF and VDR-Ff were exposed to floor dust lead loading > 10 µg/ft2. The low floor dust lead loading (mean = 3.3 µg/ft2) in the VDR-ff children may also contribute to the differences we observed in blood lead concentration. In addition, we did not examine the correlation between the other VDR polymorphisms [Bsm1, Apa1, Taq1, poly(A)] and Fok1 (Kerr Whitfield et al. 2001).
In conclusion, this study suggests that VDR-Fok1 polymorphism is an effect modifier of the relationship between floor dust lead exposure and blood lead concentration in non-African-American children, possibly through its effect on gastrointestinal absorption. Further research is necessary to evaluate the gene-environment interaction among other markers of calcium metabolism, the gene-gene interaction between the Fok1 and other VDR polymorphic sites, and blood lead concentration in children, and to examine the contribution of calcium metabolism and the VDR on racial differences in blood lead concentration.
|
|
 |
| [References Listed in PubMed]References
Adams MA. 1991. FDA Total Diet Study: dietary intakes of lead and other chemicals. Chem Speciat Bioavailab 3:37-41.
Ames SK, Ellis KJ, Gunn SK, Copeland KC, Abrams SA. 1999. Vitamin D receptor gene Fok1 polymorphism predicts calcium absorption and bone mineral density in children. J Bone Miner Res 14:740-746.
Arai H, Miyamoto K, Taketani Y, Yamamoto H, Iemori Y, Morita K, et al. 1997. A vitamin D receptor gene polymorphism in the translation initiation codon: effect on protein activity and relation to bone mineral density in Japanese women. J Bone Miner Res 12:915-921.
Bouton CM, Pevsner J. 2000. Effects of lead on gene expression. Neurotoxicology 21:1045-1055.
Canfield RL, Henderson CR Jr, Cory-Slechta DA, Cox C, Jusko TA, Lanphear BP. 2003. Intellectual impairment in children with blood lead concentrations below 10 microg per deciliter. N Engl J Med 348:1517-1526.
CDC. 1997. Screening Young Children for Lead Poisoning: Guidance for State and Local Public Health Officials. Atlanta, GA:Centers for Disease Control and Prevention.
Dawson-Hughes B, Harris SS, Finneran S. 1995. Calcium absorption on high and low calcium intakes in relation to vitamin D receptor genotype. J Clin Endocrinol Metab 80:3657-3661.
DeLuca HF. 1979. The vitamin D system in the regulation of calcium and phosphorus metabolism. Nutr Rev 37:161-193.
Faraco JH, Morrison NA, Baker A, Shine J, Frossard PM. 1989. ApaI dimorphism at the human vitamin D receptor gene locus. Nucleic Acids Res 17:2150.
Flanagan PR, Chamberlain MJ, Valberg LS. 1982. The relationship between iron and lead absorption in humans. Am J Clin Nutr 36:823-829.
Fullmer CS. 1992. Intestinal interactions of lead and calcium. Neurotoxicology 13:799-807.
------. 1997. Lead-calcium interactions: involvement of 1,25-dihydroxyvitamin D. Environ Res 72:45-55.
Gennari L, Becherini L, Mansani R, Masi L, Falchetti A, Morelli A, et al. 1999. FokI polymorphism at translation initiation site of the vitamin D receptor gene predicts bone mineral density and vertebral fractures in postmenopausal Italian women. J Bone Miner Res 14:1379-1386.
Godwin HA. 2001. The biological chemistry of lead. Curr Opin Chem Biol 5:223-227.
Gross C, Eccleshall TR, Malloy PJ, Villa ML, Marcus R, Feldman D. 1996. The presence of a polymorphism at the translation initiation site of the vitamin D receptor gene is associated with low bone mineral density in postmenopausal Mexican-American women. J Bone Miner Res 11:1850-1855.
Harris SS, Eccleshall TR, Gross C, Dawson-Hughes B, Feldman D. 1997. The vitamin D receptor start codon polymorphism (FokI) and bone mineral density in premenopausal American black and white women. J Bone Miner Res 12:1043-1048.
Heinig M, Nommsen L, Peerson J, Lonnerdal B, Dewey K. 1993. Energy and protein intakes of breast-fed and formula-fed infants during the first year of life and their association with growth velocity: the DARLING Study. Am J Clin Nutr 58:152-161.
Henry HL. 1982. The role of parathyroid hormone in the regulation of the metabolism of 25-hydroxyvitamin D3. Miner Electrolyte Metab 8:179-187.
Ingles SA, Haile RW, Henderson BE, Kolonel LN, Nakaichi G, Shi CY, et al. 1997. Strength of linkage disequilibrium between two vitamin D receptor markers in five ethnic groups: implications for association studies. Cancer Epidemiol Biomarkers Prev 6:93-98.
Institute of Medicine. 1997. Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride (Standing Committee on the Scientific Evaluation of Dietary Reference Intakes FaNB, eds). Washington DC:National Academy Press.
Johnson KB, ed. 1993. The Harriet Lane Handbook: A Manual for Pediatric House Officers. St. Louis, MO:Mosby.
Johnson NE, Tenuta K. 1979. Diets and lead blood levels of children who practice pica. Environ Res 18:369-376.
Kerper LE, Hinkle PM. 1997. Cellular uptake of lead is activated by depletion of intracellular calcium stores. J Biol Chem 272:8346-8352.
Labbok M, Krasovec K. 1990. Toward consistency in breastfeeding definitions. Stud Fam Plan 21:226-230.
Lanphear BP. 1998. The paradox of lead poisoning prevention. Science 281:1617-1618.
Lanphear BP, Burgoon DA, Rust SW, Eberly S, Galke W. 1998a. Environmental exposures to lead and urban children's blood lead levels. Environ Res 76:120-130.
Lanphear BP, Dietrich K, Auinger P, Cox C. 2000a. Cognitive deficits associated with blood lead concentrations < 10 microg/dL in US children and adolescents. Public Health Rep 115:521-529.
Lanphear BP, Eberly S, Howard CR. 2000b. Long-term effect of dust control on blood lead concentrations. Pediatrics 106:E48. Available: http://pediatrics.aappublications.org/cgi/content/full/ 106/4/e48 [accessed 1 August 2003].
Lanphear BP, Hornung R, Ho M, Howard CR, Eberle S, Knauf K. 2002. Environmental lead exposure during early childhood. J Pediatr 140:40-47.
Lanphear BP, Howard C, Eberly S, Auinger P, Kolassa J, Weitzman M, et al. 1999. Primary prevention of childhood lead exposure: a randomized trial of dust control. Pediatrics 103:772-777.
Lanphear BP, Matte TD, Rogers J, Clickner RP, Dietz B, Bornschein RL, et al. 1998b. The contribution of lead-contaminated house dust and residential soil to children's blood lead levels. A pooled analysis of 12 epidemiologic studies. Environ Res 79:51-68.
Lanphear BP, Weitzman M, Eberly S. 1996. Racial differences in urban children's environmental exposures to lead. Am J Public Health 86:1460-1463.
Lawrence RA, ed. 1994. Breastfeeding: A Guide for the Medical Profession. St. Louis, MO:Mosby.
Lin-Fu JS. 1973. Vulnerability of children to lead exposure and toxicity (second of two parts). N Engl J Med 289:1289-1293.
Mahaffey K, Gartside P, Glueck C. 1986. Blood lead levels and dietary calcium intake in 1- to 11-year-old children: the second national Health and Nutrition Examination Survey, 1976-1980. Pediatrics 78:257-262.
Manton WI, Angle CR, Stanek KL, Reese YR, Kuehnemann TJ. 2000. Acquisition and retention of lead by young children. Environ Res 82:60-80.
Melnyk LJ, Berry MR, Sheldon LS, Freeman NC, Pellizzari ED, Kinman RN. 2000. Dietary exposure of children in lead-laden environments. J Expo Anal Environ Epidemiol 10:723-731.
Miyamoto Y, Shinki T, Yamamoto K, Ohyama Y, Iwasaki H, Hosotani R, et al. 1997. 1alpha,25-dihydroxyvitamin D3-24-hydroxylase (CYP24) hydroxylates the carbon at the end of the side chain (C-26) of the C-24-fluorinated analog of 1alpha,25-dihydroxyvitamin D3. J Biol Chem 272:14115-14119.
Morrison NA, Yeoman R, Kelly PJ, Eisman JA. 1992. Contribution of trans-acting factor alleles to normal physiological variability: vitamin D receptor gene polymorphism and circulating osteocalcin. Proc Natl Acad Sci USA 89:6665-6669.
Parsons PJ, Slavin W. 1993. A rapid Zeeman graphite furnace atomic absorption spectrometric method for the determination of lead in blood. Spectrochim Acta 48B:925-939.
Pirkle J, Kaufmann R, Brody D, Hickman T, Gunter E, Paschal D. 1998. Exposure of the U.S. population to lead, 1991-1994. Environ Health Perspect 106:745-750.
Rabinowitz MB, Kopple JD, Wetherill GW. 1980. Effect of food intake and fasting on gastrointestinal lead absorption in humans. Am J Clin Nutr 33:1784-1788.
Richardt G, Federolf G, Habermann E. 1986. Affinity of heavy metal ions to intracellular Ca2+-binding proteins. Biochem Pharmacol 35:1331-1335.
Rosen JF, Chesney RW, Hamstra A, DeLuca HF, Mahaffey KR. 1980. Reduction in 1,25-dihydroxyvitamin D in children with increased lead absorption. N Engl J Med 302:1128-1131.
Saijo T, Ito M, Takeda E, Huq AH, Naito E, Yokota I, et al. 1991. A unique mutation in the vitamin D receptor gene in three Japanese patients with vitamin D-dependent rickets type II: utility of single- strand conformation polymorphism analysis for heterozygous carrier detection. Am J Hum Genet 49:668-673.
Sayre JW, Katzel MD. 1979. Household surface lead dust: its accumulation in vacant homes. Environ Health Perspect 29:179-182.
Schwartz BS, Stewart WF, Kelsey KT, Simon D, Park S, Links JM, et al. 2000. Associations of tibial lead levels with BsmI polymorphisms in the vitamin D receptor in former organolead manufacturing workers. Environ Health Perspect 108:199-203.
Schwartz J. 1994. Low-level lead exposure and children's IQ: a meta-analysis and search for a threshold. Environ Res 65:42-55.
Schwartz J, Otto D. 1991. Lead and minor hearing impairment. Arch Environ Health 46:300-305.
Six KM, Goyer RA. 1970. Experimental enhancement of lead toxicity by low dietary calcium. J Lab Clin Med 76:933-942.
Succop P, Bornschein R, Brown K, Tseng CY. 1998. An empirical comparison of lead exposure pathway models. Environ Health Perspect 106(suppl 6):1577-1583.
Watson WS, Hume R, Moore MR. 1980. Oral absorption of lead and iron. Lancet 2:236-237.
Whitfield GK, Remus LS, Jurutka PW, Zitzer H, Oza AK, Dang HT, et al. 2001. Functionally relevant polymorphisms in the human nuclear vitamin D receptor gene. Mol Cell Endocrinol 177:145-159.
Willett W, ed. 1990. Nutritional Epidemiology. Oxford, NY:Oxford University Press.
Wright RO, Shannon MW, Wright RJ, Hu H. 1999. Association between iron deficiency and low-level lead poisoning in an urban primary care clinic. Am J Public Health 89:1049-1053.
Ziegler EE, Edwards BB, Jensen RL, Mahaffey KR, Fomon SJ. 1978. Absorption and retention of lead by infants. Pediatr Res 12:29-34.
Last Updated: September 16, 2003 |
|
 |
|
| |

