Over the past 15 years, an ever-increasing number of epidemiologic studies
have shown significant associations between the mass concentration of ambient
air particulate matter (PM) and adverse respiratory and cardiovascular health
effects. These effects include PM concentration-related excess rates of daily
and annual mortality, hospital admissions, emergency room and clinician visits
for respiratory and cardiac diseases, increased use of medications, and time
lost from work and school. By the mid-1990s, the evidence for these associations
was sufficiently compelling for the U.S. Environmental Protection Agency (U.S.
EPA) to propose revised and more stringent National Ambient Air Quality Standards
(NAAQS) for PM (NAAQS 1997). Their form and stringency were endorsed by the
U.S. EPA Clean Air Scientific Advisory Committee (CASAC), a group of external
scientific advisors whose charter was established by the Clean Air Act Amendments
of 1977 (1991).
The revised PM NAAQS, promulgated in July 1997, retained with minor modification
the previous daily maxima and annual average PM NAAQS for PM whose aerodynamic
diameters were < 10 µm (PM10). It also established new PM
NAAQS for particles with aerodynamic diameters < 2.5 µm (PM2.5),
as excess mortality was found to be more strongly associated with PM2.5
than with PM10.
The U.S. EPA (and CASAC) acknowledged that the database supporting the judgments
that PM2.5 and PM10 exposures were likely causal factors
for adverse health effects was not fully supported by, or consistent with, available
knowledge of the underlying biological mechanisms. Among the likely factors
for discrepancies between observations in human populations and corresponding
observations in controlled animals and human studies are the following:
- Epidemiologic observations of adverse effects had largely been confined
to subpopulations who may have been especially susceptible because of underlying
preexisting disease or who were very young or very old. In contrast, because
of practical and ethical considerations, most human clinical studies had examined
effects in small groups of healthy individuals of intermediate ages. Similarly,
most previous animal studies focused on healthy and younger animals and used
exposures to concentrations that were much higher than those encountered in
ambient air.
- Toxicological studies have been limited, as the most active components
of PM remain a matter of speculation. Subsequent studies would need to use
more realistic ambient mixtures, e.g., concentrated ambient air particles
(CAPs) or laboratory-generated surrogates that focus on specific particle
characteristics such as particle size [e.g., ultrafines (diameters less than
0.1 µm)] and/or chemistry [e.g., metals, polycyclic aromatic hydrocarbons
(PAHs), quinones]. In addition, it is possible that responses may require
a mixture of PM components and/or the simultaneous or sequential exposure
to gaseous pollutants in the ambient air mixture [e.g., SO2, NO2,
O3, CO, and volatile organic compounds (VOCs)].
- No laboratory-based toxicological studies have been conducted involving
chronic or even subchronic exposures to ambient air PM mixtures at concentrations
at the upper end of the range of current U.S. ambient air concentrations.
As a result of the remaining scientific uncertainties, in 1998 Congress directed
the U.S. EPA to substantially increase its level of funding on PM health effects
research. It also mandated that a National Research Council (NRC) committee
(i.e., the Committee on Research Priorities for Airborne Particulate Matter)
be established to provide scientific oversight for the PM research. In the first
of its three reports, the NRC Committee on Research Priorities for Airborne
Particulate Matter recommended a multiyear research program that included the
establishment of academically based research centers to create a comprehensive
and integrated particle health effects research program. The PM centers were
intended to foster interdisciplinary collaborations within and among institutions
with extensive experience in air pollution health effects research. Research
that arose through these collaborations were in turn intended to help the U.S.
EPA address scientific issues about PM health effects in a timely and effective
manner.
The U.S. EPA through its Science to Achieve Results (STAR) program for investigator-initiated
research issued a request for proposals (RFPs) for centers for PM health effects
research in 1998. It received and peer-reviewed 21 applications, and awarded
five center grants in the summer of 1999. The successful applicants were (in
alphabetical order)
- A California consortium, headquartered at University of California
Los Angeles (UCLA; Los Angeles, California)
- Harvard University (Boston, Massachusetts)
- New York University (NYU; New York, New York)
- The University of Rochester (Rochester, New York)
- The University of Washington (Seattle, Washington).
The U.S. EPA in its RFP specified that a) each center have an
external scientific advisory committee to ensure that its research program addresses
important knowledge gaps and is scientifically sound (each of these committees
include members from academic institutions, industrial companies, public interest
groups, and some U.S. EPA scientists); b) center directors have an annual
meeting with U.S. EPA scientists and administrators for the exchange of information,
progress, and future plans, and for coordination of collaborative research;
and c) each center have an outreach program to communicate to the public
current knowledge of air pollution health effects and research progress being
made to address knowledge gaps.
The centers have addressed these requirements and initiated collaborative
activities. These include sponsorship of scientific workshops to further research
in key areas such as characterizing cardiac health effects associated with PM
exposures, assessing costs and health benefits of air pollution controls, examining
the health impacts of gasoline emissions in California, and apportioning PM
sources and their associated health effects.
Through their individual and collective activities during the initial years
of the PM health effects research centers, real progress has been made toward
the understanding of ambient air pollution health effects and addressing areas
of remaining scientific uncertainty. The PM centers plan to continue to conduct
both epidemiological and toxicological studies to enhance our understanding
of the health effects associated with ambient PM exposures. Of particular interest
will be investigations of chronic PM health effects. This research objective
is consistent with the U.S. EPA multiyear plan for PM research as defined in
its November 2001 presentation to the NRC Oversight Committee.
This report provides a synopsis of the centers' research accomplishments to
date. Many of the research studies presented received major support from PM
center funds. For others, primary support was from other sources, such as the
National Institute of Environmental Health Sciences (NIEHS) and the Health Effects
Institute (HEI), with the availability of PM center core services and interactions
with other PM center investigators providing important supplemental support.
This report consists of six sections. Sections 1-5 address issues relating
to 1) biological mechanisms, 2) acute effects, 3) chronic effects, 4) dosimetry,
and 5) exposure assessment. Section 6 summarizes the features of the PM centers
program that have facilitated the harnessing of the skills and resources of
the PM center investigators for collaborative research addressing the U.S. EPA
PM NAAQS development. Each of the five key area sections begins with background
summarizing the challenge faced by the PM centers as they began their centers
program in terms of what was known and what goals were established to address
key knowledge gaps. These background summaries are followed by an outline of
progress that has been and is being made by PM center investigators in resolving
knowledge gaps. The report also outlines short-term goals (during the 2.5 remaining
years of center support) and long-term goals (beyond the initial 5 years of
center support) for the PM centers.

Figure 1. PM center integration
of research disciplines. |
The PM centers' approach has been highly interactive both within each center
and across centers, combining research efforts on exposure/source characterization,
epidemiological and toxicological effects, and biological mechanisms summarized
in Figure 1. Continuous interdisciplinary dialogue among the PM center scientists
assures that the total breadth of PM research is covered and that new research
ideas have a sound scientific basis. This is accomplished partly through periodic
meetings among center members from the different scientific disciplines represented
in the centers. The exposure assessors provide data on specific ambient air
PM constituents to toxicologists; toxicologists provide information on the relative
toxicity of PM constituents to exposure assessors and epidemiologists; the in
vitro toxicologists share insights with epidemiologists, clinicians, and
animal toxicologists to help elucidate PM-specific response mechanisms. This
kind of collegial interaction and sharing of research within and among the centers
is a special feature of the PM centers program. The five research topics listed
in the center section of Figure 1 are the areas jointly addressed by the five
centers to obtain results useful for the regulatory needs of the U.S. EPA. Examples
of progress in these areas are provided in this special report.
Biological Mechanisms for Particulate Matter Health Effects
Background
Justifications for the 1997 PM10 and PM2.5 NAAQS were
based primarily on a large and coherent epidemiological database of significant
associations between ambient air PM concentrations and excess mortality and
morbidity. Although the 1996 PM criteria document provided some support for
biological plausibility of causal links between PM and health effects, evidence
from controlled human and animal exposure studies was still largely unavailable.
On the basis of this information gap, a major rationale for the establishment
of the PM centers was to explore biological plausibility and mechanisms of PM-associated
health outcomes. Over the past 2 years, substantial contributions have been
made toward developing hypotheses based on experimental observations, in large
part because of the research environment fostered by the structure of PM centers.
The following section provides examples of how the PM centers program has contributed
to the development and testing of plausible mechanistic hypotheses for the health
effects of PM.

Figure 2. Hypotheses for
health effects of PM. |
Recently, several mechanistic pathways have been investigated that may link
PM exposure with adverse health effects. Figure 2 highlights the complexity
and interdependency of some of these pathways (Utell and Frampton 2000). The
portal of entry for PM air pollution is the lung, and PM interactions with respiratory
epithelium likely mediate a wide range of effects, as indicated by the central
oval in Figure 2. These include respiratory as well as systemic and cardiovascular
effects. However, PM, or its reaction products, may stimulate airway sensory
nerves, leading to changes in lung function and in autonomic tone, which influence
cardiac function. Ultrafine particles by virtue of their extremely small size
may enter pulmonary capillary blood and be rapidly transported to extrapulmonary
tissues such as liver, bone marrow, and heart, with either direct or indirect
effects on organ function.
Progress in Biological Mechanism Research
This section is a discussion of mechanistic areas in which particular progress
has been made by the PM centers in inflammation and immunity, mechanisms for
cardiovascular effects, and the role of reactive oxygen species (ROS). This
is not intended to be a comprehensive review but rather a summary of center
research programs. Many important studies, particularly in vitro studies
of PM mechanisms, are not addressed.
Inflammation and immunity. Airway injury and inflammation is
a well-known consequence of toxic inhalation exposures. Previous studies involving
animal models have shown that instillation or inhalation of particles such as
diesel exhaust particles (DEPs) can cause inflammation and epithelial injury
at high doses and concentrations. However, there was little evidence prior to
the PM centers program that exposure to ambient concentrations of PM caused
significant airway inflammation. The presence or absence of an inflammatory
response is an important issue because inflammation may induce systemic effects,
including an acute-phase response with increased blood viscosity and coagulability
and possibly an increased risk for myocardial infarction in patients with severe
coronary artery disease. In chronic respiratory diseases such as asthma and
chronic obstructive pulmonary disease (COPD), inflammation is a key pathophysiologic
feature. Chronic, repeated inflammatory challenge of the airways may result
in airway remodeling that leads to irreversible lung disease. Thus, inflammation
may be involved in both acute and chronic effects.
Following is a list of several key findings from PM center research about
the role of inflammation and immunity in mediating the health effects of PM.
Particulate matter exposure and systemic markers of inflammation in humans.
The PM centers in southern California and Rochester, New York, have collaborated
in human clinical studies using identical crossover exposure protocols, subject
recruitment criteria, and outcome measures. The California studies used CAPs
at approximately 200 µg/m3, and Rochester used laboratory-generated
carbonaceous ultrafine particles at two concentrations--10 and 25 µg/m3.
Preliminary findings from both of these studies provide evidence for effects
on systemic markers of inflammation and leukocyte recruitment (Boscia et al.
2000; Daigle et al. 2002; Frampton et al. 2001).
In both centers, subjects were exposed for 2 hr with intermittent exercise.
Before and at intervals after exposure, symptom ratings, lung function testing,
and phlebotomy were performed. Sputum induction was performed approximately
24 hr after exposure. In both centers, blood was analyzed for markers of systemic
inflammation, acute-phase response, and blood coagulability. One such marker
is soluble intercellular adhesion molecule-1 (sICAM-1), a transmembrane protein
expressed on leukocytes and endothelial cells. sICAM-1 plays an important role
in monocyte recruitment to atherosclerotic lesions and inflamed airways, where
it is shed into plasma during leukocyte adhesion.
In the southern California CAPs studies, sICAM-1 increased significantly (p
< 0.05) after exposure to CAPs (relative to filtered-air controls) for the
pooled healthy and asthmatic population. The effect was greater at 21 hr after
exposure than at 4 hr. In the Rochester ultrafine particle studies, no significant
change was found in sICAM-1 in the plasma of healthy subjects, but blood monocyte
expression of ICAM-1 decreased immediately following particle exposure in a
concentration-response fashion compared with air exposure. These studies
suggest that exposures to either CAPs or ultrafine particles may initiate endothelial
and leukocyte activation, with shedding of surface ICAM-1, a key initial step
in leukocyte recruitment. These findings may have implications for cardiovascular
and respiratory disease. In a major cardiac epidemiological study, plasma sICAM-1
levels were predictive of future coronary events (Ridker et al. 1998).
Particulate matter exposure and inflammation in animals. Studies in
normal dogs exposed to Boston CAPs by inhalation showed increases in pulmonary
inflammation by bronchoalveolar lavage and in circulating blood neutrophils
associated with increases in specific ambient particle components. In these
experiments, mean exposure doses were 203 and 361 µg/m3 in the
lavage and blood studies, respectively (Clarke et al. 2000). These PM center
studies show that ambient particle components have significant pulmonary and
systemic inflammatory potential. Similar findings have been reported in rats
in PM center studies (Saldiva et al. 2002).
Effects of aging. Determining the mechanisms involved in increased
susceptibility to PM comprises another goal of PM center research being explored
at many levels. The role of aging is being examined, using animal and human
exposure studies as well as in vitro models. Studies were conducted examining
cytokine production by alveolar macrophages from aged rats and mice (> 20
months of age) after in vitro exposure to lipopolysaccharide (LPS) and
PM. Macrophages from aged animals were incubated with endotoxin, with laboratory-generated
mixed carbon/iron ultrafine particles, or with both. Baseline production of
cytokines was elevated 30-50% in aged cells compared with cells from young
animals (8-10 weeks of age). The response to LPS was enhanced at every
dose in aged cells. The response to ultrafine particles containing iron was
enhanced 2- to 3-fold in aged cells compared with young cells. Most significantly,
in the aged animals, coadministration of ultrafine particles and LPS led to
synergistic effects at the lowest ultrafine particle dose, whereas in young
cells this was observed only at the highest PM dose (Finkelstein et al. 2002).
These findings suggest a cellular basis for age-related increased susceptibility
that may relate to increased susceptibility to oxidative stress. Alternatively,
the results may be showing a lower threshold because of impaired protective
mechanisms (e.g., antioxidant defenses). Investigations of these age-related
differences are currently focusing on signal transduction mechanisms.
Effects on infection/pneumonia. Epidemiological studies have demonstrated
that infection, specifically pneumonia, contributes substantially to the increased
morbidity and mortality among elderly individuals after exposure to PM (Schwartz
1994), suggesting that inhaled PM can act as an immunosuppressive factor that
undermines normal host pulmonary immune responses. A combination of particle
concentrator technology and animal infectivity models is being used to investigate
this hypothesis. A single 5-hr inhalation exposure of bacterially infected rats
to New York City CAPs, at concentrations ranging from 65 to 150 µg/m3,
altered both pulmonary and systemic immunity and exacerbated the infection process
in a time-dependent manner (Zelikoff et al. 1999). Streptococcus pneumoniae-infected
rats exposed to PM demonstrated increased burdens of pulmonary bacteria, numbers
of circulating white blood cells, extent of pneumococcal-associated lung lesions,
and incidence of bacteremia, compared with air-exposed, infected control rats.
Conversely, this same PM exposure resulted in decreased levels of lavageable
polymorphonuclear neutrophils (PMNs), bronchus-associated lymphoid tissue, and
proinflammatory cytokines [i.e., tumor necrosis factor-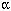 ,
interleukin (IL)-1, and IL-6] in infected rats. Subsequent studies implicated
the iron content in mediating these effects; many of the findings were reproduced
with nose-only exposure to soluble iron but not with soluble forms of other
metals (manganese, copper, or nickel). These findings suggest that PM exposure,
and specifically the soluble iron component, may affect the host immune response
during pulmonary infection and may help explain epidemiological observations.
In addition to the effects of iron, PM center investigators have shown that
DEPs and CAPs induce apoptosis in macrophages by an oxidative stress mechanism
that is dependent on the organic chemicals in the PM. Macrophage apoptosis will
lead to decreased phagocytic defenses in the lung.
,
interleukin (IL)-1, and IL-6] in infected rats. Subsequent studies implicated
the iron content in mediating these effects; many of the findings were reproduced
with nose-only exposure to soluble iron but not with soluble forms of other
metals (manganese, copper, or nickel). These findings suggest that PM exposure,
and specifically the soluble iron component, may affect the host immune response
during pulmonary infection and may help explain epidemiological observations.
In addition to the effects of iron, PM center investigators have shown that
DEPs and CAPs induce apoptosis in macrophages by an oxidative stress mechanism
that is dependent on the organic chemicals in the PM. Macrophage apoptosis will
lead to decreased phagocytic defenses in the lung.
Cardiovascular effects. Determining the mechanisms linking ambient
PM to cardiovascular effects is one of the key challenges of the PM centers.
There is growing clinical and epidemiological evidence that ambient air pollution
can precipitate acute cardiac events such as angina pectoris, cardiac arrhythmias,
and myocardial infarction, with the majority of excess PM-related deaths attributable
to cardiovascular disease (CVD). The PM centers' approach to this issue is multifaceted
and multidisciplinary. There are ongoing panel studies of susceptible subjects
involving cardiovascular monitoring at three PM centers, animal exposure studies
at four PM centers, and human clinical studies at three PM centers. Three PM
centers share a cardiac monitoring and analysis protocol for human clinical
studies.
A major step forward in this area was made with the convening of a workshop,
"Cardiovascular Effects of Air Pollution: Potential Mechanisms and Methods of
Testing," which met in Rochester, New York, in March 2001. The proceedings have
now been published (Utell et al. 2002), and important research needs in this
area have been identified. The workshop featured presentations by PM center
and other investigators, along with clinical and research cardiologists. Each
of the five U.S. EPA PM centers participated in the workshop along with representatives
from the U.S. EPA. New hypotheses and research directions were developed, and
practical issues of cardiac monitoring methodology currently in use at each
PM center were reviewed and optimized. This workshop grew directly from one
of the annual PM center directors meetings, where the need for intercenter collaboration
on this issue was identified. The interchange of ideas served as an important
turning point in our thinking about the mechanisms involved in cardiac effects,
and new collaborative efforts were initiated.
Key observations in both human and animal studies of cardiovascular effects
have been made since the PM center program was initiated. A few examples follow.
Human studies. Investigation of cardiovascular effects of PM has required
multidisciplinary collaboration. For human exposure studies, analysis of
cardiac monitoring includes a detailed analysis of heart rate variability (HRV)
and repolarization intervals before, during, and for a period of 48 hr after
exposure. In one study (Frampton et al. 2002), healthy subjects were exposed
by mouthpiece for 2 hr with intermittent exercise on three separate occasions
to air or ultrafine carbon particles at 10 and 25 µg/m3. Frequency-domain
analysis of the continuously recorded electrocardiogram (EKG) indicated that
response of the parasympathetic nervous system was blunted during recovery from
exercise immediately after exposure to ultrafine particles, compared with air.
This diminished vagal response was not observed 3.5 hr later. Monitoring also
indicated that exposure to ultrafine particles altered cardiac repolarization,
as indicated by the corrected QT interval (QTc) on the cardiogram. The increase
in QTc following exercise during air exposure was blunted with PM exposure,
and this persisted to at least 21 hr after exposure. This change in repolarization
was not explained by changes in heart rate (Frampton 2001; Zareba et al. 2001).
It is plausible that ultrafine particle exposure imposes an effect on repolarization,
either through an indirect effect via the autonomic nervous system or by directly
affecting ion channel function in ventricular myocardium through a yet unknown
mechanism. This observation is important because changes in the QT interval
have been implicated in susceptibility to cardiac arrhythmias in patients with
heart disease.
These human clinical studies are complemented by a major panel study involving
patients with preexisting coronary artery disease in Erfurt, Germany. Analysis
of the EKG recordings and blood parameters are under way, including detailed
analyses of HRV and repolarization, and acute-phase proteins using methodology
identical to the human clinical studies. These clinical/epidemiological/toxicological
collaborations are examples of how the PM centers program has fostered research
among diverse disciplines and locales.
Animal studies. Rats and mice are being instrumented for continuous
cardiac and blood pressure monitoring. Algorithms have been developed for analysis
of heart rate and blood pressure variability using continuous 24-hr recordings
in up to eight rodents simultaneously (Couderc et al. 2002). A crossover study
with aging, spontaneously hypertensive rats exposed to carbonaceous ultrafine
particles is ongoing.
PM center investigators are evaluating both inflammation and cardiovascular
effects in animal models. The hypothesis being tested is that inhaled PM causes
release of inflammatory mediators from cells in the lung that then become bloodborne
and target the cardiovascular system. The research plan uses transgenic mouse
strains with specific cardiovascular genetic alterations to create susceptibility
models. Initial studies have been using a mouse model of atherosclerosis, the
apolipoprotein E-deficient mouse generated via a targeted disruption of
the mouse apo-E gene. The deficiency of apo-E leads to a spontaneous
hypercholesterolemia. The nonhypertensive animals form atherosclerotic lesions
throughout the vasculature, which resemble, in part, human atherosclerotic lesions
at 3-5 months of age. Animals are instrumented and monitored for blood
pressure and heart rate using radiotelemetry. Individual mice were dosed with
125 µg Washington, DC, urban dust in 50 µL saline by oropharyngeal
aspiration into the lungs. Heart rate was reduced after exposure to PM in normal
and in apoE-/- mice. ApoE-/- mice showed a trend to increased blood pressure
and increased variability of blood pressure after PM exposure that did not differ
significantly from that of the normal mice (Luchtel et al. 2002). Additional
experiments are being performed as the control and apoE-/- mice age, and studies
involving more realistic exposures are being planned.
In another PM center study, male Fischer 344 rats 18 months of age with implanted
EKG transmitters were used to determine the effects of PM on the frequency of
spontaneous arrhythmias. As old rats (18 months of age) were found to have many
spontaneous arrhythmias, a standardized definition for each type of arrhythmia
was developed, and a procedure for quantifying the frequency of spontaneous
arrhythmias was established. Rats were exposed to New York City CAPs or filtered
air for 4 hr. The rats were exposed twice with a crossover design so each rat
could serve as its own control. The CAP concentration was 160 µg/m3
and 200 µg/m3 for the first and second exposures, respectively.
EKG tracings demonstrated a significant increase in the frequency of supraventricular
arrhythmias after exposure to CAPs compared with filtered air-exposed control
animals. The same rats were also exposed twice to 1 ppm SO2 and twice
to air in a repeated crossover design. No significant change in the frequency
of any category of spontaneous arrhythmia after exposure to SO2 (or
filtered air) was observed (Nadziejko et al. 2001).
The effects of PM on myocardial ischemia have also been the focus of PM center
research. Inhaled PM exacerbated ischemia in a clinically relevant model of
coronary artery occlusion in conscious dogs. Exposures to CAPs significantly
increased peak electrocardiographic ST-segment elevation during a 5-min coronary
artery occlusion compared with sham exposures in two different protocols using
conscious dogs (Godleski et al. 2000; Wellenius et al. 2003). The relationship
of ambient particle components to the degree of ischemia in dogs is the focus
of ongoing PM center research (Wellenius et al. 2003). Other PM center studies
have used an improved model of myocardial infarction in the rat and demonstrated
increased ventricular arrhythmias with aerosol exposure to oil fly ash particles
but not to carbon black particles (Wellenius et al. 2002). This model of myocardial
infarction is being used to study the effects of combinations of CAPs and carbon
monoxide. In other studies of the vascular response to inhaled particles, a
variety of cell and molecular biologic methods have been used.
Reactive oxygen species. A major finding has been that PM generates
ROS, which provide proinflammatory stimuli to bronchial epithelial cells and
macrophages. These cellular targets respond with cytokine and chemokine production,
which can enhance the response to allergens. PM may therefore act as an adjuvant
that strengthens the response of the immune system to environmental allergens.
Hallmarks of allergic inflammation include increased immunoglobin E (IgE) production,
eosinophilic bronchial inflammation, airway hyperresponsiveness, and an increase
of nitric oxide (NO) in exhaled air.

Figure 3. Thiol antioxidant
interferes in the adjuvant effects of DEPs during OVA sensitization in a
murine allergic inflammation model. |
This hypothesis is being tested using in vitro and animal studies (Whitekus
et al. 2002). In one study using an allergic mouse model, animals were exposed
at high doses to nebulized DEPs (2000 mg/m3) for 1 hr, followed by
nebulized antigen [ovalbumin (OVA)] for 20 min daily for 10 days. A control
group received saline instead of DEPs followed by OVA. To determine the role
of ROS, the same exposure groups were pretreated every day with intraperitoneal
N-acetylcysteine (320 mg/kg). Control animals received saline intraperitoneally.
Two days after the last exposure, blood was obtained and assayed for IgE and
IgG1, and the lungs were assayed for carbonyl proteins and lipid peroxides.
DEPs markedly enhanced the antibody response (Figure 3) and lipid peroxidation,
and these effects were abrogated by antioxidant treatment. Follow-up studies
using more realistic exposure levels of DEPs and/or CAPs are being planned.
Furthermore, human nasal challenge studies confirmed the role of DEPs as an
adjuvant in already established allergic responses, as well as in exposure to
neoallergens. Taken together, these findings may explain the increased number
and severity of asthma attacks in an urban setting after a surge in PM levels,
and may implicate DEPs as a factor in asthma exacerbations.
ROS associated with exposure to PM may play a role in cardiovascular effects.
9,10-Phenanthroquinone (9,10-PQ) is a potent inhibitor of neuronal form of nitric
oxide synthase (NOS). 9,10-PQ also inhibits the endothelial form of NOS, which
plays a critical role in vascular tone, thereby causing the suppression of NO-dependent
vasorelaxation of aorta and significant increase in blood pressure in rats.
Therefore, quinones and other compounds producing ROS, for example, nitro-PAHs,
may contribute to diseases related to vascular dysfunction caused by exposure.
In addition to the production of ROS, quinones, PAHs, nitro-PAHs, and related
compounds may also undergo electrophilic addition to macromolecules producing
complementary toxicity.
The key role of the PM centers in facilitating this line of investigation
on ROS has been the collaboration between scientists with diverse expertise.
For example, the collective expertise in the southern California PM center facilitated
the use of CAPs to replace DEPs for mechanistic in vitro and in vivo
studies. Asthma animal models are now being used to compare the prooxidative
and proinflammatory effects of CAPs collected on California freeways and various
source-receptor sites. In addition, human panel studies and CAPs exposure studies
now typically include the role of oxidative stress in airway inflammation (e.g.,
assays for NO and CO content in the expired air, and measures of cytokines in
induced sputum, blood, and breath condensate).
Another important development by the PM centers is collaboration among chemists,
engineers, and biologists in exploring how chemical constituents of CAPs contribute
to ROS generation and inflammation. An important observation has been that organic
components present in the organic carbon fraction generate ROS through their
ability to undergo redox cycling. The in vitro reactions correlate well
with the ability of organic PM components to generate oxidative stress in epithelial
cells and macrophages. Preliminary evidence indicates that PAHs and their oxidized
derivatives (quinones and ketones) play a key role in ROS generation at the
cellular level. The in vitro toxicity studies predict a hierarchical
or stratified oxidative stress response in which the biological effects range
from a) protective (e.g., expression of antioxidant enzymes); b)
proinflammatory (e.g., production of cytokines and chemokines); and c)
cytotoxic (e.g., cellular apoptosis and necrosis), depending on the level of
oxidative stress. The ability to relate the inherent redox-cycling and oxidative
stress capabilities of a PM sample to specific biological effects allows a more
rational interpretation of the in vivo toxicity data being generated
in the community, freeway, and source/receptor studies.
Short-Term Particulate Matter Research Goals in Biological Mechanisms
Objectives of the PM centers during the remaining funding period will include
a greater utilization of intercenter collaborations. In addition to the collaborative
efforts described in this section, new interactive center initiatives will elucidate
mechanisms of PM effects. These include sharing of PM collected from various
sources at different PM centers for use in animal and in vitro studies,
sharing of exposure technology, and developing common laboratory protocols.
An important objective will be to identify additional mechanistic links. For
example, neural pathways may play a role in mediating cardiovascular effects.
If studies currently under way confirm that PM affects autonomic regulation
of cardiovascular function, we will need to further define the mechanisms by
which those effects are initiated, including the cells and cell mediators responsible.
Research on these mechanisms will provide vital clues about individual susceptibility
and potential approaches to the prevention of adverse health effects.
Animal studies based on the hierarchical or stratified oxidative stress model
are being planned and will consider the possible identification of susceptible
human subjects with weakened oxidative stress defenses. This could involve polymorphisms
of the heme oxygenase 1 gene, which is a very sensitive antioxidant defense
mechanism that protects cells against redox-cycling DEPs chemicals and contributes
to CO and NO production during in vivo DEP exposure. The elucidation
of susceptible individuals who can be studied with rational end points will
enhance epidemiological studies and will also help to monitor the impact of
regulatory measures to reduce adverse health effects.
A continuing research objective is to further examine the role of PM composition,
size, surface area, gaseous co-pollutants, and other factors in mediating effects.
This involves a variety of experimental approaches, from detailed morphologic
and chemical analysis of ambient air PM to in vivo and in vitro
exposure studies using both CAPs and laboratory-generated PM of carefully defined
composition. A related longer-term goal is to determine the role of complex
mixtures in eliciting health effects using factor analysis to identify the sources
of PM and associated gaseous air pollutants.
Determining the mechanisms underlying individual susceptibility to PM effects
is another major objective under way in each of the PM centers. Host susceptibility
factors being investigated include age, gender, underlying disease, infections,
and genetic factors.
A key goal of PM centers research is to develop biologic markers of specific
mechanistic pathways that can be used to link findings from animal, human, and
epidemiological studies. Many examples are currently being investigated, including
plasma sICAM-1 and monocyte ICAM-1 as indicators of enhanced leukocyte endothelial
interaction, and measurement of changes in HRV as a measure of cardiovascular
autonomic effects.
Long-Term Particulate Matter Research Goals in Biological Mechanisms
One major long-term objective of the PM centers is to determine the mechanisms
involved in chronic health effects of ambient PM exposure. Epidemiological studies
have indicated that exposure to PM2.5 leads to a shortening of life
span, and this finding was a major impetus in the establishment of the annual
average PM2.5 NAAQS. Does PM2.5 exposure exacerbate underlying
disease or contribute to the genesis of disease (or both)? Some of the key findings
currently being investigated in the PM centers, and summarized here, have implications
for chronic (long-term) effects.
For example, one hypothesis is that recurrent inflammatory or allergic challenges
to the airway leads to airway remodeling, a key feature in the development of
irreversible airways obstruction in asthma and COPD. Second, there is growing
evidence that atherosclerotic CVD is an inflammatory process. PM centers are
testing the hypothesis that immune or inflammatory effects of PM exposure may
promote or accelerate atherosclerosis. Third, diabetes is associated with severe,
accelerated atherosclerotic vascular disease and increased susceptibility to
infection, and a recent study identified diabetics as particularly susceptible
to the health effects of PM. Determining the mechanisms involved in this susceptibility
of diabetics may help shed new light on PM effects mechanisms in general.
Ambient air PM contains carcinogens, and recent epidemiological evidence indicates
that mortality due to lung cancer is increased in relation to PM2.5
exposure. Investigating the PM components and mechanisms for carcinogenesis
will be an important long-term goal of the PM centers.
Finally, major long-term goals of the PM centers are to contribute new scientific
data and risk assessment tools and information that will assist in refining
air quality standards for PM, and to evaluate the public health benefits of
reductions in PM exposure. This will involve more comparisons of PM potency
across animal, clinical, and in vitro end points coupled with ever-increasing
specificity as to aerosol characteristics, leading to better indicators of biological
mechanism and links of particular sources of PM to specific health effects.
Acute Health Effects of Particulate Matter
Background
The acute health effects of PM exposures have been extensively examined by
a large number of epidemiological studies conducted worldwide. These studies
have consistently shown significant associations between daily average ambient
PM concentrations and corresponding cardiopulmonary mortality, morbidity, and
functional impairments. When the U.S. EPA promulgated its 24-hr PM2.5
NAAQS in 1997, it relied primarily on this large body of epidemiological data
relating PM exposures to daily deaths and hospital admissions. However, critics
of the revised PM NAAQS raised a number of key issues in their challenges to
the credibility of, or need for, a new 24-hr PM2.5 NAAQS, such as
a) the associations represented deaths of frail individuals whose deaths
were brought forward by only a few days or weeks (harvesting), and thus had
little public health significance; b) particles originating from different
sources have varying toxicities; therefore, the relative health impacts of particle
sources should be assessed prior to regulating their emissions; c) the
associations were potentially confounded by season, weather, and other gaseous
pollutants; d) the associations were implausible because ambient PM2.5
concentrations were not appropriate surrogates of personal PM2.5
exposures; e) the associations were implausible because there was limited
support from controlled human and animal studies; f) the identification
of populations susceptible to PM2.5 health effects is necessary prior
to promulgating a new PM2.5 standard; and g) the exposure-response
curves, showing no thresholds, were unlikely from a biological standpoint.
Many of these issues have been addressed in recent epidemiological and toxicological
investigations examining the acute health effects associated with short-term
PM exposures. Collaborations between epidemiological and toxicological communities
have led to the development of common study hypotheses and common health end
points such as lung function, arterial oxygen saturation, heart rate, HRV, blood
pressure, tissue biomarkers of effects, exhaled nitric oxide (eNO), cardiac
dysrhythmias, and respiratory symptoms. As a result, our understanding of ambient
PM acute health effects has been advanced. For example, results from recent
controlled laboratory studies have shown that short-term exposures to CAPs,
compared with artificially generated particles, result in acute health effects
comparable to those reported in the epidemiological literature. These toxicological
findings provided some evidence about biological plausibility and mechanisms
that will be of paramount importance to our efforts to examine the validity
of the observed epidemiological study findings.
Progress Made in Acute Effects Research
Since the establishment of the PM centers, substantial progress has been made
in our understanding of PM health effects. Collectively, the PM centers have
addressed a large number of scientific issues regarding acute PM health effects.
These investigations are broadly categorized as either observational or controlled
studies. It has recently been reported that the S-Plus Software (MathSoft, Inc.,
Cambridge, MA) fails to adequately meet the convergence criteria nominally applicable
to multiply smoothed data in studies of acute health effects of air pollution
(Dominici et al. 2002). Center investigators who have used this software have
reanalyzed many of their recent studies using the new default criteria of Dominici
et al. (2002), and although small changes in effect size estimates have been
made, none of them necessitated changes in the conclusions drawn from these
studies.
Observational studies. Harvesting. If air pollution-related
deaths mainly affect already frail or sick individuals whose deaths are being
brought forward by only a few days (the harvesting hypothesis), then PM-related
mortality would have little or no importance on total mortality rates. Studies
examining the harvesting hypothesis were conducted by the PM centers. Using
a series of moving averages of mortality and exposure data (7-day, 15-day, etc.),
Schwartz (2000a) examined whether the strength of the associations changed over
the various averaging periods. On the basis of the harvesting hypothesis, one
would expect that estimated PM-mortality associations would become weaker as
the averaging periods increased. However, the results from this analysis showed
that the association between PM10 and mortality remained significant,
and the estimated relative risks were, in fact, higher for the longer averaging
periods. Similar results were found for hospital admissions data (Schwartz 2001).
An alternate analytical approach was used in a 10-city meta-analysis study,
where averaging periods were increased incrementally from 1 to 45 days. Results
from this analysis showed that the PM effect increased by a factor of 2.5, again
suggesting significant shortening of life (Zanobetti et al. 2002). Similar analyses
are currently being conducted to examine cause-specific mortality.
Threshold/exposure response. Analytical methods were developed to combine
smoothed exposure-response curves from multiple locations to examine whether
a threshold in the PM10 and daily death relationship exists. Results
from multiple cities in the United States and Spain suggest that the PM10-mortality
relationships are linear down to the lowest observed exposure concentrations,
supporting the no-threshold hypothesis (Schwartz and Zanobetti 2000; Schwartz
et al. 2001). Similar results were found in subsequent studies of PM2.5
and mortality in six U.S. cities and of PM10 and hospital admissions.
In the PM10-hospital admissions study, the methodology was modified
to examine sources of heterogeneity in the exposure-response relationship
by calculating a random slope for each city. In the near future, we plan to
apply the random slope model to mortality data and to examine other responses
to PM exposures, such as EKG changes.

Figure 4. Association of
motor vehicle PM10 and CVD hospital admissions. The size of
the symbol reflects the size of the population group studied.
|
Particle-specific associations. PM center investigators have examined
the relationship between mortality and source-specific PM concentrations. Laden
et al. (2000) used source-apportionment techniques to group elemental PM concentrations
from six U.S. cities into a small number of categories, or "factors." These
factors were attributed to different PM sources such as vehicular emissions,
oil combustion, and soil. For each factor, a daily score was calculated. Subsequently,
excess daily mortality was regressed against the daily scores using multiple
regression analyses. Significant associations were found between mortality and
the traffic and coal combustion factors, with the largest effect size for the
traffic factor. No significant associations were observed for the oil and soil
factors. Mar et al. (2000) applied factor analysis to PM2.5, PM10,
PM10-2.5, sulfates, non-soil PM2.5, organic
carbon, elemental carbon, total carbon, gaseous co-pollutants, and cardiac mortality
data in Phoenix, Arizona. Results from this study showed that combustion-related
pollutants and sulfates were associated with cardiovascular mortality. Source-specific
effects were also examined by Janssen et al. (2002), who used source emission
and home characteristics to explain observed variability in the city-specific
PM10-hospital admissions coefficients. As shown in Figure 4, the
PM10 coefficient for CVD-related hospital admissions increased with
the fraction of PM10 emissions from traffic-related sources, suggesting
higher relative risks from PM10 for cities with a greater number
of traffic-related PM10 sources.
Susceptibility. Several studies have shown higher PM-associated health
risks for susceptible subpopulations such as the elderly or those with existing
cardiopulmonary diseases. PM center investigators have attempted to identify
important susceptible subpopulations. Results from these studies have shown
that socioeconomic factors and race do not affect susceptibility to PM-associated
mortality (Zanobetti and Schwartz 2000); however, females were shown to be at
greater risk.
The elderly have long been thought to be particularly susceptible to PM pollution.
Consequently, several panel studies have focused on examining the impact of
PM exposures on the elderly. In Seattle, Washington, the relationship between
outdoor PM concentrations and cardiopulmonary health effects was examined for
three panels of elderly subjects (healthy, with CVD, and with COPD). Results
from this study showed that during paced breathing exercises, an increase in
outdoor PM2.5 (lagged by 1 hr) was associated with a decrease in
the median of high frequency heart rate variability (HF HRV) in subjects with
CVD (Sullivan et al. 2002). Decreases in HF HRV were also observed for 4- and
24-hr lagged periods. No effects were observed either for healthy subjects or
those with COPD. However, results from these studies have not been entirely
consistent, as a recent study showed no significant association between PM2.5
exposure (0- to 2-day lag) and coached FEV1 (forced expiratory volume
in 1 sec) for subjects with COPD or CVD. In contrast, local outdoor PM2.5
concentration was associated with a decrement in FEV1 for normal
subjects without reported COPD or CVD (Trenga et al. 2002).
Other PM center-sponsored studies examining whether individuals with
preexisting disease are at increased risk from PM exposures are currently under
way. Data collection and analyses for a study in Erfurt, in eastern Germany,
of patients with preexisting CVD or COPD is currently being conducted to examine
whether ultrafine particles exacerbate CVDs through different mechanisms than
PM2.5 (Wichmann and Peters 2000). Analyses of 683 EKG recordings
and blood parameter analyses are ongoing. Fieldwork for the study on COPD patients
started in October 2001 and was completed in spring 2002.
Children may also be particularly susceptible to PM exposures. Koenig et al.
(2002) conducted a study of 16 children (6-12 years of age) with mild to
moderate asthma living in Seattle. Daily samples of eNO, a marker of pulmonary
inflammation, were collected for each of these children for up to 10 consecutive
days. Results from this study showed that an increase in PM2.5 was
associated with an increase in eNO, suggesting that PM2.5, at ambient
concentrations, can act as an inflammatory agent.
In an additional study conducted in Seattle, 133 children (5-13 years
of age) with asthma were observed for an average of 58 days (range 28-112
days) while they were screened for enrollment in the Childhood Asthma Management
Program study (Yu et al. 2000). In single-pollutant models, the population average
estimates indicated an increase of 18% in the odds of an asthma symptom corresponding
to a 10 µg/m3 increment in same-day PM10 and an increase
of 11% for PM10 lagged 1 day.
More recently, diabetics have been identified as an important susceptible
population. In several single-city studies, the risk of PM-associated hospital
admissions for heart disease for diabetics was double that for the general population
(Zanobetti and Schwartz 2001, 2002). In addition, diabetics had an increased
risk of PM-associated mortality (Bateson and Schwartz 2001). Individuals with
other preexisting diseases were also at higher risk. Respiratory illness, for
example, modified the risk of cardiovascular hospital admissions, whereas heart
failure was found to modify the risk of COPD hospital admissions (Zanobetti
et al. 2000).
Confounding. A new hierarchical model was used to examine the potential
confounding effect of gaseous co-pollutants (Schwartz 2000b). The results of
this study suggest that the association between PM10 and daily deaths
was not confounded by gaseous air pollutants. A subsequent examination of the
season-specific relationship between PM and mortality data for Philadelphia,
Pennsylvania, confirmed that mortality was predominantly associated with PM2.5
and not SO2 (Schwartz 2000b). Additional information about confounding
was provided by exposure studies, which showed that ambient gaseous air pollutant
concentrations are not well correlated with their respective personal exposures
but are significantly correlated to personal PM exposures, suggesting that the
ambient gaseous concentrations are acting as surrogates for personal exposures
to PM (Sarnat et al. 2001; also see "Exposure Assessment").
Controlled animal and human studies. A large number of controlled
exposure studies in humans and animals have been conducted by the PM centers
in an effort to investigate the acute biological responses induced by PM exposures.
Some of these studies have used laboratory-generated PM, whereas others have
relied on particle concentrators developed by aerosol scientists at the PM centers
to specifically serve the needs of their health effects colleagues. This makes
it possible to simulate upper-bound PM2.5 concentrations on a regular
and prolonged basis to examine the cardiac and pulmonary function, response
biomarkers, and other health end points before, during, and after exposures.
Results from these studies have repeatedly shown associations between PM2.5
exposures and a variety of acute responses, providing direct evidence that PM2.5
is biologically active at current peak exposure levels. As described in "Biological
Mechanisms for Particulate Matter Health Effects," health outcomes that may
warrant further examination in epidemiological studies include heart rate, HRV,
QTc interval changes, arrhythmias and ischemia, blood ICAM-1 and IL-6, and oxidant
stress markers. Some brief descriptions follow controlled acute health effects
of animals and humans conducted by the PM centers.

Figure 5. Responses of mice
exposed approximately 50 meters downwind of freeway to CAPs and OVA. Group
sizes = 9. Error bars represent mean ± SE.
*Significantly different from control (p < 0.05).
|
Animal studies. Ovalbumin-sensitized mice were exposed in a specially
equipped van located 50 meters downwind of a Los Angeles freeway. Groups of
mice were exposed to CAPs at two concentrations (400 µg/m3 and
800 µg/m3) for 5 or 10 days. Control mice were exposed to purified
air under the same conditions. All mice received an inhalation challenge of
OVA 2 weeks after their last CAPs or air exposure. As illustrated in Figure
5, eosinophils and OVA-IgE were increased, relative to controls, after the 5-
and 10-day exposures at 400 µg/m3 (p = 0.05 for 10-day
exposure) but not after exposure to 800 µg/m3 for 5 days. The
mice exposed to 800 µg/m3 for 5 days were not different from
controls and were suppressed with respect to eosinophils and IgE compared with
400 µg/m3 for 10 days. These preliminary results are consistent
with a multiphasic immunological response to CAPs. It is possible that exposures
to high CAPs concentrations can have cytotoxic effects that suppress allergic
responses, whereas exposures at lower CAPs concentrations can stimulate allergic
responses.
The age-resolved response of sensitized rodents was examined using 6-hr exposures
to laboratory-generated ultrafine carbon particles (100-150 µg/m3)
with and without O3 co-exposures (Elder et al. 2000, 2002). Results
showed that age, ultrafine particle concentration, ozone concentration, and
sensitizing agents were significantly associated with cellular and biochemical
lung lavage analysis and oxidative stress-related parameters of lavage and blood
cells, and acute-phase proteins. Compared with young animals, old rodents were
at increased risk for greater oxidative stress from combined ultrafine carbon
and ozone exposure.
Human studies. Eleven human volunteer subjects who were either healthy
or had asthma were exposed to inhaled carbon ultrafine particles by mouthpiece
for 2 hr (~ 2  106 particles/cm3; count median diameter = 22 nm) (Frampton
et al. 2002). Healthy subjects showed no significant response to exposure, as
determined by symptoms, spirometry, pulse oximetry, exhaled NO, or sputum cell
differential (counts compared with air control). PMN expression of ICAM-1 and
leukocyte function-associated antigen-1 decreased 3.5 hr after exposures but
to a greater degree after ultrafine particle exposure than filtered air exposure.
106 particles/cm3; count median diameter = 22 nm) (Frampton
et al. 2002). Healthy subjects showed no significant response to exposure, as
determined by symptoms, spirometry, pulse oximetry, exhaled NO, or sputum cell
differential (counts compared with air control). PMN expression of ICAM-1 and
leukocyte function-associated antigen-1 decreased 3.5 hr after exposures but
to a greater degree after ultrafine particle exposure than filtered air exposure.
For healthy subjects (n = 12), an ultrafine count concentration-response
decrease in soluble CD40L was seen compared with air exposure (p = 0.006)
(Daigle et al. 2002). An interim analysis was performed on eight subjects with
asthma. CD40L expression on monocytes, a marker of adverse inflammatory responses,
increased after PM exposure relative to air exposure (p = 0.047). There
were no changes in lung function or airway production of NO. These observations
are the first evidence of CD40-CD40L changes following exposures to ultrafine
particles and are consistent with previous findings of altered leukocyte-endothelial
interactions after such exposures.
Human volunteers were exposed to PM2.5 CAPs in Los Angeles. Exposure
to PM2.5 induced decreased HRV in healthy young adults; increased
blood ICAM-1 (indicating increased inflammation and coagulability), which was
similar in healthy and asthmatic adults; marginally significant increases in
serum IL-6, an acute-phase reactant, in asthmatics; transient decreases in arterial
oxygen saturation in elderly subjects; decreased HRV in elderly subjects; a
greater number of supraventricular and ventricular ectopic heartbeats in elderly
subjects; and increased IL-6 in induced sputum, suggesting increased airway
inflammation.
As many of these studies continue, their results are beginning to elucidate
the roles of key exposure variables on biological responses. These findings
not only support the epidemiological observations but also provide new information
about the biological outcomes that can be measured in field studies.
Short-Term Goals in Acute Effects
During the next several years, the PM centers will continue to develop and
apply new analytical techniques to address important scientific issues relating
to harvesting, confounding, exposure-response relationships, and susceptibility.
The novel statistical approaches developed for evaluating harvesting will be
applied to additional U.S. populations for a variety of health outcomes. In
addition, these new methodologies will be used to examine the relationship between
acute and subacute effects. These investigations will, in turn, provide valuable
insights for our efforts to quantify chronic PM health effects.
Furthermore, we will continue to investigate the potential for confounding
by gaseous co-pollutants and the exposure-response relationships in a variety
of urban environments with different mixtures of gaseous and particulate pollutants.
Over the past few years, the PM centers have been successful in identifying
susceptible populations. These studies suggest that individuals with cardiac
and pulmonary diseases are more vulnerable to PM exposures, and diabetics have
been identified as an important susceptible population. Observational studies
will continue to be critical in our efforts to understand susceptibility. Indeed,
on the basis of the results of previous observational studies, PM centers have
been able to develop animal models replicating human susceptibility.
Investigation of the relationship between particle composition or source characterization
and adverse health outcomes will continue to be a main focus of the PM center
research agendas, and the data now being generated by the U.S. EPA speciation
and Supersite monitoring programs will be used in this endeavor. We will also
investigate the impact of DEP exposures on the exacerbation of asthma. Similarly,
we will examine the relationship between freshly generated ultrafine particles
from mobile sources and children's acute health effects, using the residence
location relative to major interstate roadways as a marker of exposure.
The PM centers observational studies will be supplemented by a series of animal
toxicology studies that will also be completed during the next few years. The
main objective of these controlled studies will be to investigate the pulmonary
and cardiovascular effects induced by exposures to concentrated fine and ultrafine
particles and gaseous co-pollutant mixtures. Of particular interest will be
identification and evaluation of susceptible models. For example, we will conduct
studies exposing mice and rats to concentrated fine and ultrafine particles
at varying distances from high-volume interstate highways in southern California
to determine the effects associated with freshly generated vehicular emissions.
In addition, collaborative studies will be performed to examine the variability
in the PM-induced biological responses in animal hosts (e.g., pulmonary immunomodulation)
as a function of PM composition. Toward this end, PM centers will collect and
exchange PM2.5 samples containing particles of substantially different
physicochemical characteristics.
Collectively, the acute observational and controlled investigations will enable
us to enhance our understanding about the nature of transient cardiac function
changes associated with short-term peaks in PM exposures; the role of underlying
ischemia on associations between short-term peaks of PM exposure and incident
myocardial infarction; and the role of underlying respiratory disease on associations
between PM exposures and HRV.
Long-Term Goals in Acute Effects
We anticipate that many of the scientific issues addressed in the above list
of short-term goals will not be adequately resolved during the next 2-3
years. Therefore, it is likely that research on many of these issues will comprise
much of our long-term agenda on acute PM health effects. Among these issues
will be research-investigating factors, both environmental and genetic, that
explain human susceptibility. In addition, we expect that future collaborative
investigations between epidemiologists and toxicologists will lead to the development
of better biomarkers of PM effects. Finally, PM center efforts aimed at quantifying
the acute health effects of PM exposures will ultimately assist in the creation
of new chronic PM health effects models.
Chronic Health Effects of Particulate Matter
Background
As discussed in the previous section, a plethora of PM acute health effects
studies have been conducted to date. In contrast, much less information is available
on chronic effects associated with PM exposures. This is because of the complexity
and cost of chronic effect studies.
The most frequently cited PM chronic health effect studies are the Harvard
Six Cities Study (Dockery et al. 1993) and American Cancer Society (ACS) Cohort
Study (Pope et al. 1995). They found increased mortality rates (decreased life
expectancy) in cities with higher average ambient PM2.5 and sulfate
concentrations. Differences in city-specific mortality rates were not explained
by data on personal risk factors. Because these studies provided most of the
justification for the annual PM2.5 NAAQS in 1997, they generated
a number of concerns that were expressed in public comments that questioned
a) the validity of underlying study findings due to the lack of public
access to raw data; b) the adequacy consideration of city-specific characteristics
and other alternative explanations of the observed differences in mortality
rates; c) the adequacy of controls for the individual characteristics
such as age, smoking, occupation, obesity, and socioeconomic factors; and d)
the appropriateness of ambient PM measurements as surrogates of community personal
exposures.
In addition to mortality in adults, increased respiratory effects in children
associated with long-term PM exposures were reported. The Harvard Six and Twenty-four
Cities cross-sectional studies observed higher rates of respiratory symptoms
(Dockery et al. 1996) and lower lung function (Raizenne et al. 1996) for children
in cities with higher average PM2.5 and acidic sulfate concentrations.
Concerns expressed in public comments at CASAC reviews of the PM Criteria Document
and Staff Paper were similar to those expressed for the mortality studies, except
for the influence of smoking and occupation.
There were effectively no animal studies of mortality or other morbidity end
points of chronic PM exposures consistent with the epidemiological findings.
Long-term studies had shown increased lung cancer in rats exposed to very high
concentrations of DEPs or carbon particles (possibly related to the particle
clearance overload they produced).
Because of the high visibility of the Six Cities and ACS cohort studies and
their influence in the standard setting process, the HEI undertook sponsorship
of a comprehensive independent validation and reanalysis of these studies. The
HEI project (Krewski et al. 2000) validated the quality of the original annual
mortality study data, replicated the original findings, and demonstrated the
validity and robustness of Six Cities and ACS Mortality studies findings. For
both of the cohorts studied, PM-associated mortality risk was highest for individuals
with less than high school educations.
Data from other chronic exposure mortality studies have been limited. Survival
analyses of nonsmokers in California (the Adventist Health and Smog Study) reported
increased mortality associated with PM2.5 concentrations (Abbey et
al. 1999). Preliminary analyses of male veterans being treated for hypertension
reported no statistically significant increased mortality associated with fine
particle concentrations (Lipfert et al. 2000).
Progress Made in Chronic Effects

Figure 6. Mean increase and
95% confidence intervals for annual mortality rate increases per 10 µg/m3
increment of PM2.5 for the Six Cities and ACS cohorts based
on 16 years of mortality data.
|
Annual mortality cohort studies. Two of the PM centers have been
engaged in continued follow-up of the original Six Cities and ACS cohorts, respectively,
for an additional 9 years. New data on occupation and other individual characteristics
as well as new analytic methods developed in the HEI reanalyses were applied
in both studies. These recent findings are illustrated in Figure 6. The updated
ACS study involves the analysis of more extensive PM2.5, sulfate,
and gaseous co-pollutant data. For the Six Cities cohort, PM2.5 and
sulfate concentrations continued to be associated with decreased survival and
with increased mortality from cardiovascular and pulmonary causes (Laden et
al. 2001). In addition, increased lung cancer deaths were significantly associated
with PM2.5 and sulfates in the extended follow-up. For the ACS cohort,
PM2.5 and sulfate also continued to be associated with increased
cardiovascular and pulmonary mortality, and there was also an excess of lung
cancer (Pope et al. 2002). The excess risks in the ACS cohort were seen only
among individuals with less than high school educations in these extended analyses.
Effects of subchronic exposures. Recent PM center analyses have
attempted to bridge the time spans for health effects observed in studies of
acute daily exposures versus long-term chronic exposures. Analyses of the association
between daily mortality and hospital admissions with PM concentrations during
the preceding weeks to months have highlighted the importance of subchronic
exposures. In Boston, for example, an increase in the 2-day average PM2.5
of 10 mg/m3 was associated with an increase in mortality by 2.1%,
while for the same increase in monthly average, mortality increased by 3.8%
(Schwartz 2000a). In the ACS cohort study, this same increase in annual average
PM2.5 was associated with a 6.8% increase in mortality .
Children's lung growth cohort studies. Studies of chronic effects
of PM exposure on the respiratory health of children have been conducted by
two of the PM centers. Recently reported results from the Children's Health
Study (CHS) in southern California showed that PM2.5 is significantly
associated with slower growth of lung function in children residing in communities
with higher than average annual PM2.5 concentrations. Furthermore,
children who have moved from the high PM areas into areas with lower PM did
not recover their lost lung function but had subsequent rates of lung function
growth equal to that of the children in their new communities. Children who
moved from low PM areas to communities with higher PM levels had less lung function
growth than children who remained in the low PM areas (Avol et al. 2001). The
results of these analyses have provided a basis for follow-on studies of intracommunity
variations in response that are supported by PM center funds. The focus of these
recent and future studies is on proximity to heavily traveled express roads
as an independent risk factor.
These results have also stimulated PM center plans for follow-up studies of
the cohorts of children from the Six Cities and Twenty-four Cities studies.
Fourth and fifth grade children in the Twenty-four Cities study showed lower
lung function associated with community-specific PM2.5 concentrations.
Many of these children have now been retested as mature twelfth graders, and
their lung growth will be compared with community air pollution levels as was
done in the southern California study.
Short-Term Goals in Chronic Effects
The follow-up and data analysis on the Six Cities and ACS study participants
will continue to assess source-specific characteristics of PM responsible for
chronic mortality (longevity reduction) effects. This will include the assessment
of individual characteristics (disease state, socioeconomic status, smoking,
occupational exposures) that make study participants more susceptible to effects
of PM on survival. Air pollution will be treated as a time-dependent covariate
in the Six Cities cohort to determine how much of the association with mortality
in the prospective cohort study is due to exposure in the past year as opposed
to over a lifetime. Estimates of the distribution of years of life lost associated
with PM air pollution in the Six Cities cohort will also be computed. Further
studies of the ACS cohort longevity reduction will focus on the relationship
between mortality and PM composition and/or source contribution. This will be
accomplished by using particle speciation data, both the historic dichotomous
sampler network data and the new speciation site data, and source apportionment
techniques. The association between PM2.5 and/or sulfate and local
airport visibility will be determined for each community where suitable data
are available. City-specific relationships between pollution and visibility
measurements will be used to estimate PM2.5 levels prior to and after
the limited period for which direct measurement data are available in an effort
to investigate the relative roles of past and current exposures.
Analyses of data previously collected on cohorts of children from studies
performed by PM center investigators (Twenty-four Cities and southern California
CHS) will be pooled to assess effects of PM from different source classes (e.g.,
vehicular vs. power plant) and composition (e.g., nitrate vs. sulfate). Each
of these cohort studies assessed fourth and fifth grade school children using
the same questionnaires and pulmonary function measurements. The Twenty-four
Cities study assessed the effects of sulfate particles from coal-fired power
plants. The southern California study assessed the effects of nitrate particles
from vehicular traffic. The combined data set will assess chronic health effects
in these children versus PM2.5 mass and composition data.
A new epidemiological study of participants in the National Heart, Lung, and
Blood Institute Women's Health Initiative Observational Study (93,000 women
50-79 years of age from 40 centers around the country) is currently under
way. The women are being observed for the occurrence of CVD and will be matched
with community air pollution levels from ambient monitors. The effects of chronic
exposure to ambient PM on the incidence of CVD will be assessed in this cohort
using a proportional hazards model approach.
In prior epidemiological studies in Erfurt and Augsburg, Germany, acute effects
of ultrafine particles on daily mortality (Wichmann et al. 2000) and on myocardial
infarction (Peters et al. 2001) were observed. Cohorts that were established
in these cities will be used in the future to investigate whether there are
long-term effects of exposures to ultrafine particles.
A PM center subchronic animal inhalation study using New York CAPs began in
the spring of 2002. Normal and cardiac disease-susceptible mice will be
exposed for 6 hr/day, 5 days per week, for 6 months, with or without intermittent
ozone exposure. Cardiac and respiratory function will be monitored periodically,
and biochemical and genetic biomarkers of responses will be investigated in
tissues and lung lavage cells from the exposed animals. In another study, mice
will undergo infectious bacterial challenge prior to CAPs exposures to determine
possible PM exposure-related enhancement of their susceptibility to infection.
These studies will thereby assess health end points being evaluated during acute
CAPs exposures of animals at several of the PM centers and of humans at the
U.S. EPA Clinical Studies Laboratory in Chapel Hill, North Carolina, and determine
whether such acute effects progress to chronic changes that could lead to enhanced
susceptibility to PM exposures.
Long-Term Goals in Chronic Effects
Based on previous experience, repeated follow-up of existing cohorts can be
of paramount importance in understanding the association between chronic PM
exposures and human health. Results from the Six Cities and Children's Health
studies, for example, have enabled us to understand the effects of air pollution
on children's lung growth. Collectively, the PM centers have already investigated
numerous cohorts that span ages, ethnicities, and geographic locations and that
are exposed to varying PM levels and compositions. Health and exposure history
data for these cohorts are also available. Information from these numerous study
locations will be combined to increase the statistical power of existing chronic
health effects models. The range of data will, in turn, allow us to better examine
the role of PM composition, gaseous co-pollutants, ethnicity, exposure history,
age, and climate on chronic PM health effects. Therefore, it would be advantageous
for the PM centers to allocate a large fraction of their resources during the
next 5-10 years to continue with the follow-up for these existing cohorts.
As new scientific information emerges, providing better indicators of air
pollution health effects, questionnaires and medical examinations will be amended
accordingly. For example, the early cohort studies of populations initially
recruited in the mid-1970s focused on the impact of air pollution exposures
on respiratory health. Therefore, investigations centered on collecting detailed
information on respiratory health. Recent studies have demonstrated the influence
of particles and air quality on cardiovascular health. Correspondingly, information
on cardiac health has assumed a more central role in current studies. Although
limited information currently exists on the effect of air pollution among susceptible
populations, future knowledge may require a better understanding of factors
predisposing these individuals to adverse health outcomes. Of particular interest
is the gene-environment interaction. Research that integrates the findings
from the CHS with new information on traffic density and the organic constituents
in traffic-related PM will also be pursued as a long-term goal.
For chronic studies using CAPs-exposed animal models, the specific hypothesis
to be addressed in future years will be based, in part, on the findings of the
first subchronic exposure study to be conducted in New York. This study will
examine the possible acceleration of atherosclerotic plaque development, increased
reactive or obstructive airway disease, reduced lung capacities, enhanced susceptibility
to infectious agent exposures, and altered gene expression. Moreover, efforts
will be made to determine whether variations in responses during the exposures
can be related to the day-to-day variations in the chemical composition of the
CAPs. Based on the results from this study, the following parameters may be
considered in future chronic exposure studies: a) including additional
animal models to replicate human susceptibility; b) increasing exposure
durations; and c) examining coexposures to CAPs with one or more gaseous
co-pollutants. Of additional importance will be the investigation of chronic
health effects thresholds by conducting subchronic exposure studies at multiple
PM concentrations to determine the existence of thresholds, if any, and replication
of one or more of these studies in areas where the PM composition is different.
Finally, we expect that the findings from the controlled animal studies will
assist in further refining study designs for our human chronic health effects
investigations.
Dosimetry
Background
A critical link in the evaluation of the relationship between individual exposures
to PM and health responses is dosimetry. Dosimetry research investigates not
only the amounts and distribution of the deposited PM in the respiratory tract
but also the pathways by which this material is translocated to other sites
in both the respiratory tract and to more distal organ systems. Furthermore,
knowledge of inter- and intraspecies differences is critical to our effort to
extrapolate results from experimental studies to the population at large.
Prior to the establishment of the PM centers, studies of PM dosimetry in lung
airways concentrated largely on healthy adult humans, although there were a
few studies of children and adults with chronic respiratory disease. However,
very little research has examined deposition in animal models that are currently
being used in studies of PM health effects as surrogates of susceptible human
populations. Mathematical deposition models derived from experimental data had
been developed and were largely used for predicting total and regional deposited
doses. Mechanisms for clearance of deposited particles had also been studied,
and major clearance pathways in the respiratory tract regions had been well
described. However, several major gaps in our understanding of particle deposition
and clearance remained, including detailed analyses of size-resolved PM deposition
and clearance, especially for ultrafine particles, and clearance mechanisms
for PM components as well as their translocation pathways.
Research addressing these gaps is important for the interpretation of toxicological
and epidemiological findings suggesting effects on cardiopulmonary systems,
which could be due to indirect (via biological mediators initiated from acute
pulmonary effects) or direct or neuronal function changes. The multidisciplinary
teams of PM centers investigators provides a great advantage in addressing these
questions.
Progress Made in Dosimetry
Significant advances have been made at the PM centers in a number of critical
areas. These include development of techniques for the production of hollow
lung airway casts of potentially susceptible people and evaluation of dosimetry
of specific fractions of ambient PM that may be especially toxic. There has
also been work on dosimetry in humans and in animal models being used in PM
studies, using both ambient PM and surrogate particles.

Figure 7. Cross-sectional
luminal airway surface of original hollow airway cast made from a normal
human tracheobronchial tree in square millimeters versus corresponding
airway surface in square millimeters of a replica produced by STL using
data obtained by CT scanning of the original cast.
|
To address the paucity of data regarding PM deposition in the lungs of people
with preexisting pulmonary disease, a pilot PM center project investigated the
potential for retrieval of morphometric data from three-dimensional images of
tracheobronchial airways obtained in vivo by X-ray computerized tomography
(CT). The study also explored the potential for the use of stereolithography
(STL) to produce hollow airway casts of normal and abnormal lung airways for
the determination of site-specific deposition and for experimental verification
of particle deposition models. A volumetric rendering of the interior surface
of a hollow airway cast (used in previous studies) was generated, producing
a surface representation of the airway tree. These three-dimensional images
were then converted to an STL file format required for the rapid prototyping
of airway casts. The STL unit uses a computer-controlled arm connected to a
plastic extrusion device to build volumetric structures layer by layer. Figure
7 demonstrates the close concordance between an original hollow airway cast
and the replicate made of it by STL (Won et al. 2000a, 2000b).
Thin, multislice, helical CT scanning allows for the acquisition of high-resolution
volumetric image data sets of the lung during a breath-hold, or at multiple
phases within a respiratory cycle for children, the elderly, and people of all
ages having asthma or chronic obstructive lung disease. From these scans, hollow
airway casts that include five or six bronchial generations can be created and
replicated for use in studies of inhaled particle deposition in replicate casts
of both healthy and diseased airways, using realistic air flow rates.
In another study, PM center methodology was developed to determine total lung
deposition of CAPs in dogs. These studies are relevant to animal models of cardiac
disease used in the PM center research. The methods included the use of a breath-by-breath
respiratory monitoring system that provided both rate and tidal volume as well
as measurement of inhaled and exhaled particle number, classified by size. These
measurements used both a TSI Scanning Mobility Particle Sizer for particles
in the ultrafine and smaller fine ranges, and a TSI Aerosizer (TSI, Inc., Shoreview,
MN) for particles in the fine particle size range. The canine studies showed
deposition of ambient particles followed a pattern similar to that described
in the International Commission on Radiological Protection (ICRP) human particle
deposition model (ICRP 1994) but was slightly greater than that predicted by
the ICRP model. This method was subsequently used in a study of six human subjects
breathing ambient air without any enhancement of particle concentration. In
all subjects, total respiratory deposition fraction (DF) for each measured size
range (> 10 nm) followed the same pattern predicted by the ICRP model, and
in some individuals, deposition exceeded the predicted fraction. Human studies
of ambient particle deposition are important to validate deposition models.
In vivo deposition studies of ultrafine particles are currently being
conducted in humans and rats. For the human studies, a system was developed
for mouthpiece exposures, with alternating rest and exercise every 15 min. Total
respiratory DFs for particle number and mass were determined down to 7-nm particles.
Ultrafine carbon particles (22 nm) were generated. Eleven nonsmoking, healthy
subjects and 11 nonsmoking subjects with mild asthma were exposed for 2 hr.
The total respiratory deposition of inhaled ultrafine particles was high relative
to fine particles in healthy subjects (DF = 0.63 ± 0.03), and was further
increased during exercise (DF = 0.83 ± 0.04), with enhanced deposition
in asthmatics (rest DF = 0.77 ± 0.05; exercise DF = 0.86 ± 0.04).
These results suggest that asthmatics may be at greater risk from exposure to
ultrafine particles because of increased deposition in central airways.
Deposition rates for size-resolved ultrafine particle data showed that 7-nm
particles had the highest total deposition (~ 80%). Deposition efficiency (DE)
for 100-nm particles was ~ 45%. These deposition rates are in good agreement
with the ICRP particle deposition model (ICRP 1994). This model predicts that
ambient ultrafine particles of 20 nm will have the highest overall DE, as well
as the highest deposition in the gas-exchange region (Chalupa et al. 2002a,
2000b; Daigle et al. 2003; Frampton et al. 2000).
Experiments in rats were performed using two types of ultrafine 192Ir
particles and carbon particles containing the stable isotope 13C
particles. The basic design of both rat studies was the same, i.e., exposure
to the ultrafine particles followed by serial sacrifice in the postexposure
period and analysis of excised lungs and extrapulmonary organs for 13C
and 192Ir, respectively. However, there were major differences. The
13C exposure occurred for 6 hr in compartmentalized whole-body exposure
chambers, whereas the 192Ir exposures occurred via a 1-hr intratracheal
inhalation in anesthetized rats.
For the 13C exposures, the median particle size was in the range
of 20-29 nm. Exposure concentrations ranged from 80 to 180 µg/m3,
and organ burdens were determined in the first series of experiments at 0.5,
18, and 24 hr after exposure. Six unexposed rats served as controls. Lung lobes,
liver, heart, kidney, and brain sections (olfactory, cerebrum, cerebellum) were
analyzed for 13C by isotope ratio mass spectrometry (MS). On average,
approximately 9 ng 13C/g lung per µg/m3 of exposure
was found in the lung, much less than expected based on predictive lung deposition
models. Significant amounts of 13C had accumulated in the liver by
0.5 hr postinhalation only at the high exposure concentration of 180 µg/m3,
whereas by 18 and 24 hr postexposure, all liver samples showed significantly
increased 13C. Indeed, the amount of total 13C in the
liver at those time points exceeded the amount retained in the lung, demonstrating
a fast translocation of ultrafine carbon particles to the liver. Other extrapulmonary
organs analyzed for 13C did not show significant increases by 24
hr postexposure (Oberdörster et al. 2002).
Results were different for ultrafine Ir particles. Ir was found, in pretest
studies, to be essentially insoluble. In these studies, the median particle
size was approximately 15 nm. Gamma-spectroscopic analysis for 192Ir
was performed at time points ranging from 6 hr to 7 days postexposure. The ultrafine
192Ir particles, after an initial fast tracheobronchial clearance,
were essentially retained in the lung; only minute amounts were found in extrapulmonary
tissues [~ 0.5% in bone and soft tissue (muscle), < 0.1% in liver, and even
less in heart and brain]. Soluble Ir was excreted via urine (60% over 7 days),
8% remained in the lungs, and 10% was retained in both soft tissue and bone
(Kreyling et al. 2002). These results show that translocation of poorly soluble
ultrafine particles to extrapulmonary sites appears to be highly dependent on
the chemical nature of the particles. Carbon particles were to a large degree
translocated to the liver, whereas metal iridium particles were not. These results
raise a number of questions that need to be addressed in future studies, including
the evaluation of pathways for ultrafine particle translocation (lymphatic,
blood vessels, gastrointestinal tract, nasal vs. tracheobronchial vs. alveolar
region) and also binding of ultrafines to proteins.
Because currently available inhaled particle dose models provide estimates
of averaged doses (i.e., environmental aerosol concentrations), they do not
take into account the true PM heterogeneity of tissue doses that occur in the
lungs. Higher-than-average doses can be expected for those people exposed near
local particle sources, those who have high specific ventilations (ratio of
air intake to body mass), and those who have heterogeneous deposition of particles
within various regions of the respiratory tract. Thus, exercising adults and
children with lung diseases who live near heavily used freeways may be receiving
greatly elevated particle doses at numerous sites within their lungs. Similarly,
the available software used for computing inhaled particle deposition cannot
identify local populations of cells that may be of toxicological interest. The
numbers of cells that must be significantly damaged to seriously impact vulnerable
individuals is not yet known. These issues were addressed by a PM center workshop
in October 2001 that developed a framework for estimating expected local tissue
dose concentrations. This framework was applied to southern California Supersite
data sets, and local doses to sites containing the highest exposed 100 cells
at each bifurcation in the tracheobronchial tree were calculated for heavily
exposed individuals. These local bifurcation area doses were compared with the
doses applied to cells in in vitro studies of PM effects. The high in
vitro doses corresponded to estimated airway bifurcation PM doses potentially
occurring in the lungs of individuals living in the Los Angeles basin.
Short-Term Goals in Dosimetry
In a successor project to the pilot feasibility study for the production of
hollow airway casts that faithfully reproduce the in vivo dimensions,
in vivo studies are being conducted of particle deposition in anesthetized
sheep at the facilities of study collaborators at the University of Iowa (Iowa
City, Iowa) using 5-µm diameter radio-opaque droplets. This involves development
of analytical programs to examine the CT scan data. The second phase will be
to examine changes in the deposition pattern and efficiency when inhalation
is done at different points of the inspiratory cycle. The computer-controlled
respirator can produce almost any breathing pattern and can key the aerosol
delivery to any point in the respiratory cycle of the anesthetized sheep. When
quantitation has been accomplished in vivo, hollow airway casts of the
sheep lung will be produced using the methods developed in the initial PM center
pilot study. PM deposition patterns and efficiencies will then be determined
using an artificial thorax and methods and models previously used at NYU to
measure particle deposition in the hollow-cast systems. Successful development
of the test aerosol as well as quantitation methods will ultimately allow future
exploitation of these techniques for in vivo studies in both healthy
people and those with compromised lung function. In addition to determining
DE and pattern studies, PM center studies will explore the potential for identifying
bolus deposition in the sheep lung and the extent to which assumptions of standard
bolus deposition experiments are valid. The system will allow testing of the
hypothesis proposed by C. Kim and colleagues at the U.S. EPA--that regional
deposition can be accurately assessed in people via a series of bolus inhalation
experiments (Kim 2000).
Other PM center dosimetry studies will continue to focus on ambient particles
and their diurnal and temporal variation in concentration and composition. The
number of subjects will be expanded as well as the number of repetitions per
subject. This will permit definition of the range of individual differences
in particle DFs, including male-female and age differences. In addition,
the relationship of DF to day-to-day compositional differences will be explored
to determine the factors important in variations in DF.
Research will continue on ultrafine particle deposition, disposition, and
chemistry. Investigations will include the continued study of susceptible groups
(e.g., COPD, elderly) in terms of total respiratory tract deposition; the effects
of changes in tidal volume and respiratory rate in deposition; deposition efficiencies
in different anatomical compartments of the respiratory tract; clearance and
translocation pathways via the respiratory and gastrointestinal tracts; translocation
following the initial phase of accumulation in the liver; the liver as a storage
organ for distribution to other organs; the mechanisms of particle translocation;
the role of particle chemistry; the fate of organic carbon particles; the efficiency
of ultrafine particle translocation along sensory nerves in the conducting airways;
and the long-term consequences of ultrafine particle translocation to extrapulmonary
organs like the heart.
Answers to these questions are important for the interpretation of results
of toxicological and epidemiological studies with respect to effects of inhaled
particles on the cardiovascular system that could be due to indirect (via biological
mediators initiated from acute pulmonary effects) or direct (ultrafine particles
interacting with cardiac or endothelial cells) or neuronal functions effects.
To better understand the generalizability of controlled animal PM exposures
to human health, the regional inhaled particle depositions in mice from the
freeway study will be determined. Toward this end, replica airway casts of 30
Balb/c mice (15 controls and 15 sensitized) will be prepared, and morphometric
measurements of airways will be conducted. The mice will be exposed to three
sizes of electrically discharged fluorescent monodisperse polystyrene latex
particles. Their lungs will be digested, and particles deposited in the lungs
will be recovered and counted using a fluorescence-equipped microscope. These
measurements will be used for examining the toxicologic responses of the sensitized
model, as well as for performing extrapolations to humans.
Long-Term Goals in Dosimetry
The results of the in vivo particle deposition studies in sheep will
provide a firm basis for a study of human subjects. The future human in vivo
studies will focus on a) application of accepted mathematical dosimetry
models to individuals, using the morphometric data retrieved from the images.
This will be assisted by respiratory function test results that will be available
for these individuals; b) measurement of detailed particle deposition
data for a range of breathing patterns and particle sizes in hollow-cast models
of the airways of individuals representative of the various groups of interest;
and c) reconciliation of the results calculated from the current models
with experimental data to produce verified empirical deposition models that
can be used to better predict inhaled dose on the basis of metrics of exposure
to ambient PM.
Future research to relate airborne PM to improved dosimetric models will be
directed toward defining the population receiving the highest local doses in
their tissues, and defining the appropriate particle doses for use in in
vitro mechanistic studies. Research is also needed to address a)
the environmental heterogeneity of PM-associated pollutants; b) how much
time susceptible subpopulations spend in such high-concentration areas; c)
ventilation rates of those exposed; d) heterogeneous deposition patterns
of inhaled particles; e) dose implications of impaired clearance (as
is seen in some disease states) and sequestration of particle associated substances;
f) vulnerable target cells in the tissues; and g) realistic target
particle doses that can be used for the design and interpretation of in vitro
mechanistic research.
Exposure Assessment
Background
As noted in the section "Acute Health Effects of Particulate Matter," results
from daily mortality and morbidity time-series analyses provided much of the
scientific basis for setting the PM standards in 1997. These epidemiological
studies typically used PM mass concentrations measured at outdoor monitoring
sites as surrogates of population exposures to ambient air PM. The extent to
which outdoor measurements accurately reflect PM exposures has been the subject
of considerable scientific debate. Results from early exposure studies, such
as those conducted as part of the Harvard Six Cities Study and the U.S. EPA
Particle Total Exposure Assessment Methodology Study (Clayton et al. 1993),
for example, suggested that personal PM exposures may differ substantially from
outdoor concentrations because of contributions from indoor sources. Cross-sectional
analyses of these data showed weak associations between daily outdoor PM concentrations
and corresponding personal exposures, which were attributed to intersubject
variability and the limited number of measurements (~ 1-2 days) for each
individual. Consequently, the existing PM exposure database was considered inadequate
for investigating associations between personal exposures and outdoor concentrations,
and longitudinal exposure studies were proposed.
Upon completion of the 1997 PM NAAQS review and prior to the establishment
of the five PM centers, a series of longitudinal PM exposure studies were funded
by the U.S. EPA, HEI, Electric Power Research Institute, U.S. Department of
Energy, and American Petroleum Institute. The main objective of these studies
was to investigate associations over time between personal PM exposures and
outdoor PM concentrations, with specific focus on characterizing exposures for
individuals thought to be especially susceptible to PM-associated health effects.
U.S. EPA-sponsored research projects were conducted by groups from Harvard University,
NYU, and the University of Washington. The three research groups collaborated
closely in the design and preparation of field studies and shared similar sampling
procedures and questionnaires in an effort to create compatible data sets. As
part of these early longitudinal studies, the exposures of several cohorts of
susceptible individuals, including senior citizens, children, and individuals
with COPD or CVD, were measured for periods ranging from 1 to 2 weeks. Field
studies were conducted during both the summer and winter and in a variety of
urban and suburban environments including Atlanta, Georgia; Baltimore, Maryland;
Boston, Massachusetts; Nashville, Tennessee; New York, New York; and Seattle,
Washington. Although analyses of the entire body of data are still in progress,
several papers have been published and have been cited in the 2001 PM Criteria
Document external-review draft. The U.S. EPA Office of Research and Development
is planning to use this rich database to develop both acute and chronic exposure
models.
A major finding of these studies is that stronger personal-outdoor PM correlations
exist when data are analyzed by individual, over time. However, the degree of
this association varies by individual, with some individuals having significant
associations and others not. A major deficiency in exposure assessment research,
which still persists, relates to the lack of accurate chronic exposure models
to be used by epidemiological studies.
Progress Made in Exposure Assessment
The findings from the longitudinal PM exposure studies, described above, were
critical to the evolution of our collective research efforts on PM exposure
assessment. Results from the centers research pertaining to PM exposures are
presented briefly below, with many of the findings already published, in press,
or submitted for publication in peer-reviewed journals.
Considerable research was conducted to identify factors that contribute to
observed differences between outdoor concentrations of PM and corresponding
population exposures. The differences have traditionally been referred to as
"exposure error" because PM epidemiological studies typically use outdoor PM
concentrations as surrogates of exposure. PM centers exposure research efforts
have focused on the following topics:
- Characterization of spatio-temporal variability of PM components
and gaseous co-pollutants measured at centrally located outdoor sites as a
function of site characteristics using the entire U.S. air monitoring network.
The models will enable epidemiologists to quantify the effects of exposure
error on health risk estimates.
- Assessment of the contribution of outdoor and indoor PM sources on
personal exposures. This area of research is critical for determining the
relative toxicities of PM of outdoor and indoor origin.
- Measurement of exposure to specific toxic PM components and gaseous co-pollutants.
This information will be of great value to epidemiologists and toxicologists
in their efforts to identify the causal agents of air pollution-related toxicity.
Investigation of the association between outdoor particulate matter
concentrations and corresponding personal exposures. Ambient PM2.5

Figure 8. Distribution of
subject-specific correlation coefficients: ambient PM2.5 concentration
versus personal PM2.5 exposures in Baltimore, Maryland, 1998-1999.
n = 14 for both sampling seasons (Sarnat et al. 2000). These box
plots show median values and the 25th and 75th percentiles. The vertical
lines extend to the 10th and 90th percentiles and the circles indicate
extreme values.
|
concentrations were significant predictors of corresponding personal exposures
over time for a cohort of healthy senior citizens (Figure 8) (Sarnat et al.
2000). Although the strength of these associations varied by individual and
season, the results suggest that for certain individuals, ambient PM2.5
concentrations are appropriate surrogates of personal PM2.5 exposures.
These findings are consistent with results from recent longitudinal PM exposure
studies conducted in Boston (Rojas-Bracho et al. 2000) and the Netherlands (Janssen
et al. 1998). When the subject-specific data were aggregated and analyzed together
(i.e., cross-sectionally), the association between personal exposures and outdoor
concentration was weaker, further highlighting the inadequacy of cross-sectional
analytical methods for assessing true personal-ambient PM2.5 associations
for time-series studies.
Because most of these subjects spent the majority of their time (> 95%)
indoors during both seasons, seasonal differences in personal-ambient associations
were likely due to changes in indoor ventilation conditions between the summer
and winter. Summertime records of open window status provided direct evidence
of the effect of indoor ventilation on personal-ambient PM2.5 associations,
with ambient concentrations shown to be strong predictors of corresponding personal
exposures for subjects who spent the majority of their time in well-ventilated
indoor environments. This was illustrated in the plot of personal PM2.5
exposures on ambient PM2.5 concentrations, which had a slope that
was close to 1 and a limited amount of scatter. Personal-ambient PM2.5
associations were also significant for subjects who spent the majority of their
time in poorly ventilated indoor environments, but the association was much
weaker. The plot of personal exposures on ambient concentrations for poorly
ventilated environments had a slope well below 1, with a considerable amount
of scatter due to the influence of poor ventilation on both reducing the penetration
efficiencies of ambient particles and increasing the contribution of indoor
source particles on personal exposures.
Spatial variability of outdoor particulate matter concentrations.
Some of the interpersonal differences in personal-outdoor PM associations may
be due to
the spatial variability in outdoor PM concentrations. Ito et al. (2001) used
nationwide PM10 and gaseous pollutant

Figure 9. Smoothed monitor-to-monitor
temporal correlation in seven north-central U.S. sites. |

Figure 10. Most NYC summer
PM2.5 mass at health center (in Manhattan) is explained by
regional PM2.5 at Sterling Forest, a state park northwest of
Manhattan. y = 0.9895x + 3.1262; R2 = 0.843.
|

Figure 11. Relative PM2.5 mass [measured
by DataRam nephelometer (MIE, Inc., Bedford, MA)], particle number (measured
by CPC), black carbon (BC), and carbon monoxide (CO) concentrations versus
downwind distance from the Los Angeles 405 freeway (Zhu et al. 2002).
|

Figure 12. Diagram of the
multipollutant personal sampler (Demokritou et al. 2001). Abbreviations:
EC, elemental carbon; MSC, mini personal sampler; MSD, mini denuder; OC,
organic carbon; SPM, suspended particulate matter. |

Figure 13. Summary of associations
between ambient concentrations and corresponding personal exposures and
among pollutant exposures and concentrations in Baltimore, Maryland (Sarnat
et al. 2001). “3” indicates strong correlations; “¥”
indicates weak correlations. |
measurements to examine relationships between concentration measured at various
urban and suburban sites. Figure 9 shows a smoothed monitor-to-monitor temporal
correlation for air pollution and weather variables as a function of distance
in seven northern and central U.S. states. As shown, monitor-to-monitor correlations
varied by pollutant. Stronger spatial correlations were found for PM
10,
NO
2, and O
3 compared with those found for CO and SO
2.
PM
2.5 data will be incorporated within this analysis as it becomes
available.
Previous studies in the Northeastern United States have shown that mean daily
PM2.5 concentrations are reasonably uniform within large Eastern
U.S. metropolitan areas such as Washington, DC, Philadelphia, and Baltimore.
Findings from PM center-supported studies further indicate that there is relatively
little variation in ambient PM2.5 concentrations between the center
of New York City and a rural upwind location (Figure 10). Together, these results
suggest that most of the PM2.5 in the Northeastern United States
originates from distant upwind sources (Thurston et al. Unpublished data). However,
this may not be the case in other locations in the United States. PM2.5
mass concentrations in Seattle exhibited modest, yet significant, spatial variability
within a radius of 20 km. These differences were associated with both proximity
to major highways and the elevation of the monitoring location (Goswami et al.
2002). In Los Angeles, PM2.5 and PM10 concentrations measured
at various distances from highways (10-1,000 meters) showed little spatial
variability. However, particle number and black carbon concentrations decreased
rapidly with distance from highways (Zhu et al. 2002) (Figure 11).
The spatial variability of reactive chemical species in PM2.5,
such as quinones and their precursors in the Los Angeles basin, showed considerable
variability from west to east, reflecting the direction of the prevailing winds.
The naphthoquinones decreased, but the concentration of 9,10-phenanthroquinone
increased. As quinones are known to be toxic, their concentrations in PM fractions
may be a more sensitive measure of exposure. Levels of quinones in the outlying
areas were, as expected, extremely low.
Impact of indoor particulate matter sources on personal exposures. As
shown in the earlier cross-sectional PM exposure studies, variability in personal-outdoor
PM associations is due, in part, to contributions from nonambient particle sources
(Ozkaynak et al. 1996). PM center-supported longitudinal exposure studies
have provided additional evidence of the impact of non-outdoor PM sources
for both cohorts of healthy individuals and susceptible subpopulations. Results
from Seattle and Baltimore showed that nonambient PM2.5 sources contributed
49 and 67% of the total personal PM2.5 exposures, respectively (Allen
et al. 2002; Sarnat et al. 2000). In Seattle the pediatric subjects experienced
significantly higher exposures to particles of indoor origin than did adults.
Their average fraction of ambient PM contribution to total human exposures was
no higher, however (Allen et al. 2002). In Baltimore, indoor ventilation influenced
nonambient PM contributions to personal exposures, where nonambient PM2.5
sources contributed 29, 30, and 45% to total personal PM2.5 exposures
for subjects spending their time in well-ventilated, moderately ventilated,
and poorly ventilated indoor environments, respectively.
Variability of outdoor particulate matter penetration efficiencies.
People spend the majority of their time indoors. Therefore, quantifying
the effective penetration efficiencies of outdoor particles into indoor environments
is critical for understanding the variability of personal-outdoor PM relationships.
Until recently, particle penetration efficiencies were thought to be constant
and were often assumed to be 100%. As a result, personal/outdoor concentration
ratios higher than one were assumed to be indicative of the influence from non-outdoor
PM sources. Results from current PM center exposure studies have shown that
particle penetration efficiencies vary substantially by residence type (Larson
et al. 2001). In Seattle for example, particle penetration efficiency exhibited
significant interhome and intrahome variability for the 30 homes monitored.
Because home characteristics, including ventilation, vary by season and locale,
these findings may be used to explain some of the heterogeneity in PM-associated
risk factors in various epidemiological studies, as reported in the section
"Acute Health Effects of Particulate Matter."
Measurement of personal exposures to specific toxic particulate matter
components. Identifying the specific component(s) of outdoor PM responsible
for the numerous observed adverse health effects remains an important objective
of our exposure assessment research. Results presented in "Biological Mechanisms
for Particulate Matter Health Effects" of this review suggest that specific
components of outdoor PM, such as transition metals, ultrafine particles, and
PAHs, may be related to allergic airway disease and cardiovascular effects.
Funding has facilitated the development of novel personal multiple pollutant
sampling methods currently being used in many of the PM center field studies
(Figure 12) (Demokritou et al. 2001). The Multiple Pollutant Personal Sampler
provides data on concurrent size- and species-resolved personal PM exposures
as well as exposures to numerous gaseous co-pollutants (NO2, O3,
SO2, and numerous VOCs). To date, the Multiple Pollutant Personal
Sampler has been used in the Baltimore; Atlanta; Steubenville, Ohio; and Los
Angeles field studies. Other methods are currently under development at the
PM centers to examine exposures to specific PM components.
The PM centers are conducting a series of studies to identify and quantify
specific toxic PM components as well as examine their spatial and temporal distributions.
Organic PM constituents such as quinones may play a particularly important role
in generating oxidative stress, inflammation, and immunomodulating effects in
the lungs and airways. Currently, PM seasonal and spatial distributions of outdoor
PAH, aldehyde, and quinone concentrations are being characterized for 12 southern
California communities participating in the CHS (Cho et al. Unpublished data).
Center investigators expect to link spatial intercommunity variability of specific
organic (i.e., quinones) and carbonyl concentrations with reported respiratory
health effects of school children.
Measurement of personal exposures to gaseous co-pollutants. Ambient
concentrations of PM and its gaseous co-pollutants are frequently correlated,
making it difficult for epidemiological investigations to determine whether
observed PM-health effect associations are confounded by these co-pollutants.
The role of gaseous co-pollutants as potential confounders or surrogates of
personal PM2.5 exposures was investigated for cohorts of healthy
senior citizens, children, and individuals with COPD living in Baltimore (Sarnat
et al. 2001). Investigators used the Multiple-Pollutant Personal Sampler, which
allowed for associations between personal particulate and gaseous exposures
to be examined directly. Consistent with results from previous monitoring studies,
strong correlations existed between ambient PM2.5 and personal gaseous
co-pollutant concentrations (i.e., O3, NO2) (Figure 13).
In contrast, weak correlations were found between personal PM2.5 and
personal gaseous co-pollutant exposures, suggesting that the gaseous co-pollutants
are unlikely confounders of PM2.5. Finally, strong correlations between
personal exposures to PM2.5 and ambient concentrations of the co-pollutants
existed, indicating that the gaseous co-pollutants may serve as appropriate
surrogates of personal PM2.5 exposures.
Short-Term Goals in Exposure Assessment
Exposure studies conducted to date have provided a solid basis for future
research. Goals of the PM centers during the next 2-3 years include the
following:
- Complete exposure studies currently being conducted in St. Louis,
Los Angeles, Steubenville, and Seattle. These additional data sets will enable
us to further characterize personal PM exposures in locations with diverse
study populations and meteorological and air quality conditions. Of particular
importance are potential differences in exposures among susceptible subpopulations.
To date, cohort-specific differences in PM exposures have not been examined
because of sample-size limitations.
- Examine associations between personal exposures to PM and its gaseous
co-pollutants in various locations throughout the United States. Results from
Baltimore provide important initial information concerning the role of gaseous
pollutants as confounders and/or surrogates of PM. Exposure data from studies
in Los Angeles, Atlanta, Steubenville, and St. Louis will enable us to examine
these relationships in additional urban environments.
- Continue the characterization of short-term (24-hr) PM exposures
and identify factors that influence their relationship to outdoor concentrations.
This information will assist in creating models of short-term personal exposures
to be used in estimating chronic exposures to PM.
- Collect data on outdoor PM concentrations and personal exposures
to specific PM components such as metals, PAHs, elemental and organic carbon,
sulfate, and nitrate as part of our ongoing field studies. In addition, efforts
will continue to expand the range of personal exposure measurements, including
ultrafine particles and carbon monoxide.
- Determine concentrations of quinones, carbonyls, and PAHs in ambient
air samples at selected sites in the Los Angeles basin. The objective is to
provide quantitative chemical data on specific chemical entities relevant
to human health and to the development of a model for atmospheric changes
in the chemical constituents of air pollution.
- Continue data analyses for the investigation of exposure error using
air pollution data from 1985 to 2000 for the entire United States. More detailed
characterization and comparison of PM10 and PM2.5 data
will be conducted in areas with multiple monitors.
- Continue collection of outdoor PM samples to be used in toxicological
studies of fine and ultrafine ambient particles. Initial studies to examine
the hypothesis that freshly generated fine plus ultrafine emissions from a
freeway with heavy diesel traffic increases the severity of asthma and inflammatory
responses in a sensitive animal model are under way.
- Validate and refine models of local within-community exposures based on
already available data sets on traffic patterns in metropolitan areas and
further assessment of the chemical composition of traffic-associated particulate
exposures.
Long-Term Goals in Exposure Assessment
The PM centers' long-term goals involving exposure assessment include the
following: a) Development of chronic PM exposure models. Chronic exposure
models will be based on relevant advances in short-term exposure models, reexaminations
of historical and newly generated data on air pollution composition and levels,
time-activity information, and indirect measures of PM exposure, such as
covarying gaseous pollutant concentrations; geographical information system
data, and housing characteristics may also be used as input data for these chronic
exposure models. Individual or population estimates of long-term exposures will
be used by epidemiological studies to assess chronic health effects. Of particular
importance will be the development of chronic exposure models for specific toxic
components of PM. b) Development and validation of source-apportionment
techniques to link health outcomes with specific PM source types. The NYU PM
center in collaboration with colleagues at the other PM centers and at the U.S.
EPA will host a source apportionment workshop to standardize source-apportionment
methodologies for PM epidemiological studies. c) Development of new and
more sensitive biomarkers of PM exposure. This will enable us to assess human
exposures for prospective population studies in a timely and cost-effective
manner. Some preliminary work on a urinary biomarker for exposure to wood smoke
is under way (Dills et al. 2001).
Coordination of Research within the Particulate Matter Centers
Program
Background
Research centers are most productive where there is a well-defined set of
scientific questions, and where the approaches needed to answer those questions
cross traditional disciplinary boundaries of chemists, engineers, aerosol scientists,
toxicologists, epidemiologists, pulmonologists, cardiologists, immunologists,
molecular biologists, statisticians, and experts in exposure and risk assessment.
Going beyond what individual centers can accomplish, the U.S. EPA-sponsored
PM Health Effects Research centers, with encouragement from the U.S. EPA, have
created a coordinated PM centers program that keeps each center aware of work
in progress and plans for the other centers, enabling them to more fully address
the critical knowledge gaps confronting the U.S. EPA.
Linking the activities of the five PM centers and the U.S. EPA labs on a conceptual
and operational basis to maximize productivity and relevance of the scientific
findings represents a new direction for university-based research, with long-term
significance. For example, for biological mechanisms, investigators from all
five PM centers participated in discussions about unifying hypotheses of biological
mechanisms for PM health effects, which include inflammation and immunity, mechanisms
for cardiovascular effects, and the role of ROS.
There are other valuable features of the PM centers programs.
- Acceleration of the research process. In a rapidly evolving field
such as PM research, investigators need to initiate new projects that may
have a significant impact on the field. Conducting hypothesis-generating studies
can be of paramount importance. The PM centers program has used pilot project
funds to expedite the process of conducting exploratory research.
- Use of science advisory committees. The PM centers have effectively
used groups of experts to oversee their research programs, and most of the
PM centers' external science advisory committees include researchers from
other PM centers. This has helped to coordinate research efforts among the
PM centers, avoid duplication, and share resources.
- Leveraging funds to start new research initiatives. The PM centers
can provide seed funds for future research initiatives and create opportunities
for young investigators to be integrated into the overall research of the
PM centers.
- Development of new technologies. The PM centers have the resources
to develop complex new technologies that are available to the individuals
in PM centers as well as to other affiliated research groups. The creation
of the new ultrafine concentrator and multipollutant sampler are examples
of innovative technology that derived from the creation of the PM centers.
Oversight of the application of new technologies across the PM centers facilitates
their validation and optimal utilization.
- Flexibility to reallocate funds as needed and to change research
direction. The PM centers have sometimes been faced with unexpected research
opportunities and/or expenses. On the basis of the input from their science
advisory committees, the PM centers can reduce or expand activities in certain
projects in favor of others.
Particulate Matter Center Strengths
Each of the PM centers has particular strengths, as outlined below.
Strengths of the NYU PM center include a capacity to conduct studies of the
acute and cumulative effects of concentrated ambient air particles on cardiac
and respiratory system effects in laboratory animals; a focus on particle dosimetry
in the respiratory tract in both normal and abnormal human lungs; capabilities
for the generation of exposure atmospheres of freshly formed ultrafine and fine
monodisperse aerosols of acid aerosols, elemental and organic particles, and
metal oxides; and an array of inhalation chambers and head-only exposure chambers
for controlled exposure studies.
Strengths of the Rochester PM center include an interdisciplinary approach
in ambient characterization studies, epidemiological studies, clinical studies,
animal studies, and in vitro mechanistic studies; a focus on the role
of ultrafine particles compared with other particle-size fractions; and experience
in performing studies using laboratory-generated surrogate particles as well
as real-world particles.
Strengths of the southern California PM center include a well-defined focus
on mobile source pollution and health effects, with emphasis on investigation
of the biological mechanisms of PM effects in relation to PM characteristics;
emission sources and related adverse health effects; and the varying spatial
and temporal patterns of ambient PM and co-pollutants and resulting health effects,
with emphasis on field measurements and modeling.
Other strengths include the uniqueness of the Los Angeles basin airshed and
close interaction with the Los Angeles Supersite; expertise from five leading
universities in the region as well as the University of California Davis (Davis,
California) and Michigan State University (East Lansing, Michigan); extensive
ties with the CHS; the availability of mobile ultrafine, fine, and coarse concentrators
for sample collection and in vivo animal and human clinical studies;
and a focus on ROS acting as electrophilic agents and playing a central role
in allergic airway disease including asthma and cardiovascular effects through
their ability to generate oxidative stress, inflammation, and immunomodulating
effects in the lungs and airways.
Strengths of the University of Washington PM center include experience in
exposure assessment; experience in studies of acute health effects; a mouse
model of CVD; a new chronic cohort mortality study (the Women's Health Initiative);
and access to an airshed in the western United States without the sulfate common
to the east and with a higher contribution from vegetative burning.
Strengths of the Harvard PM center include close interaction among biological,
epidemiological, exposure, and engineering research groups, which has led to
the development of sensitive animal models based on our epidemiology studies--in
turn, the controlled toxicology studies have developed refined outcomes that
are currently being used in epidemiological and exposure group studies (Steubenville
and St. Louis); experience in conducting panel studies examining the relationships
between outdoor and indoor PM concentrations and corresponding personal exposures;
development of statistical models for use in time-series analyses addressing
harvesting, confounding, and dose-response relationships; a focus on methods
development for measuring continuous sulfate and nitrate concentrations, personal
particulate, and gaseous exposures; a PM concentrator for coarse, fine and ultrafine
particle modes; and development of novel inhalation technologies for exposing
individuals to ambient concentrations of PM and improving dosimetric techniques
in animal exposure models.
Particulate Matter Center Interactions
PM center personnel have met on an annual basis since the inception of the
centers. At the first PM center directors meeting, the directors and their colleagues
described the research planned or under way within their respective PM centers.
This was a period of familiarization and was characterized by informative reports.
At the third meeting in Boston (July 2001), there was a sea change in the approach
of the PM centers to their mission. It became apparent that there needed to
be greater interaction across PM centers in an intellectual context as well
as greater collaboration in a wide range of research areas. There was a clear
need expressed to communicate more effectively on an ongoing basis and to interact
with the U.S. EPA in a collaborative context to assist in the U.S. EPA research
program more fully.
Since July 2001, the PM center directors and selected colleagues have held
conference calls on a regular basis to discuss directions in research and collaborations.
These discussions have resulted in development of both short- and long-term
research goals, with intercenter collaborations as an important mechanism in
achieving these goals. In this report, both short-term and long-term research
goals are described for the PM centers overall and form the basis for collaboration
and interaction over a second 5-year period of funding. There are now plans
for a number of joint research efforts and workshops. The PM center directors
believe that funding of individual research grants should continue through the
STAR program and other sources, e.g., NIEHS and HEI, and one challenge would
be to find a way to link the research of individual investigators and research
teams to the unified efforts of the PM centers. We believe that a series of
defined workshops organized by the PM centers would bring together investigators
from the PM centers and the STAR program to discuss research findings in the
context of the U.S. EPA regulatory needs and to seek out opportunities for further
collaboration. This approach would be best organized through a joint U.S. EPA-centers
effort. The ability to link a wide range of investigators from around the country,
both intellectually and practically, represents a clear advantage of the PM
centers structure.
Particulate Matter Centers Research and Its Implications for Public Health
and Regulatory Policy
Using complementary approaches, the centers are addressing key policy issues
such as
- What properties of PM are responsible for health effects? This issue
was identified by the NRC committee as of critical importance. In testing
hypotheses concerning PM toxicity and attribution to specific constituents
or characteristics, work in the PM centers is addressing particle mass versus
surface area versus number concentration (ultrafines); the role of transition
metals; and the contributions to toxicity of acidity versus organic compounds
versus elemental carbon. Toxicological approaches are employing exposures
to CAPs or laboratory-generated PM in humans and animals; in vitro
approaches are using a number of cell lines and specific particles; and epidemiological
studies are focusing on different PM mixtures. This work should contribute
not only to our understanding of mechanisms of response but also to development
of more targeted control strategies.
- Are there specific risk factors or effect modifiers for PM effects?
This is another high priority issue. Using a variety of approaches, PM center
programs are examining risk factors such as age, gender, nutrition, and preexisting
cardiopulmonary disease. All of the PM centers are currently conducting work
to identify and characterize susceptible populations, a clear prerequisite
of any regulatory policy.
- What types of PM pose the greatest health risks? What effects are
directly attributable to PM, and what health responses may be modified by
other air pollutants? These are areas of intense research focus within the
PM centers using the different research strengths housed within each of them.
It is the output from these integrated programs that will help set the next
PM NAAQS and address issues raised by the public, industry, government, and
public advisory bodies such as CASAC.
References
Abbey DE, Nishino N, McDonnell WF, Burchette RJ, Knutsen SF,
Lawrence Beeson W, et al. 1999. Long-term inhalable particles and other air
pollutants related to mortality in nonsmokers. Am J Respir Crit Care Med 159:373-382.
Allen R, Larson T, Wallace L, Liu L-JS. In Press. Investigation
of indoor and outdoor contributions to total indoor particulate matter exposure.
Environ Sci Technol.
Avol EL, Gauderman WJ, Tan SM, London SJ, Peters JM. 2001.
Respiratory effects of relocating to areas of differing air pollution levels.
Am J Respir Crit Care Med 164:2067-2072.
Bateson TF, Schwartz J. 2001. Who is sensitive to the effects
of particles on mortality? A case-crossover analysis. Epidemiology 12(4):448.
Boscia JA, Chalupa D, Utell MJ, Zareba W, Konecki JA, Morrow
PE, et al. 2000. Airway and cardiovascular effects of inhaled ultrafine carbon
particles in resting, healthy, nonsmoking adults [Abstract]. Am J Respir Crit
Care Med 161:A239.
Chalupa D, Gibb FR, Morrow PE, Oberdörster G, Riesenfeld
E, Gelein R, et al. 2002a. A facility for controlled human exposures to ultrafine
particles. In: Crucial Issues in Inhalation Research--Mechanistic, Clinical
and Epidemiologic (Heinrich U, Mohr U, eds). INIS Monographs. Stuttgart, Germany:Fraunhofer
IRB Verlag, 241-253.
Chalupa DC, Morrow PE, Oberdörster G, Speers D, Daigle
C, Utell MJ, et al. 2002b. Deposition of ultrafine carbon particles in subjects
with asthma [Abstract]. Am J Respir Crit Care Med 165:A829.
Clarke RW, Coull B, Reinisch U, Catalano P, Killingsworth CR,
Koutrakis P, et al. 2000. Inhaled concentrated ambient particles are associated
with hematologic and bronchoalveolar lavage changes in canines. Environ Health
Perspect 108:1179-1187.
Clayton CA, Perritt RL, Pellizzari ED, Thomas KW, Whitmore
RW, Wallace LW, et al. 1993. Particle total exposure assessment methodology
(PTEAM) study. J Expo Anal Environ Epidemiol 3:227-250.
Clean Air Act. 1991. Clean Air Act Section 108 Air Quality
Criteria, Section 109 National Ambient Air Quality Standards. U.S.C. 42: Sections
7408-7409.
Couderc J-P, Elder ACP, Cox C, Zareba W, Oberdörster G.
2002. Limitation of power spectrum and time-domain analysis of heart rate variability
in short-term ECG recorded using telemetry in unrestrained rats. Comput Cardiol
29:589-592.
Daigle CC, Chalupa DC, Gibb FR, Morrow PE, Oberdörster
G, Utell MJ, et al. 2003. Ultrafine particle deposition in humans during rest
and exercise. Inhal Toxicol 15:539-552.
Daigle CC, Speers DM, Chalupa D, Stewart JC, Frasier LM, Azadniv
M, et al. 2002. Ultrafine particle exposure alters expression of CD40 ligand
(CD154) in healthy subjects and subjects with asthma [Abstract]. Am J Respir
Crit Care Med 165:A214.
Demokritou P, Kavouras IG, Ferguson ST, Koutrakis P. 2001.
Development and laboratory performance evaluation of a personal multipollutant
sampler for simultaneous measurements of particulate and gaseous pollutants.
Aerosol Sci Technol 35:741-752.
Dills RL, Zhu X, Kalman DA. 2001. Measurement of urinary methoxyphenols
and their use for biological monitoring of wood smoke exposure. Environ Res
85:145-158.
Dockery DW, Cunningham J, Damokosh AI, Neas LM, Spengler JD,
Koutrakis P, et al. 1996. Health effects of acid aerosols on North American
children: respiratory symptoms. Environ Health Perspect 104:500-505.
Dockery DW, Pope CA III, Xu X, Spengler JD, Ware JH, Fay ME,
et al. 1993. An association between air pollution and mortality in six U.S.
cities. N Engl J Med 329:1753-1759.
Dominici F, McDermott A, Zeger SL, Samet JM. 2002. On the use
of generalized additive models in time-series studies of air pollution and health.
Am J Epidemiol 156(3):193-203.
Elder ACP, Gelein R, Azadniv M, Frampton M, Finkelstein JN,
Oberdörster G. 2002. Systemic interactions between inhaled ultrafine particles
and endotoxin. Ann Occup Hyg 46(suppl 1): 231-234.
Elder ACP, Gelein R, Finkelstein JN, Cox C, Oberdörster
G. 2000. Pulmonary inflammatory response to inhaled ultrafine particles is modified
by age, ozone exposure, and bacterial toxin. Inhal Toxicol 12:227-246.
Finkelstein JN, Reed C, Johnston C, Oberdörster G. 2002.
Age alters macrophage responses to endotoxin and particle-induced cytokine gene
expression [Abstract]. Am J Respir Crit Care Med 165(8):A828.
Frampton MW. 2001. Systemic and cardiovascular effects of airway
injury and inflammation: ultrafine particle exposure in humans. Environ Health
Perspect 109:529-532.
Frampton MW, Azadniv M, Chalupa D, Morrow PE, Gibb FR, Oberdörster
G, et al. 2001. Blood leukocyte expression of LFA-1 and ICAM-1 after inhalation
of ultrafine carbon particles [Abstract]. Am J Respir Crit Care Med 163:A264.
Frampton MW, Chalupa D, Morrow PE, Gibb FR, Oberdörster
G, Boscia J, et al. 2000. Deposition of inhaled ultrafine carbon particles in
resting healthy nonsmoking adults [Abstract]. Am J Respir Crit Care Med 161:A257.
Frampton MW, Zareba W, Daigle CC, Oberdörster G, Utell
MJ. 2002. Inhalation of ultrafine particles alters myocardial repolarization
in humans [Abstract]. Am J Respir Crit Care Med 165:B16.
Godleski JJ, Verrrier RL, Koutrakis P, Catalano P, Coulll BA,
Reinisch U, et al. 2000. Mechanisms of morbidity and mortality from exposure
to ambient air particulate. Research Report no 91. Cambridge, MA:Health Effects
Institute
Goswami E, Larson T, Lumley T, Liu LJS. 2002. Spatial characteristics
of fine particulate matter: identifying representative monitoring locations
in Seattle. J Air Waste Manage Assoc 52:324-333.
ICRP (International Commission on Radiological Protection).
1994. Human Respiratory Tract Model for Radiological Protection. ICRP Publ no
66. Ann ICRP 24(1-3):1-482.
Ito K, Thurston GD, Nádas A, Lippmann M. 2001. Monitor-to-monitor
temporal correlation of air pollution and weather variables in the North-Central
U.S. J Expo Anal Environ Epidemiol 11:21-32.
Janssen NA, Hoek G, Brunekreef B, Harssema H, Mensink I, Zuidhof
A. 1998. Personal sampling of particles in adults: relation among personal,
indoor and outdoor air concentrations. Am J Epidemiol 147(6):537-547.
Janssen NA, Schwartz J, Zanobetti A, Suh HH. 2002. Air conditioning
and source-specific particle as modifiers of the effect of PM10 on
hospital admissions for heart and lung disease. Environ Health Perspect 110:43-49.
Kim CS. 2000. Methods of calculating lung delivery and deposition
of aerosol particles. Respir Care 45:695-711.
Koenig JQ, Jansen K, Mar TF, Kaufman J, Sullivan J, Liu L-JS.
2002. Measurement of offline nitric oxide in an air pollution health effect
study [Abstract]. Am J Respir Crit Care Med 165:A306.
Krewski D, Barnett RJ, Goldberg MS, Hoover K, Siemiatycki J,
Jerrett M, et al. 2000. Reanalysis of the Harvard Six Cities study and the American
Cancer Society study of particulate air pollution and mortality. Cambridge,
MA:Health Effects Institute.
Kreyling WG, Semmler M, Erbe F, Mayer P, Takenaka S, Schulz
H, et al. 2002. Ultrafine insoluble iridium particles are negligibly translocated
from lung epithelium to extrapulmonary organs. J Toxicol Environ Health 65:1513-1530.
Laden F, Neas LM, Dockery DW, Schwartz J. 2000. Association
of fine particulate matter from different sources with daily mortality in six
U.S. cities. Environ Health Perspect 108:941-947.
Laden F, Schwartz J, Speizer FE, Dockery DW. 2001. Air pollution
and mortality: a continued follow-up in the Harvard Six Cities study [Abstract].
Epidemiology 12:S81.
Larson T, Allen R, Liu LJS. 2001. Indoor and outdoor contributions
to indoor light scattering coefficient at 54 residences in Seattle, WA. Presented
at 20th Annual AAAR Conference, 15-19 October 2001, Portland, Oregon.
Lipfert FW, Perry HM Jr, Miller JP, Baty JD, Wyzga RE, Carmody
SE. 2000. The Washington University-EPRI Veterans' cohort mortality study: preliminary
results. Inhal Toxicol 12:41-73.
Luchtel D, Fu C, Ghatpande P. 2002. A mouse model to study
toxicity of particulate matter (PM) [Abstract]. Am J Respir Crit Care Med 165:A301.
Mar TF, Norris GA, Koenig JQ, Larson TV. 2000. Association
between air pollution and mortality in Phoenix, 1995-1997. Environ Health
Perspect 108:347-353.
NAAQS. 1997. National Ambient Air Quality Standards for Particulate
Matter; Final Rule. Fed Reg 62:38, 652-38, 752.
Nadziejko C, Fang K, Gordon T, Chen LC. 2001. Quantitative
analysis of arrhythmias. Presented at the Workshop on Cardiovascular Effects
Associated with Air Pollution, 7-8 March 2001, Rochester, New York.
Oberdörster G, Sharp Z, Atudorei V, Elder A, Gelein R,
Lunts A, et al. 2002. Extrapulmonary translocation of ultrafine carbon particles
following whole-body inhalation exposure of rats. J Toxicol Environ Health 65:1531-1543.
Ozkaynak H, Xue J, Spengler J, Wallace L, Pellizzari E, Jenkins
P. 1996. Personal exposure to airborne particles and metals: results from the
particle TEAM study in Riverside, CA. J Expo Anal Environ Epidemiol 6:57-78.
Peters A, Dockery DW, Muller JE, Mittleman MA. 2001. Increased
particulate air pollution and the triggering of myocardial infarction. Circulation
103(23):2810-2815.
Pope CA III, Burnett RT, Thun MJ, Calle EE, Krewski D, Ito
K, et al. 2002. Lung cancer, cardiopulmonary mortality and long-term exposure
to fine particulate air pollution. J Am Med Assoc 287:1132-1141.
Pope CA III, Thun MJ, Namboodiri MM, Dockery DW, Evans JS,
Speizer FE, et al. 1995. Particulate air pollution as a predictor of mortality
in a prospective study of U.S. adults. Am J Respir Crit Care Med 151:669-674.
Raizenne M, Neas LM, Damokosh AI, Dockery DW, Spengler JD,
Koutrakis P, et al. 1996. Health effects of acid aerosols on North American
children: pulmonary function. Environ Health Perspect 104:506-514.
Ridker PM, Hennekens CH, Roitman-Johnson B, Stampfer MJ, Allen
J. 1998. Plasma concentration of soluble intercellular adhesion molecule 1 and
risks of future myocardial infarction in apparently healthy men. Lancet 351:88-92.
Rojas-Bracho L, Suh HH, Koutrakis P. 2000. Relationships among
personal, indoor, and outdoor fine and coarse particle concentrations for individuals
with COPD. J Expo Anal Environ Epidemiol 10:294-306.
Saldiva PHN, Clarke RW, Coull BA, Stearns R, Lawrence J, Koutrakis
P, et al. 2002. Acute pulmonary inflammation induced by concentrated ambient
air particles is related to particle composition. Am J Respir Crit Care Med
165:1610-1617.
Sarnat JA, Koutrakis P, Suh H. 2000. Assessing the relationship
between personal particulate and gaseous exposures of senior citizens living
in Baltimore, MD. J Air Waste Manage Assoc 50:1184-1198.
Sarnat JA, Schwartz J, Catalano PJ, Suh HH. 2001. Gaseous pollutants
in particulate matter epidemiology: confounders or surrogates? Environ Health
Perspect 109:1053-1061.
Schwartz J. 1994. Total suspended particulate matter and daily
mortality in Cincinnati, Ohio. Environ Health Perspect 102:186-189.
------. 2000a. Harvesting and long term exposure effects in
the relation between air pollution and mortality. Am J Epidemiol 151:440-448.
------. 2000b. Daily deaths are associated with combustion
particles rather than SO2 in Philadelphia. Occup Environ Med 57:692-697.
------. 2001. Is there harvesting in the association of airborne
particles with daily deaths and hospital admissions? Epidemiology 12:55-61.
Schwartz J, Ballester F, Saez M, Pérez-Hoyos S, Bellido
J, Cambra K, et al. 2001. The concentration-response relation between air pollution
and daily deaths. Environ Health Perspect 109:1001-1006.
Schwartz J, Zanobetti A. 2000. Using meta-smoothing to estimate
dose-response trends across multiple studies, with application to air pollution
and daily death. Epidemiology 11(6):666-672.
Sullivan J, Schildcrout J, Budge M, Ishikawa N, Trenga C, Liu
S, et al. 2002. Association between short-term exposure to fine particulate
matter and heart rate variability in older subjects with and without heart disease
[Abstract]. Am J Respir Crit Care Med 165:A304.
Trenga CA, Slaughter C, Sullivan J, Jansen K, Neas LM, Kaufman
J, et al. 2002. The effect of fine particulate (PM2.5) air pollution
exposure on pulmonary function in elderly subjects [Abstract]. Am J Respir Crit
Care Med 165:A305.
Utell MJ, Frampton MW. 2000. Acute health effects of ambient
air pollution: the ultrafine particle hypothesis. J Aerosol Med 13:355-359.
Utell MJ, Frampton MW, Zareba W, Devlin RB, Cascio WE. 2002.
Cardiovascular effects associated with air pollution: potential mechanisms and
methods of testing. Inhal Toxicol 14:101-117.
Wellenius GA, Coull BA, Godleski JJ, Koutrakis P, Okabe K,
Savage ST, et al. 2003. Inhalation of concentrated ambient air particles exacerbates
myocardial ischemia in conscious dogs. Environ Health Perspect 111:402-408.
Wellenius GA, Saldiva PH, Batalha JR, Krishna Murthy GG, Coull
BA, Verrier RL, et al. 2002. Electrocardiographic changes during exposure to
residual oil fly ash (ROFA) particles in a rat model of myocardial infarction.
Toxicol Sci 66:327-335.
Whitekus MJ, Li N, Zhang M, Wang M, Horwitz MA, Nelson SK,
et al. 2002. Thiol antioxidants inhibit the adjuvant effects of aerosolized
diesel exhaust particles in a murine model for ovalbumin sensitization. J Immunol
168:2560-2567.
Wichmann HE, Peters A. 2000. Epidemiological evidence of the
effects of ultrafine particle exposure. Philos Trans R Soc 358:2751-2769.
Wichmann HE, Spix C, Tuck T, Wolke G, Peters A, Heinrich J,
et al. 2000. Daily mortality and fine and ultrafine particles in Erfurt, Germany.
Part 1: Role of particle number and particle mass. HEI Report no 98. Cambridge,
MA:Health Effects Institute.
Won C-H, Cook-Granroth J, Cohen BS, Hoffman EA. 2000a. X-Ray
CT-based assessment of variations in human airway geometry: implications for
evaluation of particle deposition and dose to different populations. Presented
at the 86th Scientific Assembly and Annual Meeting of the Radiological Society
of North America, November 26-1 December 2000, Chicago, Illinois.
Won C-H, Cook-Granroth J, Hoffman EA, Cohen BS. 2000b. X-Ray
CT-based assessment of variations in human airway geometry and the implications
for the evaluation of particle deposition and dose to different populations.
Presented at the 19th Annual Conference of the American Association for Aerosol
Research, 7 November 2000, St. Louis, Missouri.
Yu O, Sheppard L, Lumley T, Koenig JQ, Shapiro GG. 2000. Effects
of ambient air pollution on symptoms of asthma in Seattle-area children enrolled
in the CAMP study. Environ Health Perspect 108:1209-1214.
Zanobetti A, Schwartz J. 2000. Race, gender and social status
as modifiers of the effects of PM10 on mortality. J Occup Environ
Med 42:469-474.
------. 2001. Are diabetics more susceptible to the health
effects of airborne particles? Am J Respir Crit Care Med 164(5):831-833.
------. 2002. Cardiovascular damage by airborne particles:
are diabetics more susceptible? Epidemiology 13:588-592.
Zanobetti A, Schwartz J, Dockery DW. 2000. Airborne particles
are a risk factor for hospital admissions for heart and lung disease. Environ
Health Perspect 108:1071-1077.
Zanobetti A, Schwartz J, Samoli E, Gryparis A, Touloumi G,
Atkinson R, et al. 2002. The temporal pattern of mortality response to air pollution:
a multicity assessment of mortality displacement. Epidemiology 13:87-93.
Zareba W, Nomura A, Couderc JP. 2001. Cardiac effects of air
pollution: what to measure in ECG? Environ Health Perspect 109:533-538.
Zelikoff JT, Nadziejko C, Fang K, Gordon T, Premdass C, Cohen
MD. 1999. Short-term, low-dose inhalation of ambient particulate matter exacerbates
ongoing pneumococcal infections in Streptococcus pneumoniae-infected
rats. In: Proceedings of the Third Colloquium on Particulate Air Pollution and
Human Health, 6-8 June 1999, Durham, North Carolina (Phalen R, Bell, Y,
eds). Irvine, CA:University of California, 8-94-8-101.
Zhu Y, Hinds WC, Kim S, Sioutas C. 2002. Concentration and
size distribution of ultrafine particles near a major highway. J Air Waste Manage
Assoc 52:1032-1042.
Last Updated: June 12, 2003







