In the last decade many studies have assessed the association between daily deaths or hospital admissions and air pollution, both in Europe and in the United States (
1-12). Almost all of these studies reported associations between airborne particles (and sometimes other pollutants) and deaths or hospital admissions within a few days of exposure, but they have differed in the exact lag between exposure and outcome used. They have also differed in whether they examined only associations with a 24-hr averaged exposure or considered effects spread out over several days.
When studies have considered the possibility of lags or multiday effects, they usually have used ad hoc approaches based on the best fit in individual cities, which can be subject to substantial variability due to stochastic error. A systematic approach, which used a multicity analysis to overcome stochastic variability, would help clarify this situation. This has recently been applied successfully to the association between particulate matter 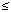 10 µm (PM10) and mortality (13). Past studies have traditionally relied on simple moving averages of pollution to assess the potential for the effect of air pollution on health to persist for more than 1 day after exposure. However, it is quite possible that the effect of air pollution decreases gradually over several days, perhaps after first rising to a peak. In that case, a weighted moving average, with weights that decline to zero after several days, would be more appropriate than a simple moving average or single day's exposure (13).
10 µm (PM10) and mortality (13). Past studies have traditionally relied on simple moving averages of pollution to assess the potential for the effect of air pollution on health to persist for more than 1 day after exposure. However, it is quite possible that the effect of air pollution decreases gradually over several days, perhaps after first rising to a peak. In that case, a weighted moving average, with weights that decline to zero after several days, would be more appropriate than a simple moving average or single day's exposure (13).
It is possible to include air pollution values on multiple days to directly estimate the effect of different lags, but this approach is limited in single-city analyses because multicollinearity makes the estimated effects of different lags very noisy. Although these estimates have large variance, they are unbiased, and hence a multiple-city analysis, which can average out the noise, makes this approach feasible (13). We have applied such a multicity approach to estimate the association between PM10 and hospital admissions for heart and lung disease, including the distribution of effects over time.
A multicity approach estimating the association between air pollution and hospital admissions has several other advantages. The National Academy of Sciences has stated that identifying individuals most sensitive to the adverse effects of particulate air pollution is a research priority (14). Multicity analyses allow us to investigate whether demographic or economic factors are modifiers of the
pollution effect. In addition, multicity approaches provide opportunities to separate the effect of different air pollutants, analyses which are of limited utility in single-city analyses (15). The present analysis examined distributed lag effects on hospital admissions, confounding by copollutants, and effect modification by socioeconomic factors in 10 locations from across the United States with daily measurements of PM10 but widely varying relationships between PM10 and other pollutants.
Data
To examine the effect of PM10 at multiple lags, we needed cities with daily PM10 monitoring, rather than the more usual 1 day in 6 monitoring schemes. We selected 10 cities from across the United States that met this criterion: Canton, Ohio; Birmingham, Alabama; Chicago, Illinois; Colorado Springs, Colorado; Detroit, Michigan; Minneapolis/ St. Paul, Minnesota; New Haven, Connecticut; Pittsburgh, Pennsylvania; Seattle, Washington; and Spokane, Washington. We chose the metropolitan county containing each city, except for Minneapolis and St. Paul, which were combined and analyzed as one city. We analyzed daily counts of hospital admissions for cardiovascular disease [CVD; International Classification of Disease, 9th revision (ICD-9) 390-429], chronic obstructive pulmonary disease (COPD; ICD-9 490-496, except 493), and pneumonia (ICD-9 480-487), in persons  65 years of age. The data were extracted from the Health Care Financing Administration (Medicare; Baltimore, MD) billing records, which we obtained for the years 1986-1994. The Medicare system provides hospital coverage for all U.S. citizens aged 65 and over.
65 years of age. The data were extracted from the Health Care Financing Administration (Medicare; Baltimore, MD) billing records, which we obtained for the years 1986-1994. The Medicare system provides hospital coverage for all U.S. citizens aged 65 and over.
Daily meteorologic measurements such as mean temperature, relative humidity, and barometric pressure, were obtained from the nearest National Weather Service surface station for each county (EarthInfo CD NCDC Surface Airways, EarthInfo Inc., Boulder, CO).
Air pollution data for PM10 were obtained from the U.S. Environmental Protection Agency's Aerometric Information Retrieval System (AIRS) (16). Many of the cities have more than one monitoring location. To ensure that our exposure measure best represented general population exposure and not local conditions, monitors within the lowest 10th percentile of the correlation among monitors across all counties were excluded. Some monitors only measure PM10 1 day in 6, and different monitors have different means and standard deviations. Therefore, we needed a scheme for computing the daily pollution value that did not change our exposure estimates day to day because of which monitors reported, as opposed to differences in actual ambient levels. Thus, the annual mean was computed for each monitor for each year and subtracted from the daily values of that monitor. We then standardized these daily deviances from each monitor's annual average by dividing by the standard deviation for that monitor. The daily standardized deviations for each monitor on each day were averaged, producing a daily averaged standardized deviation. We multiplied this by the standard deviation of all of the monitor readings for the entire year, and added back in the annual average of all of the monitors. This approach has been described previously (13).
We excluded days when PM10 exceeded the ambient air quality standard of 150 µg/m3 for the 24-hr mean in order to study the association at common concentrations. We also excluded days with hospital admissions outliers, defined as those days with daily counts more than four times the interquartile range above the median for pneumonia and CVD. For COPD, the outliers were defined as values that were three times the interquartile range above the median, or when the observations were at least 100% higher than the mean of the nearby data. These can occur for clerical reasons; for example, records without the date of admission are coded to the first of the month or year. Alternatively, these outliers may represent epidemics. This exclusion eliminated a total of 2 days of data for CVD, 44 days for pneumonia, and 13 days of data for COPD in all the 10 cities. The exclusion of these outliers did not have a marked effect on the regression coefficients for the PM10 effect.
Methods
In each city the associations between hospital admissions and PM10 were investigated with a generalized additive robust Poisson regression model (17). In the generalized additive model, the outcome is assumed to depend on a sum of nonparametric smooth functions for each variable. This allows us to better model the nonlinear dependence of daily admission on weather and season. The model is of the form:
log[E(Yt)] =  0+ S1 (X1) + ... + Sp (Xp),
0+ S1 (X1) + ... + Sp (Xp),
where E(Yt) is the expected value of the daily count of admissions (Yt) and Si are the locally weighted, running-line, smooth functions (Loess) of the covariates Xi (18).
All nonparametric smoothing functions are characterized by a smoothing parameter, which determines the smoothness of the fit. To control for weather variables (24-hr means of temperature, relative humidity, and barometric pressure) and day of the week, we chose the smoothing parameters in each city that minimized the Akaike's Information Criteria (19).
We chose city-specific smoothing parameters for season, which assure seasonal patterns have been removed, and to minimize autocorrelation of residuals. In some cases it was necessary to use autoregressive terms to eliminate serial correlation (12,20).
PM10 was treated as a linear term in our analysis to allow examination of how its effects were distributed over different lags and to allow the use of meta-analytic techniques to combine results across cities.
It has been argued that there are thresholds for the effects of air pollution and that no adverse responses occur on most days. To test this we repeated our analysis, restricting it to days when PM10 was < 50 µg/m3, which is one-third of the current U.S. 24-hr mean national ambient air quality standard (21).
Distributed lag models. Distributed lag models were introduced by Almon (22) and have been mainly applied in econometrics and social sciences. These models allow us to examine the possibility that air pollution can influence hospital admissions on the same day, but also on subsequent days.
The unconstrained distributed lag model of order q is
log[E(Yt)] =  + covariates + ß0Zt
+ covariates + ß0Zt
+ ß1Zt-1 + . . . + ßqZt-q. [1]
Hence, the outcome Yt at time t may depend on the exposure (Zt) measured not only on the current day but also on previous days. The overall impact of a unit change in exposure on one day is the sum of its impact on that day and its impacts on subsequent days (i.e., ß0 + ß1 + . . . + ßq). The problem is that Zt is correlated with Zt-1, ... , Zt-q and the high degree of collinearity will result in unstable estimates of the ßj. However, both the ßj and the sum of all ßj will be unbiased estimators of the effects at each lag and of the overall effects. Because they are unbiased, combining results across cities will produce more stable unbiased estimates.
A 1-day exposure model can be seen as a constrained model, where ßj = 0 for j = 1 ... q. If we have no strong biological reason for that constraint, it is preferable to let the data tell us what the actual pattern looks like. While the 1-day model may be an unreasonably strong constraint, which risks introducing bias, a more flexible constraint may reduce the variance of the individual ß with less risk of bias. One common approach is to constrain the ß values to follow a flexible polynomial (13,23-25).
We have used the unconstrained model as our primary approach, relying on the combined results across cities to cancel out noise and provide stable estimates. We used quadratic distributed lag models as a sensitivity analysis. In both cases we estimated lags of up to 5 days between exposure and hospitalization. For comparison to previous results, we estimated the effect of PM10 on the same day, and on the mean of the same and previous day as exposure variables.
Hierarchical modeling. Hierarchical modeling is a multistage approach in which a set of models are fit in (in our case) individual cities, and the results of those regressions are analyzed in a second-stage regression to examine issues of effect modification and confounding (26). In the second stage of the analysis we first used inverse-variance-weighted averages to combine results across cities. These were computed for both the estimated overall effect (the sum of the ßi) and for the effect of each lag. More formally, we assumed the effect of PM10 in city i (i = 1-10) was  i ~ N(µ,V), and we estimated µ from the 10 city-specific
i ~ N(µ,V), and we estimated µ from the 10 city-specific  i values and their variances by computing an inverse-
variance-weighted average. We then extended this approach to a full second-stage regression. To examine effect modification by socioeconomic status, for example, we fit a weighted, least-squares regression:
i values and their variances by computing an inverse-
variance-weighted average. We then extended this approach to a full second-stage regression. To examine effect modification by socioeconomic status, for example, we fit a weighted, least-squares regression:
 i = ß * +
i = ß * +  Pi +
Pi +  i, [2]
i, [2]
where  i is the estimated PM10 effect in city i, Pi is the socioeconomic index in that city, and, again, inverse variance weighting is done. The variable
i is the estimated PM10 effect in city i, Pi is the socioeconomic index in that city, and, again, inverse variance weighting is done. The variable  then tells us how much the effect of PM10 changes for a unit increase in the social index. We examined the percentage of the population living below the federal poverty level and the percentage of the population that was nonwhite as potential modifiers of the effect of PM10 on hospital admissions of the elderly.
then tells us how much the effect of PM10 changes for a unit increase in the social index. We examined the percentage of the population living below the federal poverty level and the percentage of the population that was nonwhite as potential modifiers of the effect of PM10 on hospital admissions of the elderly.
Confounding is usually examined by including potential confounders in what is here the first stage of a hierarchical regression model. However, because weather tends to increase or decrease all pollutants in parallel, that approach risks substantial collinearity problems. Although most pollutants increase and decrease together, the incremental increase in one pollutant (in micrograms per cubic meter) that is associated with each microgram per cubic meter increase in another pollutant varies substantially across locations. We have used this variation to examine confounding in the second stage of our analysis.
To illustrate this approach, suppose the true association is between our outcome and pollutant X1:
Y = ß0 + ß1 X1 +  t. [3]
t. [3]
Assume X1 is correlated with another pollutant, X2, that is not causal for Y. It is possible to quantify the association between them by
X1 =  0 +
0 +  1X2 +
1X2 +  t. [4]
t. [4]
Substituting Equation 4 in Equation 3 it follows that:
Y = ß0 + ß1 0 + ß1
0 + ß1 1X2 +
1X2 +  t,
t,
and we see that the induced coefficient for the noncausal variable X2 depends on  1, the slope of the relationship between X1 and X2. From this, we can see that it is natural to extend our meta-regression approach to use the slope between pollutants as an explanatory factor in the second-stage model. That is,
1, the slope of the relationship between X1 and X2. From this, we can see that it is natural to extend our meta-regression approach to use the slope between pollutants as an explanatory factor in the second-stage model. That is,
 i* = ß* +
i* = ß* + 
 i +
i +  i
i
where  i is the slope between SO2 and PM10, for example. ß*, the intercept term in this regression, is the estimated effect of PM10 in a city where it had no correlation with SO2. This is the unconfounded effect of PM10. This approach has recently been applied to mortality data (27).
i is the slope between SO2 and PM10, for example. ß*, the intercept term in this regression, is the estimated effect of PM10 in a city where it had no correlation with SO2. This is the unconfounded effect of PM10. This approach has recently been applied to mortality data (27).
Simulation study. To test the power of our two-stage approach to detect confounding, we did a simulation study. We simulated the case where one pollutant was really standing for another, and looked to see whether the association with the noncausal pollutant disappeared in our two-stage approach. Specifically, we examined a scenario where analyses were done in 10 cities, with 2,000 days of data in each location. This is somewhat fewer data than we have. In each location, we generated two exposure variables that were multivariate normal, with a correlation coefficient of 0.70. However, the regression coefficient between the two pollutants was chosen from a uniform distribution with a 3-fold variation in slopes. This is less variation than is present in the actual data we were analyzing.
We then generated a random Poisson count with a log relative risk for one pollutant of 0.05, and no true association with the other pollutant. We fit a Poisson regression in each of the 10 locations and estimated the regression coefficient of the noncausal pollutant in each location. Then we regressed those 10 coefficients against the 10 slopes relating the two pollutants and took the intercept term in that regression as the estimate of the nonconfounded effect of the noncausal pollutant. We repeated this 500 times and looked at the median and 95% confidence interval for the noncausal pollutant to see if they were centered on zero and with magnitude that would clearly distinguish them from 0.05.
Table 1 shows the 25th, 50th, and 75th percentiles of each of the variables used in the analysis in each city. Colorado Springs had the lowest median PM10 concentration, and Spokane had the highest. Table 1 also shows the dates during which daily PM10 measurements were available in each city. Table 2 presents the population  65 years of age and the percentile values for the hospital admissions data. Table 3 shows the correlation between PM10 and the weather variables. The correlations were always modest and, for temperature and barometric pressure, include both positive and negative correlations. In one city (Spokane) PM10 was essentially uncorrelated with temperature.
65 years of age and the percentile values for the hospital admissions data. Table 3 shows the correlation between PM10 and the weather variables. The correlations were always modest and, for temperature and barometric pressure, include both positive and negative correlations. In one city (Spokane) PM10 was essentially uncorrelated with temperature.



Overall effects of PM10. Table 4 shows the combined overall estimate for the constrained (1-day mean, 2-day mean, quadratic distributed lag) and the unconstrained distributed lag model, for a 10 µg/m3 increase in PM10. The effect size estimate for the 2-day mean and the quadratic distributed lag are similar to the effect estimate using the unconstrained model, and all three are always higher than the 1-day lag. When the analysis using the 2-day mean of PM10 was repeated using only days with PM10 < 50 µg/m3, the effect size increased by  20% for all three outcomes.
20% for all three outcomes.

Distributed lag over time. Figures 1-3 show the combined city estimate of the unconstrained distributed lag association between PM10 and the three analyzed causes of admissions. For COPD admissions (Figure 1) the effect is similar for PM10 on the concurrent day and the previous day and goes to near zero at lag 2 and subsequent days. For pneumonia admissions (Figure 2) the effect decreases continuously for lags 0-2 and then oscillates about zero for further lags. Cardiovascular admissions (Figure 3) show a higher effect at lag 0, dropping to a more modest effect at lags 1 and 2, and then oscillate about zero.

|
|
Figure 1. The combined city estimate of the unconstrained distributed lag between air pollution and COPD admissions. CL, confidence limits. The effect size plotted is the percent increase for a 10 µg/m3 increase in PM10. |

|
|
Figure 2. The combined city estimate of the unconstrained distributed lag between air pollution and pneumonia. CL, confidence limits. The effect size plotted is the percent increase for a 10 µg/m3 increase in PM10. |

|
|
Figure 3. The combined city estimate of the unconstrained distributed lag between air pollution and CVD. CL, confidence limits. The effect size plotted is the percent increase for a 10 µg/m3 increase in PM10. |
Second-Stage Models
Social factors. Neither the percentage of the population living in poverty nor the percentage of the population that was nonwhite was a significant modifier of the PM10 slopes in our cities. Table 5 shows the change from the baseline PM10 effect size (as percent increase in admission per 10 µg/m3 increase in concentration) associated with a 5-point increase in the percentage of the population living below the federal poverty level or the percentage of the population that is not white.

Copollutants. Figures 4 and 5 show the data for of the meta-regression. Figure 4 shows, for COPD and pneumonia, the effect of PM10 in each city plotted against the regression coefficients relating SO2 and ozone to PM10 in each city. Figure 5 presents the CVD results, where we considered also the regression coefficients of CO versus PM10.

Figure 4. The effect of PM10 on COPD and pneumonia in each city plotted against the regression coefficients relating SO2 and O3 to PM10.

Figure 5. The effect of PM10 on cardiovascular admissions in each city plotted against the regression coefficients relating SO2, CO, and O3 to PM10.
These plots give an idea of the range of the results by city. These vary from a negative effect to effects higher than a 6% increase for 10 µg/m3 PM10 for COPD or pneumonia, while for CVD the higher effects are around 2%. They also show the range of regression coefficients relating PM10 to the other pollutants. For O3 they include both positive and negative slopes and vary considerably within each sign, with a wider range among the positive slopes. For SO2 and CO the slopes are always positive, but vary by almost an order of magnitude.
As explained in "Methods," if the PM10 effect were due to confounding with other pollutants, the plots would show a significantly increasing trend with increasing slope between the pollutants. Figures 4 and 5 show little evidence of such a pattern. These results are confirmed by the meta-regression estimates, shown in Figure 6. Here the baseline estimate is the result of the distributed lag meta-analysis. Plotted above each pollutant is the estimated intercept in the meta-regression of the PM10 coefficients against the slopes between that copollutant and PM10. For all three outcomes the results appear quite stable to control for confounding by gaseous pollutants. Moreover, there are no consistent patterns indicative of confounding. For example, the effect of PM10 on pneumonia admissions increases somewhat after control for SO2 and decreases after control for O3; for COPD the pattern is the opposite. None of the copollutants was a significant predictor of the PM10 slope.

Figure 6. The results of the meta-regression estimates for CVD, COPD, and pneumonia. The baseline estimate is the result of a simple meta-analysis. Plotted above each pollutant is the estimated intercept in the meta-regression of the PM10 coefficients against the slopes between that copollutant and PM10.
Weather variables. The wide range of weather patterns, shown in Table 3, give considerable support to the conclusion that these results are not confounded by inadequate control for weather. Figure 7, plotting the effect size estimates for the distributed lag PM10 versus the correlation of PM10 with temperature and relative humidity, shows similar effects sizes across a broad range of correlations. Hence, these results are unlikely to be confounded by weather. In the formal meta-regression we found that the coefficient for temperature was not significant for all the three outcomes, but for relative humidity we found some negative confounding with COPD. The effect size of PM10 is not modified by temperature; the percentage increase of 10 µg/m3 of PM10 is 1.2% for CVD (SE = 0.2); 3.3% for COPD (SE = 0.7), and 2.1% for pneumonia (SE = 0.5). There is no effect modification due to relative humidity for CVD (1.8%; SE = 0.4) and for pneumonia (1.7%; SE = 1.1), while the PM10 effect increased for COPD with a 5.5% increase (SE = 1.2).

Figure 7. The effect-size estimates for the 2-day mean PM10 versus the correlation of PM10 with temperature and relative humidity for the three causes.
Simulation. The 95% confidence interval for the slopes between the two simulated pollutants ranged from 0.48 to 1.27, reflecting the 3-fold range that was our target. In the meta-regression, the intercept term was taken as the non-confounded effect of the noncausal pollutant, as in our analysis of real data. The median estimate for this was -0.00008, and the 95% confidence interval was -0.0098-0.0102. This demonstrates that our approach has the power to detect significant confounding in a 10-cities study, with a smaller range of variation in pollutant-pollutant slopes than was seen in the study.
There are four main findings of this study. First, PM10 is associated with increased hospital admissions for CVD, COPD, and pneumonia. Second, the effect of a 24-hr increase in PM10 is spread over that day and several subsequent days, and single-day analyses underestimate the impact of PM10. Third, these effects are not modified by poverty rates or minority status and are relatively stable to control for potential confounding by SO2, O3, and CO. And fourth, these effects persist at common concentrations well below the current air quality standards. We discuss each of these findings in turn.
The finding that airborne particles are associated with hospital admissions for heart and lung disease has been reported in many other studies. In general, the effect-size estimate reported here is consistent with those previous studies. The advantage of this study is that it involves more years of follow-up than most previous studies and 10 cities spread across the continent, with a wide range of coincident weather and copollutants.
For all three outcomes, the effect of PM10 appears to be spread over more than 1 day, and Table 4 shows that the use of a single exposure day will underestimate the effect of PM10, sometimes by a substantial factor. This suggests that integrative summaries of the health data need to address this issue. Most studies of air pollution have used multiday averages but some have not, and this will need to be taken into account in any future meta-analysis. A recent analysis of daily deaths in these same cities found the use of a single day's exposure underestimated the effect of PM10 on daily deaths by more than a factor of 2, for instance (13).
Confounding by gaseous pollutants has been raised as a major issue regarding previous studies (28). We found that the effect-size estimate for PM10 and hospital admissions for CVD, COPD, and pneumonia changed little after control for potential confounding by gaseous air pollutants in our second-stage regression. The standard errors increased because our second-stage analysis had a limited sample size (10 points in a regression estimating an intercept and a slope), but overall the evidence for confounding was small. Temperature did not appear to confound the PM10 association either, whereas for relative humidity there seemed to be some negative confounding for COPD admissions.
We have not found evidence that obvious socioeconomic factors such as poverty and race are modifiers of these effects. There may be several reasons for this. First, it is important to realize that Poisson models are relative risk models. They have multiplicativity built in. That is, a given change in PM10 is associated with a given percent increase in admissions. If a town with more poverty or larger percentage of nonwhites has a higher baseline rate of admission, then a 3% increase in the admissions rate from baseline will be a greater increase (per 10,000 persons  65 years of age) in that town than in a town with a lower baseline rate.
65 years of age) in that town than in a town with a lower baseline rate.
It may be that the medical conditions that predispose to higher risk are not well captured by these socioeconomic factors and that more specific medical conditions, rather than social factors, are needed to explore effect modification. Finally, we used county-level data for these social factors because our admissions data are on that level. But the variation in socioeconomic status within the typical urban county is usually considerably larger than the variation across counties. Our social factors may be too ecologic to be meaningful. In this case, future studies using finer geographical data may be able to find some modification.
If these associations are causal, as we have argued, then it is crucial for public health impact assessment to know whether the associations are dominated by only a few high pollution days or whether they persist at the concentrations seen on most days. When we restricted our analysis to days with concentrations of one-third of current air quality standard or less (< 50 µg/m3), we still found a significant association between PM10 and admissions for all three illnesses. Moreover, the effect size increased by 20% or more. This increase in effect size at lower concentrations has been noted previously in a mortality study (6). For a significant association to persist, and grow in size, on days with levels < 50 µg/m3, any threshold would have to be far below that level, and likely down to background levels. The more likely scenario is that the true concentration-response curve is curvilinear, with higher slopes at lower concentrations and no threshold.
In addition to this statistical evidence, there has been a substantial increase in
evidence for the biological plausibility of these effects. Recent studies have reported that particulate air pollution is associated with reduced heart rate variability and increased fibrinogen levels in animals (29-31). These are known risk factors for arrhythmia and ischemic events, which are the major sources of hospital admissions for heart disease. Human studies have reported airborne particles associated with increased plasma viscosity (32) and decreased heart rate variability (33-35), paralleling animal studies. Airborne particles have also been associated with increased fibrinogen and platelet levels in humans (36); and they are associated with increased heart rate (37,38). These changes in risk factors for arrhythmia are supported by a recent study of patients with implanted cardiac defibrillators. Defibrillator discharges to halt arrhythmic events were associated with particulate air pollution and NO2 (39). Further, the increase in mortality associated with airborne particles was particularly strong for sudden death (40), which is again consistent with these recent animal and human results.
Animals with COPD or chronic lung inflammation have been shown to have increased vulnerability to combustion particles (41-44). And exposure to concentrated air particles of animals previously infected with strep pneumonia resulted in a doubling of lung area involved with pneumonia, and of bacterial burdens (45). Influenza infections have similarly been shown to be exacerbated by air pollution (46).
Given the consistent epidemiologic evidence, the indications of little, if any, confounding by gaseous copollutants and weather, the mechanistic animal studies showing airborne particles can exacerbate these illnesses, and the more recent mechanistic human studies, we believe that there is a strong case for causal associations between PM10 and heart and lung diseases.

