Biological Product and HCT/P Deviation Reports
Annual Summary for Fiscal Year 2006
Table of Contents
- Executive Summary
- BPD Reports Submitted By Blood And Plasma Establishments
- Reporting Issues
- Most Frequent BPD Reports Submitted by Licensed Blood Establishments
- Most Frequent BPD Reports Submitted by Unlicensed Blood Establishments
- Most Frequent BPD Reports Submitted by Transfusion Services
- Most Frequent BPD Reports Submitted by Plasma Centers
- Timeliness of BPD Reports
- BPD Reports Submitted by Manufacturers of Biological Products Other Than Blood and Blood Components (Non-Blood)
- Timeliness of BPD Reports
- HCT/P Deviation Reports Submitted by Manufacturers of 361 HCT/Ps
- Timeliness of BPD Reports
- Attachments
- We received more than 38,618 reports, but did not capture data for reports that did not meet the reporting threshold. We notified the reporter that a report was not required.
- The total number of reports we received in FY06 was approximately the same as the number received in the previous year (FY05 – 38,757, FY06 – 38,618) {Table #2}.
- The number of reporting establishments increased by 4% (1,409 establishments in FY05 and 1,481 establishments in FY06) {Table #2}. There were 80 additional HCT/P establishments reporting in FY06 that did not submit reports in FY05.
- Unregistered transfusion services typically report few BPDs (68% of those reporting in FY06 submitted 1 or 2 reports) and may file no reports in a given year. Only 15% of the transfusion services submitted more than 5 reports during FY06.
- There was an increase of 6 percentage points in the number of establishments reporting electronically (FY05 – 69% {974/1,409}; FY06 – 75% {1118/1481}). We continue to encourage electronic reporting.
- The number of reports submitted electronically increased by 10 percentage points (FY05 – 62%, FY06 – 72%) {Table #7}. There was an increase in the number of reports submitted electronically from licensed blood establishments (FY05 – 51%, FY06 – 79%), plasma centers (FY05 – 52%, FY06 – 61%), derivative manufacturers (FY05 – 30%, FY06 – 51 %), and in vitro diagnostic manufacturers (FY05 – 53%, FY06 – 68%).
- Reports of post-donation information (PDI) continue to represent the largest subset of BPD reports submitted by blood and plasma establishments (72%) {Table #8}. Most often (90%), the blood collector becomes aware of disqualifying information during a subsequent donation interview {Table 10}. In 90% of the PDI reports the donor was aware of the information, but the donor screening process failed to elicit the information {Table #11}. It is unclear why blood establishments are unable to elicit information during the first donor interview, but successfully elicit the information during a subsequent interview. It is clear that the most common PDI relates to travel (45%). Eliciting proper information regarding a donor candidate’s travel history is apparently the most problematic part of the donor qualification process.
- 2,046 (5.3%) of the reports received by CBER were sent to FDA District Offices for follow-up/evaluation as potential recall situations {Table #1}.
- Of the 1,982 reports submitted by blood and plasma establishments, deviations and unexpected events that occur during the donor screening process continue to be the leading cause of potential recall situations (41%) {Table #9}.
- There was an 18% decrease from the previous year in the number of reports submitted by licensed blood establishments involving donor screening that were potential recall situations.
- The number of reports in which a donor provided disqualifying information regarding travel to a malarial endemic area or vCJD risk area and was inappropriately accepted decreased by 23% from the previous year.
- There was a 23% increase, from the previous year, in the number of reports submitted by blood and plasma establishments in which they distributed a unit collected from a donor who subsequently tested confirmed positive for a viral marker. The majority of this increase in reports involved donors who subsequently tested confirmed positive for Hepatitis B or Hepatitis C. Based on the number of questions we received regarding the requirement to report this event and discussions at public meetings, we believe the increase over the past two years was most likely due to under reporting of this type of event in the previous years, rather than an increase in the number of donors who subsequently tested confirmed positive for any viral marker.
- There was a 20% increase, from the previous year, in the number of reports submitted by plasma centers involving post donation information related to behavior or history.
- There was a 34% increase in the number of post donation information reports in which the donor had a history of piercing.
- There was a 29% increase in the number of post donation information reports in which the donor had a history of receiving a tattoo.
- There was a 26% decrease, from the previous year, in the number of reports submitted by blood establishments involving collection.
- There was a 25% decrease in the number of reports in which a distributed product was found to be clotted, but was not identified prior to distribution.
- The number of reports in which a product was found to be hemolyzed, but was not identified prior to distribution decreased by 35 reports.
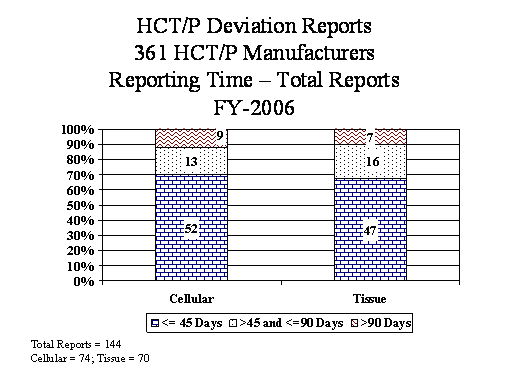
- FY06 was the first full year since the implementation of the deviation reporting requirement for HCT/Ps. We included data on HCT/P deviation reporting, for the first time, in this annual summary. We categorized the HCT/P deviation reports as either Cellular, which includes peripheral and cord stem cells or Tissue, which includes all other HCT/Ps, such as bone, skin, cornea, etc.
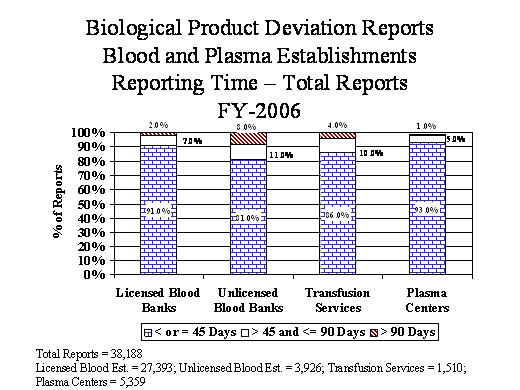
- Manufacturers must submit deviation reports within 45 calendar days of the date of discovery of the reportable event. In FY06, manufacturers submitted 90% of the blood BPD reports, 77% of the non-blood BPD reports, and 69% of the HCT/P deviation reports within 45 days {Tables #26, #29, and #31}. FDA investigators review reporting practices during establishment inspections, and we continue to publicize reporting requirements through professional meetings and publications.
- BPD Reports Submitted By Blood And Plasma Establishments:
- Reporting Issues
- Establishment distributed product collected from a donor who provided post donation information of cold or flu symptoms.
- Establishment distributed product collected from a donor who did not meet suitability criteria related to donor safety only, such as donor's weight, age, donating within 56 days of last donation, or more than 24 pheresis donations within 12 months.
- Establishment distributed product labeled with a shortened expiration date. This includes associated labeling, such as crossmatch tag or transfusion record.
- Establishment distributed an otherwise unsuitable product, such as product with incomplete testing, through appropriate emergency release procedures.
- Hospital staff transfused the wrong patient or transfused the patient with the wrong product. This event is not associated with manufacturing.
- Establishment distributed an allogeneic product when an autologous product was available.
- Recipient had a transfusion reaction unrelated to an event in manufacturing, such as Transfusion Related Acute Lung Injury (TRALI).
- Establishment distributed products collected from a donor who tested negative. The donor returned and tested reactive or repeat reactive, but not confirmed positive for a viral marker (HIV, HBV or HCV) for which we require or recommend product quarantine or consignee notification (i.e., lookback).
- Establishment distributed plasma for further manufacture which was collected from a donor who provided a history of travel to a malarial endemic area.
- Donor Screening (DS) vs. Post Donation Information (PD)
- If the blood establishment does not know the disqualifying information at the time of donation, the correct code is post donation information. If the establishment knows the disqualifying information or the information is available, but does not appropriately defer the donor, the event is a donor screening deviation.
- Routine Testing (RT) vs. Quality Control & Distribution (QC).
- A patient had a history of an antibody and the blood bank did not screen the unit for the corresponding antigen. The appropriate deviation code is QC9311 (Required testing not performed or documented for: antigen screen), not RT6106 (Testing performed, interpreted, or documented incorrectly for: antigen typing).
- Blood Collection (BC) vs. Quality Control & Distribution (QC)
- A blood establishment distributed a unit that was subsequently found to be clotted. The appropriate deviation code is BC4305.
- A blood establishment discovers a clotted component prior to distribution and discards the product. The appropriate deviation code is QC9405 if any associated products, such as FFP or Platelets, were distributed.
- Bacterial Detection Testing
- All events associated with bacterial detection testing should be coded as QC & Distribution; Distribution of product that did not meet specifications; Product with unacceptable (e.g., positive), undocumented, or incomplete product QC (QC9404).
- Most Frequent BPD Reports Submitted by Licensed Blood Establishments
- The number of these reports decreased by 2% (FY05 – 22,498).
- The number of reports in which a male donor subsequently provided information of a history of sex with another male increased by 18% (FY05 – 618).
- The number of reports in which a donor subsequently provided information regarding travel to a CJD risk area decreased by 16% (FY05 – 5,659).
- The number of reports in which a donor reported a subsequent diagnosis of cancer decreased by 23% (FY05 – 551).
- The number of reports in which a donor had a reactive test either prior to or post donation decreased by 48% (FY05 – 299). Most of these involved testing reactive post donation.
- The number of these reports increased by 3% (FY05 – 1,486).
- The number of reports involving the release of a product with unacceptable, undocumented, or incomplete product QC, specifically related to bacterial detection testing used as a quality control test, increased 16% (FY05 - 553). The industry implemented a standard for bacterial detection testing in March 2004.
- The number of these reports decreased by 7% (FY05 - 1,272).
- There was a 21% decrease in reports involving donor records which were incomplete, incorrect or not reviewed (FY05 – 337). There was a 32% decrease in reports related to the donor history questions (FY05 – 268). Of the 182 reports related to donor history questions incomplete, incorrect or not reviewed, there were 5 reports related to the use of an abbreviated donor history questionnaire when the screener should have used a full-length questionnaire, compared to 22 reports in FY05.
- The number of reports involving a donor who provided disqualifying information, specifically related to travel to a malarial endemic area, and was not deferred decreased by 20% (FY05 – 362).
- There was a 29% increase in reports in which the screener used incorrect donor identification to check the deferral file or did not check the deferral file, i.e., deferral screening not done (FY05 – 160).
- The number of these reports decreased by 6% (FY05 – 854).
- The number of reports involving the labeling of the unit or product decreased by 18% (FY05 – 443).
- The number of reports involving the labeling of the crossmatch tag or tie tag increased by 7% (FY05 – 375).
- The number of these reports decreased by 26% (FY05 - 931).
- The number of reports in which a clotted product was discovered after distribution decreased by 25% (FY05-647).
- Most Frequent BPD Reports Submitted by Unlicensed Blood Establishments
- The number of these reports increased by 7% (FY05 - 1,618). This increase was associated with an increase by 20% in the number of reports in which a product was not documented as issued in the computer (FY05 – 434). This type of event is reportable if the blood establishment uses the computer system as the only documentation of the final checks of the issue process.
- The number of reports involving the release of a product in which testing was not performed or document increased by 34% (FY05 – 181). Specifically, the number of reports involving not performing or documenting compatibility testing increased from 36 in FY05 to 50 in FY06.
- The number of these reports decreased by 6% (FY05 – 1,052).
- The number of reports involving labeling of the transfusion record decreased by 12% (FY05 – 309).
- The number of reports involving labeling of the crossmatch tag or tie tag decreased by 6% (FY05 - 467).
- The number of these reports decreased by 9% (FY05 - 530) .
- The number of reports of involving post donation illness decreased from 70 in FY05 to 47 in FY06.
- The number and distribution of these reports were similar to the reports received in the previous fiscal years (FY03 – 404, FY04 – 386, FY05 - 391).
- Most Frequent BPD Reports Submitted by Transfusion Services
- The number of these reports increased by 10% (FY05 – 727).
- The number of reports involving the release of a product in which required testing was not performed or documented increased from 94 in FY05 to 118 in FY06.
- The number of these reports decreased by 19% (FY05 - 492).
- The number of reports involving the labeling of the transfusion record decreased from 156 in FY05 to 117 in FY06.
- The number and distribution of these reports was similar to the reports received in the previous fiscal year (FY05 - 292).
- Most Frequent BPD Reports Submitted by Plasma Centers
- The number of these reports increased by 13% (FY05 - 4,424).
- The number of post donation information reports in which the donor had a history of a tattoo or piercing increased by 31% (FY05 – 2367).
- The number of these reports decreased by 7% (FY05 - 187).
- The number of reports in which the donor provided disqualifying information and was not deferred increased from 39 in FY05 to 56 in FY06.
- The number of reports in which the plasma center did not perform deferral screening decreased from 48 in FY05 to 25 in FY06.
- Timeliness of BPD Reports
- BPD Reports Submitted by Manufacturers of Biological Products Other Than Blood and Blood Components (Non-Blood)
- Allergenic manufacturers submitted 51 fewer reports (FY05 – 200).
- 116 of 125 (93%) of product specification reports were related to precipitate discovered in allergenic extracts.
- Derivative manufacturers submitted 12 fewer reports (FY05 – 47).
- In vitro diagnostic manufacturers submitted 40 fewer reports (FY05 – 100).
- Vaccine manufacturers submitted 4 more reports (FY05 – 37).
- There were 19 fewer reports identified as potential recall situations (FY05 – 33).
- Timeliness of BPD Reports
- HCT/P Deviation Reports Submitted by Manufacturers of 361 HCT/Ps
- Cellular HCT/Ps:
- Of the 27 reports involving donor testing, 22 reports involved Nucleic Acid Testing (NAT) for viral markers performed on pooled samples rather than the required individual sample.
- Of the 30 reports involving receipt, pre-distribution, shipment & distribution, 26 reports involved inappropriate distribution of product that was contaminated or potentially contaminated.
- Tissue HCT/Ps:
- Of the 30 reports involving donor eligibility submitted by tissue manufacturers, 27 reports involved the acceptance of ineligible donors.
- Timeliness of BPD Reports
- Attachments
- Table-Number of BPD Reports by Type of Blood Establishments
- List of BPD Codes for Blood and Plasma Establishments
- Table-Number of BPDs by Type of Licensed Non-Blood Manufacturer
- List of BPD Codes for Non-Blood Manufacturers
- Table-Number of HCT/P Deviations by Type of 361 HCT/P Manufacturer
- List of HCT/P Deviation Codes for 361 HCT/P Manufacturers
Licensed manufacturers of blood and blood components, including Source Plasma; unlicensed registered blood establishments; and transfusion services who had control over the product when the deviation occurred must submit Biological Product Deviation (BPD) reports to the Center for Biologics Evaluation and Research (CBER) (21 CFR 606.171). Manufacturers of licensed biological products other than blood and blood components (non-blood) who hold the biological product license for and had control over the product when the deviation occurred are also required to submit BPD reports (21 CFR 600.14). In addition, manufacturers of Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/P) regulated under section 361 of the Public Health Service Act are required to submit deviation reports (21 CFR Part 1271.350(b)). Detailed information concerning deviation reporting is available at www.fda.gov/cber/biodev/biodev.htm.
From October 1, 2005 through September 30, 2006 (Fiscal Year 2006 or FY06), CBER’s Office of Compliance and Biologics Quality/Division of Inspections and Surveillance entered 38,618 deviation reports into the BPD database:
Reports Submitted by Licensed Blood Establishments
FY04 |
FY05 |
FY06 |
|
|---|---|---|---|
| Donor Screening |
666 |
577 |
472 |
| Travel to malarial endemic area |
399 |
333 |
251 |
| Travel to vCJD risk area |
82 |
87 |
73 |
Reports submitted by Blood and Plasma Establishments
|
FY04 |
FY05 |
FY06 |
||||||
|---|---|---|---|---|---|---|---|---|---|
|
Blood |
Plasma |
Total |
Blood |
Plasma |
Total |
Blood |
Plasma |
Total |
Lookback; |
340 |
10 |
350 |
431 |
6 |
495 |
510 |
98 |
608 |
HIV |
70 |
2 |
72 |
84 |
2 |
92 |
87 |
12 |
99 |
HBV |
28 |
3 |
31 |
34 |
2 |
50 |
67 |
23 |
90 |
HCV |
239 |
5 |
244 |
293 |
2 |
333 |
309 |
63 |
372 |
Reports submitted by Plasma Centers
|
FY04 |
FY05 |
FY06 |
|---|---|---|---|
Post Donation Information |
4,304 |
3,998 |
4,826 |
Donor received tattoo |
1,593 |
1,579 |
2,045 |
Donor received ear piercing |
245 |
158 |
196 |
Donor received body piercing |
667 |
630 |
857 |
Reports submitted by Blood Establishments
|
FY04 |
FY05 |
FY06 |
|---|---|---|---|
Blood Collection |
851 |
972 |
718 |
Product contained clots |
546 |
674 |
506 |
Product hemolyzed |
80 |
51 |
16 |
FDA published two final guidance documents October 18, 2006 to assist industry in determining what events are reportable 1,2
You may submit questions concerning this summary to:
FDA/Center for Biologics Evaluation and Research
Office of Compliance and Biologics Quality
Division of Inspections and Surveillance (HFM-650)
1401 Rockville Pike, Suite 200 North
Rockville, Maryland 20852-1448
You may also contact us by email at bp_deviations@fda.hhs.gov, hctp_deviations@fda.hhs.gov, or sharon.ocallaghan@fda.hhs.gov (Sharon O’Callaghan) or by phone at 301-827-6220.
Footnotes
1Guidance for Industry - Biological Product Deviation Reporting for Blood and Plasma Establishments 10/18/2006
2 Guidance for Industry - Biological Product Deviation Reporting for Licensed Manufacturers of Biological Products Other than Blood and Blood Components 10/18/2006
Total Deviation Reports
FY06
Table 1
|
Number Of Reporting Establishments |
Total Reports Received |
Potential Recalls |
|
|---|---|---|---|---|
Blood/Plasma Manufacturers |
|
|
|
|
Licensed Blood Establishments |
231(119*) |
27,393 |
1,706 |
6.2% |
Unlicensed Blood Establishments 1 |
384 |
3,926 |
78 |
2.0% |
Transfusion Services 2 |
460 |
1,510 |
0 |
0% |
Plasma Centers |
287(54*) |
5,359 |
198 |
3.7% |
Sub-Total |
1,362 |
38,188 |
1,982 |
5.2% |
Non-Blood Manufacturers |
|
|
|
|
Allergenic |
7 |
149 |
5 |
3.4% |
Blood Derivative |
13 |
35 |
1 |
2.9% |
In Vitro Diagnostic |
9 |
60 |
7 |
11.7% |
Vaccine |
10 |
41 |
1 |
2.4% |
351 HCT/P |
1 |
1 |
1 |
100% |
Sub-Total |
40 |
286 |
15 |
5.2% |
361 HCT/P Manufacturers |
|
|
|
|
Cellular HCT/P |
41 |
74 |
5 |
6.8% |
Non-Cellular HCT/P |
38 |
70 |
44 |
62.9% |
Sub-Total |
79 |
144 |
49 |
34.3% |
Total |
1,481 |
38,618 |
2,046 |
5.3% |
1Unlicensed Blood Establishments – unlicensed blood establishments performing manufacturing of blood and blood components that require registration with FDA
2Transfusion Services – blood banks that perform limited blood and blood component manufacturing (e.g. pooling, thawing, compatibility testing), may or may not register with FDA.
*Number of license holders; one or more establishments operate under one biologics license.
Total Deviation Reports
FY04 - FY06
Table 2
Number Of Reporting Establishments |
Total Reports Received |
Potential Recalls |
|||||||
|---|---|---|---|---|---|---|---|---|---|
Blood/Plasma Manufacturers |
FY04 |
FY05 |
FY06 |
FY04 |
FY05 |
FY06 |
FY04 |
FY05 |
FY06 |
Licensed Blood Establishments |
237(117*) |
230(115*) |
231(119*) |
27,621 |
28,153 |
27,393 |
2,170 |
1,925 |
1,705 |
Unlicensed Blood Establishments |
371 |
392 |
384 |
3,502 |
3,897 |
3,926 |
58 |
44 |
78 |
Transfusion Services |
468 |
457 |
460 |
1,592 |
1,517 |
1,510 |
0 |
0 |
0 |
Plasma Centers |
357(55*) |
286(53*) |
287(54*) |
5,115 |
4,805 |
5,359 |
377 |
269 |
198 |
Sub-Total |
1,433 |
1,365 |
1,362 |
37,830 |
38,372 |
38,188 |
2,605 |
2,238 |
1,982 |
Non-Blood Manufacturers |
|
|
|
|
|
|
|
|
|
Allergenic |
9 |
8 |
7 |
158 |
200 |
149 |
5 |
13 |
5 |
Blood Derivative |
17 |
14 |
13 |
44 |
47 |
35 |
4 |
2 |
1 |
In Vitro Diagnostic |
10 |
11 |
9 |
86 |
100 |
60 |
8 |
18 |
7 |
Vaccine |
9 |
11 |
10 |
42 |
37 |
41 |
1 |
1 |
1 |
351 HCT/P |
1 |
1 |
1 |
2 |
1 |
1 |
0 |
0 |
1 |
Sub-Total |
47 |
45 |
40 |
334 |
385 |
286 |
18 |
33 |
15 |
361 HCT/P Manufacturers |
|
|
|
|
|
|
|
|
|
Cellular HCT/P |
NA |
5 † |
41 |
NA |
7 † |
74 |
NA |
2 † |
5 |
Non-Cellular HCT/P |
NA |
4 † |
38 |
NA |
6 † |
70 |
NA |
5 † |
44 |
Sub-Total |
NA |
9 † |
79 |
NA |
13 † |
144 |
NA |
7 † |
49 |
Total |
1,480 |
1,419 |
1,481 |
38,164 |
38,757 |
38,618 |
2,623 |
2,271 |
2,046 |
*Number of license holders; one or more establishments operate under one biologics license.
†Reports of events involving products manufactured on or after 5/25/05 {implementation of 21 CFR 1271.350(b)}
Blood & Plasma BPD Reports By Manufacturing System
FY04 - FY06
Table 3
MANUFACTURING SYSTEM |
FY04 | FY05 | FY06 | |||
|---|---|---|---|---|---|---|
DONOR SUITABILITY |
28,952 |
76.5% |
29,148 |
76.0% |
29,067 |
76.1% |
POST DONATION INFORMATION |
26,854 |
71.0% |
27,452 |
71.5% |
27,427 |
71.8% |
DONOR SCREENING |
2,007 |
5.3% |
1,628 |
4.2% |
1,548 |
4.1% |
DONOR DEFERRAL |
91 |
0.2% |
68 |
0.2% |
92 |
0.2% |
QC & DISTRIBUTION |
3,740 |
9.9% |
3,934 |
10.3% |
4,134 |
10.8% |
LABELING |
2,415 |
6.4% |
2,405 |
6.3% |
2,199 |
5.8% |
LABORATORY TESTING |
1,122 |
3.0% |
981 |
2.6% |
1,013 |
2.7% |
ROUTINE TESTING |
1,027 |
2.7% |
912 |
2.4% |
945 |
2.5% |
VIRAL TESTING |
95 |
0.3% |
69 |
0.2% |
66 |
0.2% |
COLLECTION |
851 |
2.2% |
972 |
2.5% |
718 |
1.9% |
COMPONENT PREPARATION |
368 |
1.0% |
407 |
1.1% |
401 |
1.0% |
MISCELLANEOUS |
382 |
1.0% |
525 |
1.4% |
658 |
1.7% |
TOTAL |
37,830 |
100% |
38,372 |
100% |
38,188 |
100% |
Non-Blood Deviation Reports By Manufacturing System
FY04 - FY06
Licensed Biological Products Other Than Blood and Blood Components
Table 4
Manufacturing System |
Allergenic | Derivative | In Vitro Diagnostic | ||||||
|---|---|---|---|---|---|---|---|---|---|
|
FY04 |
FY05 |
FY06 |
FY04 |
FY05 |
FY06 |
FY04 |
FY05 |
FY06 |
Incoming Material |
2 |
0 |
0 |
9 |
9 |
2 |
5 |
4 |
2 |
Process Controls |
2 |
1 |
9 |
9 |
5 |
5 |
20 |
18 |
10 |
Testing |
0 |
0 |
6 |
0 |
3 |
2 |
14 |
17 |
5 |
Labeling |
8 |
22 |
7 |
3 |
15 |
2 |
16 |
24 |
16 |
Product Specifications |
146 |
177 |
125 |
15 |
11 |
20 |
23 |
30 |
18 |
Quality Control & Distribution |
0 |
0 |
2 |
7 |
4 |
3 |
6 |
7 |
9 |
Miscellaneous |
0 |
0 |
0 |
1 |
0 |
1 |
2 |
0 |
0 |
TOTAL |
158 |
200 |
149 |
44 |
47 |
35 |
86 |
100 |
60 |
Table 4 (con’t)
| Manufacturing System | Vaccine | 351 HCT/P | Total | ||||||
|---|---|---|---|---|---|---|---|---|---|
|
FY04 |
FY05 |
FY06 |
FY04 |
FY05 |
FY06 |
FY04 |
FY05 |
FY06 |
Incoming Material |
1 |
1 |
1 |
0 |
1 |
0 |
17 |
15 |
5 |
Process Controls |
7 |
4 |
2 |
1 |
0 |
0 |
38 |
28 |
26 |
Testing |
4 |
1 |
5 |
0 |
0 |
0 |
18 |
21 |
18 |
Labeling |
10 |
12 |
8 |
0 |
0 |
0 |
37 |
73 |
33 |
Product Specifications |
17 |
17 |
17 |
0 |
0 |
1 |
201 |
235 |
181 |
Quality Control & Distribution |
1 |
2 |
8 |
1 |
0 |
0 |
14 |
13 |
22 |
Miscellaneous |
2 |
0 |
0 |
0 |
0 |
0 |
5 |
0 |
1 |
TOTAL |
42 |
37 |
41 |
2 |
1 |
1 |
332 |
385 |
286 |
361 HCT/Ps
Table 5
Manufacturing System |
Cellular HCT/Ps |
Non-Cellular HCT/Ps |
Total |
|||
|---|---|---|---|---|---|---|
| FY05 † | FY06 | FY05 † | FY06 | FY05 † | FY06 | |
Donor Eligibility |
3 |
2 |
5 |
30 |
8 |
32 |
Donor Screening |
0 |
0 |
0 |
12 |
0 |
12 |
Donor Testing |
0 |
27 |
0 |
8 |
0 |
35 |
Environmental Control |
0 |
0 |
0 |
1 |
0 |
1 |
Supplies and Reagents |
0 |
2 |
0 |
1 |
0 |
3 |
Recovery |
0 |
2 |
0 |
0 |
0 |
2 |
Processing and Processing Controls |
0 |
11 |
0 |
3 |
0 |
14 |
Labeling Controls |
0 |
0 |
1 |
1 |
1 |
1 |
Storage |
0 |
0 |
0 |
1 |
0 |
1 |
Receipt, Pre-Distrib., Shipment & Distrib. |
4 |
30 |
0 |
13 |
4 |
43 |
TOTAL |
7 |
74 |
6 |
70 |
13 |
144 |
†Reports of events involving products manufactured on or after 5/25/05 (implementation of 21 CFR 1271.350(b))
We implemented the on-line electronic deviation report form on June 18, 2001. The percentage of reports submitted electronically in FY06 increased by 9.8 percentage points from FY05 (from 61.9% to 71.7%). We continue to encourage all reporters to use the electronic reporting format.
Deviation Reports Submitted Electronically
Table 6
| Total Reports | # of eBPDR | % eBPDR | |
|---|---|---|---|
BLOOD/PLASMA MANUFACTURERS |
|
|
|
LICENSED BLOOD ESTABLISHMENTS |
27,393 |
19,402 |
70.8% |
UNLICENSED BLOOD ESTABLISHMENTS |
3,926 |
3,541 |
90.2% |
TRANSFUSION SERVICES |
1,510 |
1,188 |
78.7% |
PLASMA CENTERS |
5,359 |
3,275 |
61.1% |
SUB-TOTAL |
38,188 |
27,406 |
71.8% |
NON-BLOOD MANUFACTURERS |
|
|
|
ALLERGENIC |
149 |
132 |
88.6% |
DERIVATIVE |
35 |
18 |
51.4% |
IN VITRO DIAGNOSTIC |
60 |
41 |
68.3% |
VACCINE |
41 |
4 |
9.8% |
351 HCT/P |
1 |
0 |
0% |
SUB-TOTAL |
286 |
195 |
68.2% |
361 HCT/P Manufacturers |
|
|
|
Cellular HCT/P |
74 |
55 |
74.3% |
Non-Cellular HCT/P |
70 |
45 |
64.3% |
SUB-TOTAL |
144 |
100 |
69.4% |
TOTAL |
38,618 |
27,701 |
71.7% |
Percent of Electronic Deviation Reports
Table 7
|
FY03 |
FY04 |
FY05 |
FY06 |
|---|---|---|---|---|
BLOOD/PLASMA MANUFACTURERS |
|
|
|
|
LICENSED BLOOD ESTABLISHMENTS |
41.8% |
52.1% |
58.6% |
70.8% |
UNLICENSED BLOOD ESTABLISHMENTS |
84.2% |
88.9% |
90.8% |
90.2% |
TRANSFUSION SERVICES |
75.5% |
81.9% |
79.9% |
78.8% |
PLASMA CENTERS |
28.3% |
49.6% |
51.7% |
61.1% |
SUB-TOTAL |
43.7% |
56.4% |
61.8% |
71.8% |
NON-BLOOD MANUFACTURERS |
|
|
|
|
ALLERGENIC |
60.0% |
81.6% |
93.5% |
88.6% |
DERIVATIVE |
19.3% |
50.0% |
29.8% |
51.4% |
IN VITRO DIAGNOSTIC |
72.7% |
65.1% |
53.0% |
68.3% |
VACCINE |
8.9% |
14.3% |
5.4% |
9.8% |
351 HCT/P |
|
|
|
0% |
SUB-TOTAL |
45.9% |
64.2% |
66.5% |
68.2% |
361 HCT/P Manufacturers |
|
|
|
|
Cellular HCT/P |
NA |
NA |
100% † |
74.3% |
Non-Cellular HCT/P |
NA |
NA |
33.3%† |
64.3% |
SUB-TOTAL |
NA |
NA |
69.2%† |
69.4% |
TOTAL |
43.7% |
56.5% |
61.9% |
71.7% |
†Reports of events involving products manufactured on or after 5/25/05 {implementation of 21 CFR 1271.350(b)}
Total BPDRs By Manufacturing System
Table 8
Manufacturing System |
Licensed Establishments |
Unlicensed Establishments |
Transfusion Services |
Plasma Centers |
Total |
|
|---|---|---|---|---|---|---|
DS-Post Donation Information |
21,955 |
483 |
NA |
4,989 |
27,427 |
71.8% |
QC & Distribution |
1,535 |
1,739 |
800 |
60 |
4,134 |
10.8% |
Labeling |
803 |
987 |
400 |
9 |
2,199 |
5.8% |
DS-Donor Screening |
1,189 |
186 |
NA |
173 |
1,548 |
4.1% |
LT-Routine Testing |
257 |
386 |
302 |
0 |
945 |
2.6% |
Blood Collection |
693 |
22 |
NA |
3 |
718 |
1.9% |
Miscellaneous |
556 |
3 |
0 |
99 |
658 |
1.7% |
Component Preparation |
299 |
94 |
8 |
0 |
401 |
1.1% |
DS-Donor Deferral |
47 |
19 |
NA |
26 |
92 |
0.2% |
LT-Viral Testing |
59 |
7 |
NA |
0 |
66 |
0.2% |
TOTAL |
27,393 |
3,926 |
1,510 |
5,359 |
38,188 |
100% |
DS-Donor Suitability
LT-Laboratory Testing
NA-Not applicable: manufacturing not performed in transfusion service
Potential Recalls By Manufacturing System
Table 9
Manufacturing System |
Licensed Establishments |
Unlicensed Establishments |
Transfusion Services |
Plasma Centers |
Total |
|
|---|---|---|---|---|---|---|
DS-Donor Screening |
644 |
45 |
NA |
128 |
817 |
41.2% |
QC & Distribution |
576 |
14 |
0 |
36 |
626 |
31.6% |
Component Preparation |
147 |
2 |
0 |
0 |
149 |
7.5% |
Blood Collection |
142 |
1 |
NA |
0 |
143 |
7.2% |
Labeling |
56 |
9 |
0 |
1 |
66 |
3.3% |
DS-Donor Deferral |
33 |
5 |
NA |
15 |
53 |
2.7% |
DS-Post Donation Information |
28 |
0 |
NA |
18 |
46 |
2.3% |
LT-Routine Testing |
43 |
2 |
0 |
0 |
45 |
2.3% |
LT-Viral Testing |
36 |
0 |
NA |
0 |
36 |
1.8% |
Miscellaneous |
1 |
0 |
0 |
0 |
1 |
0.1% |
TOTAL |
1,706 |
78 |
0 |
198 |
1,982 |
100% |
DS-Donor Suitability
LT-Laboratory Testing
NA-Not applicable: manufacturing not performed in transfusion service
Post donation information (PDI) continues to be the most frequently reported event associated with the manufacturing of blood and plasma products. The most common PDI involved donors providing information concerning travel to malarial endemic areas and travel to an area at potential risk for vCJD. It is unclear why blood establishments are unable to elicit information during the first donor interview, but successfully elicit the information during a subsequent interview. Eliciting proper information regarding a donor candidate’s travel history is apparently the most problematic part of the donor qualification process.
FY06 Reports of Post Donation Information (PDI)
Table 10
PDI OBTAINED THROUGH: |
LICENSED ESTABLISHMENTS |
UNLICENSED ESTABLISHMENTS |
PLASMA CENTERS |
TOTAL |
|
|---|---|---|---|---|---|
Subsequent Donation |
20,002 |
436 |
4,150 |
24,588 |
89.6% |
Telephone Call from Donor |
1,322 |
34 |
19 |
1,375 |
5.0% |
Third Party (e.g., doctor, family) |
409 |
11 |
820 |
1,240 |
4.5% |
Telerecruitment |
222 |
2 |
0 |
224 |
0.8% |
TOTAL |
21,955 |
483 |
4,989 |
27,427 |
100% |
Table 11
THE PDI WAS: |
LICENSED ESTABLISHMENTS |
UNLICENSED ESTABLISHMENTS |
PLASMA CENTERS |
TOTAL |
|
|---|---|---|---|---|---|
Known, but not Provided at Time of Donation* |
19,950 |
422 |
4,482 |
24,854 |
90.6% |
Not Known at Time of Donation** |
2,005 |
61 |
507 |
2,573 |
9.4% |
TOTAL |
21,955 |
483 |
4,989 |
27,427 |
100% |
* Known, e.g., travel outside of U.S., tattoo or body piercing, history of cancer, male to male sexual contact, medication
**Not known, e.g., post donation illness, cancer diagnosed post donation, sex partner participated in high risk behavior or tested positive
In an effort to provide practical and useful information regarding reporting deviations, this section addresses non-reportable events, deviation code selection and product information entry.
Non-Reportable Events
Blood establishments submitted most of the non-reportable reports. The reports did not meet the reporting threshold because the events were either not associated with manufacturing, did not affect the safety, purity or potency of the product, or did not involve distributed products. Examples of non-reportable events include:
Deviation Code (BPD Code) Selection
In some cases the establishment selected the incorrect deviation code to capture the event. The most common errors in coding were:
Product Information
When identifying the number of units and components, the number of units should equal the number of donations. For example if the event involved 2 donations and each donation was manufactured into red blood cells, platelets and plasma, the number of units would be 2 and the number of components would be 6. Some reports listed the number of units as 6 and the number of components as 6.
Of the 27,393 reports submitted by licensed blood establishments, 21,955 (80.1%) reports involved post donation information.
Most Frequent BPD Reports - Post Donation Information
From Licensed Blood Establishments
Table 12
POST DONATION INFORMATION (PD) 21,955 |
# Reports |
% of Total (PD) |
|---|---|---|
Behavior/History |
19,994 |
91.07% |
Travel to malaria endemic area/history of malaria |
7,018 |
31.97% |
Risk factors associated with Creutzfeldt-Jakob Disease (CJD) – travel |
4,752 |
21.64% |
History of cancer |
1,258 |
5.73% |
Donor received tattoo within 12 months of donation |
870 |
3.96% |
Male donor had sex with another man |
732 |
3.33% |
Received Proscar, Tegison or Accutane |
557 |
2.54% |
History of disease |
462 |
2.10% |
Donor received bone graft or transplant |
436 |
1.99% |
Illness |
1,705 |
7.77% |
Post donation illness (not hepatitis, HIV, HTLV-I, STD, or cold/flu related) |
1,190 |
5.42% |
Reaction at phlebotomy site |
57 |
0.26% |
Babesiosis |
28 |
0.13% |
Post donation diagnosis of cancer |
423 |
1.93% |
Testing * |
154 |
0.70% |
Tested reactive for HIV prior to donation |
31 |
0.14% |
Tested reactive for HIV post donation |
24 |
0.11% |
Tested reactive for Hepatitis C post donation |
22 |
0.10% |
Not specifically related to high risk behavior |
102 |
0.01% |
Donated to be tested or called back for test results |
73 |
0.33% |
Donor does not want their blood used |
29 |
0.13% |
*Includes: tested positive for viral marker either prior to or post donation
Of the 27,393 reports submitted by licensed blood establishments, 1,535 (5.6%) reports involved quality control and distribution deviations and unexpected events.
Most Frequent BPD Reports - Quality Control & Distribution
From Licensed Blood Establishments
Table 13
QC & DISTRIBUTION (QC) 1,535 |
# Reports |
% of Total (QC) |
|---|---|---|
Distribution of product that did not meet specifications |
1,074 |
69.97% |
Product with unacceptable, undocumented, or incomplete product QC |
644 |
41.95% |
Bacterial Detection Testing |
412 |
26.84% |
Platelet count |
101 |
6.58% |
White Blood Cell count |
43 |
2.80% |
Product in which instrument QC or validation was unacceptable or not documented |
124 |
8.08% |
Product released prior to resolution of discrepancy |
78 |
5.08% |
Product identified as unsuitable due to a donor screening deviation or unexpected event |
54 |
3.52% |
Shipping and storage |
214 |
13.94% |
Product not packaged in accordance with specifications |
51 |
3.32% |
No documentation that product was shipped at appropriate temperature |
44 |
2.87% |
Shipped at incorrect temperature |
42 |
2.74% |
Distribution procedures not performed in accordance with blood bank transfusion service’s specifications |
132 |
8.60% |
Product not documented as issued in computer |
25 |
1.63% |
Product not irradiated as required |
25 |
1.63% |
Product not leukoreduced as required |
14 |
0.91% |
Visual inspection not performed or documented |
11 |
0.72% |
Required testing incomplete, or positive |
36 |
2.35% |
Failure to quarantine unit due to medical history: |
44 |
2.87% |
Post donation illness |
25 |
1.63% |
Required testing not performed or documented |
35 |
2.28% |
Of the 27,393 reports submitted by licensed blood establishments, 1,189 (4.3%) reports involved donor screening deviations and unexpected events.
Most Frequent BPD Reports - Donor Screening
From Licensed Blood Establishments
Table 14
DONOR SCREENING (DS) 1,189 |
# Reports |
% of Total (DS) |
|---|---|---|
Donor gave history which warranted deferral and was not deferred |
607 |
51.05% |
Travel to malaria endemic area/history of malaria |
288 |
24.22% |
Risk factors associated with Creutzfeldt-Jakob Disease (vCJD) - travel |
93 |
7.82% |
Received medication or antibiotics |
43 |
3.62% |
History of cancer |
33 |
2.78% |
History of disease |
19 |
1.60% |
Donor record incomplete or incorrect |
265 |
22.29% |
Donor history questions |
182 |
15.31% |
Donor identification |
23 |
1.93% |
Donor signature missing |
23 |
1.93% |
Incorrect ID used during deferral search |
189 |
15.90% |
Donor not previously deferred |
173 |
14.55% |
Donor previously deferred due to testing |
9 |
0.76% |
Donor previously deferred due to history |
7 |
0.59% |
Donor did not meet acceptance criteria |
107 |
9.00% |
Hemoglobin or Hematocrit unacceptable or not documented |
73 |
6.14% |
Temperature unacceptable or not documented |
25 |
2.10% |
Deferral screening not done |
17 |
1.43% |
Donor previously deferred due to testing |
6 |
0.50% |
Donor not previously deferred |
6 |
0.50% |
Donor previously deferred due to history |
5 |
0.42% |
Of the 27,393 reports submitted by licensed blood establishments, 803 (2.9%) reports involved labeling deviations and unexpected events.
Most Frequent BPD Reports - Labeling
From Licensed Blood Establishments
Table 15
LABELING (LA) 803 |
#Reports |
% of Total (LA) |
|---|---|---|
Crossmatch tag or tie tag labels incorrect or missing information |
401 |
49.94% |
Recipient identification missing or incorrect |
252 |
31.38% |
Autologous unit |
113 |
14.07% |
Crossmatch tag switched, both units intended for the same patient |
27 |
3.36% |
Unit, lot, or pool number incorrect or missing |
23 |
2.86% |
Antigen incorrect or missing |
22 |
2.74% |
Blood unit labels |
365 |
45.45% |
Volume incorrect or missing |
65 |
8.09% |
Extended expiration date or time |
53 |
6.60% |
Donor number or lot number incorrect or missing |
46 |
5.73% |
ABO and/or Rh incorrect |
42 |
5.23% |
Transfusion record (crossmatch slip) incorrect or missing information |
31 |
3.86% |
Recipient identification missing or incorrect |
12 |
1.49% |
Of the 27,393 reports submitted by licensed blood establishments, 693 (2.5%) reports involved blood collection deviations and unexpected events.
Most Frequent BPD Reports – Blood Collection
From Licensed Blood Establishments
Table 16
BLOOD COLLECTION (BC) 693 |
# Reports |
% of Total (BC) |
|---|---|---|
Collection Process |
551 |
79.51% |
Product contained clots, not discovered prior to distribution |
490 |
70.71% |
Donor sample tube mix-up or donor sample tube mislabeled |
20 |
2.89% |
Product hemolyzed, not discovered prior to distribution |
16 |
2.31% |
Sterility compromised |
85 |
12.26% |
Bacterial contamination |
35 |
5.05% |
Arm prep not performed or performed inappropriately |
34 |
4.91% |
Air contamination |
13 |
1.88% |
Collection Bag |
29 |
4.18% |
Apheresis collection device |
18 |
2.60% |
Of the 3,926 reports submitted by unlicensed blood establishments, 1,739 (44.3%) involved quality control and distribution deviations and unexpected events.
Most Frequent BPD Reports - Quality Control & Distribution
From Unlicensed Blood Establishments
Table 17
QC & Distribution (QC) 1,739 |
# Reports |
% of Total QC |
|---|---|---|
Distribution procedures not performed in accordance with blood bank transfusion service’s specifications |
1,229 |
70.67% |
Product not documented as issued in the computer |
519 |
29.84% |
Product not irradiated as required |
189 |
10.87% |
Improper ABO or Rh type selected for patient |
93 |
5.35% |
Procedure for issuing not performed or documented in accordance with specifications |
68 |
3.91% |
Improper product selected for patient |
67 |
3.85% |
Visual inspection not performed or documented |
50 |
2.88% |
Product not leukoreduced as required |
44 |
2.53% |
Required testing not performed or documented for: |
244 |
14.03% |
Antibody screen or identification |
56 |
3.22% |
Antigen screen |
51 |
2.93% |
Compatibility |
50 |
2.88% |
Distribution of product that did not meet specifications: |
175 |
10.06% |
Product with unacceptable, undocumented, or incomplete product QC |
89 |
5.12% |
Bacterial Detection Testing |
61 |
3.51% |
Outdated product |
31 |
1.78% |
Product in which instrument QC or validation unacceptable or not documented |
23 |
1.32% |
Required testing incomplete or positive: |
64 |
3.68% |
Antibody screen or identification |
20 |
1.15% |
Compatibility |
15 |
0.86% |
Shipping and storage |
21 |
1.21% |
Stored at incorrect temperature |
11 |
0.63% |
Product not packaged in accordance with specifications |
6 |
0.35% |
Of the 3,926 reports submitted by unlicensed blood establishments, 987 (25%) involved labeling deviations and unexpected events.
Most Frequent BPD Reports - Labeling
From Unlicensed Blood Establishments
Table 18
LABELING (LA) 987 |
#Reports |
% of Total LA |
|---|---|---|
Crossmatch tag or tie tag labels incorrect or missing information |
435 |
44.07% |
Recipient identification missing or incorrect |
170 |
17.22% |
Autologous unit |
3 |
0.30% |
Crossmatch tag switched, both units intended for the same patient |
88 |
8.92% |
Unit, lot, or pool number incorrect or missing |
57 |
5.78% |
Blood unit labels |
282 |
28.57% |
Extended expiration date or time |
137 |
13.88% |
Donor number or lot number incorrect or missing |
35 |
3.55% |
ABO and/or Rh incorrect |
31 |
3.14% |
Product type or code incorrect |
26 |
2.63% |
Transfusion record (crossmatch slip) incorrect or missing information |
270 |
27.36% |
Recipient identification missing or incorrect |
61 |
6.18% |
Transfusion record switched, both units intended for the same patient |
59 |
5.98% |
Unit, lot, or pool number incorrect or missing |
36 |
3.65% |
Of the 3,926 reports submitted by unlicensed blood establishments, 483 (12.3%) reports involved post donation information.
Most Frequent BPD Reports - Post Donation Information
From Unlicensed Blood Establishments
Table 19
POST DONATION INFORMATION (PD) 483 |
# Reports |
% of Total (PD) |
|---|---|---|
Behavior/History |
429 |
88.82% |
Travel to malaria endemic area/history of malaria |
164 |
33.95% |
Risk factors associated with Creutzfeldt-Jakob Disease (CJD) – travel |
109 |
22.57% |
History of cancer |
16 |
3.31% |
Donor received tattoo |
15 |
3.11% |
History of disease |
15 |
3.11% |
Illness |
47 |
9.73% |
Post donation illness (not hepatitis, HIV, HTLV-I, STD, or cold/flu related) |
26 |
5.38% |
Post donation diagnosis of cancer |
18 |
3.73% |
Testing* |
6 |
1.24% |
*Includes: tested positive for viral marker either prior to or post donation
Of the 3,926 reports submitted by unlicensed blood establishments, 386 (9.8%) reports involved routine testing deviations and unexpected events.
Most Frequent BPD Reports - Routine Testing
From Unlicensed Blood Establishments
Table 20
ROUTINE TESTING (RT) 386 |
# Reports |
% of Total RT |
|---|---|---|
Incorrectly tested for: |
221 |
57.25% |
Antibody screening or identification |
75 |
19.43% |
Compatibility |
63 |
16.32% |
Antigen typing |
32 |
8.29% |
Sample (used for testing) identification |
118 |
30.57% |
Sample used for testing was incorrectly or incompletely labeled |
93 |
24.09% |
Unsuitable sample used for testing (e.g., too old) |
14 |
3.63% |
Incorrect sample tested |
9 |
2.33% |
Reagent QC unacceptable or expired reagents used |
46 |
11.92% |
Antigen typing |
14 |
3.63% |
Antibody screening or identification |
10 |
2.59% |
Multiple testing |
7 |
1.81% |
Of the 1,510 reports submitted by transfusion services, 800 (53.0%) reports involved quality control and distribution deviations and unexpected events.
Most Frequent BPD Reports - Quality Control & Distribution
From Transfusion Services
Table 21
QC & Distribution (QC) 800 |
# Reports |
% of Total QC |
|---|---|---|
Distribution procedures not performed in accordance with blood bank transfusion service’s specifications |
578 |
72.25% |
Product not documented as issued in the computer |
258 |
32.25% |
Product not irradiated as required |
67 |
8.38% |
Procedure for issuing not performed or documented in accordance with specifications |
39 |
4.88% |
Improper ABO or Rh type selected for patient |
39 |
4.88% |
Product not leukoreduced as required |
32 |
4.00% |
Unit issued from the blood bank to the wrong patient |
31 |
3.88% |
Product released prior to obtaining current sample for ABO, Rh, antibody screen or compatibility testing |
30 |
3.73% |
Required testing not performed or documented for: |
118 |
14.75% |
Antigen screen |
34 |
4.25% |
ABO and Rh |
30 |
3.75% |
Antibody screen or identification |
17 |
2.13% |
Distribution of product that did not meet specifications: |
46 |
5.75% |
Outdated product |
19 |
2.38% |
Product with unacceptable, undocumented or incomplete product QC - pH for bacterial detection testing |
18 |
2.25% |
Required testing incomplete or positive: |
30 |
3.75% |
ABO and Rh |
9 |
1.13% |
Compatibility |
8 |
1.00% |
Shipping and storage |
27 |
3.38% |
Stored at incorrect temperature |
11 |
1.38% |
Temperature not recorded or unacceptable upon receipt, unit redistributed |
6 |
0.75% |
Of the 1,510 reports submitted by transfusion services, 400 (26.4%) reports involved labeling deviations and unexpected events.
Most Frequent BPD Reports - Labeling
From Transfusion Services
Table 22
LABELING (LA) 400 |
# Reports |
% of Total (LA) |
|---|---|---|
Crossmatch tag or tie tag labels incorrect or missing information |
232 |
58.00% |
Recipient identification incorrect or missing |
94 |
23.50% |
Unit or pool number incorrect or missing |
38 |
9.50% |
Crossmatch tag switched, both units intended for the same patient |
32 |
8.00% |
Crossmatch tag incorrect or missing |
18 |
4.50% |
Unit ABO and/or Rh incorrect or missing |
12 |
3.00% |
Expiration date or time extended or missing |
8 |
2.00% |
Transfusion record (crossmatch slip) incorrect or missing information |
117 |
29.25% |
Recipient identification incorrect or missing |
39 |
9.75% |
Transfusion record switched, both units intended for the same patient |
19 |
4.75% |
Transfusion record released w/unit incorrect or labeled with incorrect or missing information |
14 |
3.50% |
Unit or pool number incorrect or missing |
11 |
2.75% |
Blood unit labels |
51 |
12.69% |
Expiration date or time extended or missing |
19 |
4.75% |
Product type or code incorrect |
7 |
1.75% |
Donor number or lot number incorrect or missing |
6 |
1.50% |
Of the 1,510 reports submitted by transfusion services, 302 (20%) reports involved routine testing deviations and unexpected events.
Most Frequent BPD Reports - Routine Testing
From Transfusion Services
Table 23
ROUTINE TESTING (RT) 302 |
# Reports |
% of Total RT |
|---|---|---|
Incorrectly tested for: |
177 |
58.61% |
Antibody screening or identification |
57 |
18.87% |
Compatibility |
46 |
15.23% |
Rh typing |
23 |
7.62% |
Antigen typing |
20 |
6.62% |
Sample (used for testing) identification |
83 |
27.48% |
Sample used for testing was incorrectly or incompletely labeled |
62 |
20.53% |
Unsuitable sample used for testing |
11 |
3.64% |
Incorrect sample tested |
10 |
3.31% |
Reagent QC unacceptable or expired reagents used |
39 |
12.91% |
Antibody screening or identification |
10 |
3.31% |
Multiple testing |
8 |
2.65% |
Rh typing |
5 |
1.66% |
Antigen typing |
5 |
1.66% |
Of the 5,359 reports submitted by Source Plasma centers, 4,989 (93%) involved post donation information.
Most Frequent BPD Reports - Post Donation Information
From Plasma Centers
Table 24
POST DONATION INFORMATION (PD) 4,989 |
# Reports |
% of Total (PD) |
|---|---|---|
Behavior/History |
4,826 |
96.73% |
Donor received tattoo within 12 months of donation |
2,045 |
40.99% |
Donor received body piercing within 12 months of donation |
857 |
17.18% |
Incarcerated |
392 |
7.86% |
Risk factors associated with Creutzfeldt-Jakob Disease (CJD) – travel |
237 |
4.75% |
Donor received ear piercing within 12 months of donation |
196 |
3.93% |
Non-sexual exposure to Hepatitis C |
182 |
3.65% |
Donor received tattoo and piercing within 12 months of donation |
134 |
2.69% |
IV drug use |
116 |
2.33% |
Sex partner tested reactive for HCV |
114 |
2.29% |
Testing * |
116 |
2.33% |
Tested reactive at another center, specific testing unknown |
71 |
1.42% |
Tested reactive for HCV post donation |
18 |
0.36% |
Tested reactive for HIV post donation |
11 |
0.22% |
Illness |
47 |
0.94% |
*Includes testing positive for viral marker prior to or post donation
Of the 5,359 reports submitted by Source Plasma centers, 173 (3.2%) reports involved donor screening deviations and unexpected events.
Most Frequent BPD Reports - Donor Screening
From Plasma Centers
Table 25
DONOR SCREENING (DS) 173 |
# Reports |
% of Total (DS) |
|---|---|---|
Donor gave history which warranted deferral and was not deferred |
56 |
32.37% |
Donor received tattoo within 12 months of donation |
15 |
8.67% |
Risk factors associated with Creutzfeldt-Jakob Disease (vCJD) - travel |
7 |
4.05% |
Donor received vaccine or immune globulin |
5 |
2.89% |
History of disease or surgery |
5 |
2.89% |
Donor record incomplete or incorrect |
54 |
31.21% |
Donor history questions |
29 |
16.76% |
Arm inspection |
11 |
6.36% |
Donor signature missing |
6 |
3.47% |
Donor did not meet acceptance criteria |
32 |
18.50% |
Medical review or physical not performed or inadequate |
18 |
10.40% |
Temperature unacceptable or not documented |
8 |
4.62% |
Deferral screening not done |
25 |
14.45% |
Donor previously deferred due to history |
18 |
10.40% |
Deferred by another center |
5 |
2.89% |
IV drug user |
4 |
2.31% |
Donor previously deferred due to testing |
7 |
4.05% |
Incorrect ID used during deferral search |
6 |
3.47% |
Donor previously deferred due to history |
3 |
1.73% |
Donor previously deferred due to testing |
2 |
1.16% |
Donor not previously deferred |
1 |
0.58% |
BLOOD AND PLASMA ESTABLISHMENTS
Adherence To 45 Day Required Timeframe For Reporting
(Reporting Time = Date of FDA receipt – Date of discovery of BPD)
Table 26
Reporting Time (days) |
Licensed Establishments |
Unlicensed Establishments |
Transfusion Services |
Plasma Centers |
Total |
|||||
|---|---|---|---|---|---|---|---|---|---|---|
< or = 45 |
24,993 |
91% |
3,179 |
81% |
1,304 |
86% |
5,005 |
93% |
34,481 |
90% |
> 45 and <=90 |
1,902 |
7% |
424 |
11% |
151 |
10% |
275 |
5% |
2,752 |
7% |
> 90 |
498 |
2% |
323 |
8% |
54 |
4% |
79 |
1% |
954 |
2% |
Total |
27,393 |
100% |
3,926 |
100% |
1,509 |
100% |
5,359 |
100% |
38,187 |
100% |
*Reporting time=0 |
31 |
|
98 |
|
51 |
|
0 |
|
180 |
|
*Reporting time = 0 - reports were submitted electronically on the day discovered.

Non-blood manufacturers submitted 26% fewer reports in FY06 than in the previous year (FY05 – 384) {Table 2}.
Total BPD Reports By Manufacturing System
Table 27
MANUFACTURING SYSTEM |
Allergenic |
Derivative |
In Vitro Diagnostic |
Vaccine |
351 HCT/P |
TOTAL |
|
|---|---|---|---|---|---|---|---|
Incoming Material |
0 |
2 |
2 |
1 |
0 |
5 |
1.8% |
Process Controls |
9 |
5 |
10 |
2 |
0 |
26 |
9.1% |
Testing |
6 |
2 |
5 |
5 |
0 |
18 |
6.3% |
Labeling |
7 |
2 |
16 |
8 |
0 |
33 |
11.5% |
Product Specifications |
125 |
20 |
18 |
17 |
1 |
181 |
63.3% |
Quality Control & Distribution |
2 |
3 |
9 |
8 |
0 |
22 |
7.7% |
Miscellaneous |
0 |
1 |
0 |
0 |
0 |
1 |
0.3% |
Total |
149 |
35 |
60 |
41 |
1 |
286 |
100% |
Potential Recalls By Manufacturing System
Table 28
MANUFACTURING SYSTEM |
Allergenic |
Derivative |
In Vitro Diagnostic |
Vaccine |
TOTAL |
|
|---|---|---|---|---|---|---|
Incoming Material |
0 |
0 |
0 |
0 |
0 |
0% |
Process Controls |
1 |
1 |
0 |
0 |
2 |
14.3% |
Testing |
2 |
0 |
0 |
0 |
2 |
14.3% |
Labeling |
2 |
0 |
1 |
1 |
4 |
28.6% |
Product Specifications |
0 |
0 |
6 |
0 |
6 |
42.9% |
Quality Control & Distribution |
0 |
0 |
0 |
0 |
0 |
0% |
Miscellaneous |
0 |
0 |
0 |
0 |
0 |
0% |
Total |
5 |
1 |
7 |
1 |
14 |
100% |
NON-BLOOD MANUFACTURES
Adherence To 45 Day Required Time For Reporting
(Reporting Time = Date of FDA receipt – Date of discovery of BPD)
Table 29
Reporting Time (days) |
Allergenics |
Derivatives |
In Vitro Diagnostics |
Vaccines |
351 HCT/P |
Total |
||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
< or = 45 |
139 |
93% |
23 |
66% |
46 |
77% |
12 |
29% |
1 |
100% |
221 |
77% |
> 45 and <=90 |
6 |
4% |
11 |
31% |
10 |
17% |
23 |
56% |
0 |
0% |
50 |
17% |
> 90 |
4 |
3% |
1 |
3% |
4 |
7% |
6 |
15% |
0 |
0% |
15 |
5% |
Total |
149 |
100% |
35 |
100% |
60 |
100% |
41 |
100% |
1 |
100% |
286 |
100% |
*Reporting time=0 |
5 |
|
0 |
|
0 |
|
0 |
|
0 |
|
5 |
|
*Reporting time = 0 - reports were submitted electronically on the day discovered.


The deviation reporting requirement for HCT/Ps regulated under section 361 of the PHS Act became effective on May 25, 2005. FY06 is the first full fiscal year in which manufacturers submitted reports. Cellular HCT/Ps includes peripheral and cord stem cells. Tissue HCT/Ps includes all other HCT/Ps, such as bone, skin, cornea, etc.
Total Reports and Possible Recalls
By Manufacturing System
Table 30
|
Cellular HCT/P |
Tissue HCT/P |
Total |
Possible Recall |
||||
|---|---|---|---|---|---|---|---|---|
HCT/P Deviation Code |
Total |
Poss. Recall |
Total |
Poss. Recall |
|
|
||
Donor Eligibility |
2 |
0 |
30 |
26 |
32 |
22.2% |
26 |
53.1% |
Donor Screening |
0 |
0 |
12 |
4 |
12 |
8.3% |
4 |
8.2% |
Donor Testing |
27 |
1 |
8 |
5 |
35 |
24.3% |
6 |
12.2% |
Environmental Control |
0 |
0 |
1 |
0 |
1 |
0.7% |
0 |
0.0% |
Supplies and Reagents |
2 |
0 |
1 |
1 |
3 |
2.1% |
1 |
2.0% |
Recovery |
2 |
0 |
0 |
0 |
2 |
1.4% |
0 |
0.0% |
Processing and Processing Controls |
11 |
4 |
3 |
1 |
14 |
9.7% |
5 |
10.2% |
Labeling Controls |
0 |
0 |
1 |
1 |
1 |
0.7% |
1 |
2.0% |
Storage |
0 |
0 |
1 |
0 |
1 |
0.7% |
0 |
0.0% |
Receipt, Pre-Distribution, Shipment & Distribution |
30 |
0 |
13 |
6 |
43 |
29.9% |
6 |
12.2% |
Total |
74 |
5 |
70 |
44 |
144 |
100% |
49 |
100% |
361 HCT/P MANUFACTURES
Adherence To 45 Day Required Time For Reporting
(Reporting Time = Date of FDA receipt – Date of discovery of BPD)
Table 31
Reporting Time (days) |
Cellular |
Tissue |
Total |
|||
|---|---|---|---|---|---|---|
< or = 45 |
52 |
70% |
47 |
67% |
99 |
69% |
> 45 and <=90 |
13 |
18% |
16 |
23% |
29 |
20% |
> 90 |
9 |
12% |
7 |
10% |
16 |
11% |
Total |
74 |
100% |
70 |
100% |
144 |
100% |
*Reporting time=0 |
1 |
|
0 |
|
1 |
|
*Reporting time = 0 - reports were submitted electronically on the day discovered.