Appendix A to § 1910.134: Fit Testing Procedures (Mandatory)
Part I. OSHA-Accepted Fit Test Protocols
A. Fit Testing Procedures -- General Requirements
The employer shall conduct fit testing using the following
procedures. The requirements in this appendix apply to all OSHA-accepted
fit test methods, both QLFT and QNFT.
1. The test subject shall be allowed to pick the most acceptable
respirator from a sufficient number of respirator models and sizes
so that the respirator is acceptable to, and correctly fits, the
user.
2. Prior to the selection process, the test subject shall be
shown how to put on a respirator, how it should be positioned on the
face, how to set strap tension and how to determine an acceptable
fit. A mirror shall be available to assist the subject in evaluating
the fit and positioning of the respirator. This instruction may not
constitute the subject's formal training on respirator use, because
it is only a review.
3. The test subject shall be informed that he/she is being asked
to select the respirator that provides the most acceptable fit. Each
respirator represents a different size and shape, and if fitted and
used properly, will provide adequate protection.
4. The test subject shall be instructed to hold each chosen
facepiece up to the face and eliminate those that obviously do not
give an acceptable fit.
5. The more acceptable facepieces are noted in case the one
selected proves unacceptable; the most comfortable mask is donned
and worn at least five minutes to assess comfort. Assistance in
assessing comfort can be given by discussing the points in the
following item A.6. If the test subject is not familiar with using a
particular respirator, the test subject shall be directed to don the
mask several times and to adjust the straps each time to become
adept at setting proper tension on the straps.
6. Assessment of comfort shall include a review of the following
points with the test subject and allowing the test subject adequate
time to determine the comfort of the respirator:
(a) Position of the mask on the nose
(b) Room for eye protection
(c) Room to talk
(d) Position of mask on face and cheeks
7. The following criteria shall be used to help determine the
adequacy of the respirator fit:
(a) Chin properly placed;
(b) Adequate strap tension, not overly tightened;
(c) Fit across nose bridge;
(d) Respirator of proper size to span distance from nose to
chin;
(e) Tendency of respirator to slip;
(f) Self-observation in mirror to evaluate fit and respirator
position.
8. The test subject shall conduct a user seal check, either the
negative and positive pressure seal checks described in Appendix B-1
of this section or those recommended by the respirator manufacturer
which provide equivalent protection to the procedures in Appendix B-1.
Before conducting the negative and positive pressure checks, the
subject shall be told to seat the mask on the face by moving the
head from side-to-side and up and down slowly while taking in a few
slow deep breaths. Another facepiece shall be selected and retested
if the test subject fails the user seal check tests.
9. The test shall not be conducted if there is any hair growth
between the skin and the facepiece sealing surface, such as stubble
beard growth, beard, mustache or sideburns which cross the
respirator sealing surface. Any type of apparel which interferes
with a satisfactory fit shall be altered or removed.
10. If a test subject exhibits difficulty in breathing during
the tests, she or he shall be referred to a physician or other
licensed health care professional, as appropriate, to determine
whether the test subject can wear a respirator while performing her
or his duties.
11. If the employee finds the fit of the respirator
unacceptable, the test subject shall be given the opportunity to
select a different respirator and to be retested.
12. Exercise regimen. Prior to the commencement of the fit test,
the test subject shall be given a description of the fit test and
the test subject's responsibilities during the test procedure. The
description of the process shall include a description of the test
exercises that the subject will be performing. The respirator to be
tested shall be worn for at least 5 minutes before the start of the
fit test.
13. The fit test shall be performed while the test subject is
wearing any applicable safety equipment that may be worn during
actual respirator use which could interfere with respirator fit.
14. Test Exercises.
(a) Employers must perform the following test exercises
for all fit testing methods prescribed in this appendix, except for
the CNP quantitative fit testing protocol and the CNP REDON quantitative
fit testing protocol. For these two protocols, employers must ensure that
the test subjects (i.e., employees) perform the exercise procedure
specified in Part I.C.4(b) of this appendix for the CNP quantitative fit
testing protocol, or the exercise procedure described in Part I.C.5(b)
of this appendix for the CNP REDON quantitative fit-testing protocol. For
the remaining fit testing methods, employers must ensure that employees
perform the test exercises in the appropriate test environment in the
following manner:
(1) Normal breathing. In a normal standing position, without
talking, the subject shall breathe normally.
(2) Deep breathing. In a normal standing position, the subject
shall breathe slowly and deeply, taking caution so as not to
hyperventilate.
(3) Turning head side to side. Standing in place, the subject
shall slowly turn his/her head from side to side between the extreme
positions on each side. The head shall be held at each extreme
momentarily so the subject can inhale at each side.
(4) Moving head up and down. Standing in place, the subject
shall slowly move his/her head up and down. The subject shall be
instructed to inhale in the up position (i.e., when looking toward
the ceiling).
(5) Talking. The subject shall talk out loud slowly and loud
enough so as to be heard clearly by the test conductor. The subject
can read from a prepared text such as the Rainbow Passage, count
backward from 100, or recite a memorized poem or song.
Rainbow Passage
When the sunlight strikes raindrops in the air, they act like a
prism and form a rainbow. The rainbow is a division of white light
into many beautiful colors. These take the shape of a long round
arch, with its path high above, and its two ends apparently beyond
the horizon. There is, according to legend, a boiling pot of gold at
one end. People look, but no one ever finds it. When a man looks for
something beyond reach, his friends say he is looking for the pot of
gold at the end of the rainbow.
(6) Grimace. The test subject shall grimace by smiling or
frowning. (This applies only to QNFT testing; it is not performed
for QLFT)
(7) Bending over. The test subject shall bend at the waist as if
he/she were to touch his/her toes. Jogging in place shall be
substituted for this exercise in those test environments such as
shroud type QNFT or QLFT units that do not permit bending over at
the waist.
(8) Normal breathing. Same as exercise (1).
(b) Each test exercise shall be performed for one minute except
for the grimace exercise which shall be performed for 15 seconds.
The test subject shall be questioned by the test conductor regarding
the comfort of the respirator upon completion of the protocol. If it
has become unacceptable, another model of respirator shall be tried.
The respirator shall not be adjusted once the fit test exercises
begin. Any adjustment voids the test, and the fit test must be
repeated.
B. Qualitative Fit Test (QLFT) Protocols
1. General
(a) The employer shall ensure that persons administering QLFT
are able to prepare test solutions, calibrate equipment and perform
tests properly, recognize invalid tests, and ensure that test
equipment is in proper working order.
(b) The employer shall ensure that QLFT equipment is kept clean
and well maintained so as to operate within the parameters for which
it was designed.
2. Isoamyl Acetate Protocol
Note: This protocol is not appropriate to use for the fit
testing of particulate respirators. If used to fit test particulate
respirators, the respirator must be equipped with an organic vapor
filter.
(a) Odor Threshold Screening
Odor threshold screening, performed without wearing a
respirator, is intended to determine if the individual tested can
detect the odor of isoamyl acetate at low levels.
(1) Three 1 liter glass jars with metal lids are required.
(2) Odor-free water (e.g., distilled or spring water) at
approximately 25 deg. C (77 deg. F) shall be used for the solutions.
(3) The isoamyl acetate (IAA) (also known at isopentyl acetate)
stock solution is prepared by adding 1 ml of pure IAA to 800 ml of
odor-free water in a 1 liter jar, closing the lid and shaking for 30
seconds. A new solution shall be prepared at least weekly.
(4) The screening test shall be conducted in a room separate
from the room used for actual fit testing. The two rooms shall be
well-ventilated to prevent the odor of IAA from becoming evident in
the general room air where testing takes place.
(5) The odor test solution is prepared in a second jar by
placing 0.4 ml of the stock solution into 500 ml of odor-free water
using a clean dropper or pipette. The solution shall be shaken for
30 seconds and allowed to stand for two to three minutes so that the
IAA concentration above the liquid may reach equilibrium. This
solution shall be used for only one day.
(6) A test blank shall be prepared in a third jar by adding 500
cc of odor-free water.
(7) The odor test and test blank jar lids shall be labeled
(e.g., 1 and 2) for jar identification. Labels shall be placed on
the lids so that they can be peeled off periodically and switched to
maintain the integrity of the test.
(8) The following instruction shall be typed on a card and
placed on the table in front of the two test jars (i.e., 1 and 2):
"The purpose of this test is to determine if you can smell banana
oil at a low concentration. The two bottles in front of you contain
water. One of these bottles also contains a small amount of banana
oil. Be sure the covers are on tight, then shake each bottle for two
seconds. Unscrew the lid of each bottle, one at a time, and sniff at
the mouth of the bottle. Indicate to the test conductor which bottle
contains banana oil."
(9) The mixtures used in the IAA odor detection test shall be
prepared in an area separate from where the test is performed, in
order to prevent olfactory fatigue in the subject.
(10) If the test subject is unable to correctly identify the jar
containing the odor test solution, the IAA qualitative fit test
shall not be performed.
(11) If the test subject correctly identifies the jar containing
the odor test solution, the test subject may proceed to respirator
selection and fit testing.
(b) Isoamyl Acetate Fit Test
(1) The fit test chamber shall be a clear 55-gallon drum liner
suspended inverted over a 2-foot diameter frame so that the top of
the chamber is about 6 inches above the test subject's head. If no
drum liner is available, a similar chamber shall be constructed
using plastic sheeting. The inside top center of the chamber shall
have a small hook attached.
(2) Each respirator used for the fitting and fit testing shall
be equipped with organic vapor cartridges or offer protection
against organic vapors.
(3) After selecting, donning, and properly adjusting a
respirator, the test subject shall wear it to the fit testing room.
This room shall be separate from the room used for odor threshold
screening and respirator selection, and shall be well-ventilated, as
by an exhaust fan or lab hood, to prevent general room
contamination.
(4) A copy of the test exercises and any prepared text from
which the subject is to read shall be taped to the inside of the
test chamber.
(5) Upon entering the test chamber, the test subject shall be
given a 6-inch by 5-inch piece of paper towel, or other porous,
absorbent, single-ply material, folded in half and wetted with 0.75
ml of pure IAA. The test subject shall hang the wet towel on the
hook at the top of the chamber. An IAA test swab or ampule may be
substituted for the IAA wetted paper towel provided it has been
demonstrated that the alternative IAA source will generate an IAA
test atmosphere with a concentration equivalent to that generated by
the paper towel method.
(6) Allow two minutes for the IAA test concentration to
stabilize before starting the fit test exercises. This would be an
appropriate time to talk with the test subject; to explain the fit
test, the importance of his/her cooperation, and the purpose for the
test exercises; or to demonstrate some of the exercises.
(7) If at any time during the test, the subject detects the
banana-like odor of IAA, the test is failed. The subject shall
quickly exit from the test chamber and leave the test area to avoid
olfactory fatigue.
(8) If the test is failed, the subject shall return to the
selection room and remove the respirator. The test subject shall
repeat the odor sensitivity test, select and put on another
respirator, return to the test area and again begin the fit test
procedure described in (b) (1) through (7) above. The process
continues until a respirator that fits well has been found. Should
the odor sensitivity test be failed, the subject shall wait at least
5 minutes before retesting. Odor sensitivity will usually have
returned by this time.
(9) If the subject passes the test, the efficiency of the test
procedure shall be demonstrated by having the subject break the
respirator face seal and take a breath before exiting the chamber.
(10) When the test subject leaves the chamber, the subject shall
remove the saturated towel and return it to the person conducting
the test, so that there is no significant IAA concentration buildup
in the chamber during subsequent tests. The used towels shall be
kept in a self-sealing plastic bag to keep the test area from being
contaminated.
3. Saccharin Solution Aerosol Protocol
The entire screening and testing procedure shall be explained to
the test subject prior to the conduct of the screening test.
(a) Taste threshold screening. The saccharin taste threshold
screening,
performed without wearing a respirator, is intended to determine
whether the individual being tested can detect the taste of
saccharin.
(1) During threshold screening as well as during fit testing,
subjects shall wear an enclosure about the head and shoulders that
is approximately 12 inches in diameter by 14 inches tall with at
least the front portion clear and that allows free movements of the
head when a respirator is worn. An enclosure substantially similar
to the 3M hood assembly, parts # FT 14 and # FT 15 combined, is
adequate.
(2) The test enclosure shall have a 3/4-inch (1.9 cm) hole in
front of the test subject's nose and mouth area to accommodate the
nebulizer nozzle.
(3) The test subject shall don the test enclosure. Throughout
the threshold screening test, the test subject shall breathe through
his/her slightly open mouth with tongue extended. The subject is
instructed to report when he/she detects a sweet taste.
(4) Using a DeVilbiss Model 40 Inhalation Medication Nebulizer
or equivalent, the test conductor shall spray the threshold check
solution into the enclosure. The nozzle is directed away from the
nose and mouth of the person. This nebulizer shall be clearly marked
to distinguish it from the fit test solution nebulizer.
(5) The threshold check solution is prepared by dissolving 0.83
gram of sodium saccharin USP in 100 ml of warm water. It can be
prepared by putting 1 ml of the fit test solution (see (b)(5) below)
in 100 ml of distilled water.
(6) To produce the aerosol, the nebulizer bulb is firmly
squeezed so that it collapses completely, then released and allowed
to fully expand.
(7) Ten squeezes are repeated rapidly and then the test subject
is asked whether the saccharin can be tasted. If the test subject
reports tasting the sweet taste during the ten squeezes, the
screening test is completed. The taste threshold is noted as ten
regardless of the number of squeezes actually completed.
(8) If the first response is negative, ten more squeezes are
repeated rapidly and the test subject is again asked whether the
saccharin is tasted. If the test subject reports tasting the sweet
taste during the second ten squeezes, the screening test is
completed. The taste threshold is noted as twenty regardless of the
number of squeezes actually completed.
(9) If the second response is negative, ten more squeezes are
repeated rapidly and the test subject is again asked whether the
saccharin is tasted. If the test subject reports tasting the sweet
taste during the third set of ten squeezes, the screening test is
completed. The taste threshold is noted as thirty regardless of the
number of squeezes actually completed.
(10) The test conductor will take note of the number of squeezes
required to solicit a taste response.
(11) If the saccharin is not tasted after 30 squeezes (step 10),
the test subject is unable to taste saccharin and may not perform
the saccharin fit test.
Note to paragraph 3. (a): If the test subject eats or drinks
something sweet before the screening test, he/she may be unable to
taste the weak saccharin solution.
(12) If a taste response is elicited, the test subject shall be
asked to take note of the taste for reference in the fit test.
(13) Correct use of the nebulizer means that approximately 1 ml
of liquid is used at a time in the nebulizer body.
(14) The nebulizer shall be thoroughly rinsed in water, shaken
dry, and refilled at least each morning and afternoon or at least
every four hours.
(b) Saccharin solution aerosol fit test procedure.
(1) The test subject may not eat, drink (except plain water),
smoke, or chew gum for 15 minutes before the test.
(2) The fit test uses the same enclosure described in 3. (a)
above.
(3) The test subject shall don the enclosure while wearing the
respirator selected in section I. A. of this appendix. The
respirator shall be properly adjusted and equipped with a
particulate filter(s).
(4) A second DeVilbiss Model 40 Inhalation Medication Nebulizer
or equivalent is used to spray the fit test solution into the
enclosure. This nebulizer shall be clearly marked to distinguish it
from the screening test solution nebulizer.
(5) The fit test solution is prepared by adding 83 grams of
sodium saccharin to 100 ml of warm water.
(6) As before, the test subject shall breathe through the
slightly open mouth with tongue extended, and report if he/she
tastes the sweet taste of saccharin.
(7) The nebulizer is inserted into the hole in the front of the
enclosure and an initial concentration of saccharin fit test
solution is sprayed into the enclosure using the same number of
squeezes (either 10, 20 or 30 squeezes) based on the number of
squeezes required to elicit a taste response as noted during the
screening test. A minimum of 10 squeezes is required.
(8) After generating the aerosol, the test subject shall be
instructed to perform the exercises in section I. A. 14. of this
appendix.
(9) Every 30 seconds the aerosol concentration shall be
replenished using one half the original number of squeezes used
initially (e.g., 5, 10 or 15).
(10) The test subject shall indicate to the test conductor if at
any time during the fit test the taste of saccharin is detected. If
the test subject does not report tasting the saccharin, the test is
passed.
(11) If the taste of saccharin is detected, the fit is deemed
unsatisfactory and the test is failed. A different respirator shall
be tried and the entire test procedure is repeated (taste threshold
screening and fit testing).
(12) Since the nebulizer has a tendency to clog during use, the
test operator must make periodic checks of the nebulizer to ensure
that it is not clogged. If clogging is found at the end of the test
session, the test is invalid.
4. BitrexTM (Denatonium Benzoate) Solution Aerosol
Qualitative Fit Test Protocol
The BitrexTM (Denatonium benzoate) solution aerosol
QLFT protocol uses the published saccharin test protocol because
that protocol is widely accepted. Bitrex is routinely used as a
taste aversion agent in household liquids which children should not
be drinking and is endorsed by the American Medical Association, the
National Safety Council, and the American Association of Poison
Control Centers. The entire screening and testing procedure shall be
explained to the test subject prior to the conduct of the screening
test.
(a) Taste Threshold Screening.
The Bitrex taste threshold screening, performed without wearing
a respirator, is intended to determine whether the individual being
tested can detect the taste of Bitrex.
(1) During threshold screening as well as during fit testing,
subjects shall wear an enclosure about the head and shoulders that
is approximately 12 inches (30.5 cm) in diameter by 14 inches (35.6
cm) tall. The front portion of the enclosure shall be clear from the
respirator and allow free movement of the head when a respirator is
worn. An enclosure substantially similar to the 3M hood assembly,
parts # FT 14 and # FT 15 combined, is adequate.
(2) The test enclosure shall have a \3/4\ inch (1.9 cm) hole in
front of the test subject's nose and mouth area to accommodate the
nebulizer nozzle.
(3) The test subject shall don the test enclosure. Throughout
the threshold screening test, the test subject shall breathe through
his or her slightly open mouth with tongue extended. The subject is
instructed to report when he/she detects a bitter taste
(4) Using a DeVilbiss Model 40 Inhalation Medication Nebulizer
or equivalent, the test conductor shall spray the Threshold Check
Solution into the enclosure. This Nebulizer shall be clearly marked
to distinguish it from the fit test solution nebulizer.
(5) The Threshold Check Solution is prepared by adding 13.5
milligrams of Bitrex to 100 ml of 5% salt (NaCl) solution in
distilled water.
(6) To produce the aerosol, the nebulizer bulb is firmly
squeezed so that the bulb collapses completely, and is then released
and allowed to fully expand.
(7) An initial ten squeezes are repeated rapidly and then the
test subject is asked whether the Bitrex can be tasted. If the test
subject reports tasting the bitter taste during the ten squeezes,
the screening test is completed. The taste threshold is noted as ten
regardless of the number of squeezes actually completed.
(8) If the first response is negative, ten more squeezes are
repeated rapidly and the test subject is again asked whether the
Bitrex is tasted. If the test subject reports tasting the bitter
taste during the second ten squeezes, the screening test is
completed. The taste threshold is noted as twenty regardless of the
number of squeezes actually completed.
(9) If the second response is negative, ten more squeezes are
repeated rapidly and the test subject is again asked whether the
Bitrex is tasted. If the test subject reports tasting the bitter
taste during the third set of ten squeezes, the screening test is
completed. The taste threshold is noted as thirty regardless of the
number of squeezes actually completed.
(10) The test conductor will take note of the number of squeezes
required to solicit a taste response.
(11) If the Bitrex is not tasted after 30 squeezes (step 10),
the test subject is unable to taste Bitrex and may not perform the
Bitrex fit test.
(12) If a taste response is elicited, the test subject shall be
asked to take note of the taste for reference in the fit test.
(13) Correct use of the nebulizer means that approximately 1 ml
of liquid is used at a time in the nebulizer body.
(14) The nebulizer shall be thoroughly rinsed in water, shaken
to dry, and refilled at least each morning and afternoon or at least
every four hours.
(b) Bitrex Solution Aerosol Fit Test Procedure.
(1) The test subject may not eat, drink (except plain water),
smoke, or chew gum for 15 minutes before the test.
(2) The fit test uses the same enclosure as that described in 4.
(a) above.
(3) The test subject shall don the enclosure while wearing the
respirator selected according to section I. A. of this appendix. The
respirator shall be properly adjusted and equipped with any type
particulate filter(s).
(4) A second DeVilbiss Model 40 Inhalation Medication Nebulizer
or equivalent is used to spray the fit test solution into the
enclosure. This nebulizer shall be clearly marked to distinguish it
from the screening test solution nebulizer.
(5) The fit test solution is prepared by adding 337.5 mg of
Bitrex to 200 ml of a 5% salt (NaCl) solution in warm water.
(6) As before, the test subject shall breathe through his or her
slightly open mouth with tongue extended, and be instructed to
report if he/she tastes the bitter taste of Bitrex.
(7) The nebulizer is inserted into the hole in the front of the
enclosure and an initial concentration of the fit test solution is
sprayed into the enclosure using the same number of squeezes (either
10, 20 or 30 squeezes) based on the number of squeezes required to
elicit a taste response as noted during the screening test.
(8) After generating the aerosol, the test subject shall be
instructed to perform the exercises in section I. A. 14. of this
appendix.
(9) Every 30 seconds the aerosol concentration shall be
replenished using one half the number of squeezes used initially
(e.g., 5, 10 or 15).
(10) The test subject shall indicate to the test conductor if at
any time during the fit test the taste of Bitrex is detected. If the
test subject does not report tasting the Bitrex, the test is passed.
(11) If the taste of Bitrex is detected, the fit is deemed
unsatisfactory and the test is failed. A different respirator shall
be tried and the entire test procedure is repeated (taste threshold
screening and fit testing).
5. Irritant Smoke (Stannic Chloride) Protocol
This qualitative fit test uses a person's response to the
irritating chemicals released in the "smoke" produced by a stannic
chloride ventilation smoke tube to detect leakage into the
respirator.
(a) General Requirements and Precautions
(1) The respirator to be tested shall be equipped with high
efficiency particulate air (HEPA) or P100 series filter(s).
(2) Only stannic chloride smoke tubes shall be used for this
protocol.
(3) No form of test enclosure or hood for the test subject shall
be used.
(4) The smoke can be irritating to the eyes, lungs, and nasal
passages. The test conductor shall take precautions to minimize the
test subject's exposure to irritant smoke. Sensitivity varies, and
certain individuals may respond to a greater degree to irritant
smoke. Care shall be taken when performing the sensitivity screening
checks that determine whether the test subject can detect irritant
smoke to use only the minimum amount of smoke necessary to elicit a
response from the test subject.
(5) The fit test shall be performed in an area with adequate
ventilation to prevent exposure of the person conducting the fit
test or the build-up of irritant smoke in the general atmosphere.
(b) Sensitivity Screening Check
The person to be tested must demonstrate his or her ability to
detect a weak concentration of the irritant smoke.
(1) The test operator shall break both ends of a ventilation
smoke tube containing stannic chloride, and attach one end of the
smoke tube to a low flow air pump set to deliver 200 milliliters per
minute, or an aspirator squeeze bulb. The test operator shall cover
the other end of the smoke tube with a short piece of tubing to
prevent potential injury from the jagged end of the smoke tube.
(2) The test operator shall advise the test subject that the
smoke can be irritating to the eyes, lungs, and nasal passages and
instruct the subject to keep his/her eyes closed while the test is
performed.
(3) The test subject shall be allowed to smell a weak
concentration of the irritant smoke before the respirator is donned
to become familiar with its irritating properties and to determine
if he/she can detect the irritating properties of the smoke. The
test operator shall carefully direct a small amount of the irritant
smoke in the test subject's direction to determine that he/she can
detect it.
(c) Irritant Smoke Fit Test Procedure
(1) The person being fit tested shall don the respirator without
assistance, and perform the required user seal check(s).
(2) The test subject shall be instructed to keep his/her eyes
closed.
(3) The test operator shall direct the stream of irritant smoke
from the smoke tube toward the faceseal area of the test subject,
using the low flow pump or the squeeze bulb. The test operator shall
begin at least 12 inches from the facepiece and move the smoke
stream around the whole perimeter of the mask. The operator shall
gradually make two more passes around the perimeter of the mask,
moving to within six inches of the respirator.
(4) If the person being tested has not had an involuntary
response and/or detected the irritant smoke, proceed with the test
exercises.
(5) The exercises identified in section I.A. 14. of this
appendix shall be performed by the test subject while the respirator
seal is being continually challenged by the smoke, directed around
the perimeter of the respirator at a distance of six inches.
(6) If the person being fit tested reports detecting the
irritant smoke at any time, the test is failed. The person being
retested must repeat the entire sensitivity check and fit test
procedure.
(7) Each test subject passing the irritant smoke test without
evidence of a response (involuntary cough, irritation) shall be
given a second sensitivity screening check, with the smoke from the
same smoke tube used during the fit test, once the respirator has
been removed, to determine whether he/she still reacts to the smoke.
Failure to evoke a response shall void the fit test.
(8) If a response is produced during this second sensitivity
check, then the fit test is passed.
C. Quantitative Fit Test (QNFT) Protocols
The following quantitative fit testing procedures have been
demonstrated to be acceptable: Quantitative fit testing using a non-hazardous
test aerosol (such as corn oil, polyethylene glycol 400
[PEG 400], di-2-ethyl hexyl sebacate [DEHS], or sodium chloride)
generated in a test chamber, and employing instrumentation to
quantify the fit of the respirator; Quantitative fit testing using
ambient aerosol as the test agent and appropriate instrumentation
(condensation nuclei counter) to quantify the respirator fit;
Quantitative fit testing using controlled negative pressure and
appropriate instrumentation to measure the volumetric leak rate of a
facepiece to quantify the respirator fit.
1. General
(a) The employer shall ensure that persons administering QNFT
are able to calibrate equipment and perform tests properly,
recognize invalid tests, calculate fit factors properly and ensure
that test equipment is in proper working order.
(b) The employer shall ensure that QNFT equipment is kept clean,
and is maintained and calibrated according to the manufacturer's
instructions so as to operate at the parameters for which it was
designed.
2. Generated Aerosol Quantitative Fit Testing Protocol
(a) Apparatus.
(1) Instrumentation. Aerosol generation, dilution, and
measurement systems using particulates (corn oil, polyethylene
glycol 400 [PEG 400], di-2-ethyl hexyl sebacate [DEHS] or sodium
chloride) as test aerosols shall be used for quantitative fit
testing.
(2) Test chamber. The test chamber shall be large enough to
permit all test subjects to perform freely all required exercises
without disturbing the test agent concentration or the measurement
apparatus. The test chamber shall be equipped and constructed so
that the test agent is effectively isolated from the ambient air,
yet uniform in concentration throughout the chamber.
(3) When testing air-purifying respirators, the normal filter or
cartridge element shall be replaced with a high efficiency
particulate air (HEPA) or P100 series filter supplied by the same
manufacturer.
(4) The sampling instrument shall be selected so that a computer
record or strip chart record may be made of the test showing the
rise and fall of the test agent concentration with each inspiration
and expiration at fit factors of at least 2,000. Integrators or
computers that integrate the amount of test agent
penetration leakage into the respirator for each exercise may be
used provided a record of the readings is made.
(5) The combination of substitute air-purifying elements, test
agent and test agent concentration shall be such that the test
subject is not exposed in excess of an established exposure limit
for the test agent at any time during the testing process, based
upon the length of the exposure and the exposure limit duration.
(6) The sampling port on the test specimen respirator shall be
placed and constructed so that no leakage occurs around the port
(e.g., where the respirator is probed), a free air flow is allowed
into the sampling line at all times, and there is no interference
with the fit or performance of the respirator. The in-mask sampling
device (probe) shall be designed and used so that the air sample is
drawn from the breathing zone of the test subject, midway between
the nose and mouth and with the probe extending into the facepiece
cavity at least 1/4 inch.
(7) The test setup shall permit the person administering the
test to observe the test subject inside the chamber during the test.
(8) The equipment generating the test atmosphere shall maintain
the concentration of test agent constant to within a 10 percent
variation for the duration of the test.
(9) The time lag (interval between an event and the recording of
the event on the strip chart or computer or integrator) shall be
kept to a minimum. There shall be a clear association between the
occurrence of an event and its being recorded.
(10) The sampling line tubing for the test chamber atmosphere
and for the respirator sampling port shall be of equal diameter and
of the same material. The length of the two lines shall be equal.
(11) The exhaust flow from the test chamber shall pass through
an appropriate filter (i.e., high efficiency particulate filter) before release.
(12) When sodium chloride aerosol is used, the relative humidity
inside the test chamber shall not exceed 50 percent.
(13) The limitations of instrument detection shall be taken into
account when determining the fit factor.
(14) Test respirators shall be maintained in proper working
order and be inspected regularly for deficiencies such as cracks or
missing valves and gaskets.
(b) Procedural Requirements.
(1) When performing the initial user seal check using a positive
or negative pressure check, the sampling line shall be crimped
closed in order to avoid air pressure leakage during either of these
pressure checks.
(2) The use of an abbreviated screening QLFT test is optional.
Such a test may be utilized in order to quickly identify poor
fitting respirators that passed the positive and/or negative
pressure test and reduce the amount of QNFT time. The use of the CNC
QNFT instrument in the count mode is another optional method to
obtain a quick estimate of fit and eliminate poor fitting
respirators before going on to perform a full QNFT.
(3) A reasonably stable test agent concentration shall be
measured in the test chamber prior to testing. For canopy or shower
curtain types of test units, the determination of the test agent's
stability may be established after the test subject has entered the
test environment.
(4) Immediately after the subject enters the test chamber, the
test agent concentration inside the respirator shall be measured to
ensure that the peak penetration does not exceed 5 percent for a
half mask or 1 percent for a full facepiece respirator.
(5) A stable test agent concentration shall be obtained prior to
the actual start of testing.
(6) Respirator restraining straps shall not be over-tightened
for testing. The straps shall be adjusted by the wearer without
assistance from other persons to give a reasonably comfortable fit
typical of normal use. The respirator shall not be adjusted once the
fit test exercises begin.
(7) The test shall be terminated whenever any single peak
penetration exceeds 5 percent for half masks and 1 percent for full
facepiece respirators. The test subject shall be refitted and
retested.
(8) Calculation of fit factors.
(i) The fit factor shall be determined for the quantitative fit
test by taking the ratio of the average chamber concentration to the
concentration measured inside the respirator for each test exercise
except the grimace exercise.
(ii) The average test chamber concentration shall be calculated
as the arithmetic average of the concentration measured before and
after each test (i.e., 7 exercises) or the arithmetic average of the
concentration measured before and after each exercise or the true
average measured continuously during the respirator sample.
(iii) The concentration of the challenge agent inside the
respirator shall be determined by one of the following methods:
(A) Average peak penetration method means the method of
determining test agent penetration into the respirator utilizing a
strip chart recorder, integrator, or computer. The agent penetration
is determined by an average of the peak heights on the graph or by
computer integration, for each exercise except the grimace exercise.
Integrators or computers that calculate the actual test agent
penetration into the respirator for each exercise will also be
considered to meet the requirements of the average peak penetration
method.
(B) Maximum peak penetration method means the method of
determining test agent penetration in the respirator as determined
by strip chart recordings of the test. The highest peak penetration
for a given exercise is taken to be representative of average
penetration into the respirator for that exercise.
(C) Integration by calculation of the area under the individual
peak for each exercise except the grimace exercise. This includes
computerized integration.
(D) The calculation of the overall fit factor using individual
exercise fit factors involves first converting the exercise fit
factors to penetration values, determining the average, and then
converting that result back to a fit factor. This procedure is
described in the following equation:
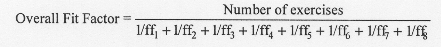
Where ff1, ff2, ff3, etc. are the
fit factors for exercises 1, 2, 3, etc.
(9) The test subject shall not be permitted to wear a half mask
or quarter facepiece respirator unless a minimum fit factor of 100
is obtained, or a full facepiece respirator unless a minimum fit
factor of 500 is obtained.
(10) Filters used for quantitative fit testing shall be replaced
whenever increased breathing resistance is encountered, or when the
test agent has altered the integrity of the filter media.
3. Ambient aerosol condensation nuclei counter (CNC)
quantitative fit testing protocol.
The ambient aerosol condensation nuclei counter (CNC)
quantitative fit testing (Portacount TM ) protocol
quantitatively fit tests respirators with the use of a probe. The
probed respirator is only used for quantitative fit tests. A probed
respirator has a special sampling device, installed on the
respirator, that allows the probe to sample the air from inside the
mask. A probed respirator is required for each make, style, model,
and size that the employer uses and can be obtained from the
respirator manufacturer or distributor. The CNC instrument
manufacturer, TSI Inc., also provides probe attachments (TSI
sampling adapters) that permit fit testing in an employee's own
respirator. A minimum fit factor pass level of at least 100 is
necessary for a half-mask respirator and a minimum fit factor pass
level of at least 500 is required for a full facepiece negative
pressure respirator. The entire screening and testing procedure
shall be explained to the test subject prior to the conduct of the
screening test.
(a) Portacount Fit Test Requirements.
(1) Check the respirator to make sure the sampling probe
and line are properly attached to the facepiece and that the respirator is fitted
with a particulate filter capable of preventing significant penetration by the
ambient particles used for the fit test (e.g., NIOSH 42 CFR 84 series 100, series 99,
or series 95 particulate filter) per manufacturer's instruction.
(2) Instruct the person to be tested to don the respirator for
five minutes before the fit test starts. This purges the ambient
particles trapped inside the respirator and permits the wearer to
make certain the respirator is comfortable. This individual shall
already have been trained on how to wear the respirator properly.
(3) Check the following conditions for the adequacy of the
respirator fit: Chin properly placed; Adequate strap tension, not
overly tightened; Fit across nose bridge; Respirator of proper size
to span distance from nose to chin; Tendency of the respirator to
slip; Self-observation in a mirror to evaluate fit and respirator
position.
(4) Have the person wearing the respirator do a user seal check.
If leakage is detected, determine the cause. If leakage is from a
poorly fitting facepiece, try another size of the same model
respirator, or another model of respirator.
(5) Follow the manufacturer's instructions for operating the
Portacount and proceed with the test.
(6) The test subject shall be instructed to perform the
exercises in section I. A. 14. of this appendix.
(7) After the test exercises, the test subject shall be
questioned by the test conductor regarding the comfort of the
respirator upon completion of the protocol. If it has become
unacceptable, another model of respirator shall be tried.
(b) Portacount Test Instrument.
(1) The Portacount will automatically stop and calculate the
overall fit factor for the entire set of exercises. The overall fit
factor is what counts. The Pass or Fail message will indicate
whether or not the test was successful. If the test was a Pass, the
fit test is over.
(2) Since the pass or fail criterion of the Portacount is user
programmable, the test operator shall ensure that the pass or fail
criterion meet the requirements for minimum respirator performance
in this Appendix.
(3) A record of the test needs to be kept on file, assuming the
fit test was successful. The record must contain the test subject's
name; overall fit factor; make, model, style, and size of respirator
used; and date tested.
4. Controlled negative pressure (CNP) quantitative fit testing
protocol.
The CNP protocol provides an alternative to aerosol fit test
methods. The CNP fit test method technology is based on exhausting
air from a temporarily sealed respirator facepiece to generate and
then maintain a constant negative pressure inside the facepiece. The
rate of air exhaust is controlled so that a constant negative
pressure is maintained in the respirator during the fit test. The
level of pressure is selected to replicate the mean inspiratory
pressure that causes leakage into the respirator under normal use
conditions. With pressure held constant, air flow out of the
respirator is equal to air flow into the respirator. Therefore,
measurement of the exhaust stream that is required to hold the
pressure in the temporarily sealed respirator constant yields a
direct measure of leakage air flow into the respirator. The CNP fit
test method measures leak rates through the facepiece as a method
for determining the facepiece fit for negative pressure respirators.
The CNP instrument manufacturer Occupational Health Dynamics of Birmingham, Alabama also provides
attachments (sampling manifolds) that replace the filter cartridges
to permit fit testing in an employee's own respirator. To perform
the test, the test subject closes his or her mouth and holds his/her
breath, after which an air pump removes air from the respirator
facepiece at a pre-selected constant pressure. The facepiece fit is
expressed as the leak rate through the facepiece, expressed as
milliliters per minute. The quality and validity of the CNP fit
tests are determined by the degree to which the in-mask pressure
tracks the test pressure during the system measurement time of
approximately five seconds. Instantaneous feedback in the form of a
real-time pressure trace of the in-mask pressure is provided and
used to determine test validity and quality. A minimum fit factor
pass level of 100 is necessary for a half-mask respirator and a
minimum fit factor of at least 500 is required for a full facepiece
respirator. The entire screening and testing procedure shall be
explained to the test subject prior to the conduct of the screening
test.
(a) CNP Fit Test Requirements.
(1) The instrument shall have a non-adjustable test pressure of
15.0 mm water pressure.
(2) The CNP system defaults selected for test pressure shall be
set at -- 15 mm of water (-0.58 inches of water) and the modeled
inspiratory flow rate shall be 53.8 liters per minute for performing
fit tests.
(Note: CNP systems have built-in capability to conduct fit testing
that is specific to unique work rate, mask, and gender situations
that might apply in a specific workplace. Use of system default
values, which were selected to represent respirator wear with medium
cartridge resistance at a low-moderate work rate, will allow inter-test
comparison of the respirator fit.)
(3) The individual who conducts the CNP fit testing shall be
thoroughly trained to perform the test.
(4) The respirator filter or cartridge needs to be replaced with
the CNP test manifold. The inhalation valve downstream from the
manifold either needs to be temporarily removed or propped open.
(5) The employer must train the test subject to hold his or her breath for at least 10 seconds.
(6) The test subject must don the test respirator without any assistance from the test administrator who is conducting the CNP fit test. The respirator must not be adjusted once the fit-test exercises begin. Any adjustment voids the test, and the test subject must repeat the fit test.
(7) The QNFT protocol shall be followed according to section I.
C. 1. of this appendix with an exception for the CNP test exercises.
(b) CNP Test Exercises.
(1) Normal breathing. In a normal standing position, without
talking, the subject shall breathe normally for 1 minute. After the
normal breathing exercise, the subject needs to hold head straight
ahead and hold his or her breath for 10 seconds during the test
measurement.
(2) Deep breathing. In a normal standing position, the subject
shall breathe slowly and deeply for 1 minute, being careful not to
hyperventilate. After the deep breathing exercise, the subject shall
hold his or her head straight ahead and hold his or her breath for
10 seconds during test measurement.
(3) Turning head side to side. Standing in place, the subject
shall slowly turn his or her head from side to side between the
extreme positions on each side for 1 minute. The head shall be held
at each extreme momentarily so the subject can inhale at each side.
After the turning head side to side exercise, the subject needs to
hold head full left and hold his or her breath for 10 seconds during
test measurement. Next, the subject needs to hold head full right
and hold his or her breath for 10 seconds during test measurement.
(4) Moving head up and down. Standing in place, the subject
shall slowly move his or her head up and down for 1 minute. The
subject shall be instructed to inhale in the up position (i.e., when
looking toward the ceiling). After the moving head up and down
exercise, the subject shall hold his or her head full up and hold
his or her breath for 10 seconds during test measurement. Next, the
subject shall hold his or her head full down and hold his or her
breath for 10 seconds during test measurement.
(5) Talking. The subject shall talk out loud slowly and loud
enough so as to be heard clearly by the test conductor. The subject
can read from a prepared text such as the Rainbow Passage, count
backward from 100, or recite a memorized poem or song for 1 minute.
After the talking exercise, the subject shall hold his or her head
straight ahead and hold his or her breath for 10 seconds during the
test measurement.
(6) Grimace. The test subject shall grimace by smiling or
frowning for 15 seconds.
(7) Bending Over. The test subject shall bend at the waist as if
he or she were to touch his or her toes for 1 minute. Jogging in
place shall be substituted for this exercise in those test
environments such as shroud-type QNFT units that prohibit bending at
the waist. After the bending over exercise, the subject shall hold
his or her head straight ahead and hold his or her breath for 10
seconds during the test measurement.
(8) Normal Breathing. The test subject shall remove and re-don
the respirator within a one-minute period. Then, in a normal
standing position, without talking, the subject shall breathe
normally for 1 minute. After the normal breathing exercise, the
subject shall hold his or her head straight ahead and hold his or
her breath for 10 seconds during the test measurement. After the
test exercises, the test subject shall be questioned by the test
conductor regarding the comfort of the respirator upon completion of
the protocol. If it has become unacceptable, another model of a
respirator shall be tried.
(c) CNP Test Instrument.
(1) The test instrument must have an effective audio-warning device, or a visual-warning device in the form of a screen tracing, that indicates when the test subject fails to hold his or her breath during the test. The test must be terminated and restarted from the beginning when the test subject fails to hold his or her breath during the test. The test subject then may be refitted and retested.
(2) A record of the test shall be kept on file, assuming the fit
test was successful. The record must contain the test subject's
name; overall fit factor; make, model, style and size of respirator
used; and date tested.
5. Controlled negative pressure (CNP) REDON quantitative fit testing protocol.
(a) When administering this protocol to test subjects, employers must comply
with the requirements specified in paragraphs (a) and (c) of Part I.C.4 of this
appendix ("Controlled negative pressure (CNP) quantitative fit testing protocol"),
as well as use the test exercises described below in paragraph (b) of this
protocol instead of the test exercises specified in paragraph (b) of Part I.C.4
of this appendix.
(b) Employers must ensure that each test subject being fit tested using this
protocol follows the exercise and measurement procedures, including the order
of administration, described below in Table A-1 of this appendix.
Table A-1. -- CNP REDON Quantitative Fit Testing Protocol
| Exercises(1) |
Exercise procedure |
Measurement procedure |
Facing Forward
|
Stand and breathe normally, without talking, for 30 seconds.
|
Face forward, while holding breath for 10 seconds. |
| Bending Over |
Bend at the waist, as if going to touch his or her toes, for 30 seconds. |
Face parallel to the floor, while holding breath for 10 seconds |
| Head Shaking |
For about three seconds, shake head back and forth vigorously several times while shouting.
|
Face forward, while holding breath for 10 seconds. |
| REDON 1 |
Remove the respirator mask, loosen all facepiece straps, and then redon the respirator mask.
|
Face forward, while holding breath for 10 seconds. |
| REDON 2 |
Remove the respirator mask, loosen all facepiece straps, and then redon the respirator mask again. |
Face forward, while holding breath for 10 seconds. |
1 Exercises are listed in the order in which they are to be administered.
(c) After completing the test exercises, the test administrator must question
each test subject regarding the comfort of the respirator. When a test
subject states that the respirator is unacceptable, the employer must
ensure that the test administrator repeats the protocol using another
respirator model.
(d) Employers must determine the overall fit factor for each test
subject by calculating the harmonic mean of the fit testing
exercises as follows:

Where:
N = The number of exercises;
FF1 = The fit factor for the first exercise;
FF2 = The fit factor for the second exercise; and
FFN = The fit factor for the nth exercise.
Part II. New Fit Test Protocols
A. Any person may submit to OSHA an application for approval of
a new fit test protocol. If the application meets the following
criteria, OSHA will initiate a rulemaking proceeding under section
6(b)(7) of the OSH Act to determine whether to list
the new protocol as an approved protocol in this Appendix A.
B. The application must include a detailed description of the
proposed new fit test protocol. This application must be supported
by either:
1. A test report prepared by an independent government research
laboratory (e.g., Lawrence Livermore National Laboratory, Los Alamos
National Laboratory, the National Institute for Standards and
Technology) stating that the laboratory has tested the protocol and
had found it to be accurate and reliable; or
2. An article that has been published in a peer-reviewed
industrial hygiene journal describing the protocol and explaining
how test data support the protocol's accuracy and reliability.
C. If OSHA determines that additional information is required
before the Agency commences a rulemaking proceeding under this
section, OSHA will so notify the applicant and afford the applicant
the opportunity to submit the supplemental information. Initiation
of a rulemaking proceeding will be deferred until OSHA has received
and evaluated the supplemental information.
[63 FR 20098, April 23, 1998; 69 FR 46993, August 4, 2004]
|

