Aggression-related Gene Weakens Brain’s Impulse
Control Circuits
A version of a gene previously linked to impulsive violence appears
to weaken brain circuits that regulate impulses, emotional memory
and thinking in humans, researchers at the National Institutes
of Health’s (NIH) National Institute of Mental Health (NIMH) have
found. Brain scans revealed that people with this version — especially
males — tended to have relatively smaller emotion-related
brain structures, a hyperactive alarm center and under-active impulse
control circuitry. The study identifies neural mechanisms by which
this gene likely contributes to risk for violent and impulsive
behavior through effects on the developing brain.
NIMH intramural researchers Andreas Meyer-Lindenberg, M.D., Ph.D.,
Daniel Weinberger, M.D., and colleagues report on their magnetic
resonance imaging (MRI) study online in the Proceedings of the
National Academy of Sciences during the week of March 20, 2006.
“These new findings illustrate the breathtaking power of ‘imaging
genomics’ to study the brain’s workings in a way that helps us
to understand the circuitry underlying diversity in human temperament,” said
NIH Director Elias A. Zerhouni, M.D., who conducted MRI studies
earlier in his career.
“By itself, this gene is likely to contribute only a small amount
of risk in interaction with other genetic and psychosocial influences;
it won’t make people violent,” explained Meyer-Lindenberg. “But
by studying its effects in a large sample of normal people, we
were able to see how this gene variant biases the brain toward
impulsive, aggressive behavior.”
The gene is one of two common versions that code for the enzyme
monoamine oxydase-A (MAO-A), which breaks down key mood-regulating
chemical messengers, most notably serotonin. The previously identified
violence-related, or L, version, contains a different number of
repeating sequences in its genetic code than the other version
(H), likely resulting in lower enzyme activity and hence higher
levels of serotonin. These, in turn, influence how the brain gets
wired during development. The variations may have more impact on
males because they have only one copy of this X-chromosomal gene,
while females have two copies, one of which will be of the H variant
in most cases.
Several previous studies had linked increased serotonin during
development with violence and the L version of MAO-A. For example,
a 2002 study* by NIMH-funded researchers discovered
that the gene’s effects depend on interactions with environmental
hard knocks: men with L were more prone to impulsive violence,
but only if they were abused as children. Meyer-Lindenberg and
colleagues set out to discover how this works at the level of brain
circuitry.
Using structural MRI in 97 subjects, they found that those with
L showed reductions in gray matter (neurons and their connections)
of about 8 percent in brain structures of a mood-regulating circuit
(cingulate cortex, amygdala) among other areas. Volume of an area
important for motivation and impulse regulation (orbital frontal
cortex) was increased by 14 percent in men only. Although the reasons
are unknown, this could reflect deficient pruning — the withering
of unused neuronal connections as the brain matures and becomes
more efficient, speculates Meyer-Lindenberg.
The researchers then looked at effects on brain activity using
functional MRI (fMRI) scans. While performing a task matching emotionally
evocative pictures — angry and fearful faces — subjects
with L showed higher activity in the fear hub (amygdala). At the
same time, decreased activity was observed in higher brain areas
that regulate the fear hub (cingulate, orbital frontal, and insular
cortices) — essentially the same circuit that was changed
in volume.
While these changes were found in both men and women, two other
experiments revealed gene-related changes in men only. In a task
which required remembering emotionally negative information, men,
but not women, with L had increased reactivity in the fear (amygdala)
and memory (hippocampus) hubs. Men with L were also deficient during
a task requiring them to inhibit a simple motor response; they
failed to activate a part of the brain (cingulate cortex) important
for inhibiting such behavioral impulses. This region was, conspicuously,
the cortex area that was most reduced in volume.
The findings echo those of a 2005 NIMH study** showing
how another serotonin-related gene variant shapes the same mood-regulating
circuit. In this study also, the gene version that boosts serotonin
levels resulted in impaired emotion-related lower brain structures,
increased fear hub activation and a weaker response of its regulatory
circuits. Yet, the effects of the L version of MAO-A were more
extensive, perhaps reflecting the fact that it also impacts another
key mood-regulating neurotransmitter, norepinephrine.
The weakened regulatory circuits in men with L are compounded
by intrinsically weaker connections between the orbital frontal
cortex and amygdala in all men, say the researchers.
“Heightened sensitivity in brain circuits important to cognitive
inhibition and memory for negative emotional information may contribute
to increased vulnerability of men with L exposed to abuse during
childhood,” suggested Weinberger. “Since only men showed gene effects
in several of these circuits, this could lead to a situation where
multiple brain control mechanisms are impaired and contribute to
manifestly violent behavior, a kind of genetic double jeopardy.”
Also participating in the study were: Joshua Buckholtz, Bhaskar
Kolachana, Ahmad Hariri, Lukas Pezawas, Giuseppe Blasi, Ashley
Wabnitz, Robyn Honea, Beth Verchinski, Joseph Callicott, Michael
Egan, and Venkata Mattay, NIMH Clinical Brain Disorders Branch.
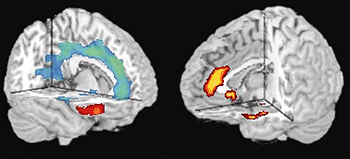 |
Structural (left) and
functional (right) MRI scan data shows that subjects with
the violence-related version of the MAO-A gene (MAOA-L)
had reduced volume and activity of the anterior cingulate
cortex (blue area in front part of brain at left and corresponding
yellow area in at right), which is thought to be the hub
of a circuit responsible for regulating impulsive aggression.
The color- coded areas show where subjects with the L gene
type differed from subjects with the H gene type.
Source: NIMH Clinical
Brain Disorders Branch |
NIMH is part of the National Institutes of Health (NIH), the
Federal Government's primary agency for biomedical and behavioral
research. NIH is a component of the U.S. Department of Health
and Human Services.
The National Institutes of Health (NIH) — The Nation's
Medical Research Agency — includes 27 Institutes and
Centers and is a component of the U.S. Department of Health and
Human Services. It is the primary federal agency for conducting
and supporting basic, clinical and translational medical research,
and it investigates the causes, treatments, and cures for both
common and rare diseases. For more information about NIH and
its programs, visit http://www.nih.gov. |

