 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
Anaplasma phagocytophilum Transmitted Through Blood Transfusion --- Minnesota, 2007Anaplasma phagocytophilum, a gram-negative, obligate intracellular bacterium of neutrophils, causes human anaplasmosis, a tickborne rickettsial disease formerly known as human granulocytic ehrlichiosis (1). In November 2007, the Minnesota Department of Health was contacted about A. phagocytophilum infection in a hospitalized Minnesota resident who had recently undergone multiple blood transfusions. Subsequent investigation indicated the infection likely was acquired through a transfusion of red blood cells. This report describes the patient's clinical history and the epidemiologic and laboratory investigations. Although a previous case of transfusion-transmitted anaplasmosis was reported (2), this is the first published report in which transfusion transmission of A. phagocytophilum was confirmed by testing of the recipient and a donor. Although polymerase chain reaction (PCR) assays provided reliable evidence of transmission in this case, no cost-effective method for screening blood donors for A. phagocytophilum exists. Screening donors for a recent history of tick bite is not likely to be sensitive or specific because such exposures are common and often not recalled by persons with anaplasmosis (3). Physicians should consider the possibility of anaplasmosis in patients who develop posttransfusion acute thrombocytopenia, especially if accompanied by fever, and should report suspected transfusion-associated cases to health authorities. Case ReportThe patient, a male aged 68 years with a medical history of chronic renal insufficiency, psoriatic arthritis, ankylosing spondylitis, and corticosteroid therapy, underwent elective knee arthroplasty and synovectomy on October 12, 2007. Three weeks before his hospitalization, the patient had traveled to an area where blacklegged ticks (Ixodes spp.) were endemic, but he did not spend time outdoors and had no known tick bites. Several hours after the procedure, the patient developed bleeding at the surgical site and associated coagulopathy, indicated by elevated international normalized ratio (INR) and partial thromboplastin time (PTT) and by decreased fibrinogen and platelet counts. The extensive hemorrhage required two surgical evacuations of hematoma from the knee, popliteal artery embolization, and transfusion of multiple blood components. During October 12--21, the patient received 34 units of nonleukoreduced red blood cells (RBC), 4 units of leukocyte-reduced apheresis platelets, 14 units of fresh frozen plasma (FFP), and 7 units of cryoprecipitate. The components came from 59 individual blood donors; all donations were collected by Memorial Blood Centers (St. Paul, Minnesota). On October 19, the patient developed sepsis and multisystem failure. He was treated empirically with antibiotics (cefazolin, piperacillin/tazobactam, vancomycin, and levofloxacin). Blood cultures were negative on October 18, 20, and 31, and urine cultures were negative on October 19 and 25. On October 31, the patient was found to have worsening thrombocytopenia. His platelet count declined from 178,000/mm3 on October 31 to 54,000/mm3 on November 5. On November 1, he developed hypotension and fever attributed to urinary tract infection. He was treated with levofloxacin and sulfamethoxazole/trimethoprim and was afebrile by November 3. On November 3, 22 days after admission, a peripheral blood smear from the patient demonstrated inclusions compatible with A. phagocytophilum morulae in neutrophils. Retrospective review of an October 15 blood smear from the patient showed no evidence of intracellular morulae. Whole blood specimens from November 3--5 were positive for A. phagocytophilum DNA by PCR assays conducted at the Mayo Medical Laboratory, Minnesota Department of Health, and CDC. Serum from November 3--5 was tested at CDC and found to be weakly positive by indirect immunofluorescence assay (IFA) (titer 1:64) for immunoglobulin G (IgG) antibodies to A. phagocytophilum. Doxycycline treatment was begun on November 5. The patient's platelet count steadily improved and returned to a normal level of 163,000/mm3 on November 10. Pretransfusion blood samples and serum from the patient's convalescence period were not available for further testing. The patient improved clinically and was transferred to a rehabilitation unit on November 13. After rehabilitation, the patient was discharged on December 3, 2007. Epidemiologic and Laboratory InvestigationIn early November, Memorial Blood Centers began an investigation to identify whether any of the 59 blood donors associated with the 34 RBC, 4 platelet, 14 FFP, and 7 cryoprecipitate units had evidence of A. phagocytophilum infection. Paired whole blood specimens from the original donations had been retained from all 34 RBC donors and eight of 14 FFP donors and were available for PCR testing. During November 2007--March 2008, Memorial Blood Centers also collected postdonation blood samples for serologic testing and information on recent illness history and potential tick exposure from 53 of the 59 donors. In addition, plasma components from two FFP donors and two cryoprecipitate donors who donated again during December 2007--January 2008 were retained for serologic testing. The whole blood specimens retained from initial donation were tested by PCR, followed by sequencing of the PCR amplicons at CDC. Serum and plasma specimens were tested by IFA for IgG antibodies to A. phagocytophilum. PCR and IFA tests on samples from a female RBC donor aged 64 years were positive for A. phagocytophilum infection (Table). A. phagocytophilum DNA was found in an RBC product donated by this woman on September 28 and transfused to the patient on October 13. IgG IFA titers to A. phagocytophilum were 1:512 and 1:256, respectively, in subsequent sera collected November 17 and December 18. The donor did not recall being bitten by a tick, but had spent time in wooded areas of northeast Minnesota where anaplasmosis is endemic within the month before her donation. She reported no history of fever during the month before or after her donation. No other patients received blood components from her donation. No whole blood samples from other tested donors were PCR positive for A. phagocytophilum. Sera from two RBC donors were weakly positive by IFA (titer 1:64), but their respective whole blood samples from the original transfused units were PCR negative. These two donors did not live on wooded property and reported they had no tick exposure or illness during the 2 months before donation. Available postdonation serum samples from other donors were negative for A. phagocytophilum by IFA (titer <1:32). Reported by: M Kemperman, MPH, D Neitzel, MS, Minnesota Dept of Health; K Jensen, J Gorlin, MD, E Perry, MD, Memorial Blood Centers, Saint Paul; T Myers, MD, T Miley, MD, Park Nicollet Methodist Hospital, Saint Louis Park, Minnesota. J McQuiston, DVM, ME Eremeeva, MD, PhD, ScD, W Nicholson, PhD, J Singleton, National Center for Zoonotic, Vector-Borne, and Enteric Diseases; J Adjemian, PhD, EIS Officer, CDC. Editorial Note:A. phagocytophilum, the causative agent of anaplasmosis, typically is transmitted to humans by infected Ixodes spp. ticks. In wooded areas of the United States, A. phagocytophilum is transmitted by the blacklegged tick (Ixodes scapularis) in the Northeast and upper Midwest and by the western blacklegged tick (Ixodes pacificus) on the West Coast. In infected persons who are symptomatic, illness onset occurs 5--21 days after a bite from an infected tick. Initial presentation typically includes sudden onset of fever, headache, malaise, and myalgia, often accompanied by thrombocytopenia, leukopenia, and elevated liver transaminases. Severe infections can include prolonged fever, shock, confusion, seizures, pneumonitis, renal failure, hemorrhages, opportunistic infections, and death (1). Anaplasmosis and other tickborne diseases, including human ehrlichiosis, Rocky Mountain spotted fever, and babesiosis, caused by Ehrlichia chaffeensis or Ehrlichia ewingii, Rickettsia rickettsii, and Babesia spp., respectively, represent a potential risk for transmission via blood transfusion in the United States (2--6). The case described in this report provides strong presumptive evidence that A. phagocytophilum infection in this patient was acquired through blood transfusion. Pretransfusion blood samples and convalescent serum from the transfusion recipient were not available for PCR or serologic testing to demonstrate conclusively that the patient was free of A. phagocytophilum infection before his hospitalization on October 12. However, the patient reported limited outdoor exposure that might include potential tick contact during the 3 weeks before hospitalization, and a blood smear collected 3 days after hospital admission showed no evidence of intracellular morulae. The timing of events and the expected incubation period for anaplasmosis (5--21 days) suggest that the patient's exposure most likely occurred during hospitalization. A. phagocytophilum DNA was found in a retained sample from the implicated RBC product that was transfused to the recipient, providing strong evidence that this was the likely route of disease transmission to the blood transfusion recipient. Some blood transfusion recipients (i.e., those who are immune compromised) likely are at increased risk for developing severe complications associated with tickborne diseases. Both A. phagocytophilum and E. chaffeensis can survive in refrigerated RBCs, and possible transfusion-transmission cases have been reported for anaplasmosis (Minnesota Department of Health, unpublished data, 1998) (2,3,5,6). However, because of the rarity of transfusion-associated cases, concerns regarding the specificity of available tests, (none of which are approved by the Food and Drug Administration), and the economic costs associated with implementation, the U.S. blood supply is not routinely screened for tickborne disease using laboratory methods (7). As a method to reduce the risk for certain pathogens in blood products, blood banks often defer donations if the potential donor is ill at the time of donation. However, persons infected with tickborne disease might experience mild illness or have asymptomatic infection, as was the case with the implicated donor in this report (1,3). Screening donors for a recent history of tick bite is unlikely to identify high-risk donors, because this type of exposure frequently is not recalled by persons with anaplasmosis (3). In this case, the implicated donor did not recall a tick bite, although she did report contact with wooded habitat in an anaplasmosis-endemic area. Nearly 75% of the other blood donors in this investigation reported similar outdoor contact, making the screening of blood donors for tick-related exposures poorly predictive for possible infection. Because Ehrlichia and Anaplasma are associated with white blood cells, leukoreduction techniques would be expected to reduce the risk for Ehrlichia and Anaplasma transfusion-transmission through RBC components (5,8). In the absence of effective screening tools to identify donors or products infected with the organisms, physicians should weigh the benefits of using leukoreduced blood components, to potentially reduce the risk for Ehrlichia and Anaplasma transmissions. Although transfusion-associated transmission of A. phagocytophilum appears to be rare, reported incidences of anaplasmosis and other tickborne diseases are increasing in the United States (1). A record 322 cases of anaplasmosis were reported in Minnesota in 2007 (6.2 cases per 100,000 population) (9). As the incidence of tickborne diseases increases, physician vigilance for possible transmission of these agents via transfusions also should increase. In addition to other more common etiologies, physicians should suspect possible rickettsial infection if transfusion recipients develop acute thrombocytopenia posttransfusion, especially if accompanied by fever. Such signs should lead to rapid assessment for rickettsial agents and empiric treatment with doxycycline (1). Although insensitive, blood smear can provide timely support for a presumptive diagnosis of anaplasmosis, followed by IFA or PCR to confirm the diagnosis (1). Similarly, babesiosis should be suspected in patients who develop hemolytic anemia and fever posttransfusion (3,4). Anaplasmosis and ehrliciosis are nationally notifiable diseases. Suspected cases of tickborne rickettsial diseases should be reported promptly to the state or local health department, and suspected transfusion-associated transmission should be reported to the supplying blood center and appropriate public health authorities. Acknowledgments The findings in this report are based, in part, on contributions by G Liu, PhD, and K Smith, DVM, PhD, Minnesota Dept of Health; M Kuehnert, MD, National Center for Preparedness, Detection, and Control of Infectious Diseases, and S Holzbauer, DVM, Coordinating Office for Terrorism Preparedness and Emergency Response, CDC. References
Table 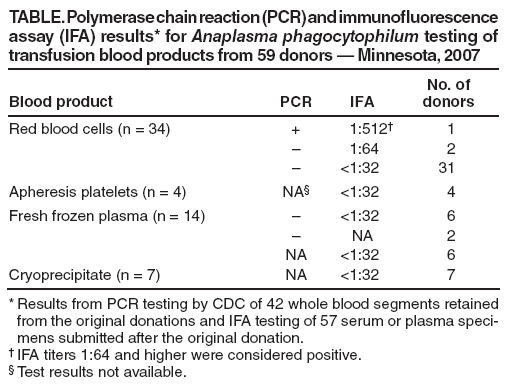 Return to top.
All MMWR HTML versions of articles are electronic conversions from typeset documents. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Date last reviewed: 10/22/2008 |
|||||||||
|