|
<<< Back to Sampling and Analytical Methods |
 Printing Instructions
Printing Instructions
|
For problems with accessibility in using figures, illustrations and PDF in
this method, please contact
the SLTC at (801) 233-4900. These procedures were designed and tested for
internal use by OSHA personnel.
Mention of any company name or commercial product does not constitute
endorsement by OSHA. |
Xylenes (o-, m-, p-isomers)
Ethylbenzene
[200
KB PDF]
Related Information: Chemical Sampling -
Xylene,
Ethyl Benzene
|
| Method number: |
1002 |
| |
| Target concentration: |
| Xylenes: |
100 ppm (435 mg/m3) |
| Ethylbenzene: |
17 ppm (73 mg/m3) |
| |
| OSHA PEL: |
| Xylenes: |
100 ppm (435 mg/m3) (TWA) |
| Ethylbenzene: |
100 ppm (435 mg/m3) (TWA) |
| |
| ACGIH TLV: |
| Xylene: |
100 ppm (TWA)
150 ppm (STEL/C) |
| Ethylbenzene: |
100 ppm (TWA)
125 ppm (STEL/C) |
| |
| Procedure: | Active samples are collected by drawing workplace air through coconut
shell charcoal sampling tubes with personal sampling pumps. Diffusive
samples are collected by exposing SKC 575-002 Passive Samplers to
workplace air. Samples are extracted with carbon disulfide and analyzed
by GC using a flame ionization detector. |
| |
| Recommended sampling time and sampling rate: |
| Charcoal tubes: |
240 min at 50 mL/min |
| SKC 575-002 Passive Samplers: |
240 min |
| |
Reliable quantitation limit
(RQL) and Standard error
of estimate (SEE): |
|
|
xylenes
|
ethylbenzene
|
|
RQL |
SEE |
RQL |
SEE |
|
(ppb) |
(µg/m3) |
(%) |
(ppb) |
(µg/m3) |
(%) |
|
charcoal tubes
SKC 575-002 Passive Samplers |
20.6
194.1 |
89.3
842.7 |
5.5
9.3* |
8.3
72.8 |
35.9
316.0 |
5.4
9.4* |
|
|
|
*For samples when sampling site pressure and temperature are known. See
Section 4.4.2 for applicable SEEs when either or both of these values are unknown. |
| |
| Special requirement: |
Sampling site temperature and barometric pressure (station pressure)
must be reported when diffusive samplers (such as SKC 575-002) are
used to sample workplace air. |
| |
| Status of method: |
Evaluated method. This method has been subjected to the established
evaluation procedures of the Methods Development Team. |
| |
| August 1999 | Warren Hendricks |
| | |
| |
Methods Development Team
Industrial Hygiene Chemistry Division
OSHA Salt Lake Technical Center
Sandy UT 84070 |
1. General Discussion
1.1 Background
1.1.1 History
Xylenes is a collective term for a mixture of m-, o-, and p- isomers of xylene. These
isomers differ only in placement of two methyl groups on a benzene ring. Technical and
commercial grades of xylenes often contain substantial amounts of ethylbenzene (10-50%), and perhaps minor amounts of other solvents as well. Mixtures of xylenes and
ethylbenzene are occasionally termed mixed xylenes.1,
2
Most occupational exposure to xylenes also results in exposure to ethylbenzene because
technical and commercial grades of xylenes are often used by industry. Therefore, test
atmospheres used in this work were prepared with a commercial source of xylenes to
simulate workplace environment. This source of xylenes contained 43% m-xylene, 20%
o-xylene, 19% p-xylene, and 15% ethylbenzene. Xylenes target concentration in test
atmospheres were approximately 100 ppm for the sum of the three isomers. These
xylenes air concentrations resulted in approximately 17 ppm ethylbenzene because of its
level in the commercial xylenes. Xylenes and ethylbenzene can be present in the
workplace in any combination and level, and this method should be satisfactory to monitor
exposures to xylenes, individual xylene isomers, and ethylbenzene. The method
recommends charcoal tubes for active sampling, and SKC 575-002 Passive Samplers for
diffusive sampling. Samples are extracted with carbon disulfide, and are analyzed by GC
using a flame ionization detector.
Determination of xylenes is well documented in the literature3,
4, and one may question why
this work was necessary. SLTC has begun to develop sampling and analytical methods
which permit the use of diffusive, as well as, active sampling. One criterion for selection
of chemicals for this evaluation program is the number of sample requests. Analysis of
xylenes is one of the most requested solvent determinations performed at SLTC.
1.1.2 Toxic effects (This section is for information only and should not be taken as the basis of OSHA policy.)
Xylenes
There is no appreciable difference in the toxicological effects of the individual xylene
isomers and those of mixed isomers. Xylenes are eye, skin, and mucous membrane
irritants. They can cause narcosis at high levels. Xylenes can cause liver and kidney
damage. There is little (if any) evidence for the carcinogenicity of xylenes in experimental
animals. The ACGIH TLV-TWA was set at 100 ppm, and the STEL at 150 ppm, for mixed
xylene isomers and for individual isomers. It was anticipated that irritant effects would be
minimal, and that neither narcosis nor chronic injury would result from exposures at these
levels.5
Ethylbenzene
Ethylbenzene is a skin and mucous membrane irritant. It has acute and possibly chronic
central nervous system effects that include vertigo, unconsciousness, tremors, and
changes in respiration. Animal experiments suggest that ethylbenzene causes damage
to the liver, kidneys, and testes. It was the opinion of the ACGIH TLV Committee that no
systemic effects would be expected at concentrations which produce skin and eye
irritation. The ACGIH TLV-TWA was set at 100 ppm and the STEL at 125 ppm to prevent
such irritation.6 ACGIH published in the 1998 TLVs and BEIs
booklet7 a "Notice of
Intended Changes" to add the A3 notation to ethylbenzene. A3 is defined as "Confirmed
Animal Carcinogen with Unknown Relevance to Humans".
1.1.3 Workplace exposure
The main source of mixed xylenes since World War II has been reformed petroleum
fractions. Earlier, xylenes were produced from coal. Coal may again become an important
source as the large coal reserves in the United States are developed for petrochemical
uses.8 U.S. production of xylenes in 1995 was 9.4 billion pounds, and that for
ethylbenzene was 13.7 billion pounds.9
Most mixed xylenes are used to blend gasoline. Mixed xylenes are also used in the paint
and coatings industry. m-Xylene is used to produce isophthalic acid, which is used in
polyesters; o-xylene is used to produce phthalic anhyride, which is used in plasticizers;
p-xylene is used to produce terephthalic acid and dimethyl terephtalate, both of which are
used to produce polyesters. o-Xylene and p-xylene are used in vitamin and
pharmaceutical synthesis, and to produce insecticides. Ethylbenzene is used to produce
styrene.10,
11
1.1.4 Physical properties (12 unless otherwise noted)
|
|
xylenes |
m-xylene |
o-xylene |
p-xylene |
ethylbenzene |
|
| CAS number13 |
1330-20-714 |
108-38-3 |
95-47-6 |
106-42-3 |
100-41-4 |
| IMIS number15 |
2590 |
|
|
|
1080 |
| molecular weight16 |
106.17 |
106.17 |
106.17 |
106.17 |
106.17 |
| boiling point (°C) |
137-145 |
138.8 |
144 |
138.5 |
136.19 |
| melting point (°C) |
|
-47.4 |
-25 |
13.2 |
-95.0117 |
| density (°C) |
about 0.86 |
0.868 (15) |
0.880 (20/4) |
0.861 (20) |
0.867 (20) |
| molecular formula |
C8H10 |
C8H10 |
C8H10 |
C8H10 |
C8H10 |
| flash point (°F) |
81-115 |
85 |
115 |
81 |
59 |
vapor pressure
(kPa, (°C))18,
19 |
|
1.1 (25) |
0.9 (25) |
1.2 (25) |
0.9 (20) |
|
Xylenes (dimethylbenzene, xylol) are soluble in alcohol and ether, but insoluble in water.
Each of the mixed xylenes is a clear, colorless liquid at room temperature, however, p-xylene forms crystals at a relatively high temperature. The xylene isomers: m-xylene (1,3-dimethylbenzene), o-xylene (1,2-dimethylbenzene), and p-xylene (1,4-dimethylbenzene)
are soluble in alcohol and ether; but they are insoluble in water. Ethylbenzene
(phenylethane) is soluble in alcohol, benzene, carbon tetrachloride, and ether; it is but
almost insoluble in water.20
Structural formulas:

m-xylene |
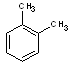
o-xylene |
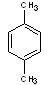
p-xylene |
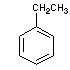
ethyl benzene |
This method was evaluated according to OSHA SLTC "Evaluation Guidelines for Air Sampling Methods
Utilizing Chromatographic Analysis21. The Guidelines define analytical parameters and specify required
laboratory tests, statistical calculations and acceptance criteria. The analyte concentrations throughout this
method are based on the recommended sampling and analytical parameters. Air concentrations in ppm and
ppb are referenced to 25 °C and 101.3 kPa (760 mmHg).
1.2 Limit defining parameters
1.2.1 Detection limit of the analytical procedure
The detection limits of the analytical procedure (DLAP) are shown in Table 1.2.1. These
are the amounts of analyte that will give detector responses significantly different from
the response of a reagent blank. (Section 4.1)
|
Table 1.2.1
DLAP (pg per sample) |
|
| xylenes | m-xylene | o-xylene | p-xylene | ethylbenzene |
|
| 14.5 | 2.1 | 8.4 | 14.0 | 5.7 |
|
|
1.2.2 Detection limit of the overall procedure
Charcoal tubes
The detection limits of the overall procedure (DLOP) are shown in Table 1.2.2.1. These are
the amounts of analyte spiked on the samplers that will give detector responses significantly
different from the response of a sampler blank. (Section 4.2)
|
Table 1.2.2.1
DLOP for Charcoal Tubes |
|
| analyte | ng | ppb | µg/m3 |
|
xylenes
m-xylene
o-xylene
p-xylene
ethylbenzene |
322
159
239
100
129 |
6.2
3.0
4.6
1.9
2.5 |
26.8
13.2
19.9
8.3
10.8 |
|
|
SKC 575-002 Passive Samplers
The detection limits of the overall procedure (DLOP) are shown in Table 1.2.2.2. These are
the amounts of analyte spiked on the samplers that will give detector responses significantly
different from the response of a sampler blank. (Section 4.2)
|
Table 1.2.2.2
DLOP for SKC 575-002
Passive Samplers |
|
| analyte | ng | ppb | µg/m3 |
|
xylenes
m-xylene
o-xylene
p-xylene
ethylbenzene |
847
448
325
437
315 |
58.2
31.1
21.9
30.1
21.9 |
252.8
134.9
95.0
130.8
94.9 |
|
|
1.2.3 Reliable quantitation limit
Charcoal tubes
The reliable quantitation limits (RQL) are shown in Table 1.2.3.1. These are the amounts of
analyte that will give detector responses that are considered the lower limits for precise
quantitative measurements. (Section 4.2)
|
Table 1.2.3.1
RQL for Charcoal Tubes |
|
| analyte | ng | ppb | µg/m3 |
|
xylenes
m-xylene
o-xylene
p-xylene
ethylbenzene |
1072
531
795
334
431 |
20.6
10.2
15.3
6.4
8.3 |
89.3
44.2
66.2
27.8
35.9 |
|
|
SKC 575-002 Passive Samplers
The reliable quantitation limits (RQL) are shown in Table 1.2.3.2. These are the
amounts of analyte that will give detector responses that are considered the lower limits
for precise quantitative measurements. (Section 4.2)
|
Table 1.2.3.2
RQL for SKC 575-002 Passive Samplers |
|
| analyte | ng |
ppb | µg/m3 |
|
xylenes
m-xylene
o-xylene
p-xylene
ethylbenzene |
2823
1495
1084
1456
1049 |
194.1
103.7
73.0
100.4
72.8 |
842.7
450.3
317.0
435.9
316.0 |
|
|
1.2.4 Instrument calibration
The coefficients of determination (r2) and of nondetermination
(k2) for the calibration curves are shown in Table 1.2.4. The
calibrated range was 0.5 to 2 times the OSHA PEL. (Section 4.3)
|
Table 1.2.4
Coefficients of Determination (r2)
and Nondetermination (k2) |
|
| analyte | charcoal tubes | SKC 575-002
Pass Samplers |
| r2 | k2 | r2 | k2 |
|
| m-xylene |
0.9994 |
6×10-4 |
0.9998 |
2×10-4 |
| o-xylene |
0.9995 |
5×10-4 |
0.9998 |
2×10-4 |
| p-xylene |
0.9994 |
6×10-4 |
0.9998 |
2×10-4 |
| ethylbenzene |
0.9994 |
6×10-4 |
0.9998 |
2×10-4 |
|
|
1.2.5 Precision (overall procedure)
Charcoal tubes
The precision of the overall procedure at the
95% confidence interval for the ambient
temperature 16-day storage test (at the target
concentration) are shown in Table 1.2.5.1.
Each precision includes an additional 5% for
sampling pump variability. (Section 4.4)
|
Table 1.2.5.1
Precision of the Overall Procedure for Charcoal Tubes |
|
| analyte | precision (±%) |
|
xylenes
m-xylene
o-xylene
p-xylene
ethylbenzene |
10.8
10.7
11.0
11.1
10.6 |
|
|
SKC 575-002 Passive Samplers
The precision of the overall procedure at the 95% confidence interval for the ambient
temperature 16-day storage tests (at the target concentration) are given in Table 1.2.5.2.
They each include an additional 8.7% for sampling rate variability. There are different
values given, depending on whether both, either, or neither temperature or atmospheric
pressure are known at the sampling site. If the sampling-site temperature (T) is unknown,
it is assumed to be 22.2 ± 15 °C (72 ± 27 °F) and a variability of ±7.7% is included. If the
atmospheric pressure (P) is unknown, it is estimated from sampling-site elevation and a
variability of ±3% is included. (Section 4.4)
Table 1.2.5.2
Precision of the Overall Procedure for SKC 575-002 Passive Samplers (±%)
|
| condition |
xylenes |
m-xylene |
o-xylene |
p-xylene |
ethylbenzene |
|
both T and P known
only T known
only P known
neither T nor P known |
18.2
19.1
23.7
24.4 |
18.3
19.2
23.7
24.4 |
18.1
19.0
23.6
24.3 |
18.3
19.2
23.7
24.4 |
18.4
19.3
23.8
24.5 |
|
1.2.6 Recovery
The recoveries from samples used in 16-day ambient temperature storage
tests remained above those shown in Table 1.2.6. (Section 4.5)
|
Table 1.2.6
Recovery (%) |
|
| analyte | charcoal
tubes | SKC 575-002
Passive Samlers |
|
xylenes
m-xylene
o-xylene
p-xylene
ethylbenzene |
99.1
99.6
98.5
98.5
100.2 |
97.7
98.2
96.6
97.8
99.5 |
|
|
1.2.7 Reproducibility
Twelve samples (six active and six diffusive) collected from test atmospheres were submitted
for analysis by SLTC. The samples were analyzed according to instructions presented in a
draft copy of this method after 16 and 22 days of storage at ambient temperature for the
active and diffusive samplers, respectively. No individual result deviated from its
theoretical value by more than the precision reported in Section 1.2.5. (Section 4.6)
2. Sampling procedure
All safety practices that apply to the work area being sampled should be followed. The sampling
equipment should be attached to the worker in such a manner that it will not interfere with work
performance or safety.
2.1 Apparatus
2.1.1 Charcoal tubes
Samples are collected with a personal sampling pump calibrated, with the sampler
attached, to within ±5% at 50 mL/min.
Samples are collected with 7-cm × 4-mm i.d. × 6-mm o.d. flame sealed glass sampling
tubes containing two sections of coconut shell charcoal. The front section contains 100
mg and the back section contains 50 mg of charcoal. The sections are held in place and
separated with glass wool and polyurethane plugs. Commercially prepared sampling
tubes were purchased from SKC for this evaluation (SKC Catalog no. 226-01, Lot 2000).
2.1.2 SKC 575-002 Passive Samplers
Samples are collected with SKC 575-002 Passive Samplers. These samplers contain
500 mg of Anasorb 747. Lot numbers 347, 764, and 872 were used in this evaluation.
A thermometer and a barometer are needed to determine sampling site temperature and pressure.
2.2 Reagents
None required.
2.3 Technique
2.3.1 Charcoal tubes
Immediately before sampling, break off both ends of the flame sealed sampling tube to
provide openings approximately half the internal diameter of the tube. Wear eye protection
when breaking tubes. Use sampling tube holders to shield the employee from the sharp,
jagged ends of the sampling tubes. All sampling tubes should be from the same lot.
Use the smaller charcoal section of the sampling tube as a back-up and position it nearest
the sampling pump. Attach the sampling tube to the sampling pump so that the tube is
in an approximately vertical position with the inlet down during sampling. Position the
sampling tube so that it does not impede work performance or safety.
Draw air to be sampled directly into the tube inlet. Sampled air is not to pass through any
hose or tubing before entering the sampling tube.
Remove the sampler and seal the tube with plastic end caps after sampling for the appropriate time. Seal
each sample end-to-end with an OSHA-21 form as soon as possible.
Submit at least one blank sample with each set of samples. Handle the blank sampler
in the same manner as the other samples, except draw no air through it.
Record sample air volume in liters for each sample, and record any potential interference.
Submit the samples to the laboratory for analysis as soon as possible after sampling.
Store the samples at reduced temperature if delay is unavoidable. Ship any bulk samples
separate from air samples.
2.3.2 SKC 575-002 Passive Samplers (In general, follow the manufacturer's instructions)
Remove the sampler from the clear package just before sampling. CAUTION - The
monitor begins to sample as soon as it is removed from this package. Retain the O-ring,
press-on cover, cover retainer, port plugs, and PTFE tube for later use.
Record the start time on the sampler label, or on the Form OSHA-91A.
Attach the sampler near the worker's breathing zone with the perforations in the sampler
facing out. Assure that the area directly in front of the sampler is unobstructed throughout
the sampling period.
Remove the sampler from the worker immediately at the end of the sampling period.
Attach the cover with the O-ring in place onto the sampler using the cover retainer.
Inspect the O-ring to be sure it is forming a good seal around the entire circumference of
the sampler. Record the stop time on the sampler label, or on the Form OSHA-91A.
Prepare a blank sample by removing it from its clear package, and then immediately
attaching a cover with the O-ring in place onto it.
Seal each sample with an OSHA-21 form.
Verify that sampling times are properly recorded on Form OSHA-91A for each sample.
Identify blank samples on this form.
Record sampling site temperature and atmospheric pressure (station pressure) on Form OSHA-91A.
List any chemicals that could be considered potential interferences, especially solvents,
that are in use in the sampling area.
Submit the samples to the laboratory for analysis as soon as possible. Store the samples
in a refrigerator if delay is unavoidable. Include the port plugs and PTFE tubes which will
be used in the laboratory analysis.
Ship any bulk samples separate from air samples.
2.4 Sampler capacity
2.4.1 Charcoal tubes
The sampling capacity of SKC Lot 2000 charcoal tubes was tested by sampling from a dynamically
generated test atmosphere of mixed xylenes (1027 mg/m3 or 237 ppm).
Samples were collected at 50 mL/min and the relative humidity was about 78% at 21 °C.
No breakthrough from the front to the back section was observed, even after sampling for ten hours.
The 5% breakthrough sampling time was determined to be in excess of 600 min. (Section 4.7.1)
2.4.2 SKC 575-002 Passive Samplers
The sampling rate and capacity of SKC 575-002 Passive Samplers were determined by
sampling from dynamically generated test atmospheres of mixed xylenes (1027 mg/m3
or 237 ppm, at 78% relative humidity and 21 °C) for increasing time intervals. Sampling
rates of 13.82 mL/min for m-xylene, 14.24 mL/min for o-xylene, 13.94 mL/min
for p-xylene, and 13.83 mL/min for ethylbenzene at 760 mmHg and 25 °C were obtained from
these tests. Sampler capacity was never exceeded, even after sampling for ten hours. (Section 4.7.2)
2.5 Extraction efficiency
It is the responsibility of the analytical laboratory to occasionally determine or confirm extraction
efficiency because the adsorbent material, reagents, or technique may be different from those
presented in this method.
2.5.1 Charcoal tubes
Mean extraction efficiencies (EE) of the analytes from SKC Lot 2000 charcoal are
presented in Table 2.5.1. The range studied was from the RQL to 2 times the 100 ppm OSHA
PEL for each xylene isomer, and for ethylbenzene. The extraction efficiency was not
affected by the presence of water. (Section 4.8.1)
Table 2.5.1
Extraction Efficiency from Charcoal (%)
|
| m-xylene | o-xylene | p-xylene | ethylbenzene |
|
| 96.3 | 93.8 | 96.1 | 97.2 |
|
2.5.2 SKC 575-002 Passive Samplers
Mean extraction efficiencies (EE) of the analytes from SKC Anasorb 747 (the adsorbent in
SKC 575-002 Passive Samplers) are presented in Table 2.5.2. The range studied
was from the RQL to 2 times the 100 ppm OSHA PEL for each xylene isomer, and for ethylbenzene.
The extraction efficiency was not affected by the presence of water. (Section 4.8.2)
Table 2.5.2
Extraction Efficiency form Anasorb 747 (%)
|
| m-xylene | o-xylene | p-xylene | ethylbenzene |
|
| 96.1 | 89.4 | 95.3 | 99.1 |
|
2.6 Recommended sampling time and sampling rate
2.6.1 Charcoal tubes
Sample for up to 4 hours at 50 mL/min when using SKC 226-01 charcoal tubes to collect
long-term samples. Sample for more than 5 min at 50 mL/min to collect short-term samples.
2.6.2 SKC 575-002 Passive Samplers
|
Sample for up to 4 hours when using SKC 575-002 Passive Samplers to collect
long-term samples. Sample for more than 5 min to collect short-term samples. |
Table 2.6.2
Sampling Rates for SKC 575-002 Passive
Samplers (mL/min) at 760 mmHg and 25 °C |
|
| m-xylene | o-xylene | p-xylene | ethylbenzene |
|
| 13.82 | 14.24 | 13.94 | 13.83 |
|
|
2.6.3 The air concentration equivalent to the reliable quantitation limit becomes larger when
short-term samples are collected. For example, the reliable quantitation limit for xylenes
is 733 ppb (3180 µg/m3) when 0.25 L of air is sampled using charcoal tubes.
2.7 Sampling interferences (Section 4.9)
2.7.1 Charcoal tubes
Retention
The ability of the sampler to retain the analytes following collection was tested. The
retention efficiency of all analytes for all samples was above 100.8% when three charcoal
tubes containing 3 mg of mixed xylenes were used to sample 9 L of contaminant-free air
having a relative humidity of 80% at 20 °C.
Low relative humidity
The ability of the sampler to collect and retain the analytes at low relative humidity was
tested. The collection efficiency of all analytes for all samples was above 99.2% when
three charcoal tubes were used to sample 12 L of air containing two times the target
concentration of mixed xylenes and having a relative humidity of 5% at 20 °C.
Low concentration
The ability of the sampler to collect and retain the analytes at low concentration was
tested. The collection efficiency of all analytes for all samples was above 94.6% when
three charcoal tubes were used to sample 12 L of air containing 0.1 times the target
concentration of mixed xylenes and having a relative humidity of 80% at 22 °C.
Interference
The ability of the sampler to collect and retain the analytes in the presence of potential
sampling interferences was tested. The collection efficiency of all analytes for all samples
was above 101.2% when three charcoal tubes were used to sample 12 L of air containing
one times the target concentration of mixed xylenes, 365 mg/m3 toluene, 372 mg/m3 butyl
acetate, and a relative humidity of 81% at 21 °C.
2.7.2 SKC 575-002 Passive Samplers
Reverse diffusion
The sampling method was tested for reverse diffusion. The retention efficiency of all
analytes for all samples was above 99.6% when three SKC 575-002 Passive Samplers
containing 0.9 mg of mixed xylenes were used to sample contaminant-free air having a
relative humidity of 80% at 20 °C for three hours.
Low relative humidity
The sampling method was tested to determine if the sampling rates remained constant
at low relative humidity. The sampling rate of all analytes for all samples was above
97.8% of the sampling rate reported in Section 2.6.2 when three SKC 575-002 Passive
Samplers were used to sample air containing two times the target concentration of mixed
xylenes and having a relative humidity of 5% at 20 °C for four hours. Low humidity did not
affect the sampling rates.
Low concentration
The sampling method was tested to determine if the sampling rates remained constant
at low concentration. The sampling rate of all analytes for all samples was above 95.0%
of the sampling rate reported in Section 2.6.2 when three SKC 575-002 Passive Samplers
were used to sample air containing 0.1 times the target concentration of mixed xylenes
and having a relative humidity of 80% at 22 °C for four hours.
Interference
The sampling method was tested to determine if the sampling rates remained constant
in the presence of sampling interferences. The sampling rate for all analytes for all
samples was above 94.3% of the sampling rate reported in Section 2.6.2 when three SKC
575-002 Passive Samplers were used to sample air containing one times the target
concentration of mixed xylenes, 365 mg/m3 toluene, 372 mg/m3 butyl acetate, and a
relative humidity of 81% at 21 °C for four hours.
3. Analytical procedure
Adhere to the rules set down in your Chemical Hygiene Plan (which is mandated by the OSHA
Laboratory Standard). Avoid skin contact and inhalation of all chemicals.
3.1 Apparatus
3.1.1 A GC equipped with a flame ionization (FID) detector. A Hewlett-Packard Model 5890
Series II GC equipped with a ChemStation, an automatic sample injector, and an FID
were used in this evaluation.
3.1.2 A GC column capable of separating mixed xylenes from the extraction solvent, internal
standards, and potential interferences. A J&W Scientific 60-m ×
0.32-mm i.d. DB-Wax (0.5 µm df) capillary column was used in this evaluation.
3.1.3 An electronic integrator or other suitable means of measuring GC detector response. A
Waters Millennium Chromatography Manager system was used in this evaluation.
3.1.4 Two and four-milliliter glass vials with PTFE-lined septum caps.
3.1.5 One and two-milliliter volumetric pipets.
3.1.6 A SKC Desorption Shaker with rack (226D-03K) was used to extract SKC 575-002 Passive Samplers in this evaluation.
3.2 Reagents
3.2.1 Xylenes, Isomers plus ethylbenzene, 98.5+%, A.C.S. reagent, Aldrich Chemical Co., Lot TR 02505LR, was used in this evaluation.
3.2.2 m-xylene, 99+%, anhydrous, Aldrich Chemical Co., Lot 00249MQ, was used in this evaluation.
3.2.3 o-Xylene, 98%, Spectrophotometric Grade, Aldrich Chemical Co., Lot 07946PN, was used in this evaluation.
3.2.4 p-Xylene, 99+%, anhydrous, Aldrich Chemical Co., Lot TQ 25949MQ, was used in this evaluation.
3.2.5 Ethylbenzene, 99.8%, anhydrous, Aldrich Chemical Co., Lot DR 03249JQ, was used in this evaluation.
3.2.6 Carbon disulfide (CS2), 99.9+%, low benzene content, Aldrich Chemical Co., Lot 07546PN, was used in this evaluation.
3.2.7 1-Phenylhexane (hexylbenzene), 97%, Aldrich Chemical Co., Lot 03006PZ, was used as
an internal standard for SKC 575-002 Passive Samplers in this evaluation.
3.2.8 p-Cymene, 99%, Aldrich Chemical Co., Lot 11703TR, was used as an internal standard
for charcoal tube samples in this evaluation.
3.2.9 The extraction solvent used for this evaluation consisted of 1 µL of the appropriate internal
standard per milliter of CS2. CAUTION: extraction efficiency of the internal standard from
the sampling medium has an effect on sample results. This effect is especially significant
for SKC 575-002 Passive Samplers. Do not substitute internal standards unless
extraction efficiencies are confirmed. Both internal standards can be present in the same
extraction solvent if the appropriate internal standard is used to calibrate the GC, and to
calculate sample results.
3.2.10 GC grade nitrogen, air, and hydrogen were used in this evaluation.
3.3 Standard preparation
3.3.1 Prepare stock mixed standards by weighing 1-mL aliquots of all four analytes into the
same container. For example: a neat mixed standard was prepared that contained 212.7 mg/mL
of m-xylene, 212.6 mg/mL of o-xylene, 213.4 mg/mL of p-xylene,
and 215.3 mg/mL of ethylbenzene.
3.3.2 Prepare working range standards for the analysis of charcoal tubes by injecting microliter
quantities of the stock mixed standard into 1-mL aliquots of extraction solvent (containing
1 µL p-cymene internal standard per milliter of CS2). For example, a working range
standard was prepared by injecting 6.0 µL of the stock mixed standard into extraction
solvent. This standard contained 1276 µg/mL of m-xylene, 1276 µg/mL of o-xylene, 1280
µg/mL of p-xylene, and 1292 µg/mL of ethylbenzene.
3.3.3 Prepare working range standards for the analysis of SKC 575-002 Passive Samplers by
first diluting the stock mixed standard (Section 3.4.1) 1 to 4 with CS2, and then injecting
microliter quantities of the diluted stock mixed standard into 2-mL aliquots of extraction
solvent (containing 1 µL 1-phenylhexane internal standard per milliter of CS2). For
example, a working range standard was prepared by injecting 6.0 µL of the diluted stock
mixed standard into extraction solvent. This standard contained 319.0 µg/mL of m-xylene,
318.9 µg/mL of o-xylene, 320.1 µg/mL of p-xylene, and 323.0 µg/mL of ethylbenzene.
3.3.4 Prepare a sufficient number of standards so that sample results will likely be bracketed
with standards. If sample results are outside the range of prepared standards, prepare
and analyze additional standards, or dilute high samples with extraction solvent and then
reanalyze the diluted samples.
3.4 Sample preparation
3.4.1 Charcoal tubes
Remove the plastic end caps from the sampling tube and carefully transfer each section
of the adsorbent into separate 2-mL glass vials. Check to be certain that no charcoal is
trapped in the glass-wool plug. Discard the end caps, glass tube, glass wool plug, and foam plugs.
Add 1.0 mL of extraction solvent to each vial and immediately seal each vial with a PTFE-lined septum cap.
Shake the vials vigorously several times during the one-hour extraction time.
3.4.2 SKC 575-002 Passive Samplers
Cut off the ends of the two protruding tubes of each sampler with a razor blade or a sharp knife.
Secure the sampler by clipping it to a rail of the detachable SKC Desorption Shaker rack.
Carefully and slowly add 2.0 mL of extraction solvent through the protruding tube nearest
the outside edge of the sampler using a volumetric pipet. The tip of the pipet should fit
just inside the sampler tube. Immediately seal the sampler tubes with the plugs supplied
by the manufacturer.
Replace the rack onto the SKC Desorption Shaker and shake the samples for one hour.
Do not allow the extracted sample to remain in the sampler. Transfer the extracted
sample into 2-mL glass vials by removing the plugs from the protruding tubes, inserting
the tapered end of the PTFE tube supplied by the manufacture into the protruding tube
nearest the outside edge of the sampler, and carefully pouring the solution into a 2-mL
glass vial. Immediately seal the vials with PTFE-lined septum caps.
3.5 Analysis
3.5.1 GC conditions
| zone temperatures: |
column |
40 °C, hold 1 min, program at 4 °C/min to 140 °C, and hold as necessary to clear column |
| injector | 220 °C |
| detector | 220 °C |
| gas flows: | hydrogen (carrier) | 4.4 mL/min (115 kPa head pressure) |
| nitrogen (makeup) | 38 mL/min |
| hydrogen (FID) | 35 mL/min |
| air (FID) | 455 mL/min |
| signal range: | | 3 |
| injection volume: | | 1 µL (20:1 split) |
| column: | | 60 m × fused silica 0.32-mm i.d. DB Wax 0.5-µm df |

Figure 3.5.1 Chromatogram of a sample desorbed from charcoal. Concentration is approximately
the 100-ppm PEL for each analyte. Key: (1) CS2, (2) ethylbenzene, (3) p-xylene,
(4) m-xylene, (5) o-xylene, (6) p-cymene, (7) 1-phenylhexane. |
3.5.2 Measure peak areas with an electronic integrator or other suitable means.
3.5.3 An internal standard (ISTD) method is used to calibrate the instrument in terms of
micrograms of analyte per sample. Prepare a calibration curve by analyzing standards,
and constructing calibration curves by plotting ISTD-corrected detector response versus
mass of analyte. Bracket sample results with standards.
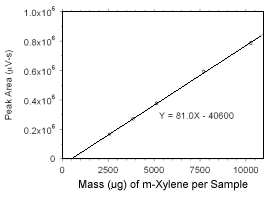
Figure 3.5.3.1 Calibration curve for m-xylene standards used to analyze charcoal tubes constrcted from the data in Table 4.3.1. |
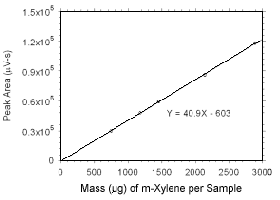
Figure 3.5.3.2 Calibration curve for m-xylene standards used to analyze SKC 575-002 Passive Samplers constructed from the data in Table 4.3.5. |
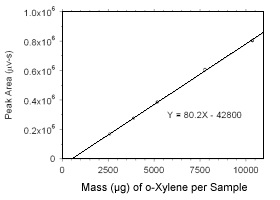
Figure 3.5.3.3 Calibration curve for o-xylene standards used to analyze charcoal tubes constructed from the data in Table 4.3.2. |
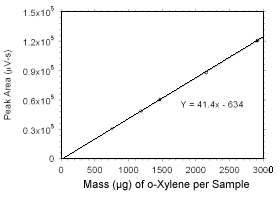
Figure 3.5.3.4 Calibration curve for o-xylene standards used to analyze SKC 575-002 Passive Samplers constructed from the data in Table 4.3.6. |

Figure 3.5.3.5 Calibration curve for p-xylene standards used to analyze charcoal tubes constructed from the data in Table 4.3.3. |
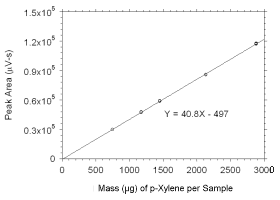
Figure 3.5.3.6 Calibration curve for p-xylene standards used to analyze SKC 575-002 Passive Samplers constructed from the data in Table 4.3.7. |
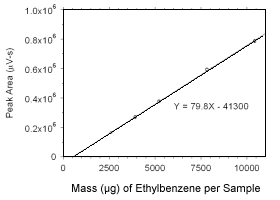
Figure 3.5.3.7 Calibration curve for ethylbenzene standards used to analyze charcoal tubes constructed from the data in Table 4.3.4. |
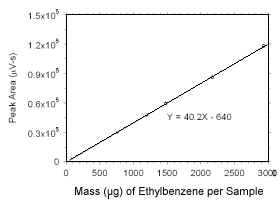
Figure 3.5.3.8 Calibration curve for ethylbenzene standards used to analyze SKC 575-002 Passive Samplers constructed from the data in Table 4.3.8. |
3.6 Interferences (analytical)
3.6.1 Any chemical that produces an FID response and has a similar retention time as any of the
analytes or internal standard is a potential interference. Any reported potential
interferences should be considered before samples are extracted. Generally,
chromatographic conditions can be altered to separate an interference from an analyte or
an internal standard.
3.6.2 The identity or purity of an analyte peak can be confirmed with additional analytical data. (Section 4.10)
3.7 Calculations
3.7.1 Charcoal tubes
Obtain separate amounts of each analyte (m-xylene, o-xylene, p-xylene, ethylbenzene) per
sample from the appropriate calibration curve in terms of micrograms per sample. These
amounts are uncorrected for extraction efficiency. Be certain that the correct internal
standard was used to calculate results (See Section 3.2.9). The back section of the
sampling tube is analyzed primarily to determine the extent of sampler saturation. If any
analyte is found on the back section, it is added to the amount found on the front section.
This amount is then corrected by subtracting the total amount (if any) found on the blank.
The air concentrations are then calculated using the following formulas. Calculate xylenes
exposure by summing the individual xylene isomer results.
| mg/m3 = |
micrograms of analyte per sample
liters of air sampled × extraction efficiency
|
| |
ppm = |
24.46 × mg/m3
106.17
|
| Where |
24.46 is the molar volume at 25 °C and 101.3 kPa (760 mmHg)
106.17 is the molecular weight of m-xylene, o-xylene, p-xylene, and ethylbenzene |
3.7.2 SKC 575-002 Passive Samplers
Obtain separate amounts of each analyte (m-xylene, o-xylene, p-xylene, ethylbenzene) per
sample from the appropriate calibration curve in terms of micrograms per sample. These
amounts are uncorrected for extraction efficiency. Be certain that the correct internal
standard was used to calculate results. This amount is then corrected by subtracting the
amount (if any) found on the blank.
Table 3.7.2
Sampling Rates for SKC 573-002 Passive
Samplers (mL/min) at 760 mmHg and 25 °C
|
| m-xylene | o-xylene | p-xylene | ethylbenzene |
|
| 13.82 | 14.24 | 13.94 | 13.83 |
|
Sampling time, sampling site temperature (°C), and sampling site pressure (mmHg) is
information given by the person submitting the samples. Sampling rates at 760 mmHg and
25 °C (SRNTP) are given in Table 3.7.2. These
sampling rates must be converted to their equivalent (SRamb) at
sampling site temperature (T) and sampling site pressure (P) by the following formula:
Assume sampling site temperature is 22.2 °C if it is not given. If sampling site pressure
is not given, it can be calculated by the following formula:
| P = ( 3.887 × 10-7 ) E2 - ( 2.7467 × 10-2 ) E + 760 |
Where
P is the approximate sampling site barometric pressure. E is the sampling site elevation.
E can be estimated from airports near the sampling site location using a web site such as
AirNav.com
Liters of air sampled is calculated by multiplying the appropriate SRamb by sampling time
and dividing that result by 1000.
Air concentrations are then calculated using the following formulas. Calculate xylenes
exposure by summing the individual xylene isomer results.
| mg/m3 = |
micrograms of analyte per sample
liters of air sampled × extraction efficiency
|
|
ppm = |
24.46 × mg/m3
106.17
|
| Where |
24.46 is the molar volume at 25 °C and 101.3 kPa (760 mmHg)
106.17 is the molecular weight of m-xylene, o-xylene, p-xylene, and ethylbenzene |
4. Backup Data
General background information about determination of detection limits and precision of the overall
procedure is found in OSHA SLTC "Evaluation Guidelines for Air Sampling Methods Utilizing
Chromatographic Analysis"22. The Guidelines
define analytical parameters and specify required laboratory tests, statistical calculations
and acceptance criteria.
4.1 Detection limit of the analytical procedure (DLAP)
The DLAP is measured as the mass of analyte introduced into the chromatographic column.
Ten standards were prepared in equal descending increments of analyte, such that the highest
standard produced a peak approximately 10 times the response of a reagent blank. These
standards, and a reagent blank, were analyzed using the recommended analytical parameters
(1-µL injection with a 20:1 split), and the data obtained were used to
determine the required parameters (A and SEEDL) for the
calculation of the DLAP. Xylenes DLOP was calculated by summing masses and areas for
individual xylene isomers. The extraction solvent contained a contaminant that eluted at
the same time as p-xylene. The amount of this contaminant was small, but sufficient
to cause a higher DLAP for p-xylene than for the other analytes.
Table 4.1
Detection Limit of the
Analytical Procedure
|
| analyte | DLAP (pg) |
|
xylenes
m-xylene
o-xylene
p-xylene
ethylbenzene |
14.5
2.1
8.4
14.0
5.7 |
|
Table 4.1.1
DLAP for Xylenes |
|
concn
(ng/mL) | mass on
column (pg) | area counts
(µV-s) |
|
0
512
1026
1538
2052
2564
3076
3590
4102
4616
5128 |
0
25.6
51.3
76.9
102.6
128.2
153.8
179.5
205.1
230.8
256.4 |
150
196
284
369
395
482
566
636
697
769
853 |
|
|
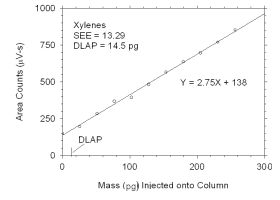
Figure 4.1.1 Plot of data used to determine DLAP for xylenes.
|
| |
Table 4.1.2
DLAP for m-Xylene |
|
concn
(ng/mL) | mass on
column (pg) | area counts
(µV-s) |
|
0
170
342
512
684
854
1024
1196
1366
1538
1708 |
0
8.5
17.1
25.6
34.2
42.7
51.2
59.8
68.3
76.9
85.4 |
8
32
52
80
102
123
145
168
196
215
240 |
|
|
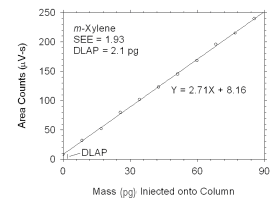
Figure 4.1.2 Plot of data used to determine DLAP for m-xylene.
|
| |
Table 4.1.3
DLAP for o-Xylene |
|
concn
(ng/mL) | mass on
column (pg) | area counts
(µV-s) |
|
0
172
342
514
684
856
1028
1198
1370
1540
1712 |
0
8.6
17.1
25.7
34.2
42.8
51.4
59.9
68.5
77.0
85.6 |
0
21
37
81
85
120
143
171
174
231
235 |
|
|
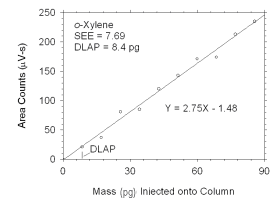
Figure 4.1.3 Plot of data used to determine DLAP for o-xylene.
|
| |
Table 4.1.4
DLAP for p-Xylene |
|
concn
(ng/mL) | mass on
column (pg) | area counts
(µV-s) |
|
0
170
342
512
684
854
1024
1196
1366
1538
1708 |
0
8.5
17.1
25.6
34.2
42.7
51.2
59.8
68.3
76.9
85.4 |
142
143
195
208
208
239
278
297
327
323
368 |
|
|
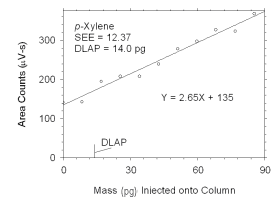
Figure 4.1.4 Plot of data used to determine DLAP for p-xylene.
|
| |
Table 4.1.5
DLAP for Ethylbenzene |
|
concn
(ng/mL) | mass on
column (pg) | area counts
(µV-s) |
|
0
174
346
520
692
866
1040
1212
1386
1558
1732 |
0
8.7
17.3
26.0
34.6
43.3
52.0
60.6
69.3
77.9
86.6 |
0
26
38
66
92
120
146
165
177
206
233 |
|
|
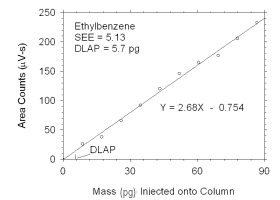
Figure 4.1.5 Plot of data used to determine DLAP for ethylbenzene.
|
4.2 Detection limit of the overall procedure (DLOP) and reliable quantitation limit (RQL)
The DLOP is measured as mass per sample and expressed as equivalent air concentrations,
based on the recommended sampling parameters. Ten 100-mg
portions of SKC Lot 2000 charcoal, and ten 500-mg portions of SKC Anasorb
747, (representing SKC 575-002 Passive Samplers) were spiked with equal
descending increments of analyte, such that the highest sampler loading would produce a
peak approximately 10 times the response for a sample blank. These spiked samples, and
sample blanks were analyzed with the recommended analytical parameters, and the data obtained
used to calculate the required parameters (A and SEEDL) for the calculation of DLOP and RQL.
Xylenes DLOP and RQL were calculated by summing masses and areas for individual xylene
isomers. Sample air volume and extraction efficiency for xylenes is the mean of those for individual
xylene isomers. Table 4.2 is a summary of DLOP results and is presented for quick reference.
Table 4.2
Detection Limit of the Overall Procedure Summary
|
| analyte | charcoal
tubes | SKC 575-002
Passive Samplers |
| ng | µg/m3 | ppb | ng | µg/m3 | ppb |
|
xylenes
m-xylene
o-xylene
p-xylene
ethylbenzene |
322
159
239
100
129 |
26.8
13.2
19.9
8.3
10.8 |
6.2
3.0
4.6
1.9
2.5 |
847
448
325
437
315 |
252.8
134.9
95.0
130.8
94.9 |
58.2
31.1
21.9
30.1
21.9 |
|
Table 4.2.1
DLOP and RQL
for Xylenes from Charcoal Tubes |
|
mass per sample
(ng) | area counts
(µV-s) |
|
0
513
1025
1538
2051
2563
3076
3590
4101
4614
5128 |
139
238
272
357
423
485
524
624
691
763
835 |
|
|
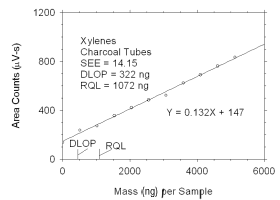
Figure 4.2.1 Plot of data used to determine DLOP and RQL for xylenes.
|
| |
Table 4.2.2
DLOP and RQL for Xylenes
from SKC 575-002 Passive Samplers |
|
mass per sample
(ng) | area counts
(µV-s) |
|
0
1025
2051
3076
4101
5128
6152
7179
8204
9228
10255 |
136
247
278
334
452
497
588
615
696
769
844 |
|
|
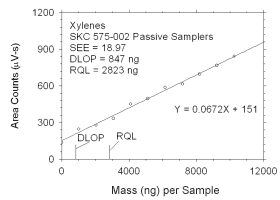
Figure 4.2.2 Plot of data used to determine DLOP and RQL for xylenes.
|
| |
Table 4.2.3
DLOP and RQL
for m-Xylene from Charcoal Tubes |
|
mass per sample
(ng) | area counts
(µV-s) |
|
0
171
341
512
683
853
1024
1195
1365
1536
1707 |
0
39
54
74
109
116
134
168
200
215
239 |
|
|
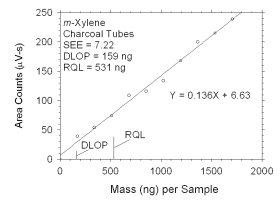
Figure 4.2.3 Plot of data used to determine DLOP and RQL for m-xylene.
|
| |
Table 4.2.4
DLOP and RQL for m-Xylene
from SKC 575-002 Passive Samplers |
|
mass per sample
(ng) | area counts
(µV-s) |
|
0
341
683
1024
1365
1707
2048
2390
2731
3072
3414 |
0
46
56
61
110
116
158
175
194
226
237 |
|
|
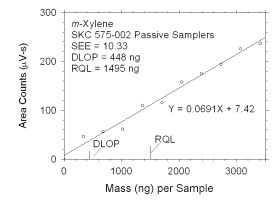
Figure 4.2.4 Plot of data used to determine DLOP and RQL for m-xylene.
|
| |
Table 4.2.5
DLOP and RQL
for o-Xylene from Charcoal Tubes |
|
mass per sample
(ng) | area counts
(µV-s) |
|
0
171
343
514
685
857
1028
1200
1371
1542
1714 |
0
31
39
70
85
121
115
161
168
210
239 |
|
|
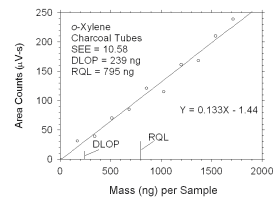
Figure 4.2.5 Plot of data used to determine DLOP and RQL for o-xylene.
|
| |
Table 4.2.6
DLOP and RQL for o-Xylene from
SKC 575-002 Passive Samplers |
|
mass per sample
(ng) | area counts
(µV-s) |
|
0
343
685
1028
1371
1714
2056
2399
2742
3084
3427 |
0
29
35
69
96
112
126
147
164
190
223 |
|
|
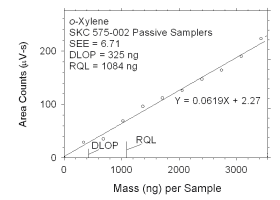
Figure 4.2.6 Plot of data used to determine DLOP and RQL for o-xylene.
|
| |
Table 4.2.7
DLOP and RQL
for p-Xylene from Charcoal Tubes |
|
mass per sample
(ng) | area counts
(µV-s) |
|
0
171
341
512
683
853
1024
1195
1365
1536
1707 |
139
168
179
213
229
248
275
295
323
338
357 |
|
|
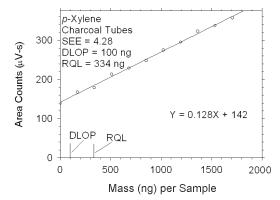
Figure 4.2.7 Plot of data used to determine DLOP and RQL for p-xylene.
|
| |
Table 4.2.8
DLOP and RQL for p-Xylene from
SKC 575-002 Passive Samplers |
|
mass per sample
(ng) | area counts
(µV-s) |
|
0
341
683
1024
1365
1707
2048
2390
2731
3072
3414 |
136
172
187
204
246
269
304
293
338
353
384 |
|
|
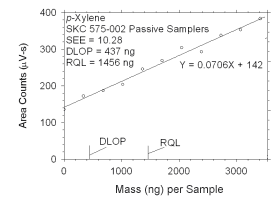
Figure 4.2.8 Plot of data used to determine DLOP and RQL for p-xylene.
|
| |
Table 4.2.9
DLOP and RQL
for Ethylbenzene from Charcoal Tubes |
|
mass per sample
(ng) | area counts
(µV-s) |
|
0
173
346
519
692
865
1038
1211
1384
1558
1731 |
0
35
44
76
88
118
136
153
182
198
231 |
|
|
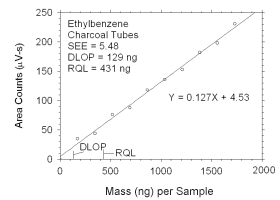
Figure 4.2.9 Plot of data used to determine DLOP and RQL for ethylbenzene.
|
| |
Table 4.2.10
DLOP and RQL for Ethylbenzene from
SKC 575-002 Passive Samplers |
|
mass per sample
(ng) | area counts
(µV-s) |
|
0
346
692
1038
1384
1731
2077
2423
2769
3115
3461 |
0
25
52
71
90
111
143
153
186
212
251 |
|
|
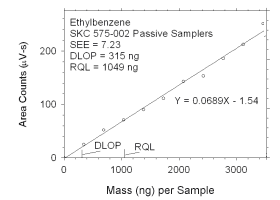
Figure 4.2.10 Plot of data used to determine DLOP and RQL for ethylbenzene.
|
The RQL is considered the lower limit for precise quantitative measurements.
It is determined from the regression line parameters obtained for calculation of
DLOP, providing the extraction efficiency (EE) is 100 ± 25% at the RQL.
Table 4.2.11
Reliable Quantitation Limits
|
|
charcoal
tubes |
SKC 575-002
Passive Samplers |
| analyte | ng | µg/m3 | ppb | EE(%) | ng | µg/m3 | ppb | EE(%) |
|
xylenes
m-xylenes
o-xylenes
p-xylenes
ethylbenzene |
1072
531
795
334
431 |
89.3
44.2
66.2
27.8
35.9 |
20.6
10.2
15.3
6.4
8.3 |
98.4
98.9
95.6
99.9
99.4 |
2823
1495
1084
1456
1049 |
842.7
450.3
317.0
435.9
316.0 |
194.1
103.7
73.0
100.4
72.8 |
93.6
96.4
84.9
95.6
97.6 |
|

Figure 4.2.11 Chromatogram of a sample containing masses of analytes approximating the
RQLs extracted from a charcoal tube. Key: (1) ethylbenzene, (2) p-xylene, (3)
m-xylene, (4) o-xylene.
| |
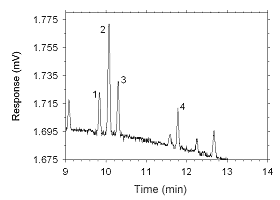
Figure 4.2.12 Chromatogram of a sample containing masses of analytes approximating the RQLs extracted from a SKC 575-002 Passive Sampler. Key: (1) ethylbenzene, (2) p-xylene,
(3) m-xylene, (4) o-xylene.
|
4.3 Instrument calibration
|
The instrument was calibrated for xylene isomers and ethylbenzene over a range of
from 0.5 to 2 times the 100 ppm PEL for each analyte. Calibration was performed
at concentrations appropriate for both active and diffusive samplers.
Calibration curves were constructed from the tabulated data and are shown in
Section 3.5.3. Coefficients of determination (r2) and of
nondetermination (k2) are shown in Table 4.3.
|
|
Table 4.3
Coefficient of Determination (r2)
and of Nondetermination (k2) |
|
| analyte |
charcoal
tubes |
SKC 575-002
Pass Sampers |
| r2 |
k2 | r2 |
k2 |
|
| m-xylene |
0.9994 |
6 × 10-4 |
0.9998 |
2 × 10-4 |
| o-xylene |
0.9995 |
5 × 10-4 |
0.9998 |
2 × 10-4 |
| p-xylene |
0.9994 |
6 × 10-4 |
0.9998 |
2 × 10-4 |
| ethylbenzene |
0.9994 |
6 × 10-4 |
0.9998 |
2 × 10-4 |
|
|
Table 4.3.1
Instrument Response to
m-Xylene for Charcoal Tubes
|
× OSHA PEL
(µg/sample) |
0.5×
2568 |
0.75×
3852 |
1×
5136 |
1.5×
7704 |
2×
10272 |
|
| area (µV-s) |
164794
164754
164983
164992
165047
165182 |
269791
269445
269434
269351
269191
268965 |
375905
375908
376753
376901
375901
375624 |
593689
592710
593187
592430
591644
593035 |
783759
784944
786776
784541
784130
783251 |
|
Table 4.3.2
Instrument Response to
o-Xylene for Charcoal Tubes
|
× OSHA PEL
(µg/sample) |
0.5×
2590 |
0.75×
3884 |
1×
5179 |
1.5×
7769 |
2×
10358 |
|
| area (µV-s) |
167753
167760
167934
167902
167961
168094 |
274897
274708
274660
274497
274406
274212 |
383480
383480
384309
384433
383472
383231 |
606183
605143
605576
605029
604460
605477 |
801612
802500
804231
802144
801744
800999 |
|
Table 4.3.3
Instrument Response to
p-Xylene for Charcoal Tubes
|
× OSHA PEL
(µg/sample) |
0.5×
2559 |
0.75×
3839 |
1×
5118 |
1.5×
7678 |
2×
10237 |
|
| area (µV-s) |
164552
164539
164734
164768
164807
164923 |
268846
268538
268527
268449
268265
268051 |
374277
374271
375116
375271
374253
374021 |
590454
589602
590062
589284
588468
589848 |
779090
780366
782170
779941
779572
778654 |
|
Table 4.3.4
Instrument Response to
Ethylbenzene for Charcoal Tubes
|
× OSHA PEL
(µg/sample) |
0.5×
2608 |
0.75×
3912 |
1×
5216 |
1.5×
7824 |
2×
10432 |
|
| area (µV-s) |
164275
164292
164473
164518
164581
164665 |
269560
269199
269224
269089
268885
268679 |
375888
375846
376676
376857
375780
375508 |
593883
593063
593502
592685
591749
593173 |
783759
785278
787146
784787
784405
783391 |
|
Table 4.3.5
Instrument Response to m-Xylene
for SKC 575-002 Passive Samplers
|
× OSHA PEL
(µg/sample) |
0.5×
749 |
0.75×
1177 |
1×
1455 |
1.5×
2140 |
2×
2889 |
|
| area (µV-s) |
29838
29955
29857
29887
29836
29842 |
47626
47689
47832
47847
47796
47774 |
58960
59183
59046
59397
59004
59284 |
86200
86119
86097
85956
85933
85854 |
117788
117903
117765
118128
117561
117760 |
|
Table 4.3.6
Instrument Response to o-Xylene
for SKC 575-002 Passive Samplers
|
× OSHA PEL
(µg/sample) |
0.5×
755 |
0.75×
1187 |
1×
1467 |
1.5×
2158 |
2×
2913 |
|
| area (µV-s) |
30464
30579
30508
30521
30486
30483 |
48643
48697
48848
48880
48780
48795 |
60209
60434
60309
60692
60248
60531 |
88024
87973
87915
87801
87757
87695 |
120324
120516
120317
120670
120081
120298 |
|
Table 4.3.7
Instrument Response to p-Xylene
for SKC 575-002 Passive Samplers
|
× OSHA PEL
(µg/sample) |
0.5×
746 |
0.75×
1173 |
1×
1450 |
1.5×
2133 |
2×
2879 |
|
| area (µV-s) |
29754
29867
29774
29817
29771
29772 |
47433
47507
47680
47672
47625
47597 |
58707
58961
58809
59157
58765
59041 |
85808
85716
85692
85590
85545
85468 |
117229
117319
117199
117578
116981
117192 |
|
Table 4.3.8
Instrument Response to Ethylbenzene
for SKC 575-002 Passive Samplers
|
× OSHA PEL
(µg/sample) |
0.5×
761 |
0.75×
1195 |
1×
1478 |
1.5×
2173 |
2×
2934 |
|
| area (µV-s) |
29797
29920
29824
29882
29834
29816 |
47592
47668
47822
47826
47764
47738 |
58904
59197
59043
59390
58958
59261 |
86193
86089
86057
85970
85917
85830 |
117774
117857
117737
118148
117530
117767 |
|
4.4 Precision (overall procedure)
4.4.1 Charcoal tubes
|
The precision at the 95% confidence level is obtained by multiplying the SEE by 1.96
(the z-statistic from the standard normal distribution at the 95% confidence
level). In Section 4.5, 95% confidence intervals are drawn about their respective
regression lines in the storage graph figures. The precisions of the overall procedure
were obtained from the ambient temperature storage tests and are shown in Table 4.4.1.
|
Table 4.4.1
SEEs and Precisions of the
Overall Procedure for Charcoal Tubes |
|
| analyte | SEE(%) | precision(±%) |
|
xylenes
m-xylene
o-xylene
p-xylene
ethylbenzene |
5.50
5.45
5.59
5.67
5.41 |
10.8
10.7
11.0
11.1
10.6 |
|
|
4.4.2 SKC 575-002 Passive Samplers
The precision at the 95% confidence level is obtained by multiplying the SEE by 1.96
(the z-statistic from the standard normal distribution at the 95% confidence
level). In Section 4.5, 95% confidence intervals are drawn about their respective
regression lines in the storage graph figures. Each precision includes an additional 8.7%
for sampling rate variability. There are different values given, depending on whether both,
either, or neither temperature or atmospheric pressure are known at the sampling site. If
the sampling-site temperature (T) is unknown, it is assumed to be
22.2 ± 15 °C (72 ± 27 °F) and a variability of ±7.7% is included.
If the atmospheric pressure (P) is unknown, it is estimated from sampling-site
elevation and a variability of ±3% is included. The precisions of the overall
procedure are shown in Table 4.4.2.
Table 4.4.2
SEEs and Precisions of the Overall Procedure for SKC 575-002 Passive Samplers
|
|
condition |
xylenes |
m-xylene |
o-xylene |
p-xylene |
ethylbenzene |
|
|
|
SEE
(%) |
precision
(±%) |
SEE
(%) |
precision
(±%) |
SEE
(%) |
precision
(±%) |
SEE
(%) |
precision
(±%) |
SEE
(%) |
precision
(±%) |
|
both T and P known
only T known
only P known
neither T nor P known |
9.29
9.76
12.07
12.43 |
18.2
19.1
23.7
24.4 |
9.32
9.79
12.09
12.46 |
18.3
19.2
23.7
24.4 |
9.24
9.71
12.03
12.40 |
18.1
19.0
23.6
24.3 |
9.33
9.80
12.10
12.46 |
18.3
19.2
23.7
24.4 |
9.39
9.86
12.14
12.51
|
18.4
19.3
23.8
24.5 |
|
4.5 Storage tests
4.5.1 Charcoal tubes
Storage stability samples were prepared by sampling (at 50 mL/min for four hours)
dynamically generated test atmospheres of mixed xylenes with SKC 226-01 sampling
tubes. These samples were collected simultaneously along with diffusive samples. The
concentrations of the test atmospheres were 207 mg/m3 (48 ppm)
for m-xylene, 96 mg/m3 (22 ppm) for
o-xylene, 90 mg/m3 (21 ppm) for p-xylene, and
73 mg/m3 (17 ppm) for ethylbenzene at 83% relative humidity
and 20 °C. Xylenes concentration was the sum of the individual isomers and was
393 mg/m3 (91 ppm). These air concentrations were approximately
one times the target concentration for xylenes and were in the same proportions as were
the analytes in the mixed xylenes used to generate the test atmospheres. Xylenes results
were calculated from summed individual isomers results. Sample results are corrected for
extraction efficiency.
Table 4.5.1.1
Storage Tests for Xylenes
|
time
(days) |
ambient storage
recovery (%)
|
refrigerated storage
recovery (%) |
|
0
3
7
10
14
16 |
97.0
101.1
102.2
96.5
101.6
99.8 |
98.9
102.5
97.4
97.9
96.7
97.0 |
96.8
100.3
103.1
99.2
97.9
101.1 |
97.0
103.1
96.3
100.4
101.3
101.7 |
98.9
101.3
100.2
98.2
99.4
93.8 |
96.8
100.3
100.8
98.8
100.2
102.0 |
|
Table 4.5.1.2
Storage Tests for m-Xylene
|
time
(days) |
ambient storage
recovery (%)
|
refrigerated storage
recovery (%) |
|
0
3
7
10
14
16 |
97.3
101.4
102.5
97.0
101.9
100.6 |
99.1
102.8
97.8
98.3
97.2
98.7 |
97.2
100.6
103.3
99.5
99.8
101.9 |
97.3
103.3
97.6
100.7
101.6
102.0 |
99.1
101.6
100.7
98.7
99.7
93.7 |
97.2
100.6
101.2
99.2
100.5
102.3 |
|
Table 4.5.1.3
Storage Tests for o-Xylene
|
time
(days) |
ambient storage
recovery (%)
|
refrigerated storage
recovery (%) |
|
0
3
7
10
14
16 |
96.1
100.7
101.5
95.2
101.0
99.5 |
98.4
101.8
96.3
97.0
95.6
95.8 |
95.9
99.5
102.6
98.4
98.5
100.6 |
96.1
102.5
95.6
99.6
100.6
101.1 |
98.4
100.7
99.2
96.9
98.8
94.4 |
95.9
99.6
100.0
97.7
99.5
101.4 |
|
Table 4.5.1.4
Storage Tests for p-Xylene
|
time
(days) |
ambient storage
recovery (%)
|
refrigerated storage
recovery (%) |
|
0
3
7
10
14
16 |
97.0
101.1
102.1
96.6
101.6
99.6 |
98.8
102.5
97.5
98.0
96.8
97.0 |
96.8
100.3
103.0
99.2
93.1
101.1 |
97.0
103.0
96.4
100.4
101.3
101.7 |
98.8
101.3
100.3
98.3
99.4
93.5 |
96.8
100.3
100.9
98.9
100.2
101.9 |
|
Table 4.5.1.5
Storage Tests for Ethylbenzene
|
time
(days) |
ambient storage
recovery (%)
|
refrigerated storage
recovery (%) |
|
0
3
7
10
14
16 |
98.2
102.1
103.1
98.2
102.5
100.0 |
99.7
103.6
99.0
99.3
98.6
97.5 |
98.3
101.6
103.9
100.3
100.8
101.4 |
98.2
104.0
97.5
101.6
102.3
102.6 |
99.7
102.3
101.9
100.1
100.6
93.5 |
98.3
101.3
102.2
100.3
101.2
103.0 |
|
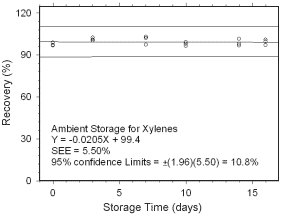
Figure 4.5.1.1 Ambient storage test for xylenes collected on charcoal tubes. |
|
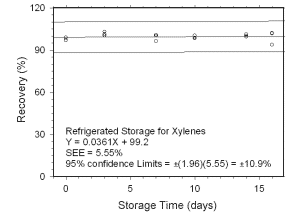
Figure 4.5.1.2 Regrigerated storage test for xylenes collected on charcoal tubes.
|
| |
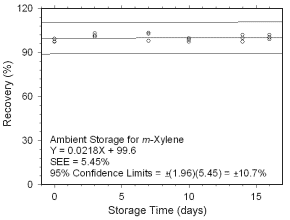
Figure 4.5.1.3 Ambient storage test for m-xylene collected on charcoal tubes. |

Figure 4.5.1.4 Regrigerated storage test for m-xylene collected on charcoal tubes.
|
| |
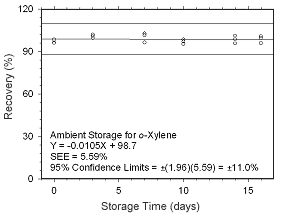
Figure 4.5.1.5 Ambient storage test for o-xylene collected on charcoal tubes. |

Figure 4.5.1.6 Regrigerated storage test for o-xylene collected on charcoal tubes.
|
| |
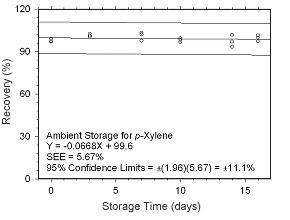
Figure 4.5.1.7 Ambient storage test for p-xylene collected on charcoal tubes. |
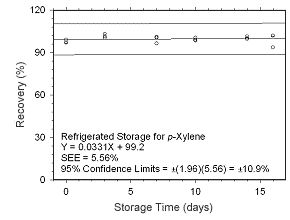
Figure 4.5.1.8 Regrigerated storage test for p-xylene collected on charcoal tubes.
|
| |

Figure 4.5.1.9 Ambient storage test for ethylbenzene collected on charcoal tubes. |

Figure 4.5.1.10 Regrigerated storage test for ethylbenzene collected on charcoal tubes.
|
4.5.2 SKC 575-002 Passive Samplers
Storage stability samples were prepared by sampling dynamically generated test
atmospheres of mixed xylenes with SKC 575-002 Passive Samplers. The face
velocity of the test atmosphere was about 0.4m/s past the diffusive samplers. The samplers
were orientated parallel to the flow direction. These samples were collected for four
hours simultaneously along with active samples under conditions described in Section
4.5.1. Xylenes results were calculated from summed isomers results. Sample results are
corrected for extraction efficiency.
Table 4.5.2.1
Storage Tests for Xylenes
|
time
(days) |
ambient storage
recovery (%)
|
refrigerated storage
recovery (%) |
|
0
3
7
10
14
16 |
96.6
101.0
104.6
101.1
98.0
97.4 |
97.3
101.6
93.5
102.8
92.9
98.9 |
97.1
102.5
99.7
97.2
99.7
94.4 |
96.6
101.2
100.9
100.5
100.6
102.4 |
97.3
102.5
100.0
99.5
99.4
100.9 |
97.1
101.4
98.6
100.7
100.5
97.9 |
|
Table 4.5.2.2
Storage Tests for m-Xylene
|
time
(days) |
ambient storage
recovery (%)
|
refrigerated storage
recovery (%) |
|
0
3
7
10
14
16 |
96.8
101.5
105.1
101.6
98.4
97.9 |
97.5
102.2
93.9
103.3
93.3
99.4 |
97.4
103.1
100.2
97.6
100.1
94.9 |
96.8
101.8
101.3
100.9
101.1
102.9 |
97.5
103.1
100.4
100.0
99.9
101.4 |
97.4
101.9
99.0
101.1
101.0
98.4 |
|
Table 4.5.2.3
Storage Tests for o-Xylene
|
time
(days) |
ambient storage
recovery (%)
|
refrigerated storage
recovery (%) |
|
0
3
7
10
14
16 |
96.2
99.7
103.4
99.9
96.9
96.2 |
96.8
100.4
92.4
101.5
92.1
97.7 |
96.6
101.1
98.6
96.1
99.0
93.1 |
96.2
99.7
99.9
99.4
99.3
101.1 |
96.8
101.1
98.8
98.4
98.2
99.6 |
96.6
100.4
97.3
99.6
99.3
96.7 |
|
Table 4.5.2.4
Storage Tests for p-Xylene
|
time
(days) |
ambient storage
recovery (%)
|
refrigerated storage
recovery (%) |
|
0
3
7
10
14
16 |
96.6
101.2
104.8
101.3
98.1
97.5 |
97.2
101.8
93.6
103.1
92.9
99.1 |
97.0
102.8
99.9
97.3
99.6
94.5 |
99.6
101.5
101.0
100.6
100.7
102.5 |
97.2
102.8
100.2
99.7
99.5
101.0 |
97.0
101.5
98.7
100.8
100.7
98.0 |
|
Table 4.5.2.5
Storage Tests for Ethylbenzene
|
time
(days) |
ambient storage
recovery (%)
|
refrigerated storage
recovery (%) |
|
0
3
7
10
14
16 |
97.4
103.0
106.5
103.0
99.6
99.1 |
98.1
103.6
95.2
104.7
94.3
100.8 |
98.0
104.4
101.5
98.8
101.0
96.1 |
97.4
103.2
102.6
102.2
102.7
104.4 |
98.1
104.4
101.7
101.3
101.2
102.9 |
98.0
103.5
100.4
102.4
102.4
99.7 |
|
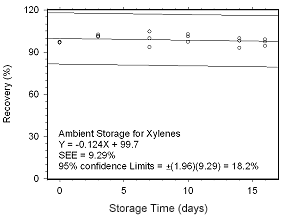
Figure 4.5.2.1 Ambient storage test for xylenes collected on SKC 575-002 Passive Samplers. |
|
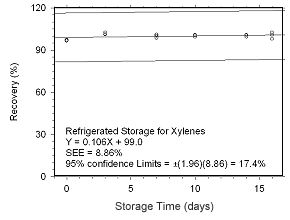
Figure 4.5.2.2 Regrigerated storage test for xylenes collected on SKC 575-002 Passive Samplers.
|
| |

Figure 4.5.2.3 Ambient storage test for m-xylene collected on SKC 575-002 Passive Samplers. |

Figure 4.5.2.4 Regrigerated storage test for m-xylene collected on SKC 575-002 Passive Samplers.
|
| |
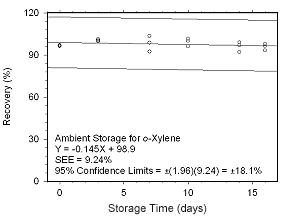
Figure 4.5.2.5 Ambient storage test for o-xylene collected on SKC 575-002 Passive Samplers. |

Figure 4.5.2.6 Regrigerated storage test for o-xylene collected on SKC 575-002 Passive Samplers.
|
| |
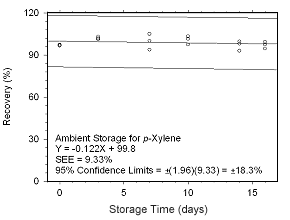
Figure 4.5.2.7 Ambient storage test for p-xylene collected on SKC 575-002 Passive Samplers. |

Figure 4.5.2.8 Regrigerated storage test for p-xylene collected on SKC 575-002 Passive Samplers.
|
| |
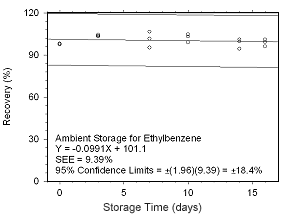
Figure 4.5.2.9 Ambient storage test for ethylbenzene collected on SKC 575-002 Passive Samplers. |
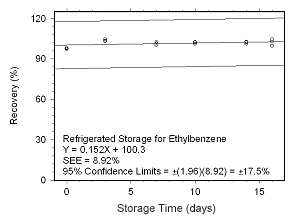
Figure 4.5.2.10 Regrigerated storage test for ethylbenzene collected on SKC 575-002 Passive Samplers.
|
4.6 Reproducibility
Twelve samples (six charcoal tubes and six SKC 575-002 Passive Samplers) were collected from
controlled test atmospheres similar to that used to collect storage stability samples. The samples
were submitted to SLTC for analysis. The charcoal tube samples were analyzed after 16 days of
storage at ambient temperature, and the SKC 575-002 Passive Samplers after 22 days of storage
at ambient temperature. Sample results were corrected for extraction efficiency. No sample result
had a deviation greater than the precision of the overall procedure reported in Section 4.4. Xylenes
results were calculated by summing results for individual isomers.
Table 4.6.1
Reproducibility Data for Xylene Isomers on Charcoal
Tubes
|
| m-xylene |
o-xylene |
p-xylene |
theo
µg/samp |
reported
µg/samp |
recovery
(%) |
deviation
(%) |
theo
µg/samp |
reported
µg/samp |
recovery
(%) |
deviation
(%) |
theo
µg/samp |
reported
µg/samp |
recovery
(%) |
deviation
(%) |
|
2555
2595
2623
2557
2564
2623 |
2552
2593
2427
2580
2593
2610 |
99.9
99.9
92.5
100.9
101.1
99.5 |
-0.1
-0.1
-7.5
0.9
1.1
-0.5 |
1189
1207
1221
1190
1193
1221 |
1245
1266
1158
1262
1263
1275 |
104.7
104.9
94.8
106.1
105.9
104.4 |
4.7
4.9
-5.2
6.1
5.9
4.4 |
1112
1129
1142
1129
1116
1142 |
1140
1158
1082
1152
1159
1166 |
102.5
102.6
94.7
102.0
103.9
102.1 |
2.5
2.6
-5.3
2.0
3.9
2.1 |
|
Table 4.6.2
Reproducibility Data for Xylenes and Ethylbenzene on Charcoal Tubes
|
| xylenes |
|
|
|
|
ethylbenzene |
theo
µg/samp |
reported
µg/samp |
recovery
(%) |
deviation
(%) |
theo
µg/samp |
reported
µg/samp |
recovery
(%) |
deviation
(%) |
|
4856
4931
4986
4876
4873
4986 |
4937
5017
4667
4994
5014
5051 |
101.7
101.7
93.6
102.4
102.9
101.3 |
1.7
1.7
-6.4
2.4
2.9
1.3 |
|
|
|
|
|
|
|
904.8
918.9
929.0
905.6
911.8
929.0 |
929.4
944.2
895.4
939.1
945.6
944.3 |
102.7
102.8
96.4
103.7
103.7
101.6 |
2.7
2.8
-3.6
3.7
3.7
1.6 |
|
Table 4.6.3
Reproducibility Data for Xylene Isomers on SKC 575-002
Passive Samplers
|
| m-xylene |
o-xylene |
p-xylene |
theo
µg/samp |
reported
µg/samp |
recovery
(%) |
deviation
(%) |
theo
µg/samp |
reported
µg/samp |
recovery
(%) |
deviation
(%) |
theo
µg/samp |
reported
µg/samp |
recovery
(%) |
deviation
(%) |
|
847.6
847.6
847.6
847.6
847.6
847.6 |
859.1
823.9
811.5
806.0
805.5
803.3 |
101.4
97.2
95.7
95.1
95.0
94.8 |
1.4
-2.8
-4.3
-4.9
-5.0
-5.2 |
401.3
401.3
401.3
401.3
401.3
401.3 |
430.8
417.1
408.8
399.0
406.3
398.9 |
107.4
103.9
101.9
99.4
101.2
99.4 |
7.4
3.9
1.9
-0.6
1.2
-0.6 |
372.8
372.8
372.8
372.8
372.8
372.8 |
404.9
389.1
383.0
381.0
380.2
380.4 |
108.6
104.4
102.7
102.2
102.0
102.0 |
8.6
4.4
2.7
2.2
2.0
2.0 |
|
Table 4.6.4
Reproducibility Data for Xylenes and Ethylbenzene on SKC 575-002 Passive
Samplers
|
| xylenes |
|
|
|
|
ethylbenzene |
theo
µg/samp |
reported
µg/samp |
recovery
(%) |
deviation
(%) |
theo
µg/samp |
reported
µg/samp |
recovery
(%) |
deviation
(%) |
|
1622
1622
1622
1622
1622
1622 |
1695
1630
1603
1586
1592
1583 |
104.5
100.5
98.8
97.8
98.2
97.6 |
4.5
0.5
-1.2
-2.2
-1.8
-2.4 |
|
|
|
|
|
|
|
302.3
302.3
302.3
302.3
302.3
302.3 |
312.2
298.3
293.9
295.0
291.7
293.7 |
103.3
98.7
97.2
97.6
96.5
97.2 |
3.3
-1.3
-2.8
-2.4
-3.5
-2.8 |
|
4.7 Sampler capacity
4.7.1 Charcoal tubes
The sampling capacity of charcoal tubes was tested by sampling dynamically generated
test atmospheres of mixed xylenes with SKC 226-01 (Lot 2000) sampling tubes. These
samples were collected simultaneously along with diffusive samples. The sampling times
were 5, 10, 15, and 30 min; and 1, 2, 3, 4, 6, 8, and 10 hours. Three active and three
diffusive samples were collected for each time period. The mean concentrations of the test
atmospheres were 456 mg/m3 (105 ppm) for m-xylene, 212 mg/m3 (49 ppm) for o-xylene,
198 mg/m3 (46 ppm) for p-xylene, and 161 mg/m3 (37 ppm) for ethylbenzene at 78%
relative humidity and 21 °C. These air concentrations were approximately two times the
target concentration for xylenes and were in the same proportions as were the analytes in
the mixed xylenes used to generate the test atmospheres. No breakthrough from the front
to the back section of the sampling tubes for any of the analytes was observed even when
samples were collected for ten hours at 50 mL/min. Sampler capacity was never
exceeded. Nearly 31 mg of mixed xylenes had been collected after ten hours. The
recommended sampling time was set at four hours and the recommended sampling rate
at 50 mL/min. These tests also showed that samples can be collected for as short a time
as 5 min at 50 mL/min and still provide excellent results.
4.7.2 SKC 575-002 Passive Samplers
The sampling rate and sampler capacity of SKC 575-002 Passive Samplers were
determined with samples collected at the increasing time intervals from the controlled test
atmospheres described in Section 4.7.1. The face velocity of the test atmosphere was
approximately 0.4 m/s, and the samplers were orientated parallel to the flow direction.
Three samples were collected at each time interval. Sampler capacity has been defined
to be exceeded when the "apparent" sampling rate decreases rapidly. The sampling rate
only appears to decrease because the sampler can collect no additional analyte at the
point when capacity is exceeded. Sampling rates are presented in mL/min at 760 mmHg
and 25 °C.
Table 4.7.2
Determination of Sampling Rate and Recommended Sampling Time
|
| time |
m-xylene |
o-xylene |
p-xylene |
ethylbenzene |
| (h) | mL/min | RSD | mL/min | RSD | mL/min | RSD | mL/min | RSD |
|
| 0.083 | 13.75 | 2.2 | 14.25 | 3.8 | 13.99 | 2.8 | 13.85 | 2.3 |
| 0.167 | 13.53 | 1.4 | 13.97 | 1.3 | 13.71 | 1.8 | 13.68 | 1.5 |
| 0.25 | 13.91 | 0.9 | 14.38 | 0.9 | 14.01 | 0.9 | 13.96 | 0.8 |
| 0.5 | 13.94 | 1.6 | 14.39 | 2.4 | 14.07 | 1.7 | 13.93 | 1.2 |
| 1 | 13.81 | 2.1 | 14.17 | 2.4 | 13.94 | 2.2 | 13.86 | 2.2 |
| 2 | 13.55 | 1.7 | 13.79 | 1.8 | 13.59 | 1.7 | 13.50 | 2.0 |
| 3 | 14.01 | 1.4 | 14.27 | 1.4 | 14.04 | 1.4 | 13.96 | 1.5 |
| 4 | 13.60 | 0.6 | 14.93 | 0.9 | 13.67 | 0.7 | 13.59 | 0.6 |
| 6 | 14.20 | 5.0 | 14.40 | 5.0 | 14.48 | 5.0 | 14.19 | 5.0 |
| 8 | 13.60 | 0.2 | 13.70 | 0.1 | 13.70 | 0.2 | 13.59 | 0.2 |
| 10 | 14.11 | 2.6 | 14.35 | 2.5 | 14.16 | 2.7 | 14.06 | 2.6 |
| |
| mean | 13.82 | | 14.24 | | 13.94 | | 13.83 | |
| RSD | 1.7 | | 2.4 | | 1.9 | | 1.6 | |
|
The preliminary sampling rate was determined by averaging the values for the 0.5, 1, and
2 hour samples. Horizontal lines were constructed 10% above and 10% below the
preliminary sampling rate. All the sampling rates were included in the calculated mean
sampling rates because all were between the two horizontal lines.
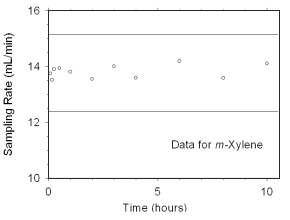
Figure 4.7.2.1 Sampler capacity data for m-xylene. |
|
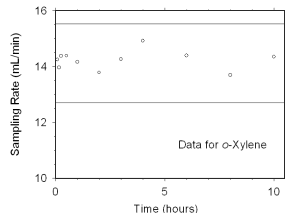
Figure 4.7.2.2 Sampler capacity data for o-xylene. |
| |
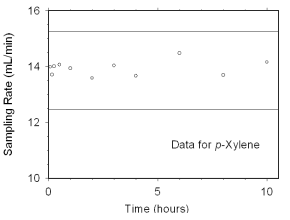
Figure 4.7.2.3 Sampler capacity data for p-xylene. |
|
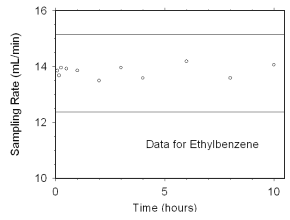
Figure 4.7.2.4 Sampler capacity data for ethylbenzene. |
4.8 Extraction efficiency and stability of extracted samples
Each laboratory must determine and confirm extraction efficiency periodically. Other solvents can
be used in conjunction with this method provided the new solvent is tested. The new solvent should
be tested as described below and the extraction efficiency must be greater than 75%.
A summary of the extraction efficiency results over the range of RQL to 2 times the target
concentration is presented in Table 4.8 for quick reference.
Table 4.8
Extraction Efficiency (%) Summary
|
| analyte |
charcoal
tubes |
SKC 575-002
Passive Samplers |
|
| m-xylene | 96.3 | 96.1 |
| o-xylene | 93.8 | 89.4 |
| p-xylene | 96.1 | 95.3 |
| ethylbenzene | 97.2 | 99.1 |
|
4.8.1 Charcoal tubes
The extraction efficiencies (EEs) of the analytes were determined by liquid-spiking
100-mg portions of SKC Lot 2000 charcoal with the analytes at levels from the
RQL to 2 times the OSHA PEL for each analyte. These samples were stored overnight at ambient
temperature, and then extracted with 1mL of CS2 (containing 1 µL
of p-cymene per mL of CS2) for 1 hour. The samples were
vigorously shaken periodically over the extraction time. The EEs of the analytes at the
target concentration were also determined from "wet" charcoal to confirm that EE remained
constant. Wet charcoal was prepared by collecting samples from a humid (about 80% RH and
22 °C) atmosphere at 50 mL/min for 4 hours. Only the front section of these samples
was used to prepare wet EE samples. The stability of extracted samples was investigated
by reanalyzing the 1 × PEL samples a day after the original analysis. Three vials
were immediately resealed with new septa caps and three vials retained their punctured
septa following the original analysis.
Table 4.8.1.1
Extraction Efficiency of m-Xylene from SKC Lot 2000 Charcoal
|
level
|
sample number
|
| × OSHA PEL | µg per sample | 1 | 2 | 3 | 4 | 5 | 6 | mean |
|
| RQL | 0.548 | 107.1 | 104.6 | 97.9 | 92.7 | 95.4 | 95.7 | 98.9 |
| 0.05 | 256 | 96.9 | 96.1 | 95.3 | 94.7 | 94.6 | 95.8 | 95.6 |
| 0.1 | 512 | 96.5 | 99.7 | 96.3 | 94.0 | 96.7 | 94.0 | 96.2 |
| 0.2 | 1024 | 96.5 | 95.2 | 94.6 | 96.5 | 95.6 | 94.4 | 95.5 |
| 0.5 | 2560 | 97.7 | 96.0 | 94.6 | 93.9 | 95.0 | 95.3 | 95.4 |
| 1.0 | 5136 | 97.0 | 96.5 | 96.5 | 97.6 | 98.1 | 97.9 | 97.3 |
| 2.0 | 10486 | 93.7 | 95.6 | 94.3 | 97.6 | 94.5 | 95.5 | 95.2 |
| wet (1.0) | 5136 | 98.0 | 96.4 | 97.7 | 97.4 | 96.6 | 97.9 | 97.3 |
The mean EE at concentrations from the RQL to 2 times the PEL (excepting the wet EE) is 96.3%. |
Table 4.8.1.2
Extraction Efficiency of o-Xylene from SKC Lot 2000 Charcoal
|
level
|
sample number
|
| × OSHA PEL | µg per sample | 1 | 2 | 3 | 4 | 5 | 6 | mean |
|
| RQL | 0.785 | 99.6 | 96.9 | 100.8 | 88.4 | 92.8 | 95.0 | 95.6 |
| 0.05 | 257 | 93.8 | 93.0 | 92.4 | 91.6 | 91.6 | 92.7 | 92.5 |
| 0.1 | 514 | 93.5 | 96.7 | 93.3 | 91.2 | 93.7 | 91.2 | 93.3 |
| 0.2 | 1028 | 93.6 | 92.3 | 91.8 | 93.6 | 92.6 | 91.4 | 92.6 |
| 0.5 | 2570 | 94.9 | 93.2 | 92.0 | 91.3 | 92.4 | 92.7 | 92.8 |
| 1.0 | 5179 | 94.6 | 94.0 | 94.0 | 95.1 | 95.4 | 95.4 | 94.8 |
| 2.0 | 10575 | 93.2 | 94.6 | 93.9 | 98.5 | 93.9 | 94.5 | 94.8 |
| wet (1.0) | 5179 | 95.2 | 93.8 | 95.1 | 94.8 | 93.9 | 95.1 | 94.7 |
The mean EE at concentrations from the RQL to 2 times the PEL (excepting the wet EE) is 93.8%. |
Table 4.8.1.3
Extraction Efficiency of p-Xylene from SKC Lot 2000 Charcoal
|
level
|
sample number
|
| × OSHA PEL | µg per sample | 1 | 2 | 3 | 4 | 5 | 6 | mean |
|
| RQL | 0.788 | 102.1 | 96.9 | 100.7 | 103.3 | 105.6 | 90.8 | 99.9 |
| 0.05 | 256 | 96.5 | 95.7 | 95.0 | 94.3 | 94.2 | 95.3 | 95.2 |
| 0.1 | 512 | 96.1 | 99.3 | 95.9 | 93.7 | 96.3 | 93.3 | 95.8 |
| 0.2 | 1024 | 96.2 | 94.8 | 94.2 | 96.1 | 95.1 | 94.0 | 95.1 |
| 0.5 | 2560 | 97.3 | 95.6 | 94.2 | 93.6 | 94.6 | 94.9 | 95.0 |
| 1.0 | 5118 | 96.6 | 96.0 | 96.0 | 97.2 | 97.7 | 97.5 | 96.8 |
| 2.0 | 10450 | 93.2 | 95.2 | 93.8 | 96.9 | 94.1 | 95.1 | 94.7 |
| wet (1.0) | 5118 | 97.5 | 95.9 | 97.3 | 96.9 | 96.2 | 97.5 | 96.9 |
The mean EE at concentrations from the RQL to 2 times the PEL (excepting the wet EE) is 96.1%. |
Table 4.8.1.4
Extraction Efficiency of Ethylbenzene from SKC Lot 2000 Charcoal
|
level
|
sample number
|
| × OSHA PEL | µg per sample | 1 | 2 | 3 | 4 | 5 | 6 | mean |
|
| RQL | 0.43 | 96.9 | 97.1 | 96.2 | 100.7 | 109.1 | 96.6 | 99.4 |
| 0.05 | 260 | 98.3 | 97.4 | 96.6 | 95.9 | 95.9 | 97.1 | 96.9 |
| 0.1 | 519 | 97.8 | 101.2 | 97.6 | 95.4 | 98.0 | 95.2 | 97.5 |
| 0.2 | 1038 | 97.9 | 96.5 | 95.8 | 97.9 | 96.8 | 95.8 | 96.8 |
| 0.5 | 2596 | 99.0 | 97.3 | 95.8 | 95.1 | 96.2 | 96.5 | 96.7 |
| 1.0 | 5216 | 98.2 | 97.7 | 99.0 | 98.8 | 99.3 | 99.1 | 98.7 |
| 2.0 | 10650 | 93.0 | 95.7 | 93.7 | 95.3 | 94.2 | 95.6 | 94.6 |
| wet (1.0) | 5216 | 99.3 | 97.7 | 99.0 | 98.6 | 97.9 | 99.3 | 98.6 |
The mean EE at concentrations from the RQL to 2 times the PEL (excepting the wet EE) is 97.2%. |
Table 4.8.1.5
Stability of m-Xylene Extracted from SKC Lot 2000 Charcoal
|
punctured septa replaced
|
punctured septa retained
|
initial EE
(%) | EE after one day
(%) | difference
(%) | initial EE
(%) | EE after one day
(%) | difference
(%) |
|
| 97.0 | 97.4 | 0.4 | 97.6 | 96.1 | -1.5 |
| 96.5 | 97.1 | 0.6 | 98.1 | 96.7 | -1.4 |
| 96.5 | 98.2 | 1.7 | 97.9 | 95.9 | -2.0 |
| mean |
mean |
| 96.7 | 97.6 | 0.9 | 97.9 | 96.2 | -1.6 |
|
Table 4.8.1.6
Stability of o-Xylene Extracted from SKC Lot 2000 Charcoal
|
punctured septa replaced
|
punctured septa retained
|
initial EE
(%) | EE after one day
(%) | difference
(%) | initial EE
(%) | EE after one day
(%) | difference
(%) |
|
| 94.6 | 95.1 | 0.5 | 95.1 | 93.6 | -1.5 |
| 94.0 | 94.6 | 0.6 | 95.4 | 94.1 | -1.3 |
| 94.0 | 95.8 | 1.8 | 95.4 | 93.2 | -2.2 |
| mean |
mean |
| 94.2 | 95.2 | 1.0 | 95.3 | 93.6 | -1.7 |
|
Table 4.8.1.7
Stability of p-Xylene Extracted from SKC Lot 2000 Charcoal
|
punctured septa replaced
|
punctured septa retained
|
initial EE
(%) | EE after one day
(%) | difference
(%) | initial EE
(%) | EE after one day
(%) | difference
(%) |
|
| 96.6 | 97.0 | 0.4 | 97.2 | 95.6 | -1.6 |
| 96.0 | 96.7 | 0.7 | 97.7 | 96.2 | -1.5 |
| 96.0 | 97.8 | 1.8 | 97.5 | 95.3 | -2.2 |
| mean |
mean |
| 96.2 | 97.2 | 1.0 | 97.5 | 95.7 | -1.8 |
|
Table 4.8.1.8
Stability of Ethylbenzene Extracted from SKC Lot 2000 Charcoal
|
punctured septa replaced
|
punctured septa retained
|
initial EE
(%) | EE after one day
(%) | difference
(%) | initial EE
(%) | EE after one day
(%) | difference
(%) |
|
| 98.2 | 98.6 | 0.4 | 98.8 | 97.2 | -1.6 |
| 97.7 | 98.2 | 0.5 | 99.3 | 97.8 | -1.5 |
| 99.0 | 99.4 | 0.4 | 99.1 | 97.0 | -2.1 |
| mean |
mean |
| 98.3 | 98.7 | 0.4 | 99.1 | 97.3 | -1.7 |
|
4.8.2 SKC 575-002 Passive Samplers
The extraction efficiencies (EE) of the analytes were determined by liquid-spiking
500-mg portions of SKC Anasorb 747 (the sorbent in SKC 575-002
Passive Samplers) with the analytes at levels from the RQL to 2 times the OSHA PEL for
each analyte. These samples were stored overnight at ambient temperature, and then
extracted with 2 mL of CS2 (containing 1 µL of
1-phenylhexane per mL of CS2) for 1 hour. The
samples were vigorously shaken periodically over the extraction time. The EEs of the
analytes at the target concentration were also determined from "wet" samplers to confirm
that EE remained constant. Wet SKC 575-002 Passive Samplers were prepared by
sampling from a humid (about 80% RH and 22 °C) atmosphere for 4 hours. These samples were
extracted with 2 mL of CS2 (containing 1 µL of 1-phenylhexane
per mL of CS2) for 1 hour on a SKC 226D-03K Desorption
Shaker. The stability of extracted samples was investigated by reanalyzing the 1 × PEL
samples a day after the original analysis. Three vials were immediately resealed with new
septa caps and three vials retained their punctured septa following the original analysis.
Table 4.8.2.1
Extraction Efficiency of m-Xylene from SKC Anasorb 747
|
level
|
sample number
|
| × OSHA PEL | µg per sample | 1 | 2 | 3 | 4 | 5 | 6 | mean |
|
| RQL | 1.422 | 93.0 | 90.1 | 99.0 | 89.9 | 105.4 | 100.9 | 96.4 |
| 0.05 | 73 | 98.6 | 97.7 | 97.6 | 97.2 | 98.2 | 97.6 | 97.8 |
| 0.1 | 145 | 95.9 | 95.9 | 93.7 | 95.0 | 95.8 | 95.3 | 95.3 |
| 0.2 | 290 | 93.2 | 93.6 | 94.2 | 94.1 | 94.9 | 95.6 | 94.3 |
| 0.5 | 725 | 94.1 | 95.1 | 95.6 | 96.3 | 94.7 | 97.1 | 95.5 |
| 1.0 | 1451 | 97.2 | 96.1 | 101.1 | 95.8 | 96.0 | 97.0 | 97.2 |
| 2.0 | 2902 | 99.3 | 95.3 | 96.0 | 95.8 | 96.4 | 95.8 | 96.4 |
| wet (1.0) | 1498 | 96.6 | 96.7 | 93.8 | 98.0 | 96.7 | 95.3 | 96.2 |
The mean EE at concentrations from the RQL to 2 times the PEL (excepting the wet EE) is 96.1%. |
Table 4.8.2.2
Extraction Efficiency of o-Xylene from SKC Anasorb 747
|
level
|
sample number
|
| × OSHA PEL | µg per sample | 1 | 2 | 3 | 4 | 5 | 6 | mean |
|
| RQL | 1.075 | 83.9 | 76.4 | 86.3 | 87.8 | 86.6 | 88.1 | 84.9 |
| 0.05 | 73 | 92.3 | 90.9 | 91.0 | 90.4 | 91.8 | 91.5 | 91.3 |
| 0.1 | 146 | 89.8 | 89.6 | 87.5 | 89.0 | 89.9 | 89.3 | 89.2 |
| 0.2 | 292 | 87.7 | 88.0 | 88.4 | 88.4 | 89.0 | 89.8 | 88.6 |
| 0.5 | 728 | 88.4 | 89.2 | 89.8 | 90.4 | 88.8 | 91.2 | 89.6 |
| 1.0 | 1456 | 91.5 | 90.5 | 95.3 | 90.1 | 90.4 | 91.3 | 91.5 |
| 2.0 | 2913 | 93.7 | 88.7 | 90.7 | 90.5 | 90.9 | 90.4 | 90.8 |
| wet (1.0) | 1511 | 91.0 | 90.8 | 88.3 | 92.2 | 91.1 | 89.9 | 90.6 |
The mean EE at concentrations from the RQL to 2 times the PEL (excepting the wet EE) is 89.4%. |
Table 4.8.2.3
Extraction Efficiency of p-Xylene from SKC Anasorb 747
|
level
|
sample number
|
| × OSHA PEL | µg per sample | 1 | 2 | 3 | 4 | 5 | 6 | mean |
|
| RQL | 1.542 | 98.2 | 95.7 | 92.2 | 90.9 | 97.3 | 99.3 | 95.6 |
| 0.05 | 73 | 97.6 | 97.2 | 96.9 | 96.0 | 97.4 | 96.7 | 97.0 |
| 0.1 | 145 | 95.0 | 94.8 | 93.0 | 94.3 | 95.1 | 94.7 | 94.5 |
| 0.2 | 290 | 92.4 | 92.9 | 93.4 | 93.3 | 94.2 | 94.7 | 93.5 |
| 0.5 | 725 | 93.3 | 94.3 | 94.8 | 95.4 | 93.9 | 96.2 | 94.7 |
| 1.0 | 1451 | 96.3 | 95.2 | 100.2 | 94.9 | 95.1 | 96.1 | 96.3 |
| 2.0 | 2902 | 99.7 | 94.5 | 95.2 | 95.0 | 95.6 | 94.9 | 95.8 |
| wet (1.0) | 1493 | 95.7 | 95.7 | 92.9 | 97.0 | 95.8 | 94.4 | 95.3 |
The mean EE at concentrations from the RQL to 2 times the PEL (excepting the wet EE) is 95.3%. |
Table 4.8.2.4
Extraction Efficiency of Ethylbenzene from SKC Anasorb 747
|
level
|
sample number
|
| × OSHA PEL | µg per sample | 1 | 2 | 3 | 4 | 5 | 6 | mean |
|
| RQL | 1.058 | 99.7 | 105.0 | 93.7 | 88.5 | 95.2 | 103.4 | 97.6 |
| 0.05 | 74 | 102.2 | 101.2 | 101.1 | 100.2 | 101.2 | 100.8 | 101.1 |
| 0.1 | 147 | 99.1 | 99.1 | 97.3 | 98.6 | 99.3 | 98.6 | 98.7 |
| 0.2 | 294 | 96.3 | 96.8 | 97.5 | 97.2 | 98.4 | 98.8 | 97.5 |
| 0.5 | 736 | 97.3 | 98.4 | 98.9 | 99.5 | 98.3 | 100.5 | 98.8 |
| 1.0 | 1471 | 100.5 | 99.3 | 104.4 | 99.0 | 99.1 | 100.3 | 100.4 |
| 2.0 | 2942 | 102.6 | 98.3 | 99.1 | 98.8 | 99.6 | 98.9 | 99.6 |
| wet (1.0) | 1521 | 99.8 | 100.1 | 97.0 | 101.3 | 100.0 | 98.4 | 99.4 |
The mean EE at concentrations from the RQL to 2 times the PEL (excepting the wet EE) is 99.1%. |
Table 4.8.2.5
Stability of m-Xylene Extracted from SKC Anasorb 747
|
punctured septa replaced
|
punctured septa retained
|
initial EE
(%) | EE after one day
(%) | difference
(%) | initial EE
(%) | EE after one day
(%) | difference
(%) |
|
| 97.2 | 97.8 | 0.6 | 95.8 | 94.2 | -1.6 |
| 96.1 | 95.4 | -0.7 | 96.0 | 94.0 | -2.0 |
| 101.1 | 100.0 | -1.1 | 97.0 | 95.7 | -1.3 |
| mean |
mean |
| 98.1 | 97.7 | -0.4 | 96.3 | 94.6 | -1.6 |
|
Table 4.8.2.6
Stability of o-Xylene Extracted from SKC Anasorb 747
|
punctured septa replaced
|
punctured septa retained
|
initial EE
(%) | EE after one day
(%) | difference
(%) | initial EE
(%) | EE after one day
(%) | difference
(%) |
|
| 91.5 | 91.9 | 0.4 | 90.1 | 88.8 | -1.3 |
| 90.5 | 89.7 | -0.8 | 90.4 | 88.7 | -1.7 |
| 95.3 | 94.2 | -1.1 | 91.3 | 90.2 | -1.1 |
| mean |
mean |
| 92.4 | 91.9 | -0.5 | 90.6 | 89.2 | -1.4 |
|
Table 4.8.2.7
Stability of p-Xylene Extracted from SKC Anasorb 747
|
punctured septa replaced
|
punctured septa retained
|
initial EE
(%) | EE after one day
(%) | difference
(%) | initial EE
(%) | EE after one day
(%) | difference
(%) |
|
| 96.3 | 96.9 | 0.6 | 94.9 | 93.5 | -1.4 |
| 95.2 | 94.6 | -0.6 | 95.1 | 93.3 | -1.8 |
| 100.2 | 99.3 | -0.9 | 96.1 | 94.9 | -1.2 |
| mean |
mean |
| 97.2 | 96.9 | -0.3 | 95.4 | 93.9 | -1.5 |
|
Table 4.8.2.8
Stability of Ethylbenzene Extracted from SKC Anasorb 747
|
punctured septa replaced
|
punctured septa retained
|
initial EE
(%) | EE after one day
(%) | difference
(%) | initial EE
(%) | EE after one day
(%) | difference
(%) |
|
| 100.5 | 101.0 | 0.5 | 99.0 | 97.3 | -1.7 |
| 99.3 | 98.7 | -0.6 | 99.1 | 97.1 | -2.0 |
| 104.4 | 103.4 | -1.0 | 100.3 | 98.8 | -1.5 |
| mean |
mean |
| 101.4 | 101.0 | -0.4 | 99.5 | 97.7 | -1.7 |
|
4.9 Interferences (sampling)
4.9.1 Charcoal tubes
Retention
The ability of charcoal tubes to retain mixed xylenes after collection was tested by
sampling a test atmosphere containing 454 mg/m3, 211
mg/m3, 198 mg/m3, and 161 mg/m3
m-xylene, o-xylene, p-xylene, and ethylbenzene,
respectively (at 80% RH and 20 °C) with six samplers for one hour at 50 mL/min. Three
samples were analyzed immediately and three were used to sample contaminant-free
humid air for an additional three hours, and then analyzed. All the samples in the second
set retained at least 101.0, 101.2, 100.8, 101.0% of the means of the first set for
m-xylene, o-xylene, p-xylene, and ethylbenzene,
respectively.
Table 4.9.1
Retention of Mixed Xylenes on Charcoal Tubes
|
|
m-xylene |
o-xylene |
p-xylene |
ethylbenzene |
|
(mg/m3) |
(% mean) |
(mg/m3) |
(% mean) |
(mg/m3) |
(% mean) |
(mg/m3) |
(% mean) |
|
1st set
1
2
3
mean
2nd set
1
2
3 |
443.9
428.5
447.4
439.9
444.3
453.4
449.5 |
101.0
103.1
102.2 |
203.9
194.6
206.1
201.5
203.9
208.3
207.2 |
101.2
103.4
102.8 |
192.6
185.7
194.1
190.8
192.4
196.7
195.0 |
100.8
103.1
102.2 |
158.6
154.8
159.5
157.6
159.2
161.9
159.5 |
101.0
102.7
101.2 |
|
Low relative humidity
The ability of charcoal tubes to collect mixed xylenes at low humidity was tested by
sampling a test atmosphere containing 468 mg/m3, 218
mg/m3, 204 mg/m3, 166
mg/m3 m-xylene, o-xylene,
p-xylene, and ethylbenzene, respectively (at 5% RH and 20 °C) with
three samplers for four hours at 50 mL/min. The samples were analyzed immediately. The
sample results (when compared to theoretical concentrations) were 102.0, 100.4, and
99.8% for m-xylene; 101.4, 100.4, and 99.2% for o-xylene; 101.6, 100.1,
and 99.6% for p-xylene; and 102.5, 100.8, and 100.4% for ethylbenzene.
Low concentration
The ability of charcoal tubes to collect mixed xylenes at low concentrations was tested
by sampling a test atmosphere containing 22 mg/m3, 10
mg/m3, 10 mg/m3, 8
mg/m3 m-xylene, o-xylene,
p-xylene, and ethylbenzene, respectively (at 80% RH and 22 °C)
with three samplers for four hours at 50 mL/min. The samples were analyzed immediately.
The sample results (when compared to theoretical concentrations) were 95.2, 97.6, and 102.2
for m-xylene; 94.6, 96.1, and 101.2% for o-xylene; 95.0, 97.2, and 101.8%
for p-xylene; and 96.1, 98.6, and 102.5% for ethylbenzene.
Interference
The ability of charcoal tubes to collect mixed xylenes in the presence of sampling
interferences was tested by sampling a test atmosphere containing 230
mg/m3, 107 mg/m3, 100
mg/m3, 82 mg/m3, 365
mg/m3, 372 mg/m3
m-xylene, o-xylene, p-xylene,
ethylbenzene, toluene, and butyl acetate respectively (at 81% RH and 21 °C) with
three samplers for four hours at 50 mL/min. The samples were analyzed immediately.
The sample results (when compared to theoretical concentrations) were 103.3, 102.9,
and 101.8 for m-xylene; 102.8, 102.5, and 101.2% for o-xylene; 103.2,
102.6, and 101.6% for p-xylene; and 104.1, 103.4, and 102.6% for ethylbenzene.
4.9.2 SKC 575-002 Passive Samplers
Reverse diffusion
The ability of SKC 575-002 Passive Samplers to retain mixed xylenes after
collection was tested by sampling a test atmosphere containing 454 mg/m3,
211 mg/m3, 198 mg/m3, and
161 mg/m3 m-xylene, o-xylene,
p-xylene, and ethylbenzene, respectively (at 80% RH and 20 °C) with six
samplers for one hour. Three samples were analyzed immediately and three were used to
sample contaminant-free humid air for an additional three hours, and then
analyzed. Sampling rates from Section 4.7 were converted to their equivalents under
experimental temperature and pressure and used to calculate results in Table 4.9.2. All
the samples in the second set retained at least 100.2, 99.6, 99.8, and 100.0% of the means
of the first set for m-xylene, o-xylene, p-xylene, and
ethylbenzene, respectively.
Table 4.9.2
Retention of Mixed Xylenes on SKC 575-002 Passive Samplers
|
|
m-xylene |
o-xylene |
p-xylene |
ethylbenzene |
|
(mg/m3) |
(% mean) |
(mg/m3) |
(% mean) |
(mg/m3) |
(% mean) |
(mg/m3) |
(% mean) |
|
1st set
1
2
3
mean
2nd set
1
2
3 |
450.6
449.4
419.5
439.8
440.7
449.0
449.4 |
100.2
102.1
102.2 |
206.9
208.0
191.9
202.3
201.4
206.0
205.9 |
99.6
101.8
101.8 |
195.7
197.0
182.0
191.6
191.2
195.0
195.3 |
99.8
101.8
101.9 |
161.2
162.8
149.9
158.0
157.9
160.8
161.4 |
100.0
101.8
102.2 |
|
Low relative humidity
The ability of SKC 575-002 Passive Samplers to collect mixed xylenes at
low humidity was tested by sampling a test atmosphere containing 468 mg/m3,
218 mg/m3, 204 mg/m3, 166
mg/m3 m-xylene, o-xylene,
p-xylene, and ethylbenzene, respectively (at 5% RH and 20 °C) with three
samplers for four hours. The samples were analyzed immediately. Sampling rates (760 mmHg
and 25 °C) were 13.92, 13.63, and 13.94 mL/min for m-xylene; 14.23, 13.93,
and 14.24 mL/min for o-xylene; 14.05, 13.77, and 14.08 mL/min for p-xylene; and
14.01, 13.73, and 14.02 mL/min for ethylbenzene.
Low concentration
The ability of SKC 575-002 Passive Samplers to collect mixed xylenes at
low concentrations was tested by sampling a test atmosphere containing 22 mg/m3,
10 mg/m3, 10 mg/m3, 8 mg/m3
m-xylene, o-xylene, p-xylene, and ethylbenzene,
respectively (at 80% RH and 22 °C) with three samplers for four hours. The samples
were analyzed immediately. Sampling rates (760 mmHg and 25 °C) were 13.44, 13.68,
and 13.16 mL/min for m-xylene; 13.90, 14.28, and 13.53 mL/min for o-xylene;
13.32, 13.69, and 13.36 mL/min for p-xylene; and 13.71, 13.76, and 13.37 mL/min
for ethylbenzene.
Interference
The ability of SKC 575-002 Passive Samplers to collect mixed xylenes in
the presence of sampling interferences was tested by sampling a test atmosphere containing
230 mg/m3, 107 mg/m3,
100 mg/m3, 82 mg/m3,
365 mg/m3, 372 mg/m3
m-xylene, o-xylene, p-xylene,
ethylbenzene, toluene, and butyl acetate respectively (at 81% RH and 21 °C) with three
samplers for four hours. The samples were analyzed immediately. Sampling rates (760
mmHg and 25 °C) were 13.73, 13.12, and13.77 mL/min for m-xylene; 14.06, 13.43, and
14.08 mL/min for o-xylene; 13.86, 13.25, and 13.90 mL/min for p-xylene; and
13.80, 13.18, and 13.86 mL/min for ethylbenzene.
4.10 Qualitative analysis
The identity of suspected mixed xylenes can be confirmed by GC/mass spectrometry.
Mass spectra for the analytes are presented below.
Figure 4.10.1 Mass spectrum for m-xylene. |
|

Figure 4.10.2 Mass spectrum for o-xylene. |
|
Figure 4.10.3 Mass spectrum for p-xylene. |
|
Figure 4.10.4 Mass spectrum for ethylbenzene. |
1. Kirk-Othmer Encyclopedia of Chemical Technology; 3rd ed.; Grayson, M Ed.; John Wiley & Sons: New York, 1984, Vol. 24; pp. 709-744.
2. Documentation of the Threshold Limits Values and Biological and Indices, 6th ed., American Conference of Governmental Industrial Hygienists, Inc: Cincinnati, OH, 1991, Vol. III, pp. 187-193.
3. OSHA Analytical Methods Manual; Vol. 1; U.S. Department of Labor, Occupational Safety and Health Administration; Directorate for Technical Support, OSHA Salt Lake Technical Center: Salt Lake City, UT, 1990; Method 7: Organic Vapors; American Conference of Governmental Hygienists (ACGIH): Cincinnati, OH; Publication No. 4542.
4. NIOSH Manual of Analytical Methods, 4th ed.; U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health: Cincinnati, OH, 1996, Method 1501: Hydrocarbons, Aromatic; (NIOSH) Cincinnati, OH; Publication No. 94-113.
5. Documentation of the Threshold Limits Values and Biological and Indices, 6th ed., American Conference of Governmental Industrial Hygienists, Inc: Cincinnati, OH, 1991, Vol. III, pp. 1732-1740.
6. Documentation of the Threshold Limits Values and Biological and Indices, 6th ed., American Conference of Governmental Industrial Hygienists, Inc: Cincinnati, OH, 1991, Vol. I, pp. 581-584.
7. 1998 TLVs and BEIs, Threshold Limit Values for Chemical Substances and Physical Agents, ISBN: 1-882417-23-2; American Conference of Governmental Industrial Hygienists, (ACGIH): Cincinnati, OH, 1998.
8. Kirk-Othmer Encyclopedia of Chemical Technology, 3rd ed.; Grayson, M Ed.; John Wiley & Sons: New York, 1984, Vol. 24; pp. 709-744.
9. ACS Publications;
Production growth sputtered in most sectors (accessed March 1999).
10. Kirk-Othmer Encyclopedia of Chemical Technology, 3rd ed.; Grayson, M Ed.; John Wiley & Sons: New York, 1984, Vol. 24; pp. 709-744.
11. The Condensed Chemical Dictionary, 8th ed.; Revised by Hawley, G., Ed., Van Nostrand Reinhold: New York, 1971, p. 358, 942, 943.
12. The Condensed Chemical Dictionary, 8th ed.; Revised by Hawley, G., Ed., Van Nostrand Reinhold: New York, 1971, p. 358, 942, 943.
13. Kirk-Othmer Encyclopedia of Chemical Technology, 3rd ed.; Grayson, M Ed.; John Wiley & Sons: New York, 1984, Vol. 24; pp. 709-744.
14. OSHA Computerized Information System Database, Chemical Sampling Information, Salt Lake Technical Center, Occupational Safety and Health Administration, Salt Lake City, UT March 1999.
15. OSHA Computerized Information System Database, Chemical Sampling Information, Salt Lake Technical Center, Occupational Safety and Health Administration, Salt Lake City, UT March 1999.
16. Kirk-Othmer Encyclopedia of Chemical Technology, 3rd ed.; Grayson, M Ed.; John Wiley & Sons: New York, 1984, Vol. 24; pp. 709-744.
17. Documentation of the Threshold Limits Values and Biological and Indices, 6th ed., American Conference of Governmental Industrial Hygienists, Inc: Cincinnati, OH, 1991, Vol. II, pp. 581-584.
18. Documentation of the Threshold Limits Values and Biological and Indices, 6th ed., American Conference of Governmental Industrial Hygienists, Inc: Cincinnati, OH, 1991, Vol. III, pp. 1732-1740.
19. Documentation of the Threshold Limits Values and Biological and Indices, 6th ed., American Conference of Governmental Industrial Hygienists, Inc: Cincinnati, OH, 1991, Vol. I, pp. 581-584.
20. The Condensed Chemical Dictionary, 8 th ed.; Revised by Hawley, G., Ed., Van Nostrand Reinhold: New York, 1971, p. 358, 942, 943.
21. Burright, D.; Chan, Y.; Eide, M.; Elskamp, C.; Hendricks, W.; Rose, M. "Evaluation Guidelines for Air Sampling Methods Utilizing Chromatographic Analysis", OSHA Salt Lake Technical Center, U.S. Department of Labor: Salt Lake City, UT, 1999 (in-revision).
22. Burright, D.; Chan, Y.; Eide, M.; Elskamp, C.; Hendricks, W.; Rose, M. "Evaluation Guidelines for Air Sampling Methods Utilizing Chromatographic Analysis", OSHA Salt Lake Technical Center, U.S. Department of Labor: Salt Lake City, UT, 1999 (in-revision).
|
| |

|

