|
|
 |
Malaria Vaccine Development Branch |
|
Product Development Timeline
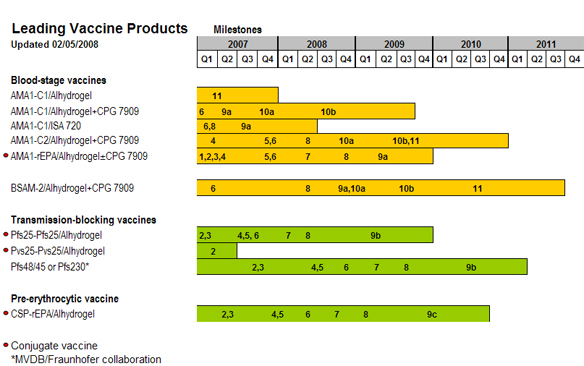
Milestones
Discovery Milestones
- Rationale for candidate antigen
- Viable process for candidate antigen production
- Resources to facilitate development of investigational vaccine based on candidate antigen
Preclinical Milestones
- Preclinical animal studies establishing potency parameters (e.g., ELISA, GIA, TBA)
- cGMP production of drug substance (MCB, PCB, manufacture and testing): release of drug substance
- Formulation optimization (including conjugation), cGMP production of drug product (manufacture and testing): release of drug product
- GLP toxicity study of the drug product in an appropriate animal model: no signs of general toxicity
Clinical Milestones
- U.S. FDA effective Investigational New Drug Application
- Malaria-Naïve Phase I Trial
- Blood-stage vaccine, malaria-naïve Phase I trial in adults: safe and immunogenic (ELISA)
- Transmission-blocking vaccine, malaria-naïve Phase I trial in adults: safe and immunogenic (TBA)
- Pre-erythrocytic vaccine, malaria-naïve Phase I/IIa trial in adults: safe, immunogenic, and protective
- Malaria-Exposed Phase I Trial
- Blood-stage vaccine, malaria-exposed Phase I trial in adults: safe
- Blood-stage vaccine, malaria-exposed Phase I trial in children or infants: safe and immunogenic (ELISA)
- Blood-stage vaccine, malaria-exposed Phase II trial in children or infants: safe, immunogenic, and biologic impact
- Transmission-blocking vaccine, malaria-exposed village Phase I/II/III trial: safe and immunogenic (TBA)
back to top
|
|