Speech before
NACDS/HDMA RFID Healthcare Adoption Summit
Remarks by
Randall Lutter, Ph.D.
Associate Commissioner
Food and Drug Administration
November 14 , 2005
This text contains Dr. Lutter's prepared remarks. It
should be used with the understanding that some material may have been
added or deleted during actual delivery.
Introduction
- I am very pleased to have this opportunity to speak at the first,
and hopefully not the last NACDS and HDMA Summit on the adoption of Radio
Frequency Identification technology in health care. We at FDA congratulate
HDMA and NACDS for organizing this meeting and greatly appreciate your
ongoing commitment and work to advance current and emerging technology
and business solutions--such as RFID--to combat counterfeit drugs and thereby
improve the integrity and safety of the U.S. drug supply.
- RFID is more than just a means to control counterfeit drugs. In
the broader scheme it is a 21st century technology to ensure patient safety. As
makers, distributors, and dispensers of drug products, you have an obligation
to maintain and guarantee the integrity of the drug products that you sell. RFID
can both help you to meet this obligation and help us ensure that all drugs
are safe and effective.
- I am here to tell you that FDA
shares your hopes about the promise that RFID brings for the distribution
and delivery of health care products into the 21st Century. We also share some of your concerns
about the need for coordinated action to overcome obstacles in implementing
this technology.
- Dr. Andy von Eschenbach, our Acting Commissioner is engaged, knowledgeable,
interested, and supportive of RFID.
- Successful adoption of electronic track and trace technologies
like RFID will require unusually high levels of cooperation among all stakeholders
in the manufacture and distribution of medical products. Meetings
like this are essential to foster and cultivate the necessary cooperation.
Without cooperation among stakeholders the full promise of RFID can not
be realized. While investments in some RFID technology may well make
sense for some individual companies, the net benefits that all stakeholders
will enjoy from RFID will be maximized only if independent organizations
adopt common standards and compatible approaches.
- Today I would like first to discuss the policy problem that electronic
track and trace technologies such as RFID can help solve, at least from
the perspective of FDA. Increasingly sophisticated counterfeit drug
organizations have posed an increasing threat to the integrity of the drug
supply in the U.S. Lack of information in the drug distribution system
is hindering efforts to ensure that each patient gets the right drug in
the right amount at the right time.
- Second, I would like to summarize for you the steps that FDA has
taken recently to remedy this problem. These include the February
2004 Report on Combating Counterfeit Drugs, and our efforts to encourage
the voluntary adoption of RFID during the stay until December 2006, of
the pedigree regulations issued under the Prescription Drug Marketing Act.
- And finally, I will tell you what our plans are for helping drive
adoption of an electronic pedigree and RFID and ensuring that Americans
have the greatest possible assurance that their medications are safe and
effective.
- But before proceeding, let me share with you a vision about how
the distribution of medical products could occur in the near future, if
we take the right steps today.
- We at FDA envision a world where devious would-be peddlers of
diverted or fake medicines are thwarted and unable to sell to unsuspecting
U.S. wholesalers, pharmacists and, most importantly, patients.
- We envision pharmacists at drug stores and hospitals being certain
of the safety and efficacy of drugs they are dispensing, including their
appropriateness for particular patients.
- This certainty comes not merely because the label conveys FDA
approval but because an up-to-date electronic database confirms that
the particular package contains the genuine authentic product--it was
tracked at each stage of the distribution chain, from the FDA-approved
manufacturing facility, to the dispensing pharmacist.
- Further, the electronic track and trace system that will provide
this certainty, offers such savings to private firms that the benefits
of greater certainty about safety and efficacy are realized without any
untoward increase in the costs of medications at a retail level.
- This final point is important. While fighting counterfeit
drugs is a key part of FDA’s mission to ensure drug safety, we
acknowledge important public concerns about the cost of medications and
the implications of high costs for access to drugs. Based on discussions
with some drug companies and retailers, we believe that RFID can offer
significant savings in the form of better inventory management to manufacturers,
wholesalers and retailers alike. Other savings would stem from
reduction in theft and product loss, improved recalls, and reduction
in paperwork burdens. While desire for these cost savings is understandably
the key motivation for your pursuit of RFID, our interests overlap. Thoughtful
adoption of RFID, while helping you financially, will also offer a lower
cost way of ensuring authenticity of drugs thereby providing key support
for our fight against diversion and counterfeiting.
- Let me elaborate why implementing this vision is important to combating
the problem of counterfeit drugs.
The Counterfeit Drug Problem
- But before describing the counterfeit drug problem, let me make
a general comment: While we are here to discuss steps to improve
the integrity of the U.S. drug supply it is important to note how good
it already is. Counterfeit drugs in the U.S. are quite rare.
- While we have no direct quantitative evidence about the prevalence
of counterfeit drugs, because they so successfully mimic genuine products
and by all accounts are rare, we are confident that the overwhelming
majority of prescription drugs sold in the U.S. are products genuinely
approved by FDA.
- We believe counterfeit drugs represent significantly less than
one percent of the total U.S. drug supply.
- The high confidence that we and the public have about the distribution
system for U.S. drug products stems from an intricate web of federal
and state laws. These laws require drugs to be treated as potentially
dangerous consumer goods that require regulations and oversight by health
professionals in order to protect the public health.
- Despite this high confidence, FDA has been concerned that the
drug supply is under increasing threat of attack from ever more sophisticated
counterfeiters. This disturbing trend is evident in the increased
efforts to introduce counterfeit drugs into the U.S. market.
- The Agency has witnessed an increase in counterfeiting activities
and a greater capacity to introduce finished dosage form counterfeits
into legitimate drug distribution channels. Illicit wholesale drug
diverters and others in the supply chain provide the window through which
most counterfeit drugs have historically entered legitimate distribution
channels.
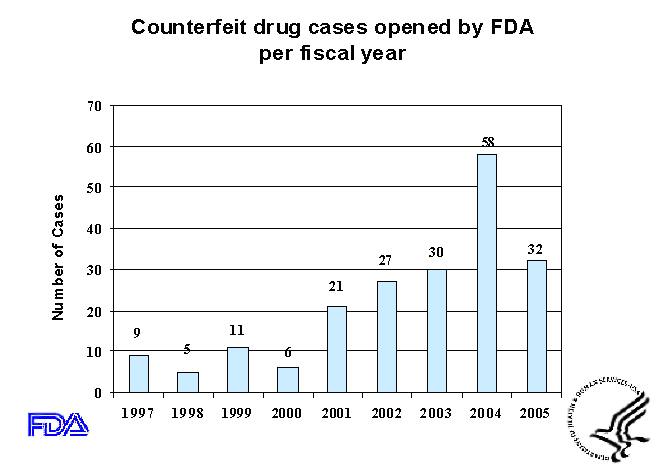
- As you can see from Slide 1, the number of newly initiated counterfeit
drug cases has risen sharply from just a few years ago, although preliminary
data from fiscal year 2005 suggest a decline relative to the peak reported for
fiscal year 2004. In fiscal year 2004, FDA's Office of Criminal Investigations
(OCI) initiated 58 counterfeit drug cases, a significant increase from the 30
cases initiated in FY 2003.
D
- Let me stress that these are estimates of the number of newly
initiated counterfeit drug cases being investigated. Since these
are ongoing cases, we have no estimate of the volume of counterfeit drugs
involved in each case--it could vary from dozens to thousands.
- We believe that the unusually high number of cases in FY2003
is in part due to an increased awareness and vigilance at all levels
of the drug distribution chain. One factor contributing to this
increased awareness and vigilance is the Counterfeit Drug Report that
FDA issued in February 2004. A second is increased referrals from,
and coordination with other state and federal law-enforcement agencies,
and communications with drug manufacturers.
- The decline from 58 cases in 2004 to 32 new cases in 2005 may
be due partly to a positive deterrent effect of the new 2004 cases on
the level of counterfeiting in 2005. But the decline may also exist
because some new cases, after further research, are found to relate to
the large number of existing ones. In reality we can not
identify the exact causes of changes in the number of new counterfeit
drug cases opened each year.
- Fortunately, most of the counterfeit drugs at issue did not reach
consumers because we focused our resources and developed proactive investigations. We
believe that this strategy enabled us to identify components of counterfeit
products and interdict finished counterfeit drug products before they
entered retail distribution.
- It is important to note that the number of cases that OCI has
opened is NOT an indication of the prevalence of drug counterfeiting
in the U.S. Almost 4 billion prescriptions were filled last year. That
means a very large volume of drugs is moving through the supply chain. The
sophistication and precision of counterfeit copies make a reliable estimate
of the number impossible. However, we believe that existing regulations
and the commitments by most supply chain stakeholders to comply with
these rules, keep very low the prevalence of drug counterfeiting in the
U.S.
- Unfortunately, not everyone abides by the rules. Counterfeit,
stolen, and otherwise fraudulently obtained pharmaceutical drugs can
enter legitimate channels through pre-existing illicit diversion networks. OCI’s
enforcement efforts targeting these diverters also have resulted in detection
and dismantling of counterfeit schemes. Without the intimate
knowledge of diversion borne of extensive investigative experience it
would be difficult, if not impossible, to effectively combat pharmaceutical
counterfeiting.
- Counterfeit drugs in other countries are a much bigger threat than
in the U.S. Strong enforcement efforts are the key to keeping counterfeits
off the U.S. market.
FDA’s Recent Activities
- FDA has tried to implement the Prescription Drug Marketing Act
while encouraging the voluntary adoption of anti-counterfeiting technologies
and practices.
- For those of you who are not familiar with PDMA, the Prescription
Drug Marketing Act is a law passed in 1987 in response to a number of counterfeit
drug incidents in the U.S. where patients received counterfeit drugs.
- PDMA requires State licensing of wholesale distributors of prescription
drugs and requires unauthorized wholesale distributors to provide purchasers
a statement (also called a pedigree) identifying each prior sale of the
drug. FDA issued final regulations implementing the PDMA in 1999.
- Shortly thereafter, the agency received comments raising a number of
concerns related to the pedigree provisions. Many of the concerns suggested
that there would be an adverse economic impact on wholesalers who have to provide
pedigrees. Comments noted the
high costs of using paper pedigrees--the best technology then available—and
the inability of wholesalers would be unable to get complete pedigrees
from sellers, even for legitimate transactions. As a result,
FDA stayed certain provisions of the final rule, and has continued
to stay these provisions. The current stay, which expires in
December 2006, was issued in part to give stakeholders time to implement
an electronic track and trace technology solution.
- Our statements to stakeholders in the February 2004 report were
- Adoption and common use of reliable track and trace technology
would be feasible in 2007, and would help secure the integrity of the
drug supply chain by providing an accurate drug “pedigree”.
- RFID is the most promising approach to provide reliable and
timely track and trace information.
- Adoption of electronic track and trace technology would help
stakeholders meet and surpass the goals of PDMA.
- Since February 2004 we have engaged in a variety of efforts to
cultivate RFID implementation/adoption.
- We are working closely with individual companies and supply
chain partnerships who are implementing or piloting RFID, including
several companies that are here today.
- EPCglobal has kindly welcomed FDA to meetings of its Health
and Life Sciences Business Action Group, where much of the standard
setting work is being done. We are pleased by the progress that
they are making in this area.
- We have led an FDA RFID working group, in which all FDA centers
are represented, information is shared within the agency, and consistent
approaches are identified and pursued.
- We have also served on an inter-governmental RFID Council,
coordinated by the Department of Defense. We participate in 4
sub-working groups on this panel, including privacy, standards, technology
and policy.
- In our February 2004 report, we took an essentially voluntary
approach toward widespread adoption of electronic track and trace.
Supply chain stakeholders assured us that there would be considerable movement
toward implementation of RFID and that widespread adoption could be
done in 2007. We believed at that time regulatory intervention might
stifle innovation and progress in adopting this emerging technology. Yet
from our vantage point today, it appears a voluntary approach may not
be enough.
- In an annual update in May of this year we said that “stakeholders
have made tremendous progress in the development and implementation of
EPC/RFID [(electronic product code for unit serialization)]. This
is a huge endeavor that requires close collaboration among all constituents
of the pharmaceutical distribution system. ”
- We also said that “we are pleased with the progress stakeholders,
standard setting bodies, and software and hardware companies have made
thus far toward implementing an electronic pedigree for drug products. We
realize that there have been, and continue to be, challenges along
the way. However, we are optimistic that this progress will continue
in an expeditious manner toward meeting our 2007 goal. If it
appears that his goal will not be met, we plan to consider the options
regarding implementation of the PDMA provisions that are the subject
of the stay.”
- At this point we have become concerned about the slow or inadequate progress
implementing an electronic pedigree. We are concerned that widespread adoption
may not be realized by December 2006, when the PDMA stay expires.
Next Steps
- Let me turn now to our plans to renew and reinvigorate our support
for RFID so as to accelerate its adoption.
- First, FDA, given its responsibilities for drug safety, must
ensure that radio-frequency exposure does not impact the quality, safety,
or efficacy of drug and biologic products.
- We asked stakeholders to share data and information regarding
the effects of RF on drug and biologic products. We haven’t
received significant amount of data to date.
- We believe, based on preliminary information and hypothesis,
that RF does not have a significant thermal effect on solid dosage
forms.
- An FDA CDRH lab has done some preliminary research looking
at the heating effect of 915 MHz RFID fields on a placebo, simulating
a pharmaceutical. The research showed a very modest rise in temperature,
of unknown significance.
- This small bit of information shows that we are just beginning
to know the impact of RF on certain products. This has given
us the impetus to develop and conduct our own studies to determine
the impact of RF on a range of products.
- We are developing protocols for further research, but we intend
to look at RF effects on various types of drug, biologics, and various
types of packaging materials under a variety of conditions. Effects
we will consider will include
- Thermal effects (the impact of heat), and
- Non-thermal effects (the impact on molecular bonds).
- We expect to share the results in spring or summer 2006.
- Second, I am pleased to announce here that the FDA Counterfeit
Drug Task Force is going to hold a Public Workshop in January or February
2006.
- The new chairpersons of the Task Force are Maggie Glavin, Associate
Commissioner for Regulatory Affairs and me. The purpose of the meeting
is to --
- Facilitate RFID standard-setting and coordination of issues,
- Discuss PDMA/pedigree issues related to the stay, and
- Reaffirm our commitment to facilitate and drive adoption
of electronic track/trace technology.
- Our general interest is in identifying the current barriers
to adoption and finding ways these barriers can be overcome. We
haven’t yet set the agenda, but I can sketch here some issues
that we’ve heard merit further public discussion. These
include:
- What type of number should be used as the unique identifier: the
embedded NDC number or a randomly generated number to protect privacy
and mask product identity;
- Should all of the transaction information be stored and accessible
at a single central database or a distributed network of information. Does
it really matter if it’s one or the other?
- What common fields/information should be included in an electronic
pedigree?
- What incentives are needed for more rapid and widespread
adoption?
- What can FDA do to further facilitate/drive adoption across
the supply chain?
- In the coming weeks, we will put out an invitation for people
to testify on these and other issues and we expect to publish more
information about this meeting in the near future both in the Federal
Register and posted on FDA’s website.
- We have been advocating the adoption and widespread use of electronic
track and trace technologies for almost 2 years now. We know that
many of you have been involved in exploring RFID technology for significantly
longer than that. With our February 2004 report, we gave this technology
a big push for use in the pharma sector. Let me be clear
that this continues to be an Agency priority and we are prepared to give
this another big push to keep the momentum moving quickly.
- I believe that the steps that I have shared with you today—the
public workshop and any follow up from that and the RFID research that
we will be conducting, as well as our continued interactions and discussions
with you and your companies, demonstrate our commitment to push this
forward.
- We want this to succeed and we continue to believe that widespread
adoption is feasible in the very near future
Thanks again for giving me this opportunity to speak. I have
a few minutes for questions.
 Get free
weekly updates about FDA press releases, recalls, speeches, testimony
and more.
Get free
weekly updates about FDA press releases, recalls, speeches, testimony
and more.





![]()
![]() Get free
weekly updates about FDA press releases, recalls, speeches, testimony
and more.
Get free
weekly updates about FDA press releases, recalls, speeches, testimony
and more.
![]()