1. General Discussion
1.1 Background
1.1.1 History of procedure
This evaluation was undertaken because the OSHA Salt Lake Technical Center laboratory received samples requesting
the analysis of methidathion. This report describes the analytical method developed for the sampling and the
analysis of methidathion.
1.1.2 Toxic effects (This section is for information only and should not be taken as the basis of OSHA policy.)
Methidathion is a hydrazine derivative (thiadiazole). Hydrazine derivatives retain some of hydrazine's toxicity.
This is the basis for perhaps the fastest growing use of hydrazine derivatives as pesticides in agriculture.
Hundreds, perhaps thousands, of hydrazine derivatives have been suggested or patented for pesticidal applications.
(Ref.5.1) Methidathion is also an organophosphate (cholinesterase-inhibiting) insecticide and its toxicity is
related to the organophosphate properties.
Symptoms of acute exposure
Symptoms of organophosphate poisoning include headache, giddiness, blurred vision, weakness, nausea, diarrhea,
cramps, discomfort in the chest, nervousness, sweating, miosis (pinpoint), tearing, salivation, uncontrollable
muscle twitches, convulsions, coma, and loss of reflexes and sphincter control. If swallowed and aspirated into
lungs, chemical pneumonia can occur. (Ref. 5.2)
Chronic toxicity studies
In long term feeding studies with mice, rats and dogs, high doses of Methidathion Technical caused hepatotoxic
effects. In mice, lifetime feeding at high doses produced liver tumors. In a reproductive study with rats,
cholinesterase inhibition caused impaired mating performance, decreased pup body weights and decreased pup
viability at the highest dose level administered. Methidathion was not teratogenic in rats or rabbits, and various
mutagenicity studies show that methidathion is not genotoxic. (Ref. 5.2)
1.1.3 Potential workplace exposure
Methidathion is an insecticide and acaricide. It is used to control alfalfa weevils and certain other insects in
alfalfa, scales in citrus, spider mites, bollworms, budworms, lygus bug, pink bollworms, and whiteflies in cotton.
It is also used on sunflower, artichokes, apples, almonds, cherries, apricots, pears, nectarines, plums, prunes,
walnuts, peaches and pecans. (Ref. 5.3) There was no information available on the number of workers potentially
exposed to methidathion.
1.1.4 Physical properties (Refs. 5.3 and 5.4 unless otherwise noted)
| Chemical name: |
O,O-dimethyl phosphorodithioate, S-ester with 4-(mercaptomethyl)-2-methoxy- 21,3,4-thiadiazolin-5-one; Phosphorodithioic acid O,O-dimethyl ester S-ester with
4-(mercaptomethyl)-2-methoxy- 21,3,4-thiadiazolin-5-one; Phosphorodithioic acid O,O-dimethyl ester S-ester with
4-(mercaptomethyl)-2-methoxy- 21,3,4-thiadiazolin-5-one; dithiophosphoric acid O,O'-dimethyl-S-[(2-methoxy-1,3,4-thiadiazol-5(4H)-on-4-yl)-methyl]
ester; dithiophosphoric acid O,O'-dimethyl-S-[(5-methoxy-1,3,4-thiadiazol-2(3H)-one-3-yl)methyl] ester; O,O'-dimethyl-S-[(2-methoxy-1,3,4-thiadiazole-5(4H)-one-4-yl)methyl]
dithiophosphate 21,3,4-thiadiazolin-5-one; dithiophosphoric acid O,O'-dimethyl-S-[(2-methoxy-1,3,4-thiadiazol-5(4H)-on-4-yl)-methyl]
ester; dithiophosphoric acid O,O'-dimethyl-S-[(5-methoxy-1,3,4-thiadiazol-2(3H)-one-3-yl)methyl] ester; O,O'-dimethyl-S-[(2-methoxy-1,3,4-thiadiazole-5(4H)-one-4-yl)methyl]
dithiophosphate |
| Common name: |
Methidathion |
| Synonyms: |
Ultracide; Supracide; somonil; Fisons NC 2964; DMTP; ENT 27193 |
| CAS number: |
950-37-8 |
| Molecular formula: |
C6H11N2O4PS3 |
Structural formula: |
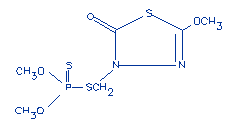 |
| Molecular weight: |
302.31 |
| Melting point: |
39-40°C |
| Vapor pressure: |
3.33 x 10-7 kPa (2.5 x 10-6 mmHg) at 25°C (technical) |
| Solubility: |
Readily soluble in benzene, acetone, methanol, xylene and other organic
solvents; solubility in water less than 1% |
| Description: |
Colorless crystals |
| Stability: |
Relatively stable to hydrolysis in neutral or slightly acidic media, less
stable in more acidic (pH 1) or alkaline media (pH 13, 50% loss in 30 min @ 25°C.) (Ref. 5.5) |
|




