|
Interim Timeline
Actions to Protect Public Health: XDR/MDR TB
Case
October 9, 2007
This is an archived document. The links
and content are no longer being updated.
PDF version
formatted for print - 103MB
Text version available below
Text Version
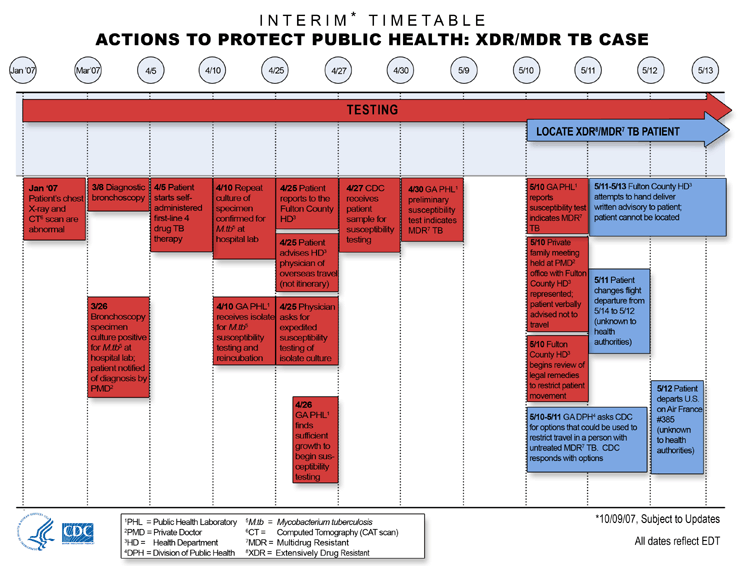
January
- Testing: Patient's chest X-ray and CT scan are
abnormal.
March 8
- Testing: Diagnostic bronchoscopy.
March 26
- Testing: Bronchoscopy specimen culture positive
for Mycobacterium tuberculosis at hospital lab;
patient notified of diagnosis by private medical doctor (PMD).
April 5
- Testing: Patient starts self-administered
first-line 4-drug TB therapy.
April 10
- Testing: Repeat culture of specimen confirmed for
Mycobacterium tuberculosis at hospital lab.
- Testing: Georgia Public Health Laboratory
receives isolate for Mycobacterium tuberculosis
susceptibility testing and reincubation.
April 25
- Testing: Patient reports to the Fulton County
Health Department.
- Testing:
Patient advises health department physician of overseas
travel (not itinerary).
- Testing:
Physician asks for expedited susceptibility testing of
isolate culture.
April 26
- Testing: Georgia Public Health Laboratory finds
sufficient growth to begin susceptibility testing.
April 27
- Testing: CDC receives patient sample for
susceptibility testing.
April 30
- Testing: Georgia Public Health Laboratory
preliminary susceptibility test indicates MDR TB.
May 10
- Testing: Georgia Public Health Laboratory
susceptibility test indicating multidrug-resistant
tuberculosis (MDR TB) reported to Fulton County Health
Department.
- Testing: Private family meeting held at private
doctor's office with Fulton County Health Department
represented. Patient verbally advised not to travel.
- Testing: Fulton County Health Department begins
review of legal remedies to restrict patient movement.
May 10-11
- Locate XDR/MDR TB
Patient: Georgia Department of
Public Health asks CDC for options that could be used to
restrict travel in a person with untreated MDR TB. CDC
responds with options.
May 11-13
- Locate XDR/MDR TB
Patient: Fulton County Health
Department attempts to hand deliver written advisory to
patient. Patient cannot be located.
May 11
- Locate XDR/MDR TB
Patient: Patient changes flight
departure from May 14 to May 12 (unknown to health
authorities).
May 12
- Locate XDR/MDR TB
Patient: Patient departs U.S.
on Air France #385 (unknown to health authorities).
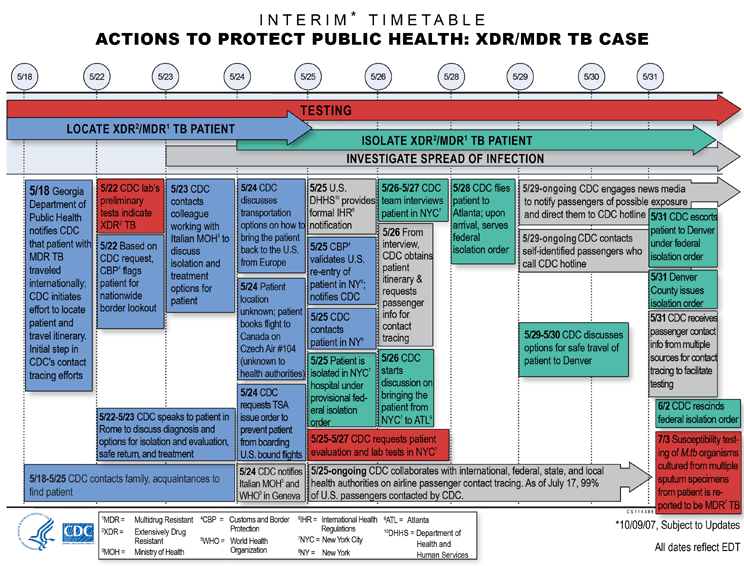
May 18
- Locate XDR/MDR TB Patient: Georgia Department of Public Health notifies CDC that patient with MDR TB traveled
internationally; CDC initiates effort to locate patient and
travel itinerary. This is the initial step in CDC's contact
tracing efforts.
May 18-25
- Locate XDR/MDR TB
Patient: CDC contacts family,
acquaintances to find patient.
May 22
- Testing: CDC lab's preliminary tests indicate
extensively drug-resistant tuberculosis (XDR TB).
- Locate XDR/MDR TB
Patient: Based on CDC request,
U.S. Customs and Border Protection (CBP) flags patient for
nationwide border lookout.
May 22-23
- Locate XDR/MDR TB
Patient: CDC speaks to patient
in Rome to discuss diagnosis and options for isolation and
evaluation, safe return to U.S., and treatment.
May 23
- Locate XDR/MDR TB
Patient: CDC contacts colleague
working with Italian Ministry of Health (MOH) to discuss
temporary isolation and treatment options for patient in
Rome.
May 24
- Locate XDR/MDR TB
Patient: CDC discusses transportation options on how to bring the patient
back to the U.S. from Europe.
May 24
- Locate XDR/MDR TB
Patient: Patient location
unknown. Patient leaves Rome on newly booked flight to
Montreal, Canada on Czech Air #104 (unknown to health
authorities).
- Locate XDR/MDR TB
Patient:
CDC
requests Transportation Security Administration (TSA) issue
order to prevent patient from boarding U.S. bound flights.
May 24
- Investigate Spread of Infection: CDC notifies
Italian Ministry of Health authorities and World Health
Organization (WHO) in Geneva.
May 25
- Investigate Spread of Infection: U.S. Department
of Health and Human Services (DHHS) provides formal
International Health Regulation (IHR) notification.
- Locate XDR/MDR TB
Patient: Customs and Border Protection (CBP) validates U.S.
re-entry of patient in New York (NY) by automobile; notifies
CDC.
- Locate XDR/MDR TB
Patient:
CDC contacts patient in New York.
- Isolate XDR/MDR TB
Patient: Patient is isolated
in New York City (NYC) hospital under provisional federal
isolation order.
May 25-27
- Testing: CDCs requests patient
evaluation and lab tests in New York City.
May 25 - ongoing
- Investigate Spread of Infection: CDC collaborates
with international, federal, state, and local health
authorities on airline passenger contact tracing. As of July
17, 99% of U.S. passengers contacted by CDC.
May 26-27
- Isolate XDR/MDR TB
Patient: CDC team interviews
patient in New York City.
May 26
- Investigate Spread of Infection: From interview,
CDC obtains patient itinerary and requests passenger info
for contact tracing.
- Isolate XDR/MDR TB Patient: CDC starts discussion
on bringing the patient from NYC to Atlanta.
May 28
- Isolate XDR/MDR TB
Patient: CDC flies patient to Atlanta; upon arrival, serves federal isolation order.
May 29 - ongoing
- Investigate Spread of Infection:
CDC engages news media to notify passengers of possible exposure
and direct them to CDC hotline.
- Investigate Spread of Infection: CDC contacts
those self-identified passengers who call CDC hotline.
May 29-30
- Isolate XDR/MDR TB
Patient: CDC discusses options for safe travel of patient to TB treatment
facility in Denver.
May 31
- Isolate XDR/MDR TB
Patient: CDC escorts patient
to TB treatment facility in Denver by air ambulance under
federal isolation order.
- Isolate XDR/MDR TB
Patient: Denver County issues
isolation order.
- Investigate Spread of Infection: CDC receives
passenger contact info from multiple sources for contact
tracing to facilitate testing.
June 2
- Isolate XDR/MDR TB
Patient: CDC rescinds federal
isolation order.
July 3
- Testing: Susceptibility testing of
Mycobacterium tuberculosis organisms cultured from
multiple induced sputum specimens from the patient is
reported to be MDR TB.
Last Reviewed: 05/18/2008
Content Source: Division of Tuberculosis Elimination
National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention
Back to Top of Page
|
You will need Adobe Acrobat™ Reader
v5.0 or higher to read pages that are in PDF format. Download the Adobe Acrobat™ Reader.
If you have difficulty accessing any
material on the DTBE Web site because of a disability, please contact
us in writing or via telephone and we will work with you to make the information
available.
Division of Tuberculosis Elimination
Attn: Content Manager, DTBE Web site
Centers for Disease Control and Prevention
1600 Clifton Rd., NE Mailstop E-10
Atlanta, GA 30333
CDC-INFO at (1-800) 232-4636
TTY: 1 (888) 232-6348
E-mail: tbinfo@cdc.gov
|
 Home | Site Map
| Contact Us
Home | Site Map
| Contact Us
Accessibility
| Privacy Policy Notice |
FOIA
| USA.gov
CDC Home |
Search |
Health Topics A-Z
Centers for Disease Control & Prevention
National Center for HIV/AIDS,
Viral Hepatitis, STD, and TB Prevention
Division of Tuberculosis Elimination
Please send comments/suggestions/requests to: CDCINFO@cdc.gov
|