Nanotechnology: Should carbon nanotubes be handled in the workplace like asbestos?
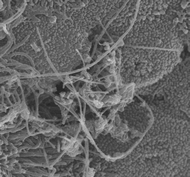 Multi-walled carbon nanotubes in conducting airways of the mouse lung after inhalation exposure.
Multi-walled carbon nanotubes in conducting airways of the mouse lung after inhalation exposure.Photo credit: Robert Mercer
Nanotechnology poses a question for occupational health and safety professionals. Does this technology, and the tiny nanoparticles that are its tools, pose an unintended risk of illness or injury for workers employed in the industry?
The National Institute for Occupational Safety and Health is at the forefront of the effort to understand the health and safety ramifications of working with nanomaterials. There have been an increasing number of scientific publications from the research community at large—including a new study issued just this week—that address one type of nanomaterial in particular, carbon nanotubes, and seek to determine if they biologically behave like asbestos. That is, if inhaled, are carbon nanotubes likely to cause irreparable and fatal effects such as those associated with asbestos exposure? The effects of asbestos include severe lung fibrosis or scarring, lung cancer, including cancer of the lining of the lungs or pleura called mesothelioma.
The question of a comparison between carbon nanotubes and asbestos arises for several reasons. Some varieties of carbon nanotubes are similar in shape to asbestos fibers, and like asbestos, some varieties of carbon nanotubes have been shown in laboratory studies to persist in the lungs of laboratory animals. Some animal studies have even shown effects similar to those of asbestos.
Carbon nanotubes are tiny, cylindrical, manufactured forms of carbon. There is no single type of carbon nanotube. One type can differ from another in terms of shape (single-walled or multi-walled) or in chemical composition (pure carbon or containing metals or other materials). Carbon nanotube exposures can potentially occur not only in the process of manufacturing them, but also at the point of incorporating these materials into polymer composites, medical nanoapplications, and electronics.
The release of two recent reports, one from a research lab in Japan (Takagi et al., J Toxicol Sci 33:105-116, 2008) and one—this week—from the United Kingdom (Poland et al., Nature Nanotechnology advance online publication, 20 May 2008;[DOI10.1038/nnano.2008.111]) contribute to the carbon nanotube/asbestos fiber comparison debate. The publication of the Japanese study contributed to the decision by the Japanese Ministry of Health, Labor and Welfare to issue a notice which instructs those involved in the manufacture, repair and inspection of nanomaterials that the processes should be carried out under either sealed, unattended or automated conditions, or a local exhaust system should be installed.1, 2, 3 The recently published British study adds to the body of work showing an asbestos-like response (more below).
What are the implications of the most recent finding to the risk assessment and risk management of carbon nanotubes in U.S. workplaces?
The question of whether carbon nanotubes pose a toxicological hazard has been investigated since at least 2003. A challenge has been in determining if carbon nanotube materials used in the workplace have the same characteristics as those associated with biological responses in laboratory studies. Earlier studies used materials with high levels of other forms of carbon such as carbon black and high levels of metal catalyst.
Carbon nanotubes can vary widely in diameter, length, number of layers, and structures. They can also vary widely in surface composition, since certain carbon nanotubes may be "coated" with specific metals or other materials in order to perform specific functions. Also, they can clump together or agglomerate, which can affect their potential for settling in the lungs if inhaled, their ability to penetrate the body's membranes and consequently move from the lungs to other organs, and their interaction with cells and tissue. Such variations bring an additional degree of complexity to risk assessment analysis for carbon nanotubes.
In 2005, NIOSH researchers showed that aspiration of single-walled carbon nanotubes in mice caused progressive fibrosis and granuloma formation.4 (Fibrosis and granuloma reduce gas-exchange area in the lung, thus making breathing difficult.) It is estimated that one month of exposure to carbon nanotubes at the airborne concentration of 5 milligrams per cubic meter of air, or mg/m3, would yield an equivalent dose in workers to that causing fibrosis in the mouse.5 The 5 mg/m3 concentration is sometimes reported on material safety data sheets as a manufacturer's suggested exposure limit for carbon nanotubes and is based on the permissible occupational exposure limit (PEL) for graphite, whose most commonly known use is as a powder for manufacturing pencils. However, the findings reported by NIOSH in 2005 suggest that, on the basis of the effects seen in the laboratory studies, it might not be appropriate to use the graphite PEL for carbon nanotubes. In fact, BSI British Standards Published Document PD 6699-2:2007 "Guide to safe handling and disposal of manufactured nanomaterials" suggests setting a benchmark exposure limit for nanoscale fibers longer than 5 µm to one tenth of the PEL for asbestos, that is at 0.01 fiber per cubic centimeter. However, this benchmark exposure limit, described as a "pragmatic guidance level only," was not rigorously developed and is derived "on the assumption that the hazard potential of the nanoparticle form is greater than the large particle form."
This weeks' British study finds that long (that is, longer than 20 micrometer or µm) multi-walled carbon nanotubes exhibit asbestos-like response in the form of injury to the linings of the body cavity in laboratory mice, while short and tangled multi-walled carbon nanotubes do not. A multi-walled carbon nanotube is composed of several nanotubes on a common axis. The Japanese study reports that multi-walled carbon nanotubes were more potent in causing mesothelioma than asbestos in laboratory mice genetically modified to be prone to cancer.
Asbestos-like responses to carbon nanotubes may not be entirely surprising to scientists, given previous toxicological and epidemiological studies of other biopersistent fibers since such studies show that once fibers are deposited in the lung, they stay there.6 However, questions have been raised about using these research findings for risk assessment analysis in the light of study limitations such as use of model animals, artificial administration methods, and sometimes extremely high doses, which are not representative of those exposures usually present in the workplace environment. Such limitations are not unusual for pioneering scientific studies. They simply mean that at this stage of the research, gaps remain that need to be closed by further study before quantitative risk assessment can be conducted.
How do we protect workers today?
In the workplace, developing and implementing a workplace risk management program (including evaluating the hazards, assessing worker exposures, installing and evaluating engineering controls, establishing procedures for personal protective equipment, and providing worker education and training programs) can minimize worker exposure to carbon nanotubes. NIOSH recommends that such prudent practices be used while scientists continue the research that is needed for better risk assessment. (See NIOSH Approaches to Safe Nanotechnology.) Use of basic engineering control systems such as enclosures and local exhaust ventilation was shown to greatly reduce exposure levels, while filters used in HVAC systems and respirators were reported to capture nanoscale particles with stated levels of efficiency.7 In addition, medical screening is part of a complete safety and health management program, and established medical surveillance approaches can help to assess whether control measures are effective and identify new or unrecognized problems and health effects.8
While the mechanisms of biological responses to carbon nanotubes are not yet fully understood, recent studies such as those from Japan and the UK add to the growing body of peer-reviewed scientific literature and remind the occupational safety and health community that carbon nanotubes should be handled prudently to minimize potential exposures in the workplace and to prevent potential adverse health effects in workers.
—Vladimir V. Murashov, Ph.D.
Dr. Murashov is a Special Assistant for Nanotechnology to the NIOSH Director. He is a member of the U.S. Nanoscale Science, Engineering, and Technology subcommittee. He also leads projects for the ISO Technical Committee 229 (Nanotechnologies) and the Organization for Economic Cooperation and Development's Working Party on Manufactured Nanomaterials.
Visit the NIOSH website for more information on nanotechnology research at NIOSH.
Endnotes
- External link: http://wwwhourei.mhlw.go.jp/hourei/doc/tsuchi/200207-a00.pdf
- External link: http://www.jniosh.go.jp/joho/nano/index.html
- External link: techon.nikkeibp.co.jp/english/NEWS_EN/20080305/ 148469/?ST=english_PRINT
- Shvedova et al. Am J Physiol Lung Cell Mol Physiol 289(5):L698-708, 2005
- Ibid.
- NIOSH Asbestos Road Map
- NIOSH Approaches to Safe Nanotechnology
- Interim Guidance for the Medical Screening of Workers Potentially Exposed to Engineered Nanoparticles
See the Comment Policy



Comments
NIOSH is the United States government agency with legislative responsibility to recommend occupational health standards to OSHA. Without a REL, this post is of minimal value to practitioners.
The post is also deficient for failing to note that laboratory bioassays of carbon black provide clear evidence of carcinogenicity, that IARC has found sufficient evidence in laboratory studies that carbon black is carcinogenic, and therefore classified carbon black as possibly carcinogenic to humans, group 2A. NIOSH has a responsibility to opine whether carbon nanotubes are at a minimum carbon black, and whether the bioassay and IARC designation must be disclosed on MSDS of materials containing > .1% carbon nanotubes.
Posted 5/26/08 at 9:45 am
Thank you for your comment. NIOSH is exploring the development of Recommended Exposure Limits (RELs) for carbon nanotubes and will request information on the topic from the public in the near future. Carbon particles need to be treated on a case by case basis. One type of carbon material does not predict how another material will behave regarding carcinogenicity. Hence, they most likely should be considered separately when developing RELs. For example, as mentioned in the blog, data on graphite (a carbon polymorph) may not be applicable to carbon nanotubes.
Posted 6/09/2008 at 10:58 am
This study does not consider the nature of the catalysts incorporated into the body along with the tubes. The catalysts used in this partiular study were, for some reason, nickel, cobalt and combinations of nickel with cobalt. Nanotubes both mulitwall or single wall can be grown with iron catalysts so these was not necessary inclusions. Since nickel and cobalt are both known to cause a very strong mutagenic reponse perhaps this is what they were seeing. In addition it is well known that CNTs can have adsorbed high molecular weight hydrocarbons some of which also will cause a mutagenic response. THis is also true of other forms of carbon unless special means are used to remove these species. Aside from these two effects, the electronic nature of CNTs is related to their Chirality, again this was not a controlled variable.
Posted 6/05/2008 at 11:29 am
We have limited experience with measurements in a nanotube production facility. We measure very low concentrations (expressed as a surface area per unit volume)in the fraction below 1 µm. I suspect coagulation of the tubes in the vortex of the cyclone.
We intend to sample a coarser fraction (still in the respirable range). This coagulation would suggest that similar processes would occur in the respiratory system??
Posted 6/20/2008 at 6:24 am
Agglomeration of carbon nanotubes can occur as they move in the air including air in the lung. Nanotube agglomeration can affect their potential for settling in the lungs if inhaled, their ability to penetrate the body's membranes and consequently move from the lungs to other organs, and their interaction with cells and tissue.
Posted 6/23/2008 at 3:16 pm
From a chemical point of view carbon nano-tubes are part of the family of polycyclic aromatic condensed rings (see Polycyclic aromatic hydrocarbons (PAHs) at http://en.wikipedia.org/wiki/Polycyclic_aromatic_hydrocarbon ). These molecules are able to form stable "radicals" that can chemically "interact" with DNA and are capable of altering the chemical structure of DNA promoting DNA miss-functioning and cancer formation (see for ex. Benzopyrene http://en.wikipedia.org/wiki/Benzopyrene). Polycyclic aromatic hydrocarbons are formed during the synthesis of CNTs and are "absorbed" on the internal and external surfaces of the nano-tubes. These substances can be released when CNTs came in contact with human body. Also directly CNTs are able to for"very stable radicals" that could interact with DNA. More deep investigations are needed before use CNTs on a large scale.
Posted 7/22/2008 at 4:32 am
Carbon nanotubes and PAHs. (see comment by Enrico Costantini)
Can the same thing not be said about common graphite? Graphite can also be considered as condensed rings, with that difference, compared to nanotubes, that the surface is more "flat".
Posted 7/25/2008 at 4:51 am
Concerning Nanotechnology: With its medical aplication and its ability to monitor the human body, is it detectable with a simple blood test? If not, what type would be required? If your agency is not able to answer this question, where would I be able to find this answer? My question is derived from personal research and an airing from the ABC Today show. The airing of this show was the week of nov. 5th to nov 12th 2007
Posted 8/9/2008 at 10:45 pm
NIOSH is the federal agency responsible for conducting research and providing guidance to protect workers from risks they face on the job. NIOSH is carrying out a program of research on nanotechnology applications to be able to make science-based recommendations to protect workers who manufacture or use nanomaterials on their job. NIOSH believes that answers to the following occupational questions are essential to understanding these implications and applications:
However, NIOSH has not conducted specific research on biomonitoring for engineered nanomaterials, such as conducting blood tests for engineered nanoparticles.
Other agencies that may be able to address your question on blood tests for nanomaterials are:
Posted 8/12/2008 at 9:23 am