 |
 |
FDA
Home Page | Search FDA Site |
FDA A-Z Index | Contact FDA
![]()
Cradas | Patents | Licensing
Our purpose is to provide current policies and procedures to assist the FDA community and potential collaborators to develop and transfer federal technology to the commercial marketplace.
The Federal Technology Transfer Act of 1986 (FTTA) which amended the Stevenson-Wydler Technology Innovation Act of 1980; Executive Order 12591 of April 10, 1987; the National Technology Transfer Advancement Act of 1995, and other Public Health Service (PHS) policies
encourage greater interactions and collaborations among Federal laboratories, state and local governments, universities, and the private sector.
FTTA created the mechanism for such collaborations, the Cooperative Research and Development Agreement (CRADA), and provided incentives for
The FTTA makes it each technical employee's responsibility as well as the Agency's management structure to develop opportunities to transfer technology. The Agency continues to strongly support collaborative research and development under CRADAs which are within FDA's mission and that may result in patents and licenses which promote public health.
FDA has internal procedures for establishing CRADAs; filing Invention Reports; and, through a formal agreement with NIH's Office of Technology Transfer, licensing patented inventions and biological materials. We also have a Fair Access and Conflict of Interest policy to safeguard the Agency and the scientist while fostering collaborative efforts. All these topics are addressed in the selections which follow.
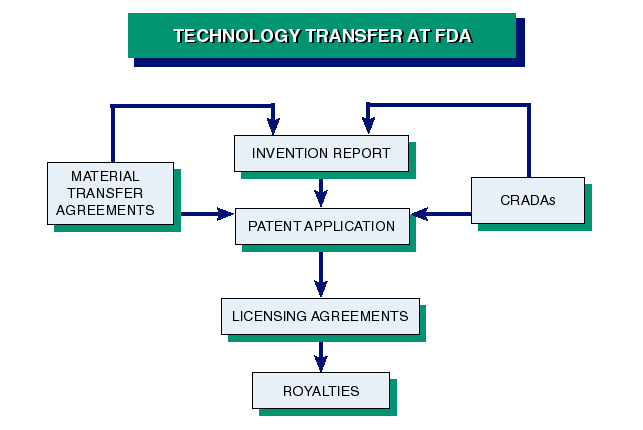
Technology transfer in FDA and throughout the Federal Government encourages partnerships for collaborative research and development. Such partnerships may strengthen the Agency's research efforts and increase resources for mission-related projects while benefiting the public, the agency, and the scientist. Fair access and conflict of interest policies apply.
![]()