Preventable Adverse Drug
Reactions:
A Focus on Drug Interactions
This learning module was developed based on a needs survey sent to all third year
medicine clerkship directors and all medicine residency program directors
in the United States. This
module was developed by the Center
for Education and Research on Therapeutics (CERT) while at
Georgetown
University
(CERT now located at the
University of Arizona Health Sciences
Center) in collaboration with the Center for Drug Evaluation and
Research at the Food and Drug Administration.
The work was sponsored by the Agency for Healthcare
Research and Quality (AHRQ). We encourage you to complete
the Module Evaluation Form, as it will provide feedback for the
development of future learning modules.
Welcome to the Adverse Drug Reaction (ADR) learning
module. The module will begin with a presentation
of a case that was published in 1990. This
case demonstrates why it is important that health
care practitioners report ADRs to the Food and
Drug Administration (FDA). It was also a pivotal
case resulting in recognition and definition of
one type of preventable adverse drug reaction— drug
interactions mediated by the cytochrome P450
pathway of drug metabolism.
After discussing this case, we will
discuss the prevalence and incidence
of adverse drug reactions. We will
then examine several well-recognized types
of drug interactions that are the causes of preventable
adverse reactions. This section will focus
primarily on cytochrome P450-mediated drug
interactions, although other types of interactions will
also be included, as well as examples of drug-drug,
drug-diet, and drug-herbal interactions. The
emphasis will be on current knowledge that can
help healthcare providers predict possible drug interactions.
This will be followed by a discussion of
ADR reporting via the FDA’s MedWatch program. Finally,
a stepwise systems approach to preventing
ADRs due to drug interactions will be
outlined.
Sample Case

The first case we will consider is that of the potentially
lethal arrhythmia, torsades de pointes, occurring
in association with terfenadine (Seldane) use
in a young woman.1
This ECG is a classic example of torsades
de pointes, which is French for
"twisting of the points."
Torsades is a form of ventricular tachycardia that
can most often be due to medications. The
QRS complexes during this rhythm tend to show
a series of "points up" followed by "points down"
often with a narrow waist between. Recognition and
reporting of this arrhythmia in association with terfenadine, astemizole (Hismanal), cisapride
(Propulsid),
grepafloxacin (Raxar), and mibefradil (Posicor) ultimately led to the removal
of these medications from the market.
1Monahan BP, Ferguson CL, Cleave
ES, Lloyd BK, Troy J, Cantilena LR. Torsade de pointes occurring in
association with terfenadineuse. JAMA 1990;264:2788–2790.

A 39-year-old female was evaluated for episodes of
syncope and light-headedness that began two days
prior to her hospital admission.1
The history was
consistent with possible cardiovascular causes, and
the patient was admitted and placed on telemetry where
the preceding rhythm strip was observed.
Ten days prior to admission she had been
prescribed terfenadine (Seldane—an antihistamine) 60
mg twice-a-day and cefaclor (Ceclor—a cephalosporin
antibiotic) 250 mg three-times-a-day. On
the eighth day of terfenadine therapy the patient
began a self-medicated course of ketoconazole (Nizoral—an
azole antifungal) at 200 mg twice-a-day
for vaginal candidiasis. She was also taking
medroxyprogesterone acetate at a dosage of
2.5 mg a-day. Upon admission to the
hospital the patient was noted to have
a QTc interval of 655 milliseconds (normal
is less than 440 milliseconds). During the hospitalization
the patient experienced near syncopal episodes
associated with torsades de pointes noted
on telemetry.
After discontinuing the medications, the QTc interval
normalized. She had no further episodes of
torsades de pointes, and she was discharged with
no recurrence of syncope.
1Monahan BP, Ferguson CL, Cleave
ES, Lloyd BK, Troy J, Cantilena LR. Torsade de pointes occurring in
association with terfenadineuse. JAMA 1990;264:2788–2790.
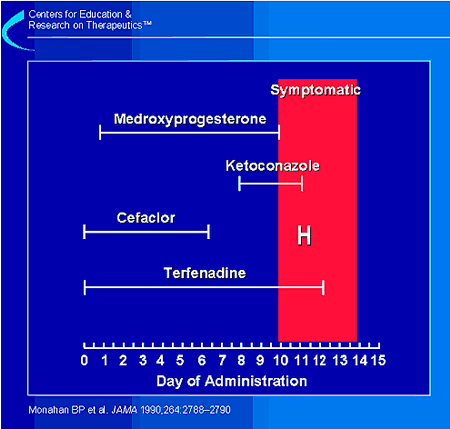
This figure illustrates the time course of the medications that
the patient took.1 In
relation to when the symptoms started,
the most recently prescribed drug was
ketoconazole. Ketoconazole has not been
associated with development of torsades de pointes
when used by itself. How did
ketoconazole interact with terfenadine to cause
QT prolongation and torsades de pointes in this
patient? That question will be answered during the
course of this module.
1Monahan BP, Ferguson CL, Cleave
ES, Lloyd BK, Troy J, Cantilena LR. Torsade de pointes occurring in
association with terfenadineuse. JAMA 1990;264:2788–2790.
ADRs:
Prevalence and Incidence

The first question healthcare providers should ask themselves
is "why is it important to learn about ADRs?"
The answer is because ADRs are one of the
leading causes of morbidity and mortality in health
care. The Institute of Medicine reported in January of 2000 that from
44,000 to 98,000 deaths occur annually
from medical errors.1 Of
this total, an estimated 7,000 deaths
occur due to ADRs. To put this in
perspective, consider that 6,000 Americans die
each year from workplace injuries.
However, other studies conducted on
hospitalized patient populations have
placed much higher estimates on the
overall incidence of serious ADRs. These
studies estimate that 6.7% of hospitalized patients
have a serious adverse drug reaction with
a fatality rate of 0.32%.2 If
these estimates are correct, then
there are more than 2,216,000 serious
ADRs in hospitalized patients, causing over
106,000 deaths annually. If true, then ADRs are
the 4th leading cause of death—ahead of pulmonary
disease, diabetes, AIDS, pneumonia, accidents,
and automobile deaths.
These statistics do not include the number of ADRs
that occur in ambulatory settings. Also, it is
estimated that over 350,000 ADRs occur in U.S. nursing
homes each year.3 The
exact number of ADRs is not certain
and is limited by methodological considerations.
However, whatever the true number is,
ADRs represent a significant public health problem
that is, for the most part, preventable.
1Committee on Quality of Health Care
in America: Institute of Medicine.
To err is human: building a safer health system. Washington, D.C.:
National
Academy Press; 2000. 2Lazarou J, Pomeranz B, Corey PN. Incidence of adverse drug reactions in hospitalized
patients: A
meta-analysis of prospective studies. JAMA 1998;279:1200–1205. 3Gurwitz JH, Field TS,
Avorn J, McCormick D, Jain S, Eckler M, et al. Incidence and preventability of
adverse drug events in nursing homes. Am
J Med 2000;109(2):87–94.

We can next ask ourselves, what are the health care
costs associated with adverse drug reactions? Again,
methodological constraints limit making completely
accurate estimates, but one estimate of the
cost of drug-related morbidity and mortality is $136
billion annually,1 which
is more than the total cost of
cardiovascular or diabetic care in the United
States. In addition, one out of 5 injuries or deaths
per year to hospitalized patients may be as a
result of ADRs.2 Finally,
a two-fold greater mean length of
stay, cost and mortality has been reported for
hospitalized patients experiencing an ADR compared
to a control group of patients without an
adverse drug reaction.3
1Johnson JA, Bootman JL. Drug-related morbidity and mortality. A cost-of-illness model.
Arch
Intern Med 1995;155(18):1949–1956. 2Leape LL,
Brennan TA Laird N, Lawthers AG ,Localio AR, Barnes BA et al. The nature of
adverse events in hospitalized patients. Results of the Harvard Medical
Practice Study II. N Engl J Med 1991;324(6):377–384. 3Classen DC
, Pestotnik SL, Evans RS, Lloyd JF, Burke JP. Adverse drug events in hospitalized
patients. Excess length of stay, extra costs, and attributable mortality. JAMA
1997;277(4):301–306.

Why are there so many ADRs? There are many reasons.
Here are just a few.
First, more drugs—and many more
combinations of drugs—are being used
to treat patients than ever before. To
exemplify this point, 64% of all patient
visits to physicians result in prescriptions.1
Secondly, 2.8 billion prescriptions were
filled in the year 2000. 2 That is about 10 prescriptions for every person in the United States.
Finally, the rate of ADRs increases
exponentially after a patient is on 4
or more medications.3
Efforts to reduce polypharmacy are
important but for many patients, the
number of medications cannot always
be reduced without doing harm. That is
why it is important to understand the basis for drug
interactions. This will allow us to make the most
appropriate choices in prescribing and avoiding
preventable ADRs.
1Schappert SM.
Ambulatory care visits to physician offices, hospital outpatient
departments, and emergency departments: United States,1997. National Center
for Health Statistics. Vital Health
Stat. 1999;13(143).
2National Association of Chain Drug Stores.
2000 community pharmacy results. 2001. Alexandria, VA. 3Jacubeit
T, Drisch D, Weber E. Risk factors as
reflected by an intensive drug monitoring system. Agents
Actions 1990;29:117–125.

It is worth considering
how well a drug’s safety is
defined prior to its approval and marketing. This
will indicate how confident practitioners can
be that a new drug’s safety profile has been fully
defined.
Most new drugs are
approved with an average of
1,500 patient exposures and usually for only relatively
short periods of time. However, some drugs
cause serious ADRs at very low frequencies and
would require many more exposures to detect
the reaction. For example, bromfenac (Duract)
was a non-steroidal anti-inflammatory agent
(NSAID) that was removed from the market in
1998, less than 1 year after it was introduced. Bromfenac
caused serious hepatotoxicity in only 1
in 20,000 patients taking the drug for longer than
10 days.1 To
reliably detect the toxic effects of
a drug with a 1 in 20,000 adverse drug reaction frequency,
the new drug application database would
have to include 100,000 patient exposures. A
drug that is tested in a few thousand people 11
may have an excellent safety
profile in those few thousand
patients. However, within a short time after
entering the market, the drug may be administered
to several million patients. That means
that for drugs that cause rare toxicity, their
toxicity can only be detected after, not before,
marketing.
If one case of
hepatotoxicity is seen during pre-marketing
testing, it can be difficult, if not impossible,
to ascertain whether it was secondary to
the drug or just the background rate of disease that
is seen in the population.
So, the safety profile for
new drugs that come on
the market is never totally defined because new
drugs are studied only in relatively small and
homogenous patient populations. The complete safety
profile of a new drug will be defined only
after it has been approved and is in use on the
market.
1Friedman MA,
Woodcock J, Lumpkin MM, Shuren JE, Hass AE, Thompson LJ. The safety of
newly approved medicines: do recent market removals mean
there is a problem? JAMA
1999;
281(18):1728–1734.

Health care
providers have misconceptions about reporting
ADRs.1–3 These misconceptions include the
ideas that: 1) All serious ADRs are documented by
the time a drug is marketed; 2) It is hard to determine
if a drug is responsible for the ADR; 3)
ADRs should only be reported if absolute certainty exists
that the ADR is related to a particular drug;
and, finally, 4) One case reported by an individual physician
does not contribute to medical knowledge.
Let’s look at each one of these points.
1) As we have
seen, rare ADRs are usually NOT documented
by the time a drug is marketed.
2) It can be hard
to determine if an individual drug
caused a reaction in a complicated patient receiving
multiple medications. However, the temporal
relationship of a reaction with regard to
the administration of a new medication can be
helpful. Also, biological plausibility (asking if
the drug’s mechanism of action makes this possible
or likely) can also be helpful. The bottom line
is, even when in doubt about whether a
drug caused the reaction, report it.
3) A
suspicion of an adverse drug reaction should be
reported. A health care provider does not have
to be absolutely certain that a drug caused a
reaction. All reports contribute to the heightening of
the awareness of FDA safety scientists as
they monitor all of the evidence to evaluate the
potential for drug-related toxicity.
4) One individual
report CAN make a difference. Many
drug withdrawals began with one clinical report
that initiated further investigation. In the example
case in this module, a single report ultimately
led to the removal of terfenadine from
the market. This report potentially saved many
lives and led to a better understanding of the
mechanism involved in causing torsades de pointes.
Almost all drugs are now evaluated prior
to being released on the market for their potential
to induce cardiac arrhythmias, also as
a result of this single case report.
1Figueiras A, Tato F,
Fontainas J, Gestal-Otero JJ. Influence of physicians’ attitudes on
reporting adverse drug events: a case-control study. Med
Care 1999;37(8):809-814. 2Eland IA, Belton KJ, van Grootheest AC, Meiners AP, Rawlins MD,
Stricker BH. Attitudinal survey of voluntary
reporting of adverse drug reactions. Br
J Clin Pharmacol 1999;48(4):623–627. 3Chyka PA, McCommon
SW. Reporting of adverse drug reactions by poison control centres in the
US. Drug Saf 2000;23(1):87–93.

The inability of the FDA to effectively warn health care
providers and patients about drug interactions and
our inability to translate existing knowledge into
changes in prescribing have resulted in huge economic
consequences for the pharmaceutical industry
and the loss from the marketplace of effective
drugs, including terfenadine, mibefradil, astemizole, and cisapride.
These 4 drugs were removed from the market or restricted in their use because it became
clear that they continued to be
prescribed in an unsafe manner, even
after multiple warning letters were disseminated
by the manufacturer and the FDA to health
care professionals concerning their proper use.
Each of these drugs has value in the pharmaceutical marketplace,
and each has value to patients.
However, because the manufacturer and the
FDA could not prevent co-prescription of these drugs
with interacting drugs resulting in fatal interactions,
the risk associated with continued widespread
availability could not be justified.
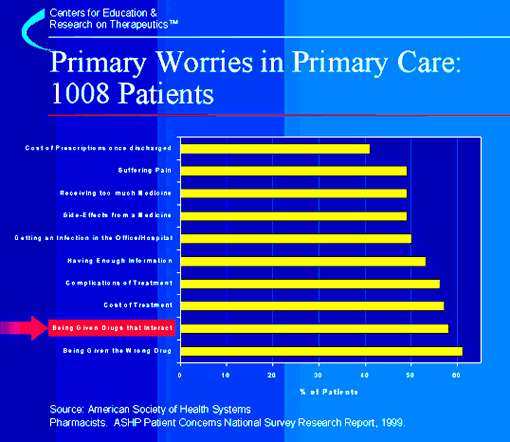
This figure shows data from a national survey conducted
in 1999 by the American Society of Health
Systems Pharmacists (ASHP)1 that
evaluated patient concerns about
health systems. This was a random
telephone survey of 1,008 adults. Although
the respondents were very concerned about
suffering from pain and the cost of filling prescriptions,
they were most concerned about being
given the wrong drug or that a drug interaction would
occur. The public in general has a much greater
level of concern about ADRs than most health
care providers would suspect. These
data demonstrate that drug interactions and reactions
are not only a concern to health care providers
but to patients as well.
1American Society of Health Systems Pharmacists.
ASHP Patient Concerns National Survey Research Report. 1999. Bethesda, MD.
Types
of Drug Interactions

The previous slides have reviewed information about
the magnitude of adverse drug reactions and the
burden they place on the health care system. How
much do drug interactions contribute to the total
number of preventable ADRs?
Again, estimates of the numbers of patients injured
due to drug interactions vary widely. However,
some reasonable estimates come from the
work of Dr. Lucien Leape and colleagues.1
In a systems analysis of
ADRs, they estimated that drug-drug
interactions represent from 3–5% of all in-hospital
medication errors. Drug interactions are
also an important cause of patient visits to emergency
departments.2
1Leape LL, Bates DW, Cullen
DJ, Cooper J, Demonaco HJ, Gallivan T, et al. Systems analysis of adverse drug events.
ADE Prevention Study Group. JAMA 1995;274(1):35–43. 2Raschetti
R, Morgutti M, Menniti-Ippolito F, Belisari A, Rossignoli A,
Longhini P, et al. Suspected
adverse drug events requiring emergency department visits or hospital
admissions. Eur J Clin Pharmacol 1999;54(12):959–963.

Recent publications have shown that many adverse
drug reactions can be prevented and detected
through the use of systems interventions. For
example, many health systems have instituted new
technologies to minimize patient injury due to
medication errors and drug-drug interactions.1–3
Tools like computerized physician order and
prescription entry 1
and bar coding systems 3 have demonstrated
tangible benefits. The potential for reducing
medication errors by using computerized medical
records as well as drug-interaction screening software
that detects and alerts the physician and/or
pharmacist to potentially serious drug interactions
has been recognized.4
These technological solutions do have
limitations, however. The
fragmentation of healthcare delivery may
result in incomplete records. More significant is
the fact that, although this information is avail-able, it
is not uniformly or optimally incorporated into
decision making. This is exemplified in the observation
by Cavuto et al. that pharmacists filled prescriptions
for drug combinations that were known
to interact even though computerized drug interaction
software was in place.5 This
problem persists as shown in the 2000
paper by Smalley et al. on
prescription errors with cisapride.6
These findings should reinforce the need
for the health care practitioner to
develop their own systems approach to
prescribing without creating undesirable
drug interactions. A fundamental understanding
of the clinical pharmacology of drug
interactions and a framework for avoiding preventable
drug interactions remains critically important.
Thus we need to overlay technologic solutions
on a base that is strong in basic principles of
clinical pharmacology and drug interactions. Incorporation
of up-to-date computerized databases is
valuable, and frequent consultation with other members
of the healthcare team, such as nurses and
pharmacists, is essential.
1Bates DW,
Leape LL, Cullen DJ ,Laird N, Petersen LA, Teich JM et al. Effect of computerized physician
order entry and a team intervention on prevention of serious medication
errors. JAMA 1998;280(15):1311–1316. 2Evans RS,
Pestotnik SL, Classen DC, Horn SD, Bass SB, Burke JP. Preventing adverse drug events in
hospitalized patients. Ann
Pharmacother 1994;28(4):523–527. 3Gebhart F. VA facility slashes drug
errors via bar-coding. Drug Topics 1999;1:44. 4Committee on Quality of Health
Care in America: Institute of
Medicine. To err is human: building a safer health system. Washington, D.C.:
National Academy Press,2000. 5Cavuto NJ, Woosley RL, Sale M.
Pharmacies and prevention of potentially fatal drug interactions. JAMA 1996;275: 1086–1087.
6Smalley W, Shatin D, Wysowski DK, Gurwitz J Andrade SE, Goodman M, et al. Contraindicated use of
cisapride: impact of food and drug administration regulatory action. JAMA
2000;284(23):3036–3039.

We will discuss an approach to prescribing drugs in
ways that avoid adverse drug interactions as a cause
for preventable medication errors.
Drug interactions can occur via several
mechanisms:
• Drugs interactions can occur even before drugs enter
the body due to formulation incompatibility, or
at any point in the process of absorption, distribution metabolism, and
elimination.
• Drugs can bind to each other in the GI tract, preventing
absorption, and reducing systemic availability.
• In theory, drugs could interact in the plasma via protein-bumping
reactions but, despite the emphasis placed
on these in many texts and pharmacology courses,
there are no known clinically relevant examples
in which this mechanism is responsible.
• A large number of important interactions do occur
in the liver and GI tract due to changes in
the rates of drug metabolism brought about by
other medicines that are inducers or inhibitors of
drug metabolism. We will be looking at this topic
in depth.
• A few interactions occur through competition at drug
transporters.
• Finally, interactions can occur at the level of drug action,
such as the combination of verapamil, a calcium
channel blocker, and a beta-blocker. Both slow
the heart rate by different mechanisms, and the
combination is relatively contraindicated because
heart block can result. Because of this interaction
many textbooks and computer pro-grams warn
against concomitant use of any beta-blocker and
any calcium channel blocker. This creates
a great deal of confusion and distrust of drug
interaction warnings, because most health care
providers know that drugs in these two classes
are often employed successfully and safely
in patients with hypertension.

The next few slides will review some of the mechanisms
for drug interactions in more detail. Several
examples of drug interactions that occur prior
to drug administration are listed here. When
phenytoin is added to solutions of dextrose, a precipitate forms and the
phenytoin falls to the bottom
of the IV bag as an insoluble salt. When this happens,
it is no longer available to control seizures. Amphotericin
is still used widely as a urinary bladder
perfusion to treat aggressive fungal infections.
If it is administered in saline, the drug precipitates
and can erode through the bladder wall if
not removed. The clinical presentation of such
cases is an acute abdomen due to perforation of
the bladder.1 Lastly,
it is recommended that aminoglycosides not
be co-mixed in IV fluids with betalactam antibiotics.
This can markedly reduce antibiotic efficacy.
1Personal
Communication, David
Flockhart, MD, PhD, University of Indiana, July 2001.

A number of interactions occur in the GI tract and
reduce the entry of drugs into the systemic circulation.
Particularly notable among these is the
ability of aluminum-containing medicines
such as sucralfate (Carafate) and antacids to reduce the absorption of expensive and potentially life-saving
antibiotics like ciprofloxacin (Cipro) and azithromycin (Zithromax).
Women taking iron supplements often do not
consider them as a medicines, and should
be specifically questioned about whether they
are taking iron if they are to be prescribed a quinolone
or azithromycin. Drugs such as ketoconazole (Nizoral)
and delavirdine (Rescriptor) require an acidic environment to be in the
non-charged form that is preferentially
absorbed. Solubility is drastically
reduced in neutral or basic medications
such as omeprazole (Prilosec),
lansoprazole (Prevacid), or H2-antagonists that raise
the stomach’s pH.

Some drugs can "bump" other drugs off proteins in
the plasma and result in an increased amount of
free drug, but this is only transient because the usual
elimination mechanisms respond by increasing the
rate of elimination. There is no clinically relevant protein-bumping interaction that has
been reported. The previously cited
examples have subsequently been shown to
be due to inhibition of elimination, not
plasma protein displacement.
Drug Metabolism

The next few slides will focus on drug metabolism. Some
important preventable drug interactions are due
to their effects on drug metabolizing enzymes, resulting
in either inhibition (reduced activity) of the
enzyme or induction (increased activity) of the enzyme.
There are many potential consequences of
changes in drug metabolism for a given drug. It
is made more complex by the fact that there are multiple
pathways of metabolism for many drugs.
The majority of drugs that are metabolized are converted to inactive metabolites. This is the
most common fate for most drugs. Of the
remaining drugs, some are converted to metabolites that retain the same activity as the
parent. An example of this is fexofenadine (Allegra),
the active metabolite of terfenadine that
has equal potency at the histamine receptor
and now is on the market and used clinically for
allergic rhinitis. However, fexofenadine is
more than 50 times less active in blocking potassium channels
in the heart and therefore, unlike terfenadine,
does not cause torsades de pointes.1
In some cases the metabolites are actually
more potent than the parent. For example,
a pro-drug such as enalapril must be
hydrolyzed to enalaprilat to become
active.
In some cases, the metabolites have entirely
new pharmacologic actions not seen with
the parent drug. Metabolites can also be toxic, such as the metabolites
of acetaminophen, which can cause liver
failure, or the metabolite of meperidine, which
can cause seizures.
Inhibition of metabolism could result in
potentially toxic concentrations of the
parent compound. On the other hand, if the
parent drug needs to be metabolized to the
active compound and metabolism is
inhibited, then a therapeutic failure could result.
This happens, for example, if codeine, a prodrug,
is not metabolized to morphine. Induction of
drug metabolizing enzymes could similarly result
in a subtherapeutic effect by reducing drug levels
below that required for efficacy.
1Woosley RL, Chen Y, Freiman JP,
Gillis RA. Mechanism of the cardiotoxic actions of terfenadine. JAMA
1993;269(12):1532–1536.

The major group of enzymes in the liver that metabolize
drugs can be isolated in a subcellular fraction
termed the microsomes. The largest and most
important of these enzymes are the cytochrome P450
family of enzymes. The origin of the term "cytochrome
P450" will be explained later. In addition
to cytochrome P450, there are other enzymes in
microsomes such as flavin monooxygenase (termed
FMO3). These are also responsible for metabolism
of some drugs, but have not been as well characterized
as the cytochrome P450 system, and will
not be discussed further in this presentation.

Drug metabolism is generally classified in two phases,
termed Phase I and Phase II.
Phase I reactions include oxidation or
reduction reactions, usually through the
actions of cytochrome P450 oxidative
enzymes or reductases. These enzymes
prepare very lipophilic molecules for Phase
II reactions by creating a conjugation site, often
a reactive group such as an hydroxyl group.
Phase II reactions "conjugate" a
water soluble entity such as acetate or
glucuronate onto the drug at the newly
created or pre-existing sites, forming a
more polar and water soluble metabolite that can be
more easily excreted in the urine and/or bile.

There are some characteristics of drug metabolism that
can help predict important interactions due to inhibition of metabolism. Since
Phase II reactions generally result in conjugation of a drug to a
water-soluble group like a sugar, peptide (glutathione) or sulfur group, and,
because there is a large excess of these groups in well nourished cells, these
reactions are rarely rate-limiting. Thus, they are rarely involved in drug
interactions. In contrast, the Phase I reactions carried out by cytochrome
P450 enzymes, flavin monooxygenases, and reductases are more frequently
rate-limiting. These are the target of clinically significant drug
interactions, such as the inhibition of cyclosporine metabolism by
erythromycin.
Six cytochrome P450 isoforms have been well
characterized in terms of drug metabolism in humans. These will be reviewed in
the next few slides. Of note, 3 of these isoforms—CYP2C9,CYP2C19, and CYP2D6—can
be genetically absent.
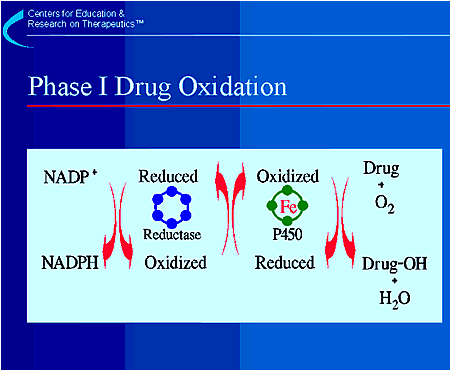
Phase I oxidative enzymes are mostly found in the
endoplasmic reticulum, a subcellular organelle in the liver. The predominant
enzymes responsible for Phase I reactions are those involving the microsomal
mixed function oxidation system. This system requires the presence of NADPH
and NADPH-cytochrome P450 reductase. "Cytochrome P450" is a
superfamily of enzymes that is the terminal oxidase of this oxidation system.
These enzymes are companions and part of a cascade that shuttles electrons
from molecular oxygen to oxidize drugs. "Cytochrome" means colored
cells, and the enzymes contain iron, which gives the liver its red color.
"P450" comes from the observation that the enzyme absorbs a very
characteristic wavelength (450 nm) of UV light when it is exposed to carbon
monoxide.
There are many different isoforms of cytochrome P450, but 6 have been
especially well characterized in terms of clinically relevant drug metabolism
and will be discussed here.
As shown in the slide, the enzymes function in a cascade of
oxidation-reduction reactions that ultimately result in one atom of oxygen
being incorporated into an oxidized metabolite, such as the hydroxylated form
of drug shown in the slide.

This slide lists the major cytochrome P450 isozymes that
are responsible for metabolism of drugs in humans. These enzymes will be
reviewed in detail. Because many drugs are metabolized principally by these
enzymes, important interactions between drugs can be predicted by using a list
of drugs that are inhibitors or inducers of that enzyme. This simplifies the
search for interacting drugs and provides a framework for prediction of
interactions. Next we will review how these enzymes are named.

Cytochrome P450s were named by molecular biologists and protein chemists.
The enzymes are named according to families that are defined by the similarity
of their amino acid sequence.
A very important principle in pharmacology applies in this case: A small
change in the structure of a drug or a protein that interacts with it can
result in major changes in the actions of the drug. Because of this great
sensitivity, small changes in amino acid sequence can result in huge changes
in substrate specificity for the cytochrome P450 enzymes. For example, 2C19 is
the principal metabolic enzyme for omeprazole (Prilosec) metabolism,
but a closely related enzyme, 2C9, has no catabolic effect on omeprazole. Thus,
little functional similarity is imparted by the similarity in amino acid
sequence on which this nomenclature is based. However, as will be seen later,
there is some concordance between classes of drugs and the P450 family that
metabolizes them. The focus of the subsequent slides will be to outline the
role of the cytochrome P450 isozymes in metabolism of commonly used drugs and
to identify tools that can be used in clinical practice to avoid cytochrome
P450-mediated drug interactions.

The graph on the left lists the major isoforms of CYP450 and their relative roles in drug metabolism (not relative amounts found in the liver) based upon the number of drugs that are known to be metabolized by that particular isozyme. CYP3A is responsible for the metabolism of the largest number of drugs followed by CYP2D6.
The graph on the right summarizes the relative quantity of specific P450 families found in the
liver.1 The CYP3A family is present in the largest amounts. CYP2D6 accounts for less than 2% of
the total content of P450 in the liver, but as shown on the
left, is responsible for the metabolism of a large fraction of drugs. A large
amount of cytochrome P450 has not yet been characterized.
There is tremendous variability between individuals in terms of expression
of cytochrome P450 isozymes. For example, CYP2D6 is not present at all in some
livers.
Note: 2C on the graph on the right refers to both CYP2C9 and CYP2C19.
1Shimada T, Yamazaki H, Mimura M, Inui
Y, Guengerich FP. Interindividual variations in human liver
cytochrome P-450 enzymes involved in the oxidation of drugs, carcinogens and
toxic chemicals: studies with liver microsomes of 30 Japanese and 30
Caucasians. J Pharmacol Exp Ther 1994;270(1):414–423.
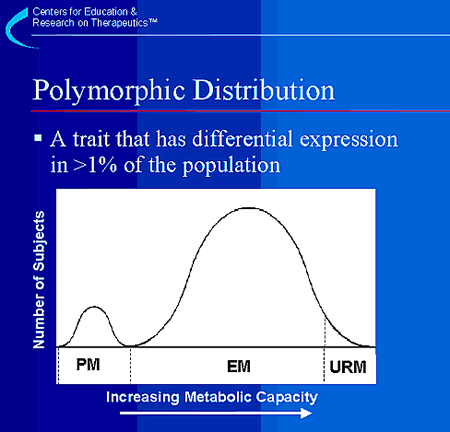
The cytochrome P450 enzymes have 3 interesting properties that often make it possible to predict drug interactions.
First, some people have mutations in one or more of the nucleic acids in the DNA sequence expressing a given cytochrome P450 isozyme. As a
result, the enzyme may be absent or have low, or no, metabolizing activity for drugs that are usually metabolized by that enzyme. If the mutation is relatively common (more than 1%) it creates a polymorphism—this is a trait that has genetic variation that results in more than a single population being represented in greater than 1% of the total population. It literally means that the distribution of the trait has
“multiple” “forms,” i.e. “poly” “morphic.” At least 3 of the cytochrome P450s that we will be discussing
(2D6, 2C19, 2C9) are polymorphic in their distribution. People expressing a polymorphism will therefore metabolize drugs at a different rate than the rest of the population.
This graph demonstrates a population drug metabolism distribution for CYP2D6. On the graph, PM means poor
metabolizer, EM means extensive metabolizer, which is the normal or usual
phenotype, and URM means ultra-rapid metabolizer. Approximately 7% of the U.S. population has a genetic defect in CYP2D6 that results in a poor metabolizer phenotype. Ultra-rapid metabolizers
usually do not appear as a separate distribution in most phenotypic data but are important because a usual dose of drug in these people will be cleared more quickly than in the rest of the population and will result in lower blood levels of the drug and,
perhaps, less therapeutic effect. For CYP2D6, it is known that these individuals have very high activity because they have multiple copies of the CYP2D6 gene (up to13 copies have been reported).
Second, people that have usual drug metabolizing ability (EM) can become phenotypic poor
metabolizers if they are given a substance (drug or food as we will see later) that inhibits the enzyme. So if two drugs are given that are metabolized by the same
enzyme, and one inhibits the enzyme, the second drug can accumulate to higher and potentially toxic levels.
Third, several of the cytochrome P450 isozymes can be “induced” to have increased activity. If this
occurs, metabolism of any drug that is a substrate for that isozyme will be metabolized more quickly resulting in lower plasma concentrations of the drug. This may also reduce the efficacy of the drug. Also, if the drug is metabolized to a toxic
compound, the toxic metabolite may accumulate to higher levels.
The P450 isozymes will now be reviewed in more detail. The laminated card in the pocket of the module can be used as a reference for the next few slides.

CYP3A is responsible for metabolizing the greatest number of marketed
drugs. These include a wide range of important medications including
cyclosporine and HIV protease inhibitors, as well as cisapride (Propulsid)
and the no longer marketed non-sedating antihistamines terfenadine (Seldane)
and astemizole (Hismanal). Although CYP3A is not polymorphic in
its distribution (it doesn’t have a distinctly separate population as seen
on the previous graph), its activity varies over 50-fold in the general
population. CYP3A has been recently reviewed.1
CYP3A is the drug metabolizing pathway involved in the case of torsades de
pointes described at the beginning of the module. Terfenadine, one of the first
non-sedating antihistamines, is metabolized by CYP3A to fexofenadine. When the
CYP3A-mediated metabolism of terfenadine is inhibited by ketoconazole, as in
the case described, terfenadine accumulates to high levels. At these high
levels, terfenadine is a blocker of potassium channels in the heart.2 Potassium
channels are important for repolarization of the heart. Once these channels
are blocked, QT interval on the electrocardiogram can be prolonged and torsades
de pointes can develop, as was seen in this
case. Many commonly used drugs can inhibit this enzyme as we will see in the
next slide. This important enzyme has been the basis for most of the fatal
drug interactions that have gained so much publicity in recent years. For
terfenadine, as well as astemizole and cisapride, recognition and reporting of
torsades de pointes in association with the drug and its interactions
ultimately led to withdrawal of these drugs from the market.
The vast majority of drugs that may cause cardiac arrhythmias by prolonging
the QT interval are metabolized by cytochrome P450 3A. While the biological
basis for this remains unclear, it does make it easier to remember!
Also note that CYP3A is found in the liver and also in the GI tract. Drugs
that are substrates of CYP3A can be extensively metabolized in the GI tract,
and ,in fact, the GI tract is responsible for a large part of the
metabolism that was formerly attributed totally to the liver! Inhibition of GI
tract CYP3A also results in higher plasma levels of substrate drugs.
1Thummel KE, Wilkinson
GR. In vitro and in vivo drug interactions involving human CYP3A. Annu
Rev Pharmacol Toxicol 1998;38:389–430. 2Woosley RL, Chen Y,
Freiman JP, Gillis RA. Mechanism of the cardiotoxic actions of terfenadine. JAMA
1993;269(12):1532–1536.

These
are the important inhibitors of CYP3A that will make patients appear
phenotypically to resemble poor metabolizers. Azole antifungal drugs, in
general, are
potent inhibitors of CYP3A, although fluconazole is a weak inhibitor and
inhibits CYP3A only at high doses. All the macrolide antibiotics, except
azithromycin, are
also potent inhibitors of this
cytochrome P450 isoform. Cimetidine is a broad, but relatively weak, inhibitor
of many cytochrome P450 enzymes. Also, notice that a food, grapefruit juice,
is listed as an inhibitor. The role of grapefruit juice in drug interactions
will be discussed later.

Several
commonly used drugs have been characterized as inducers of CYP3A. Use of these
drugs could potentially result in lack of therapeutic efficacy
of a CYP3A substrate. Drug interactions with the herbal remedy St. John’s
wort will be discussed later in the presentation.

CYP2D6 metabolizes many of the cardiovascular and neurologic drugs in use
today. Study of CYP2D6 has led to understanding the failure of codeine to
relieve pain in some patients. Codeine is actually a pro-drug that is converted
to morphine. Codeine itself is much less active as an analgesic, but causes
nausea and other adverse effects. The absence of cytochrome P450 2D6 in 7% of
Caucasians means that these individuals cannot metabolize codeine to the active
metabolite, morphine, and therefore will get little, if any, pain relief from
codeine.1 However, they will experience codeine’s adverse effects,
particularly if the dose is increased in the futile attempt to obtain pain
relief.
Thirty percent of Ethiopians studied had multiple copies of the 2D6 gene (up
to13) and increased eynzyme activity resulting in ultrarapid metabolism.2 Ultra-rapid
metabolism results in lower blood levels following a standard dose of any drug
metabolized by this enzyme. Therefore these patients may have an inadequate
response to standard dosages of ß-blockers, narcotic analgesics, or
antidepressants and may require higher dosages for clinical effectiveness.
Several commonly used medications inhibit CYP2D6. These include quinidine3
as well as haloperidol and some other antipsychotics.4,5 The
well-described pharmacokinetic interaction between selective serotonin reputake
inhibitor (SSRI) antidepressants and tricyclic antidepressants appears to be
due to the fact that fluoxetine and paroxetine are both potent inhibitors of
CYP2D66,7 and render patients metabolically equivalent to people who
do not have the enzyme. This increases the plasma levels of tricyclic
antidepressants and increases the potential for side effects. In contrast,
patients co-prescribed fluoxetine or paroxetine with codeine may experience no
analgesic benefit, since codeine requires CYP2D6 for metabolism to morphine.
1Caraco Y, Sheller J, Wood AJ.
Pharmacogenetic determination of the effects of codeine and prediction of drug
interactions. J Pharmacol Exp Ther 1996;
278(3):1165–1174. 2Aklillu E, Persson I, Bertilsson L,
Johansson I, Rodrigues F, Ingelman-Sundberg M. Frequent
distribution of ultrarapid metabolizers of debrisoquine in an ethiopian
population carrying duplicated and multiduplicated functional CYP2D6 alleles. J
Pharmacol Exp Ther 1996;278(1):441–446. 3Branch RA, Adedoyin A,
Frye RF, Wilson JW, Romkes M. In vivo modulation of CYP enzymes by quinidine
and rifampin. Clin Pharmacol Ther 2000;
68(4):401–411. 4Shin JG, Kane K, Flockhart DA. Potent inhibition of CYP2D6 by haloperidol metabolites:
stereoselective inhibition by reduced halo-peridol. Br
J Clin Pharmacol 2001;51(1):45–52. 5Shin JG, Soukhova N, Flockhart DA. Effect
of antipsychotic drugs on human liver cytochrome P-450 (CYP) isoforms in vitro:
preferential inhibition of CYP2D6. Drug
Metab Dispos 1999;27(9):1078–1084. 6Bergstrom RF, Peyton AL, Lemberger L.
Quantification and mechanism of the fluoxetine and tricyclic antidepressant
interaction. Clin Pharmacol Ther 1992;51(3):239–248. 7Leucht S,
Hackl
HJ, Steimer W, Angersbach D, Zimmer R. Effect of adjunctive paroxetine on serum
levels and side-effects of tricyclic antidepressants in depressive inpatients.
Psychopharmacology (Berl) 2000;147(4):378–383.

CYP2C9 has a polymorphic distribution in the population and is missing in 1%
of Caucasians. It is the major enzyme responsible for metabolism of many of the
non-steroidal anti-inflammatory drugs (NSAIDs), including the second generation
cyclooxygenase-2 (COX-2) specific inhibitors. A number of other important
medications have their metabolism primarily catalyzed by CYP2C9. An important
drug metabolized by this enzyme is warfarin (Coumadin), and almost
all inter-patient variability in warfarin levels and anticoagulant effects can
be explained on the basis of CYP2C9 activity (not the differences in protein
binding as originally thought).
The azole antifungal agent fluconazole (Diflucan) is a potent
inhibitor of CYP2C9. Fluconazole, at conventional doses, abolishes CYP2C9
activity.
An interaction between fluconazole and warfarin results in at least a
two-fold increase in warfarin blood level, a reduction in warfarin clearance,
and increased anticoagulation.1 Clinical studies have identified a
significant interaction between fluconazole and celecoxib (Celebrex),
leading to a twofold increase in celecoxib plasma concentrations.2 A
clinical pharmacokinetic study demonstrated an increase in phenytoin area under
the plasma concentration curve (AUC) following fluconazole administration,3
and symptomatic phenytoin toxicity has been reported with concomitant
administration of fluconazole and phenytoin.4
1Black DJ, Kunze KL , Wienkers LC, Gidal BE, Seaton
TL, McDonnell ND, et al. Warfarin-fluconazole. II.
A metabolically based drug interaction: in vivo studies. Drug
Metab Dispos 1996;24(4):422–428. 2Celebrex. Physicians’Desk
Reference. Montvale, NJ: Medical Economics Company, Inc., 2001:2482–2485.
3Touchette MA, Chandrasekar PH, Milad MA, Edwards DJ. Contrasting effects of fluconazole and ketoconazole on phenytoin
and testosterone disposition in man. Br J
Clin Pharmacol 1992;34(1):75–78. 4Cadle RM, Zenon GJ,III, Rodriguez-Barradas MC, Hamill RJ. Fluconazole-induced symptomatic phenytoin toxicity. Ann
Pharmacother 1994;28(2):191–195.

Cytochrome P450 2C19 is notable because of its genetic absence in such a
high percentage of Asians (approximately 20–30%). This enzyme metabolizes
many anticonvulsants, diazepam (Valium), omeprazole (Prilosec)
and several of the tricyclic antidepressants. Asians have reduced clearance of
diazepam compared to Caucasians,1 and, in fact, a survey of Asian
and Western physicians demonstrated the use of lower doses of diazepam in
Asians.2 Asian patients may have a lower omeprazole dosage
requirement for effective treatment of Helicobacter pylori. According to
the omeprazole package insert, Asians have about a four-fold increase in the
AUC of omeprazole compared to Caucasians, and the labeling recommends that one
should consider dosage adjustment.3 In addition, the poor metabolizer genotype for CYP2C19 resulted in a higher
cure rate for H. pylori (100%) than the rapid metabolizer genotype
(28.6%) in an Asian population treated with omeprazole as part of dual therapy.4
Similar results have been shown more recently with proton pump inhibitors
in a triple therapy regimen.5
Ketoconazole6 and omeprazole7 are inhibitors of
CYP2C19 and have the potential for clinically significant interactions with
substrates of CYP2C19 such as diazepam8 or phenytoin.9 Isoniazid,
used to treat tuberculosis, is an inhibitor of CYP2C1910 and should
be prescribed cautiously to patients taking phenytoin and other drugs
metabolized by CYP2C19.
1Ghoneim MM,
Korttila K, Chiang CK ,Jacobs L ,Schoenwald RD, Mewaldt SP, et al. Diazepam effects and kinetics in Caucasians
and Orientals. Clin Pharmacol Ther 1981;29(6):749–756. 2Rosenblat R,
Tang
SW. Do Oriental psychiatric patients receive different dosages of psychotropic
medication when compared with occidentals. Can
J Psychiatry 1987;32(4):270–274. 3Prilosec. Physicians’ Desk Reference.
Montvale, NJ: Medical Economics Company, Inc.;2001:587–591. 4Furuta T, Ohashi K, Kamata T, Takashima M ,Kosuge K, Kawasaki T, et al. Effect of genetic differences in omeprazole
metabolism on cure rates for Helicobacter
pylori infection and peptic ulcer. Ann
Intern Med 1998; 129(12):1027–1030. 5Furuta T, Shirai N, Takashima M, Xiao F, Hanai H, Sugimura H, et al. Effect of genotypic differences in CYP2C19 on cure
rates for Helicobacter pylori infection
by triple therapy with a proton pump inhibitor, amoxicillin, and clarithromycin.
Clin Pharmacol Ther 2001;69(3):158–168. 6Atiba JO, Blaschke
TF, Wilkinson GR. Effects of ketoconazole on the polymorphic 4-hydroxylations
of S-mephenytoin and debrisoquine. Br J
Clin Pharmacol 1989;28(2):161–165. 7Ko JW, Sukhova N, Thacker D, Chen P, Flockhart DA. Evaluation of omeprazole and lansoprazole as inhibitors of
cytochrome P450 isoforms. Drug Metab
Dispos 1997;25(7): 853–862. 8Ishizaki T, Chiba K, Manabe K, Koyama E, Hayashi M, Yasuda S, et al. Comparison of the interaction potential of a new
proton pump inhibitor, E3810,versus omeprazole with diazepam in extensive and
poor metabolizers of S-mephenytoin 4'-hydroxylation. Clin
Pharmacol Ther 1995;58(2):155–164. 9Prichard PJ, Walt RP, Kitchingman GK, Somerville KW ,Langman MJ, Williams J,et al. Oral phenytoin pharmacokinetics
during omeprazole therapy. Br J Clin
Pharmacol 1987;24(4):543–545. 10Desta Z, Soukhova NV, Flockhart DA.
Inhibition of cytochrome P450 (CYP450) isoforms by isoniazid: potent
inhibition of CYP2C19 and CYP3A. Antimicrob
Agents Chemother 2001;45(2):382–392.

Cytochrome
P450 1A2 is an important drug metabolizing enzyme in the liver that
metabolizes many commonly used drugs including theophylline, imipramine,
propranolol, and clozapine. CYP1A2 is induced in a clinically relevant manner by tobacco
smoking. The clearance of theophylline, imipramine, propranolol and clozapine
are all increased by smoking. Thus, people who smoke may require higher doses
of some of the medications that are substrates of CYP1A2. In contrast, a
smoker
would require a decrease in theophylline dosage if, for example, smoking
were discontinued and the enzyme no longer induced. This topic has been
recently reviewed by Zevin and Benowitz.1
Inhibitors of CYP1A2, including some fluoroquinolone
antibiotics, can increase the plasma concentrations of drugs that are
metabolized by CYP1A2,with a potential for increased toxicity.2,3
1Zevin S, Benowitz
NL. Drug interactions with tobacco smoking. An update. Clin
Pharmacokinet 1999;36(6):425–438. 2Raaska K, Neuvonen PJ. Ciprofloxacin
increases serum clozapine and N-desmethylclozapine: a study in patients with
schizophrenia. Eur J Clin Pharmacol 2000;56(8):585–589. 3Grasela TH,
Jr., Dreis
MW. An evaluation of the quinolone-theophylline interaction using the Food and
Drug Administration spontaneous reporting system. Arch
Intern Med 1992;152(3):617–621.
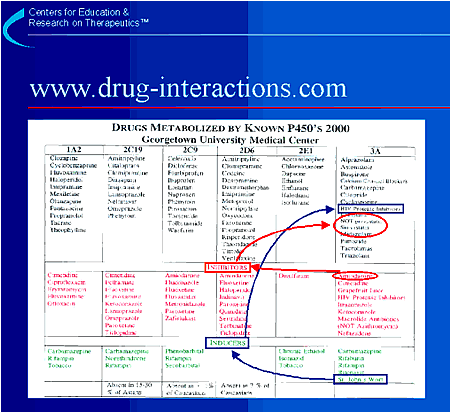
It would be impossible to memorize all of the drug interactions that have
been presented here. Fortunately there are aids to help health care providers
to anticipate and prevent drug interactions, such as the tool shown here. This
is a pocket version of a much larger CYP P450 drug interaction table. A more
complete version of this card is maintained on the Internet at www.drug-interactions.com.
This table includes a listing of the 6 major cytochrome P450 isozymes involved
in drug metabolism and the drugs that are metabolized by them. We recommend
using this or another table as a quick reference for detection of potential
drug interactions.
If 2 drugs are metabolized by the same cytochrome P450 isozyme, it is very
possible that competitive inhibition could lead to higher than usual levels of
either or both of the drugs. If a drug is metabolized by a specific cytochrome
P450 and is taken with an inhibitor or inducer of that isozyme, an interaction
is also likely.
The following are examples of how to use this card. Suppose your patient is
taking amiodarone and you want to add a statin agent to decrease the patient’s
cholesterol (follow red circles and arrows above). The card shows that
amiodarone is an inhibitor of CYP2D6 and CYP3A. We also note that lovastatin
and simvastatin are metabolized by CYP3A and that if given with amiodarone
(which is inhibiting the enzyme) a toxic level of the statin may occur. The
result may be an adverse reaction (rhabdomyolysis or liver toxicity). The best
choice would be pravastatin, which is not metabolized by CYP3A. Another
example would be if your patient were taking an HIV protease inhibitor and
wants to take St. John’s wort (follow green squares and arrows above).
According to the card, St. John’s wort induces CYP3A4, which metabolizes most
protease inhibitors. The concomitant administration of St. John’s wort with
protease inhibitors could result in the induction of CYP3A4, increased
metabolism, and subtherapeutic levels of the protease inhibitor.
Laminated versions of this card can be ordered from the website listed
above. At the website, it is possible to easily obtain the reference for a
given drug by clicking on the drug. The website hyperlinks to PubMed and
searches for a list of the relevant publications.

In
addition to the drug-drug interactions just reviewed, drug-disease
interactions can occur. These include interactions between certain drugs and
specific disease states. Severe liver disease can be associated with reduced
metabolic clearance and higher plasma levels of drugs extensively metabolized
by the liver.1 Although liver disease reduces drug clearance on
average, the
change is relatively small and usually not clinically relevant except in
patients with near terminal liver disease. The effects of renal disease on
elimination of drugs that are primarily cleared renally are more predictable,
and well-established guidelines exist for dosage of many drugs in renal
disease.2 Heart failure
reduces liver blood flow and causes a reduction in clearance for drugs such as
lidocaine or propranolol that are usually extensively cleared by the liver,3
and acute myocardial infarction reduces clearance of some drugs, such as
lidocaine, as well.4 Acute viral infection and changes in thyroid
function have been associated with altered clearance for some drugs, such as
theophylline and warfarin.5–7 However, the results are so
variable between individuals that it is difficult to predict who is at risk,
and these changes are usually only clinically important in cases of extremely
impaired organ function.
1Brouwer KLR, Dukes
GE, Powell JR. Influence of liver function on drug disposition. In: Evans WE,
Schentag JJ, Jusko WJ, editors. Applied Pharmacokinetics: Principles of
Therapeutic Drug Monitoring. Vancouver, WA: Applied Therapeutics, Inc.;
1992:6-1-6-59. 2Lam YW, Banerji S, Hatfield C, Talbert RL. Principles
of drug administration in renal insufficiency. Clin Pharmacokinet 1997;32(1):30-57.
3Shammas FV, Dickstein K. Clinical pharmacokinetics in heart
failure. An updated review. Clin Pharmacokinet 1988;15(2):94-113. 4Pieper JA, Johnson KE. Lidocaine. In: Evans WE, Schentag JJ, Jusko WJ,
editors.
Applied Pharmacokinetics:Principles of Therapeutic Drug Monitoring.
Vancouver, WA: Applied Therapeutics Inc.; 1992:21-1-21-37. 5Pokrajac M,
Simic
D,Varagic VM. Pharmacokinetics of theophylline in hyperthyroid and hypothyroid
patients with chronic obstructive pulmonary disease. Eur J Clin Pharmacol 1987;33(5):483–486.
6Stephens MA, Self TH, Lancaster D, Nash T. Hypothyroidism: effect on
warfarin anticoagulation. South Med J 1989;82(12):1585–1586. 7Yamaguchi A, Tateishi T, Okano Y, Matuda T, Akimoto Y, Miyoshi T, et al. Higher
incidence of elevated body temperature or increased C-reactive protein level
in asthmatic children showing transient reduction of theophylline metabolism. J
Clin Pharmacol 2000;40(3):284–289.

Several
drugs are known to interact with foods,1 some of which are listed
here. One of the early observations was the reduced absorption of tetracycline
when taken with milk products. The chelation of tetracycline by calcium
prevents it from being absorbed from the intestines. Dietary sources of
vitamin K, such
as spinach or broccoli, may increase the dosage
requirement for warfarin by a pharmacodynamic antagonism of its effect.
Patients should be counseled to maintain a consistent diet during warfarin
therapy. Grapefruit
juice contains a bioflavonoid that inhibits CYP3A and blocks the metabolism of
many drugs. This was first described for felodipine (Plendil)2
but has now been observed with several drugs.3 This
interaction can lead to reduced clearance and higher blood levels when the
drugs are taken simultaneously with grapefruit juice. With regular
consumption, grapefruit juice also reduces the expression of CYP3A in the GI
tract.4
1Williams L, Davis JA, Lowenthal DT. The
influence of food on the absorption and metabolism of drugs. Med
Clin N Am 1993;77(4):815–829. 2Bailey DG, Spence JD, Munoz C, Arnold JM.
Interaction of citrus juices with felodipine and nifedipine. Lancet 1991;337(8736):268–269.
3Kane GC, Lipsky JJ. Drug-grapefruit
juice interactions. Mayo Clin Proc 2000;75(9):933–942. 4Lown KS,
Bailey DG, Fontana RJ, Janardan SK, Adair CH, Fortlage LA et al. Grapefruit juice
increases felodipine oral availability in humans by decreasing intestinal
CYP3A protein expression. J Clin Invest 1997;99(10):2545–2553.

This
slide demonstrates the effects of grapefruit juice on felodipine
pharmacokinetics and pharma-codynamics.1 The left graph shows
felodipine plasma concentrations at specific time points, up
to 24 hours, following administration of a single
dose of felodipine with 250 cc of grapefruit juice or water. The right graph
shows systolic and diastolic blood
pressure from the same time points. Compared with water, there is an increase
in felodipine plasma concentrations, as well as a decrease in systolic and
diastolic blood pressure. This demonstrates a potentially clinically
significant effect of the grapefruit juice-felodipine interaction.
1Dresser GK, Bailey DG, Carruthers SG.
Grapefruit juice-felodipine interaction in the elderly. Clin
Pharmacol Ther 2000;68:28–34.

It
has been suspected that herbal remedies could interact with other herbals or
even prescription drugs. Ingestion of St. John’s wort has resulted in
several clinically significant interactions with drugs that are metabolized by
CYP1A2 or CYP3A, including indinavir (Crixivan)1 and
cyclosporin (Sandimmune and Neoral).2,3 An
interaction with digoxin (Lanoxin) has also been reported that
may be mediated by interference with P-glycoprotein (P-GP), a
transport system that pumps drugs across membranes.4 These
interactions are most likely due to induction of the cytochrome P450 isozyme
or the drug transporter and have caused decreased plasma concentrations of prescription drugs. In the case of cyclosporin, subtherapeutic levels
resulted in transplant organ rejection. Warnings about St. John’s wort drug
interactions have been extended to oral contraceptives, with labeling
suggesting the possibility of breakthrough bleeding and potential for loss of
contraceptive effect.
It is likely that many drug-herbal interactions exist but have not yet been
detected. It is therefore important that health care providers obtain a
complete drug history that includes herbal remedies and other natural products
and dietary supplements and that they be alert to potential interactions.
1Piscitelli SC, Burstein AH, Chaitt D, Alfaro RM, Falloon J. Indinavir concentrations and St John’s wort. Lancet
2000;355(9203):547–548. 2Breidenbach T, Hoffmann MW, Becker T,
Schlitt H, Klempnauer J. Drug interaction of St John’s wort with
cyclosporin. Lancet 2000;355(9218):1912. 3Ruschitzka F,
Meier PJ, Turina M, Luscher TF, Noll G. Acute heart transplant rejection due to
Saint John’s wort. Lancet 2000;355(9203):548–549. 4Johne A,
Brockmoller J, Bauer S, Maurer A, Langheinrich M, Roots I. Pharmacokinetic
interaction of digoxin with an herbal extract from St John’s wort (Hypericum
perforatum). Clin
Pharmacol Ther 1999; 66(4):338–345.
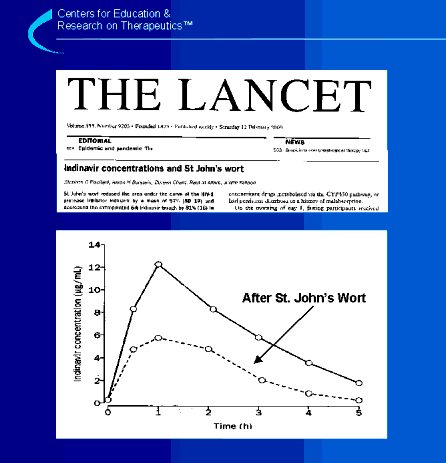
This
slide shows the mean plasma concentration time course of indinavir in 8
healthy volunteers with indinavir alone or after taking indinavir with St.
John’s wort.1 After administration of St. John’s wort, a
57% reduction was observed in the indinavir area under the plasma
concentration-time curve (AUC), indicative
of reduced exposure to indinavir. This study prompted a public health advisory
released by the FDA on February10, 2000 (www.fda.gov/cder/drug/advisory/stjwort.htm)
about the risk of drug interactions between St. John’s wort and other
medications. The potential for loss of therapeutic efficacy due to this
interaction suggests the importance of taking a complete medication history.
This history should include questions about herbal therapy and other natural
products as well as over-the-counter medications.
1Piscitelli SC, Burstein AH, Chaitt D, Alfaro RM, Falloon J. Indinavir concentrations and St John’s wort. Lancet
2000;355(9203):547–548.
ADR Reporting

Given the vital importance of postmarketing surveillance of new drugs,
MedWatch, the FDA Medical Products Reporting Program, was established in 1993.
The program has 4 general goals. The first goal is to increase awareness of
drug, device and other medical product induced disease and the importance of
reporting.
The second goal of MedWatch is to clarify what should (and should not) be
reported. Health professionals are asked to limit reporting to serious adverse
reactions. This is important both in improving the quality of individual
reports and enabling the FDA and the manufacturer to focus on the most
significant reactions. Causality is not a prerequisite for reporting;
suspicion that a medical product may be related to a serious reaction is
sufficient reason to report.
The third goal is to make it as easy as possible to report to the FDA. Only
one reporting form is necessary. The postage-paid form for voluntary reporting
is available in the back of the Physicians’ Desk Reference or from
the FDA via the toll free number (1-800-FDA-1088) or from the FDA/MedWatch
website (www.fda.gov/medwatch).
The fourth and final goal of the program is to provide feedback to health
professionals about new safety problems with pharmaceuticals and medical
devices. Safety-related labeling changes, "Dear Health Care
Professional" correspondence, safety alerts and FDA public health
advisories are posted on the FDA/MedWatch website.
1Kessler
DA. Introducing MedWatch: a new approach
to reporting medication and device adverse effects and product problems. JAMA
1993;269:2765–8.
Preventing Drug
Interactions

In closing, it is impossible to remember all of the drug interactions that
can occur. It is therefore important to develop a stepwise approach to
preventing adverse reactions due to drug interactions.
First, taking a good medication history is essential. The "AVOID
Mistakes" mnemonic presented on the next slide can help health care
practitioners to develop good habits when performing this task.
Second, it is essential that physicians develop an understanding of which
patients are at risk for drug interactions. Of course any patient taking 2
medications is at some risk. Studies show that the rate of adverse drug
reactions increases exponentially in patients taking 4 or more medications.1
Importantly, some categories of drugs are especially at high risk for
interactions. These categories include anticonvulsants, antibiotics, and
certain cardiac drugs such as digoxin, warfarin, and amiodarone.
Third, any time a patient is taking multiple drugs, we recommend that the
first step be to check a readily available pocket reference, recognizing that
the interaction may not be listed and a more complete search may be required.
Fourth, consult other members of the health care team. Depending upon the
practice setting, this may be a clinical pharmacologist, a hospital pharmacist,
a specially trained office staff nurse, or the nearby pharmacist in community
practice.
Fifth, use one of the computerized databases available. Up-to-date
databases are maintained by gsm.com and epocrates.com, and others.* The latter
can be placed on a hand-held computer (e.g. Palm Pilot) and can be configured
to automatically update each time you synchronize with the desktop computer.
The Medical Letter Drug Interaction Program is inexpensive and updated
quarterly.*
*These programs are not endorsed by the FDA.
1Jacubeit T,
Drisch D, Weber E. Risk
factors as reflected by an intensive drug monitoring system. Agents
Actions 1990;29:117–125.

Finally, use of the "AVOID Mistakes" mnemonic can help to develop
good practice habits and offers a useful way of remembering the components of
a good drug history.
A history of allergies or previous history of adverse reactions to any
drugs should be elicited in a way that will yield the most useful information.
For example, rather than asking about a history of drug allergy, the patient
should be asked whether there is any drug that should not be prescribed for
any reason. A specific question should be asked about the use of vitamins and
herbal or other natural products. Old drugs (prescription and over the
counter) should be considered as well as new drugs, since some of the effects
(either toxicity or potential for drug interactions) could be relatively
long-lasting. The potential for adverse drug interactions should be evaluated.
The need for a behavioral contract between the physician and the patient
should be considered in an effort to help the patient reach the therapeutic
goal, either in the case of drug dependence or adherence to a therapeutic
regimen, with a clear plan. Finally, a family history of benefits or problems
with any medications will help determine whether pharmacogenetics should be
considered in tailoring drug therapy.

These web sites are not endorsed by the FDA.

Post-doctoral training for physicians and
pharmacologists interested in clinical pharmacology as a career is available
at NIH-sponsored sites as well as other sites throughout the country. For a
list of available training programs and contact information, see the website
of the American Society for Clinical Pharmacology and Therapeutics (ASCPT).
Acknowledgements

Self-Assessment Questions
 Back to Top Back to Top  Back to Drug Information
Back to Drug Information  CDER Home
CDER Home
FDA/Center for Drug Evaluation and Research
Last Updated: July 31, 2002
Originator: OTCOM/DLIS
HTML by SJW |
