|
|
 |
|
 |
| |
 |
 |
| |
|
|
| |
Rac1b: A Highly Selective Marker and Target for Cancer
IB-2167
|
|
 |
|
|
| |
|
APPLICATIONS
OF TECHNOLOGY:
| |
|
|
| |
 |
|
| |
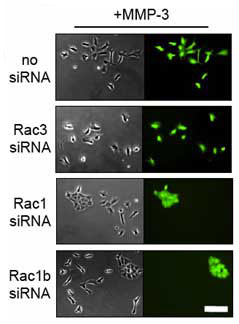
Selective knockdown of Rac1/Rac1b and Rac1b inhibits MMP-3 induced cell scattering. The cells were treated with MMP-3 for 4 days. Scale bar, 25 µm. Please click on the publication link below for more information. |
|
| |
|
|
- Early stage detection of breast, colon, prostrate, kidney, skin, stomach, ovarian, and lung cancers
- Selective therapeutic target
- A research tool for investigating MMP-induced malignancy mechanisms
ADVANTAGES:
- Target is malignant-specific and upregulated
- Elucidates a new and distinct cancer-promoting pathway
- Rac1b mouse antibody and Rac1b siRNA have been developed
ABSTRACT:
Mina Bissell and her team at Berkeley Lab have identified the Rac1b protein as a highly specific, early stage biomarker for matrix metalloproteinase (MMP)-induced malignancies, which include breast, colon, prostrate, kidney, skin, stomach, ovary, and lung cancers. Rac1b is an especially promising biomarker and therapeutic target because it is not generated in non-malignant cells, and unlike most oncogenic splice isoforms induced in cancers, it becomes more highly activated.
MMPs are near ubiquitous elements of the tumor microenvironment and are associated with epithelial-mesenchymal transition (EMT) and malignant transformation in cultured cells. The Berkeley Lab researchers have uncovered the mechanism by which these effects are produced. MMP-3 has been found to induce an alternatively spliced form of Rac1, Rac1b, which causes an increase in cellular reactive oxygen species (ROS), which in turn stimulate the expression of transcription factor Snail and EMT. Snail and EMT cause oxidative damage to DNA and genomic instability that lead to malignancy.
The Bissell lab has developed Rac1b mouse antibody and Rac1b siRNA. In collaboration with the Mayo Clinic, they have determined that expression of MMPs in transgenic animals leads to upregulation of Rac1b, confirming its significance in vivo as a diagnostic marker and therapeutic target. Rac1b is an intracellular protein lending itself to small molecule and RNAi therapeutics. |
|
|
| STATUS:
|
|
|
| FOR
MORE INFORMATION:
Radisky, D. C., Levy, D. D., Littlepage, L.E., Lui, H., Nelson, C. M., Fata, J. E., Leake, D., Godden, E. L., Albertson, D. G., Nieto, M.A., Werb, Z., Bissell, M., “Rac1b and Reactive Oxygen Species Mediate MMP-3-induced EMT and Genomic Instability,” Nature 2005, 436, 123-127. |
|
| REFERENCE
NUMBER: IB-2167 |
|
| SEE
THESE OTHER BERKELEY LAB TECHNOLOGIES IN THIS FIELD:
|
|
|
|
|
|
CONTACT:
|
Technology
Transfer Department
E.O. Lawrence Berkeley National Laboratory
MS 90-1070
Berkeley, CA 94720
(510) 486-6467 FAX: (510) 486-6457
TTD@lbl.gov |
|
|
| |
|
|
| |
|
|
|



