You are here:
Research
- Lab Overview
- Laboratory Chief Office
- Muscle Assembly Group
- Muscle Physiology Section
- Muscle Proteomics and Nanotechnology Section

Muscle Assembly Group
Robert Horowits, Ph.D.
Group Head, Muscle Assembly Group
Phone: (301) 435-8371
Fax: (301) 402-0009
E-mail: horowitr@mail.nih.gov
Research Overview
Muscles are packed with proteins that transform chemical
energy into mechanical work. Filaments of actin and myosin protein are
contained in myofibrils, the machinery responsible for shortening and
contraction in striated muscle. Dr. Horowits' research focuses on how
myofibrils are assembled and maintained and how force is transmitted
from the contractile machinery to the ends of cells, across membranes
and beyond the cells. N-RAP, a muscle-specific protein concentrated at
myotendon junctions in skeletal muscle and intercalated disks in cardiac
muscle, is the central focus of Horowits' current studies. Unlike most
proteins found at muscle connections attaching myofibrils to membranes,
N-RAP is found only in muscles that make striated myofibrils. The
characteristics and binding properties of this highly specialized
protein indicate that it may play a critical role in muscle assembly.
Horowits and NIH colleagues published a paper in 1999 exploring
N-RAP's potential to anchor the terminal actin filaments of the
myofibril to the cell membrane and transmit tension from the myofibril
to the extracellular matrix. They detected high-affinity binding of
defined regions of N-RAP to actin, talin, and vinculin. Figure 1
illustrates hypothetical membrane-myofibril links based on this study.

In a recently published paper, Horowits and his colleagues describe how they used immunofluorescence to track the localization of N-RAP during myofibrillogenesis in cultured cardiomyocytes. In accordance with their model of N-RAP's function and interactions, the protein was strongly targeted to sites of mechanical coupling during myofibrillogenesis. Prior to myofibrillogenesis and cell spreading, N-RAP was diffusely distributed in the cytoplasm. As the cell spread and pre-myofibrils were formed, N-RAP became targeted to the newly created terminal ends of these structures and incorporated into the pre-myofibrils themselves. He was especially excited to discover that, as the pre-myofibrils became mature myofibrils, N-RAP was observed at the terminal ends, but not within the mature sarcomeres, Figure 2.
|
This cover illustration accompanied an article
by Carroll and Horowits in Cell Motility and the Cytoskeleton (volume
47, number 1, 2000). The article can be viewed at http://www. interscience. wiley.com/. |
| Figure 2 | |
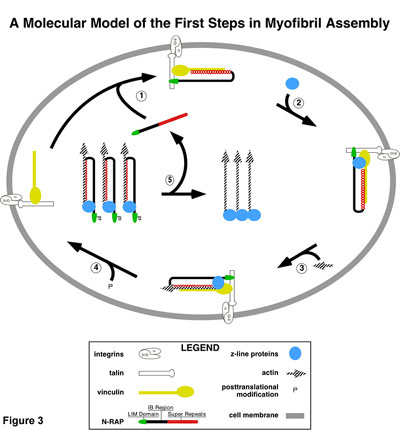
In addition, N-RAP was localized to the site of cell-cell contact, even in the absence of significant myofibril formation. The results demonstrated that myofibrillogenesis and cell contact formation each can independently target N-RAP to the longitudinal ends of cardiomyocytes. This study suggests that N-RAP may serve as a constant connection to the membrane that persists from the genesis of the myofibril to its final position in the mature heart. In a followup study, Horowits and colleagues used GFP-tagged constructs in chick cardiomyocytes to explore the targeting and functional effects of individual N-RAP domains. They found that each region of N-RAP is critical for myofibril assembly. Combining the targeting and functional effects of N-RAP domains with information in the literature, they proposed a new model for initiation of myofibrillogenesis in which N-RAP acts as an organizing center for the initial recruitment and assembly of sarcomeric actin filaments and Z-lines, Figure 3.
Horowits participated in a collaborative study to examine whether or not
cardiomyocyte cytoarchitecture is affected in two mouse models for
dilated cardiomyopathy (DCM), a widespread disease in people. In both
strains, biochemical and morphological alterations were observed at the
intercalated disks, the specialized areas of mechanical coupling between
cardiomyocytes. Of the proteins associated with myofibril attachment,
N-RAP alone was increased early in development in the DCM-susceptible
mice when compared with normal mice, even before the phenotype of DCM
became apparent. If N-RAP appears before alterations in levels of other
proteins or clinical signs of DCM, it could potentially serve as an
early marker for the disease. Further observations of the binding
properties between N-RAP and another protein concentrated at the
intercalated disk suggested the presence of a feedback mechanism that
may be involved in the onset of DCM.
Future studies will further
clarify N-RAP's role in myofibrillogenesis. Dr. Horowits plans to
continue exploring the targeting and function of GFP-labeled N-RAP
domains in cultured cardiomyocytes. Video or time-lapse microscopy will
be used to obtain a more detailed picture of the timing of key events
and may be extended to observe N-RAP domains in cultured skeletal
myoblasts, myotubes, and cells that do not normally express N-RAP, such
as those from smooth muscle or non-muscle cells. He also plans to
continue his efforts to biochemically purify and characterize N-RAP,
building on the successful enrichment of N-RAP in a fascia
adherentes fraction from cardiac muscle. The yeast two-hybrid system
will be used to screen cDNA libraries from striated muscle for proteins
that can interact with full length N-RAP and defined regions of N-RAP.
Muscle N-RAP will be assayed for post-translational modifications, with
particular emphasis on phosphorylation. Through these varied approaches,
Horowits hopes to extend our understanding of muscle assembly in health
and disease.
Selected Publications
Greenberg, C.C., P.S. Connelly, M.P. Daniels, and R. Horowits. Krp1 (sarcosin) promotes lateral fusion of myofibril assembly intermediates in cultured mouse cardiomyocytes. Exp Cell Res, 314: 1177-1191, 2008.
![]()
Horowits, R. Nebulin regulation of acting filament lengths: new angles. Trends in Cell Biology 16. 2006; 121-124.
![]()
Dhume, A., S. Lu, and R. Horowits. Targeted disruption of N-RAP gene function by RNA interference: A role for N-RAP in myofibril organization. Cell Motil Cytoskeleton. 2006; 63: 493-511.
![]()
Lu S, Borst DE, Horowits R. N-RAP expression during mouse heart development. Dev Dyn. 2005; 233: 201-212.
![]()
Carroll S, Lu S, Herrera AH, Horowits R. N-RAP scaffolds I-Z-I assembly during myofibrillogenesis in cultured chick cardiomyocytes. J Cell Sci. 2004; 117(Pt 1): 105-14.
![]()
See complete list of publications
Updated September 17, 2007