Electromagnetic Spectrum
Measuring the electromagnetic spectrum
You actually know more about it than you may think! The
electromagnetic
(EM) spectrum is just a name that scientists give a bunch of types of
radiation when
they want to talk about them as a group. Radiation is energy that travels and
spreads out as it goes--
visible light that
comes from a lamp in your house and
radio waves that
come from a radio station are two types of electromagnetic radiation. Other examples of EM radiation are
microwaves,
infrared and
ultraviolet
light,
X-rays and
gamma-rays.
Hotter, more energetic objects and events create higher energy radiation
than cool objects. Only extremely hot objects or particles moving at
very high velocities can create high-energy radiation like X-rays and
gamma-rays.
Here are the different types of radiation in the EM spectrum, in
order from lowest energy to highest:
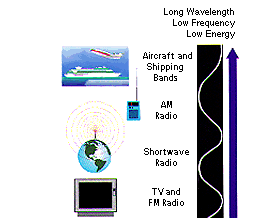
|
Radio: Yes, this is the same
kind of energy that radio stations emit
into the air for your boom box to capture and turn into your favorite
Mozart, Madonna, or Justin Timberlake tunes. But radio waves are also emitted by
other things ... such as
stars and
gases in space. You may not be able to
dance to what these objects emit, but you can use it to learn
what they are made of. |
 |
Microwaves: They
will cook your popcorn in just a few minutes! Microwaves in
space are used by astronomers
to learn about the structure of nearby galaxies, and our
own Milky Way! |
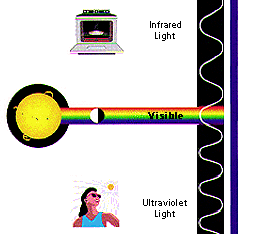 |
Infrared: We often think
of this as being the same thing as 'heat',
because it makes our skin feel warm. In space, IR light maps the
dust between stars.
Visible: Yes, this is the part that our eyes see. Visible radiation
is emitted by everything from fireflies to light bulbs to stars ...
also by fast-moving particles hitting other particles.
Ultraviolet: We know that the Sun is a source of ultraviolet (or UV)
radiation, because it is the UV rays that cause our skin to
burn! Stars and other "hot" objects in space emit UV radiation.
|
 |
X-rays: Your doctor uses
them to look at your bones and your dentist
to look at your teeth. Hot gases in the Universe also emit X-rays
. |
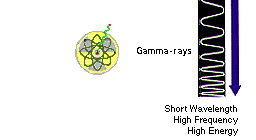 |
Gamma-rays:
Radioactive materials (some natural and others made by
man in things like nuclear power plants) can emit gamma-rays. Big
particle accelerators that scientists use to help them understand what
matter is
made of can sometimes generate gamma-rays. But the biggest gamma-ray
generator of all is the Universe! It makes gamma radiation in all
kinds of ways. |
A Radio Wave is not a Gamma-Ray, a Microwave is not an X-ray ...
or is it?

|
|
Radio waves, visible light, X-rays, and all the other parts of the electromagnetic spectrum are fundamentally the same thing, electromagnetic radiation.
|
We may think that radio waves are completely different physical
objects or events than gamma-rays.
They are produced in very different ways, and we detect them in different
ways. But are they really different things? The answer is 'no'.
Radio waves, visible light, X-rays, and all the
other parts of the electromagnetic
spectrum are fundamentally the same thing. They are all
electromagnetic
radiation.
Electromagnetic radiation can be described in terms of a stream of
photons, which are massless
particles each traveling in a wave-like pattern and moving at the
speed of
light. Each photon contains a certain amount (or bundle) of
energy, and all electromagnetic
radiation consists of these photons. The only difference between the various
types of electromagnetic radiation is the amount of energy found in the
photons. Radio waves have photons with low energies,
microwaves have a little more energy than radio waves, infrared has
still more, then visible, ultraviolet, X-rays, and ... the
most energetic of all ... gamma-rays.
 |
|
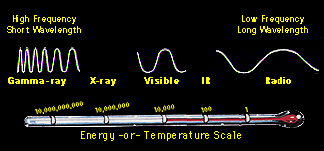
The electromagnetic spectrum can be expressed in terms of energy, wavelength, or frequency.
|
Actually, the electromagnetic spectrum can be expressed in terms of energy,
wavelength, or
frequency. Each
way of thinking about the EM spectrum is
related to the others in a precise mathematical way. So why do we have three
ways of describing things, each with a different set of physical units?
After all, frequency is measured in cycles per second (which is called a
Hertz), wavelength
is measured in
meters, and energy
is measured in
electron
volts.
The answer is that scientists don't like to use big numbers when they
don't have to. It is much easier to say or write "two
kilometers or 2
km" than "two thousand meters or 2,000 m". So generally,
scientists use whatever units are easiest for whatever they are working with.
In radio astronomy,
astronomers tend to use wavelengths or frequencies. This is
because most of the radio part of the EM spectrum falls in the range from
about 1 cm to 1 km (30 gigahertz (GHz) to 100 kilohertz (kHz)). The radio is a
very broad part of the EM spectrum. Infrared astronomers also use wavelength
to describe their part of the EM spectrum. They tend to use microns (or
millionths of meters) for wavelengths, so that they can say their part of the
EM spectrum falls in the range 1 to 100 microns. Optical astronomers use
wavelengths as well. In the older "CGS" version of the metric
system, the units used were angstroms. An angstrom is equal to 0.0000000001
meters (10-10 m in
scientific
notation)! In the newer
"SI" version
of the metric system, we think of visible
light in units of
nanometers or 0.000000001 meters (10-9 m). In this system, the
violet, blue, green, yellow, orange, and red light we know so well has
wavelengths between 400 and 700 nanometers. This range is only a small
part of the entire EM spectrum, so you can tell that the light we see is
just a little fraction of all the EM radiation around us!
By the time you get to the ultraviolet, X-ray, and gamma-ray
regions of the EM spectrum, lengths have become too tiny to think about any
more. So scientists usually refer to these photons by their energies, which
are measured in electron volts. Ultraviolet radiation falls
in the range from a few electron volts (eV) to about 100 eV. X-ray photons
have energies in the range 100 eV to 100,000 eV (or 100 keV). Gamma-rays then
are all the photons with energies greater than 100 keV.
 Show me a chart of the wavelength, frequency, and energy regimes
of the spectrum !
Show me a chart of the wavelength, frequency, and energy regimes
of the spectrum !
Why Do We Have to Go to Space to See All of the Electromagnetic
Spectrum?

Electromagnetic
radiation from space is unable to reach the surface of the Earth except at
a very few
wavelengths, such as the
visible spectrum,
radio
frequencies, and some ultraviolet wavelengths. Astronomers can get above enough of the Earth's atmosphere to
observe at some
infrared wavelengths from mountain tops or by flying their telescopes in
an aircraft. Experiments can also be taken up to altitudes as high as 35 km
by balloons which can operate for months. Rocket flights can take instruments
all the way above the Earth's atmosphere for just a few minutes before they
fall back to Earth, but a great many important first results in
astronomy and
astrophysics
came from just those few minutes of observations. For long-term observations, however, it is best to have your detector on an orbiting satellite ... and get above it all!
The vertical illustration of the different regions of the EM spectrum and
their common uses was done by Design at Work for the curriculum
supplement "Human Physiology in Space" by Barbara F. Lujan and
Ronald J. White, 1994. Used by permission.
|
