| |
|
2003 Analytical
Chemistry Division Archive News |
| |
|
———————————————————————————————
|
| |
|
NIST
Scientists Evaluate Detoxification of PCB-Contaminated Materials
Using Electron Beam Technology and Ultra-Violet Radiation
Dianne Poster,
Analytical Chemistry Division
|
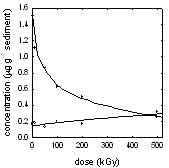
Concentration
(µg g-1) of PCBs (·) (S 29 congeners) and biphenyl
(o) in electron beam irradiated samples of SRM 1944, New York/New
Jersey Waterway Sediment, as a function of irradiation dose (kGy).
|
| |
|
The transformation
of polychlorinated biphenyls (PCBs) in environmental media into
compounds with a lower degree or absence of chlorine content has
been achieved. CSTL scientists, in collaboration with researchers
from the University of Maryland, College Park, have demonstrated
effective radiation-induced degradation of PCBs in water systems,
transformer oils, and marine sediments using electron beam technology.
This technology, which makes use of an electron beam from a linear
accelerator, has the potential to be the preferred treatment for
the cleanup of PCBs in environmental media when compared to currently
used methods such as incineration or organic solvent washing. Gaseous
and particulate waste effluents that result from incineration are
minimized and the process does not produce solvent waste streams
that must be treated or disposed of properly. In addition, radiation
processes are inherently selective to the target chlorinated compounds,
leading to either total dechlorination or to products with reduced
chlorine content that are more susceptible to bioremediation.
To investigate
the application of an electron beam to the dechlorination of PCBs
in marine sediment, samples of Standard Reference Material (SRM)
1944, New York/New Jersey Waterway Sediment, were mixed with aqueous
alcohol solutions and irradiated. Isopropanol was added to enhance
radiolytic yields of reductants and, thus, dechlorination of contaminants.
In the electron beam irradiated samples, the concentrations of 29
PCB congeners were found to decrease as a function of dose. At the
highest dose (500 kGy) the total concentration of PCBs was decreased
by 83 percent. This research has demonstrated the feasibility of
this approach and has provided initial design parameters for possible
scale-up. Further experiments are underway to examine the use of
other additives, particularly those that are more environmentally
benign (such as ascorbate) that will also enhance the dechlorination
process.
An alternative
treatment of PCBs in environmental media may be the use of ultraviolet
(UV) radiation. PCBs in transformer oils have been fully degraded
using radiation generated by a UV lamp. The application of this
method to marine sediment slurries led to little dechlorination
in the presence of isopropanol, but with added triethylamine dechlorination
was achieved (about 60 percent). It is likely that photolysis under
optimal conditions (other additives, additional exposure time) may
prove as effective as electron beam treatment for the dechlorination
of PCBs in sediment. Moreover, the photochemical efficiency can
be increased by an order of magnitude or more by using a quartz
window or a windowless configuration, using a better UV source,
and improving the irradiation geometry. These parameters and the
use of environmentally friendly additives are under study.
June 2003
|
| |
|
———————————————————————————————
|
| |
|
New
SRM Being Developed for Heart Attack Marker
David Bunk,
Analytical Chemistry Division
|
| |
|
The
clinical measurement of serum cardiac troponin I (cTnI) has become
an important tool in the diagnosis of acute myocardial infarction
and myocardial damage. Unfortunately, considerable variability in
clinical cTnI assay results has been reported. A ten-fold difference
in assay response between methods is common, but as much as a hundred-fold
difference in response has also been observed. With so much variation
between assays, physicians and clinical laboratory staff are required
to establish their own decision points for cTnI based on the assay
used. Problems can arise when different assays are being used, such
as when clinical laboratories change assays, attempt to correlate
results between point-of-care testing platforms and the central laboratory,
or when a clinician compares results from different laboratories.
Standardization of clinical cTnI measurements is needed to provide
more reliability in the use of cTnI assays for the diagnosis of myocardial
infarction and damage.
With the assistance
of the American Association of Clinical Chemistry (AACC), the International
Federation of Clinical Chemistry (IFCC), and the manufacturers of
clinical cTnI assays, NIST is developing a cTnI reference material
to address the need for assay standardization. Through two interlaboratory
comparison studies, various troponin preparations, including recombinant
and native troponin complexes of the troponin T, troponin I, and
troponin C subunits, recombinant troponin IC complex, and recombinant
and native cTnI, were evaluated using fifteen commercial cTnI assays.
Based on the results of these interlaboratory comparison studies,
NIST Standard Reference Material (SRM) 2921 will be a purified human
cardiac troponin complex of the troponin T, troponin I, and troponin
C subunits. The native troponin complex standardized clinical assays
better than the other troponin preparations evaluated. Additionally,
the native troponin complex was found to be commutable when compared
to pooled patient samples for all fifteen assays included in the
study. The troponin complex, purified from healthy human hearts
under non-denaturing conditions, is produced for NIST by HyTest,
Ltd. in Turku, Finland.
NIST SRM 2921
will be supplied as a frozen aqueous solution of the troponin complex.
Characterization of the structures of the troponin subunits will
be performed by mass spectrometry and a certified value for the
concentration of the troponin I subunit will be determined by a
variety of analytical techniques, including amino acid analysis.
Manufacturers of commercial cTnI assays will be able to use SRM
2921 for quality control purposes and to establish direct SI-traceability
of assay measurements, as well as for the value assignment of secondary
reference materials.
SRM 2921 is
expected to be available in the fall of 2003.
June 2003
|
| |
|
———————————————————————————————
|
Law
Enforcement - New NIST Standards Say ‘Hairs’ to Better
Drug Testing
Michael Welch,
Analytical Chemistry Division
|
|
|
|
Traditional
methods of screening job applicants, athletes and others for illicit
drug use increasingly are being supplanted or complemented by hair
analysis, which offers several testing advantages. The accuracy
of such hair tests now can be quality assured through the use of
two new Standard Reference Materials (SRMs) from the National Institute
of Standards and Technology (NIST).The
new NIST standards, which consist of human hair segments that have
been soaked in solutions containing target drugs, help validate
the accuracy of test methods for detecting those drugs. The first,
SRM 2379, is designed for tests of stimulants (“uppers”)
such as cocaine and PCP. Its companion SRM, number 2380, checks
tests for depressants (“downers”), such as codeine and
THC (the active ingredient in marijuana).As
new hair tissue forms in the roots, drugs and other chemicals from
the bloodstream may be absorbed into and retained by the growing
follicles. Hair’s advantages in drug testing—when compared
to using fluids such as urine—are that it can be collected
more easily, is more difficult to switch or contaminate, and retains
traces of drugs for at least 90 days (not just two or three). However,
hair analysis generally is not applicable to the detection of drug
use initiated within the past 10 days. Therefore, it may be favored
as a complement to, rather than a substitute for, traditional methods.NIST
began researching the analysis of drugs in hair in 1990 and has
conducted seven interlaboratory comparison exercises to see how
well different methods work, and in some cases, how well different
laboratories conduct such tests.
May 2003
|
|
|
———————————————————————————————
|
|
Glass
SRM May be Boon to Gumshoes
Steven
Choquette, Analytical Chemistry Division
|
|
|
|
The capability
to identify chemical substances easily and accurately at a crime
scene or other location outside a laboratory, without handling the
material or opening containers, would be a boon for many in science.
A new NIST standard that reduces calibration costs as much as 20-fold
represents a major step toward making such a tool practical. A small
piece of chromium-doped glass, Standard Reference Material (SRM)
2241, will enable users to calibrate the output of Raman spectrometers.
Without this SRM, full calibration of these instruments is so expensive
that many users skip it and, therefore, may get inaccurate results.
Raman spectroscopy reveals the chemical composition of a sample
by illuminating it with a laser and then identifying color changes
in a very small amount of the scattered light. The technique is
simple enough to use in the field and, unlike some competing methods,
can be used to measure samples through transparent containers.
May 2003
|
|
|
———————————————————————————————
|
|
|
|
SRM
1946 Lake Superior Fish Tissue
Stephen Wise, Analytical
Chemistry Division
|
|
|
This SRM has
been developed for evaluating analytical methods for determining
polychlorinated biphenyl (PCB) congeners, chlorinated pesticides,
fatty acids, extractable fat, methylmercury, total mercury, and
selected trace elements in fish tissue and similar matrices. Of
particular interest are persistent environmental and tissue contaminants
such as PCBs and chlorinated pesticides, some of which are suspected
carcinogens or have been banned for other reasons; and mercury and
methylmercury, which are highly toxic, the latter to nerve tissue.
SRM 1946 is the first to provide certified values for three non-ortho-substituted
PCBs that are considered the most toxic because of their structural
resemblance to 2,3,7,8-tetrachlorodibenzo-para-dioxin (TCDD).
In addition, this SRM is intended for analysis of food proximates
(fat, protein, and carbohydrates) and other constituents of interest
in food analysis. For example, fish containing more than a level
of methylmercury established by regulation is not to be consumed.Certified
values are provided for 30 PCB congeners and 15 chlorinated pesticides,
respectively. These values were obtained via two or more independent
analytical techniques, based principally on chromatographic and
mass spectrometric methods.
Certified values also are given for total extractable fat and 13
individual fatty acids; these are based on measurements made by
NIST and collaborating laboratories. Certified values for methylmercury,
total mercury, arsenic, and iron are based on results of two or
more independent analytical procedures, performed at NIST and collaborating
laboratories.
SRM 1946 also provides reference concentration values for 12 PCB
congeners, 2 chlorinated pesticides, 12 fatty acids, certain proximates
and elements, and caloric content. Certified, reference, and information
values are listed in the NIST Certificate of Analysis for Standard
Reference Material® 1946, issued 7 October 2002.
One unit of SRM 1946 consists of five bottles, each of which contains
approximately 10 g to 12 g (wet basis) of frozen (not freeze-dried)
tissue homogenate. This SRM is currently in preparation.
May 2003
|
|
|
———————————————————————————————
|
|
|
|
|
|
|
The Nutrition
Labeling and Education Act of 1990 requires that information for
selected nutrients is provided on labels for processed foods. In
response, NIST has been working to provide food-matrix SRMs with
values assigned for the required nutrients. SRM 2387 Peanut Butter
is the most recent SRM in this series. SRM 2387 is intended for
use as a primary control material for assigning values to in-house
control materials and to validate methods for measuring nutrients
such as fat, protein, calcium, iron, calories, and vitamins. This
is the first food-matrix reference material available from NIST
for which values are also assigned for individual amino acids and
for aflatoxins.
To study the robustness of analytical methods, AOAC International
developed a nine-sector triangle in which foods are positioned based
on their fat, protein, and carbohydrate content. The idea was that
one or two foods within each sector should be representative of
other foods within that sector when validating an analytical method.
Similarly, one or two food-matrix reference materials in each sector
can be used as control materials for other foods within that sector.
With the release of SRM 2387, one or more reference materials in
each of the nine sectors are available from NIST. This SRM is currently
in preparation.
May 2003
See a recent
article on SRM 2387 and the completed food triangle at The
New York Times.
|
|
|
———————————————————————————————
|
|
SRM
3072 Diquat Dibromide Monohydrate in Water
Lane Sander,
Analytical Chemistry Division
|
|
|
|
Standard Reference
Material (SRM) 3072 is a solution of diquat dibromide monohydrate
(Chemical Abstracts Registry Number 85-00-07) in water intended
primarily for use in the calibration of chromatographic instrumentation
used for the determination of diquat dibromide monohydrate. This
SRM can also be used to fortify aqueous samples with known amounts
of diquat dibromide monohydrate. A unit of SRM 3072 consists of
five 2 mL ampoules, each containing approximately 1.2 mL of solution.
Certified Concentration of Diquat Dibromide: The certified
concentration value [1,2], given below, is based on results obtained
from the gravimetric preparation of this solution and from the analytical
results determined by using liquid chromatography (LC). A NIST certified
value is a value for which NIST has the highest confidence in its
accuracy in that all known or suspected sources of bias have been
investigated or accounted for by NIST. Diquat
Dibromide Monohydrate 39.7 mg/kg ± 0.8 mg/kg
A unit of SRM 3072 consists of five 2 mL ampoules, each containing
approximately 1.2 mL of solution.
May 2003
|
|
|
———————————————————————————————
|
|
SRM
2702 Inorganics in Marine Sediment
Rolf
Zeisler, Analytical Chemistry Division
|
|
|
Standard Reference
Material (SRM) 2702 is a marine sediment collected at the mouth
of the Baltimore Harbor, Baltimore, MD. SRM 2702 is intended for
use in evaluating analytical methods for the determination of selected
elements in marine or fresh water sediment and similar matrices.
All of the constituents in SRM 2702, for which certified, reference,
and information values are provided, were naturally present in the
sediment material before processing.
Certified values for concentrations, expressed as mass fractions,
for 25 elements. The certified values are based on the agreement
of results from two or more chemically independent analytical techniques
obtained at NIST and collaborating expert laboratories [1]. A NIST
certified value is a value for which NIST has the highest confidence
in its accuracy in that all known or suspected sources of bias have
been investigated or accounted for by NIST.
The sediment used to prepare this SRM was collected from the Chesapeake
Bay at the mouth of the Baltimore (MD) Harbor near the Francis Scott
Key Bridge (39°12.3'N and 76°31.4'W). This location is very
near the site where SRM 1941 and SRM 1941a were collected. The sediment
was collected using a Kynar-coated modified Van Veen-type grab sampler.
A total of approximately 3300 kg of wet sediment was collected from
the site. The sediment was freeze-dried, sieved at 70 µm (100
% passing), homogenized in a cone blender, radiation sterilized
at 33 kGy to 45 kGy (60Co) dose, and then packaged in screw-capped
amber glass bottles each containing approximately 50.
A unit of SRM 2702 consists of a bottle containing 50 g of radiation-sterilized,
freeze-dried sediment material. This SRM is currently in preparation.
May 2003
|
|
|
———————————————————————————————
|
|
Tiny
Cell-Based Chemical Detectors Have Protection Potential
Laurie
Locascio,
Analytical
Chemistry Division
|
|
|
|
|
|
Laurie
Locascio places a water sample on a tiny sensing chip that detects
toxic chemicals. |
|
|
| A
highly sensitive, inexpensive “lab-on-a-chip” that provides
warning within seconds of even trace amounts of toxic chemicals in
water was designed and demonstrated recently by National Institute
of Standards and Technology (NIST) scientists and collaborators.
The prototype
sensor system monitors the natural response of bacterial cells bound
within the microscopic channels of a plastic microfluidics device—a
miniaturized chemical and biochemical analysis system. In the presence
of certain chemicals, the cells eject large amounts of potassium,
which is detected with an optical sensor that changes color. The
prototype was demonstrated as part of an early warning system for
industrial pollutants that interfere with sewage treatment, but
it also has potential homeland security applications.
Cell-based sensors
are of great interest today because they can respond to a wide range
of chemical toxins rapidly. NIST’s primary contributions to
this project involve expertise in microfluidics technology, particularly
aspects such as plastics processing. The new device has a novel
configuration in which, through the use of lasers, tiny posts are
constructed within the channels to act as a sieve and promote adhesion
of the cells.
Although this
type of chemical test could be performed in other formats, a microfluidics
device is more sensitive because of the high surface to
volume ratio, and also faster because of the close juxtaposition
of chemicals and cells. In addition, it consumes less reagent and
sample material and could be used in a distributed sensor network
for real-time field testing. The device was designed in collaboration
with scientists at Virginia Polytechnic Institute and State University
(Blacksburg, Va.) and Veridian Pacific-Sierra Research (Charlottesville,
Va.).
March 2003
|
|
|
 Return to ACD Home
Return to ACD Home
 Return to Top of Document
Return to Top of Document
Date
created: May 13, 2002
Last updated:
May 6, 2004
Contact: acd_webmaster@nist.gov
|
|
|

