 |
|
Clinical Research Program Perry J. Blackshear, M.D., D.Phil.(http://www.niehs.nih.gov/research/atniehs/labs/lst/polypeptide/index.cfm)
Principal Investigator Research SummaryThe Gene Environment Interactions Group (GEIG) conducts diverse clinical research studies aimed at understanding the complex interaction between genes and the environment in polycystic ovary syndrome, diabetes, asthma, autoimmune, respiratory and cardiovascular diseases, and neural tube defects and others. A wide variety of study designs are used including twin multivariate analyses, and case-control and pedigree study designs. 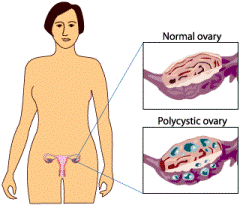
Figure A.
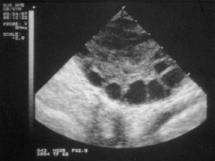
Figure B.
Figure A: About 75-80% of women with PCOS actually develop ovarian cysts. The other 20-25% of PCOS women do not. With PCOS, these appear as multiple, smaller cysts around the edge of the ovary, each being only a few millimeters in size. Figure B: On ultrasound, they give a characteristic "string of pearls" appearance.
The GEIG has combined its interest in twin research with examining the etiology of polycystic ovary syndrome (PCOS) and type 1 diabetes. For PCOS, the group is collaborating with the Mid-Atlantic Twin Registry, the University of British Columbia Twin Registry, and others to assemble a large cohort of intact female twin pairs in which at least one member has PCOS(http://www.niehs.nih.gov/research/clinical/program/groups/gei/pcos.cfm) . The twins will be studied in several phases to first estimate the relative contributions of genes and environmental to the syndrome, and then to identify specific genetic, epigenetic and environmental factors that are involved in its development. In a second unrelated twin study, the GEIG has assembled a smaller cohort of twins in collaboration with Dr. David Leslie at St. Bartholomew's Hospital in London to examine whether maternofetal microchimeric cells (cells passed from the mother to fetus during gestation) can influence the development of type 1 diabetes by stimulating an autoimmune process in these genetically susceptible individuals. Clinical research within the GEIG is also focused on characterizing human genetic variation and identifying genetic susceptibility loci for a diverse set of complex conditions. The genes under study were originally discovered and cloned by Blackshear's basic research group, and include the: 1) the tristetrapolin (TTP) family of genes (code zinc finger proteins that bind and destabilize message for TNFα and GM-CSF); and 2) the myristoylated alanine-rich C kinase substrate (MARCKS) and its related MARCKS-like protein (MLP) (developmental genes that code for protein kinase C substrates). Genetic association studies are being conducted for autoimmune conditions, respiratory and cardiovascular disease (TTP gene family), and neural tube defects and autism (MARCKS and MLP). Major areas of research:
Current projects:
|
|

