CDC Features
Current Features
Vaccine Safety
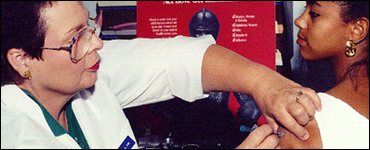
Vaccines are one of the greatest success stories in public health. Vaccines have ended smallpox, nearly ended polio, and reduced outbreaks of measles, pertussis, and other illnesses to an all-time low.
Vaccines are our best defense against infectious diseases, but no vaccine is 100% safe or effective. People react differently to vaccines, so some people have side effects or are not protected from the disease. Scientists are trying to develop safer, more effective vaccines.
Testing Vaccines Before Licensing
Vaccines must be licensed by the U.S. Food and Drug Administration (FDA) before they can be used in the United States. Before the FDA approves a license, a vaccine is tested extensively to ensure it is safe.
Monitoring Vaccines After Licensing
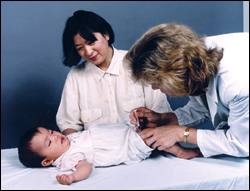
After a vaccine is licensed for public use, its safety is monitored in several ways. Manufacturers must provide the FDA with samples from each vaccine lot before its release, plus the manufacturer's test results for vaccine safety, potency, and purity. The Vaccine Adverse Event Reporting System (VAERS) and Vaccine Safety Datalink (VSD) project also monitor the safety of vaccines.
• Rare side effects and delayed reactions may not happen until the vaccine is given to millions of people. The Vaccine Adverse Event Reporting System (VAERS) takes reports of health problems that happen after vaccination so scientists can find out if a vaccine is causing a health problem.
• The Vaccine Safety Datalink (VSD) project stores medical records for 5.5 million people who are members of participating managed care organizations, and watches them for possible side effects of vaccination.
Concerns About Vaccine Safety
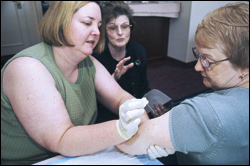
Vaccines, like any medication, can cause side effects. However, a decision not to immunize a child also involves risk. It is a decision to put the child and others who come into contact with him or her at risk of getting a disease that could be deadly.
Consider measles. One out of 30 children with measles develops pneumonia. For every 1,000 children who get the disease, one or two die from it. Thanks to vaccines, we have few cases of measles in the U.S. today. However, the disease is extremely contagious, and each year dozens of cases are imported into the U.S. from other countries.
Why It's Important to Monitor Vaccine Safety
The most important reason is to detect reactions that are so rare they are not found during testing before a vaccine is licensed. Monitoring also makes sure new vaccines are safe for groups such as the elderly, those with chronic medical conditions, and pregnant women, who may not be included in pre-licensing tests. Monitoring vaccine safety also helps maintain public confidence so enough people are vaccinated to prevent disease outbreaks.
More Information on Vaccine Safety
Page last updated: April 21, 2008
Content sources: Immunization Safety Office
Content owner: National Center for Health Marketing
URL for this page: www.cdc.gov/Features/VaccineSafety

