Back to: News & Events : Past Meetings and Workshops : NCCAM Grantsmanship 2008
Lifecycle of a Grant Application
June 2008 – Presentation delivered at NCCAM Grantsmanship Workshop
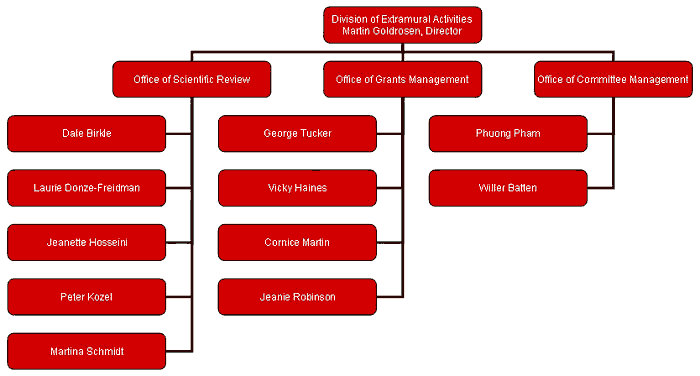
Martin Goldrosen, DEA
Dale Birkle, SRO
George Tucker, GMO
Overview
- Overall Application Process
- e-Submissions
- Receipt and Referral
- Peer Review
- Advisory Council
- Cost Analysis by Grants Management
- Just-in-Time Information
- Notice of Grant Award
Overall Application Process
Review Process for a Research Grant
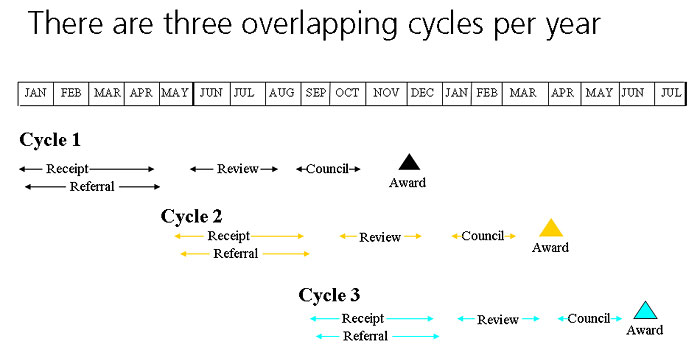
Overall Timeframe from Submission to Award
Dual Level of Review for Applications
First Level of Review
Scientific Review Group (SRG)
- Provides Initial Scientific Merit Review of Grant Applications
- Rates Applications and Makes Recommendations for Appropriate Level of Support and Duration of Award
Second Level of Review
Council
- Assesses Quality of SRG Review of Grant Applications
- Makes Recommendation to Institute Staff on Funding
- Evaluates Program Priorities and Relevance
- Advises on Policy
E-Submissions
Overall Application Process
- Prepare to Apply
- Find Opportunity and Download Application Package
- Prepare Application
- Submit Application to Grants.gov (AOR submits)
- Check Submission Status in Commons
- Check Assembled Application
- Submission Complete
Prepare to Apply
Future Transitions
All applications now require electronic submission with the exception of the following mechanisms:
- Career Development (Feb 09)
- Fellowship (April 09)
- Training (T&D, Sept 09)
- Complex, Multi-project Applications (Project/Centers)
These use the paper PHS398 application process
Two Systems Working Together
Grants.gov- Federal government’s single online portal to find and apply for Federal grant funding
- Used by all 26 Federal grant-making agencies
- Organizations must register
- Authorized organization rep (AOR) submits applications
- electronic Research Administration system
- Applicants/grantees receive and transmit application and award information
- Used by NIH and other HHS components
- Signing Officials AND Principal Investigators must register
- PIs: Fill out forms completely and keep them updated
eRA Commons
Both Organization and PI Registration Required
Registration eRA Commons –PI
- Principal Investigators (PIs) must work through their institution's Signing Official (SO) to register
- The PI and SO need separate accounts in eRA Commons because each has different privileges
- PIs have one account that follows them throughout their careers — the same account may be affiliated with multiple organizations
- CRITICAL to fill out forms completely, check NIH contributions (grant history), and keep it UPDATED
- Allow 4–5 business days to complete
Internal Process Considerations
Find Out How Your Organization Manages…
- Internal review & approval processes
- Sharing applications in progress
- Last minute queue at Sponsored Programs
…Calculate lead time needed
Make Sure You Have Necessary Software
Software Requirements
- PureEdge Viewer — free at Grants.gov
- PDF generation software — list provided on Grants.gov Web site
- New Adobe eForms will replace PureEdge
- Platform Independent
- Only requires free Adobe Reader*
Transition to begin Dec. 2008
Find Opportunity and Download Application Package
Find Opportunity & Download Package
- ALL applications must be submitted in response to a Funding Opportunity Announcement (FOA)
- NIH simultaneously posts FOAs on Grants.gov and in the NIH Guide for Grants and Contracts http://grants.nih.gov/grants/guide/index.html
- NCCAM website (www.nccam.nih.gov)
Find Opportunity & Download FOA
- Read the FOA carefully for specific application instructions
- Within the announcement — Click the Apply for Grant Electronically button
Submit Application to Grants.gov (AOR submits)
On-Time Submission
- All registrations must be completed prior to initial submission
- Initial submission must have a Grants.gov timestamp before 5:00 p.m. local time of submitting organization on the .receipt date (found in FOA)
- For STANDARD receipt dates only:
- Two week extension for members of review panels
- Continuous submission for members of standing study sections
- For details, see http://grants.nih.gov/grants/guide/notice-files/NOT-OD-08-027.html and http://grants.nih.gov/grants/guide/notice-files/NOT-OD-08-026.html
Back to NIH
You’re not done yet
Make sure you follow the application through to NIH
Check Submission Status in Commons
eRA Will Check To Ensure…
- Once NIH retrieves the application from Grants.gov, eRA systems check the application against NIH business rules
- Detailed check against the instructions in the application guide
- Validates against the instructions in the FOA
- Page limits
- Attachments, missing sections
- Missing information in Commons
- For a detailed list of NIH validations: http://era.nih.gov/ElectronicReceipt/prepare_app.htm#5
If You Receive Errors & Warnings .from NIH…
- Errors must be addressed for the application to move on to NIH Receipt & Referral
- An application must be error-free in order for eRA systems to generate an image of the application for viewing
- Warnings may be fixed at the applicant’s discretion but do not require action for the application to move on
- If the PI corrects any Warnings or Errors, they should contact the AOR/SO. The AOR/SO must submit the entire 'Changed/Corrected' application again through Grants.gov.
- Use changed/corrected check box on SF424(R&R) form
NOTE: Reviewers do not see notifications provided to applicants about errors/warnings.
Application Image — Validation and Errors
- Applicants have 2 weekdays (M-F, excluding federal holidays) to correct errors
- During this time period, the Signing Official (SO) has option to "Reject"
- If "Rejected" for any reason.other than a system formatting issue, subsequent corrected submission is subject to the NIH late policy
- PI will receive email notification if SO rejects application
Submitting Changed/Corrected Application: Cover Letter
If submitting a changed/corrected application during the 2-business day window after the receipt date, the cover letter must explain changes
- If your original submission included a cover letter then you must include all previous cover letter text in the revised cover letter attachment
- Reviewers do not see cover letters
Check Status in eRA Commons
If the application is error-free the eRA Commons system will:
- Assemble the grant image
- Generate Table of Contents and bookmark important sections
- Include headers (PI name) & footers (page numbers) on all pages
https://commons.era.nih.gov/commons/
E-mail Notifications — eRA Commons
AOR/SO and PI receive e-mails
- Validations Complete/Check Assembled Application
- Validations Complete/Check Assembled Application — Warnings Only
- Action Required: Errors and/or Warnings, Missing or Invalid Commons Username, System Error
- SO has refused application — SO rejected the application
Check Assembled Application
Application Image — Viewing Window
- After NIH receives an error-free application, an assembled application image is generated and placed in the eRA Commons
- Applicants have 2 weekdays (M-F, excluding federal holidays) to view the assembled application before it automatically moves forward to NIH's Division of Receipt and Referral
- Once the application moves to Receipt and Referral, the image cannot be replaced. Only options for changes is e-Additions to the e-Grant folder
Submission Complete
Application Complete!
Receipt and Referral
Peer Review of NIH Support Mechanisms
CSR Review
- Research Project Grant (R01, R03, R21)
- Individual Fellowships (F31, F32)
- Fogarty Fellowship (F05, F06)
- Short-Term Training (T35)
- Small Business Grants (R41, R42, R43, R44)
- Academic Research Enhancement Award (R15)
Institute Review
- Program Project Grant (P01)
- Center Grant (P50)
- Institutional Fellowship (T32)
- Career Development Awards (K01, K05, K07, K08, etc.)
- Cooperative Centers (U19)
- Conference Grant (R13)
- Contracts
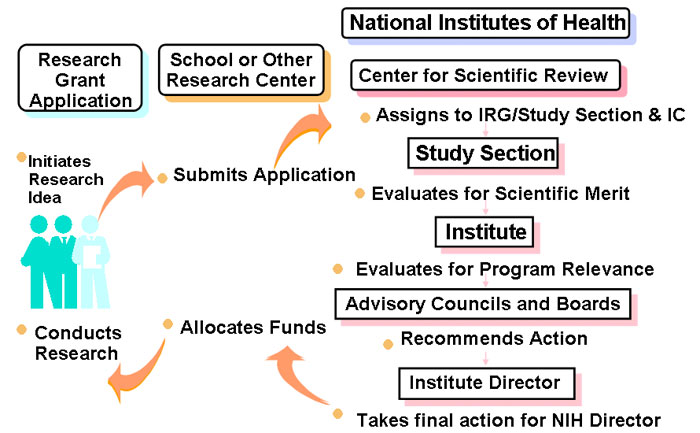
Center for Scientific Review
- Serves as central receipt point for most PHS grant applications
- Assigns applications to CSR Integrated Review Groups/Study Sections or Institute Scientific Review Groups
- Assigns applications to NIH Institute(s) as potential funding component(s)
- Conducts initial scientific merit review of most research applications submitted to the NIH
- Conducts review of NCCAM R01s
Referral Officers
- Scientists that are highly experienced Scientific Review Officers
Applications are Assigned to:
- Scientific review groups based on:
- Specific review guidelines for each scientific review group
- Grant mechanism
- Institutes based on:
- Overall mission of the Institute
- Specific programmatic mandates and interests of the Institute (PAs and RFAs)
Applications are Assigned by:
Other factors
- Self-referral in cover letter
- Word mining from cover letter and abstract
- PI history
- Requests from Program Officers
Assignment to CSR Study Sections
Applications (NCCAM R01s, SBIRs) are assigned to Integrated Review Groups (IRGs) for review. IRGs are clusters of scientifically-related study sections
- The IRG assignment is based on specific referral guidelines for each IRG
- Each of the IRGs within CSR has 5 – 8 standing study sections and several special emphasis panels (SEP)
- Interest statements for IRGs and study sections can be found at: http://cms.csr.nih.gov/PeerReviewMeetings/CSRIRGDescription/
Within an IRG, applications are assigned for review to
- Standing Study Sections when the subject matter of the application matches the referral guidelines for the study section
- Ad Hoc Special Emphasis Panels (SEPs) when the subject matter does not fit into any study section, or when assignment of an application to the most appropriate study section would create a conflict of interest. Also used for special mechanisms (e.g., fellowships, SBIRs, AREAS)
CSR Review Divisions
Division of Biologic Basis of Disease
- AIDS and Related Research IRG (AARR)
- Endocrinology & Reproductive Sciences IRG (ENR)
- Immunological Sciences IRG (IMM)
- Infectious Diseases and Microbiology IRG (IDM)
- Nutritional and Metabolic Sciences IRG (NMS)
- Oncological Sciences IRG (ONC)
Division of Molecular and Cellular Mechanisms
- Biochemical Sciences IRG (BCS)
- Biophysical and Chemical Sciences IRG (BPC)
- Cell Development and Function IRG (CDF)
- Genetic Sciences IRG (GNS)
- Molecular, Cellular and Developmental Neurosciences IRG (MCDN)
Division of Physiology and Pathology
- Cardiovascular Sciences IRG (CVS)
- Hematology IRG (HEM)
- Integrative, Functional & Cognitive Neuroscience IRG (IFCN)
- Musculoskeletal & Dental Sciences IRG (MSD)
- Pathophysiological Sciences IRG, (PPS)
Division of Clinical and Population-Based Studies
- Behavioral & Biobehavioral Processes IRG (BBBP)
- Brain Disorders and Clinical Neuroscience IRG (BDCN)
- Risk, Prevention and Health Behavior IRG (RPHB)
- Social Science, Nursing, Epidemiology and Methods IRG (SNEM)
- Surgery, Radiology, and Bioengineering IRG (SRB)
Assignment to Institutes
Applications are referred to an Institute or Center as the potential funding component:
- This assignment is based on a match between the research proposed and the overall mission of the Institute or Center
- Sponsoring Institutes are listed at the top of Funding Announcements
- Specific research interests are in the text of the Funding Announcement
- Where applications are appropriate for more than one Institute or Center, multiple assignments are made
- NCCAM's priorities http://nccam.nih.gov/research/priorities/index.htm#5
Locus of Review
CSR
- Research Project Grant (R01, R03, R21)
- Individual Fellowships F31, F32)
- Institutional Fellowship (T32)
- Fogarty Fellowship (F05, F06)
- Cooperative Centers (U19)
- Small Business Grants (R41, R42 R43, R44)
- Academic Research Enhancement Award (R15)
Institutes
- Program Project Grant (P01)
- Center Grant ( P50)
- Career Development Awards (K01, K05, K07, K08, etc.)
- Short-Term Training (T35)
- Conference Grant (R13)
- Contracts
Assignment to NCCAM Review Groups
Applications are assigned to the Integrated Review Group called ZAT1
Z = special emphasis panel
AT= Institute designation for NCCAM
Fellowships, Career Development, Exploratory, Conference, AREA, Multi-site R01s, Special Initiatives (RFAs, PAR, PAS)
Funding announcements will state the locus of review
"Applications that are complete will be evaluated for scientific and technical merit by an appropriate peer review group convened by NCCAM"
"Applications will be evaluated for scientific and technical merit by an appropriate peer review group convened by CSR"
Assignment to NCCAM Review Groups
Applications are further assigned to specific review groups
-
Basic Sciences R21s and R15s (DB00, now PK00)
Clinical Sciences R21s and multi-site R01s (JH00)
Training (all mechanisms, LD00)
Centers (SM00)
Review Group Member Conflicts
Requests for Applications
PAR: Special Review Criteria
PAS: Set-aside Funds
Sample Application Number
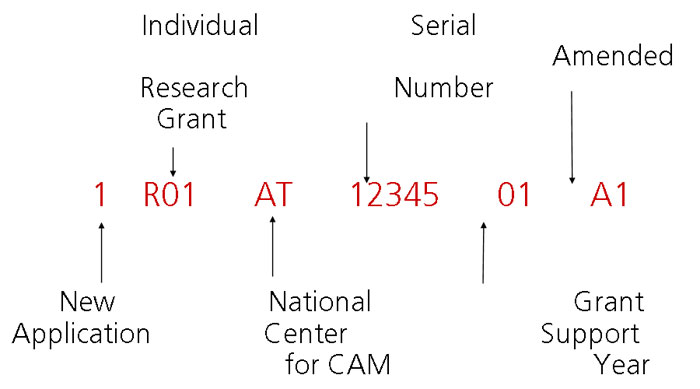
Requesting Re-assignment
- Contact your Program Officer and SRO
- Prepare to strongly justify your request
- Conflicts of Interest
- Inappropriate Expertise
- Have an alternative review group in mind
- Assignment is constrained by referral guidelines
- The SRO and PO usually know best!
- MUST be done within a few days of initial assignment
- Could be better to withdraw and re-submit than have review done by the wrong reviewers
Peer Review
- Each CSR standing review group has 12 - 24 members who are primarily from academia
- A Special Emphasis Panel (SEP) can have 6 to 60 members
- One to a hundred-plus applications are reviewed at each review meeting
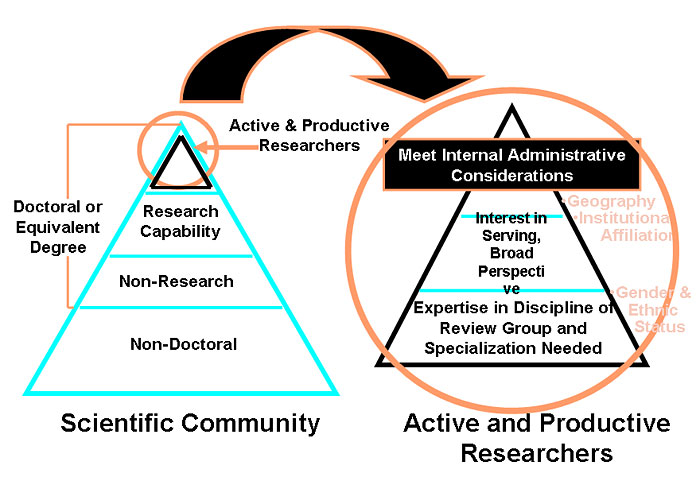
Criteria for Selection of Peer Reviewers
Certification of No Conflict of Interest
"This will certify that in the review of applications and proposals by (study section) on (date), I did not participate in the evaluation of any grant or fellowship applications from
- any organization, institution or university system in which a financial interest exists to myself, spouse, parent,child, or collaborating investigators;
- any organization in which I serve as officer, director, trustee, employee or collaborating investigator; or
- any organization which I am negotiating or have any arrangements concerning prospective employment or other such associations."
Confidentiality
- Review materials and proceedings of review meetings represent privileged information to be used only by reviewers and NIH staff.
- At the conclusion of each meeting, reviewers will be asked to destroy or return all review-related material.
- Reviewers should not discuss review proceedings outside of the meeting or with anyone except the SRO.
- Questions concerning review proceedings should be referred to the SRO.
Review Criteria
- Significance: Does the study address an important problem? How will scientific knowledge be advanced?
- Is the CAM therapy important, commonly used, traditionally used?
- Is there evidence of efficacy?
- Is there a strong rationale for doing the study?
- Approach: Are design and methods well-developed and appropriate? Are problem areas addressed?
- Don’t forget about guidelines for use of natural products http://grants.nih.gov/grants/guide/notice-files/NOT-AT-05-003.html http://grants.nih.gov/grants/guide/notice-files/NOT-AT-05-004.html
- Troubleshooting, alternative approaches
- Innovation: Are there novel concepts or approaches? Are the aims original and innovative?
- Just looking at CAM is not innovative
- Investigator: Is the investigator appropriately trained?
- Evidence of CAM interests and experience on the team
- Environment: Does the scientific environment contribute to the probability of success? Are there unique features of the scientific environment?
- Institutional support for CAM research
Additional Factors
- Use and protection of human subjects
- Minority, gender and age considerations
- Use and protection of animal subjects
- Biohazards
- Criteria specific to the FOA
- Progress for competing renewals
- Response to the prior critiques for amended
- Multiple leadership plan
Outcomes of Review
- Scored, Scientific Merit Rating
- Priority score
- Percentile (for R01 applications ONLY)
- Resume of discussion and critiques
- Unscored (lower half)
- Critiques
- Unscored
- Lower half of applications reviewed by the scientific review group
- No priority score is given
- The summary statement is a compilation of reviewers' comments
Advisory Council
NACCAM
- NCCAM's Advisory Council has 18 members consisting of CAM practitioners, conventional scientists and members of the public
- They are appointed by the Secretary of Health and Human services for 4 years
- NACCAM meets three times per year
Council Deliberations
- Is the proposed research significant from the perspective of your own area of expertise?
- Is it relevant to NCCAM's mission and areas of programmatic interest?
- How important is this individual study?
- Is the proposed research duplicative?
- Are there human subject or animal welfare concerns that have not been adequately addressed?
- Were there any serious problems with the initial review?
Council Action
- Concurrence with study section action
- Modification of study section action
- Deferral for re-review
What Determines Which Awards Are Made?
- Scientific merit
- Program considerations
- Availability of funds
Cost Analysis by Grants Management
Cost Analysis
- The grants office will ensure that a cost analysis is performed on any application that requires a detailed budget
- Cost analysis involves obtaining cost breakdowns, validating cost data, evaluating specific elements of cost, and examining data to determine the necessity for, and the reasonableness and allowability of, the costs included in the application budget
- The extent of cost analysis will depend on the type of funding instrument and award mechanism, the complexity of the project, prior experience with the applicant, and other factors
Allowable & Unallowable Costs
Costs must be…
- reasonable
- allowable
- allocable
- consistently treated
- conform to any limitations or exclusions by the agency
Direct Costs vs F&A Costs
Direct costs can be identified and charge to a specific project with relative ease and with a high degree of accuracy
Direct Costs
- Salaries and Fringe Benefits
- Equipment
- Consultants
- Travel
- Supplies
- Patient Care Costs
- Other Direct Costs
- Consortia
Applications ≥ $500,000
- Must contact & IC staff must agree to accept the application prior to a submission for any investigator-initiated competitive applications with ≥$500,000 direct cost for any single year (consortium F&A doesn't count)
- Request must be 6 weeks before deadline
see NIH Guide dated 10/16/2001
Modular Budgets
- Applications for R01, R03, R15, R21 and RFAs with no more than $250,000 direct costs/year
- Requested total direct costs in modules of $25,000 reflecting appropriate support for project. There will be no future year escalations in the traditional sense
Salaries and Wages
- Key Personnel — all individuals who contribute to the scientific development or execution of a project in a substantive, measurable way, whether or not salaries are requested (biosketch)
- Other Personnel
Which are appropriate for salaries?
- Technician
- Co-Investigator from another institution
- Graduate student researcher
- Secretary
When Direct Charging of Clerical Salaries May Be Appropriate
- Large complex programs that entail assembling and managing teams of investigators from a number of institutions
- Projects that involve extensive data accumulation, analysis and entry, surveying, tabulation, cataloging, literature searching or reporting
- Projects requiring project-specific database management; individualized graphics or manuscript preparation; human or animal protocols; and multiple project-related investigator coordination/communications
- Projects that are geographically inaccessible to normal departmental administrative services
- Projects that require making travel and meeting arrangements for a large number of participants
How do you convert percent effort to person months?
Multiply percentage of effort associated with the project, times the number of months of the appointment
- 10% of 12 mo appointment= 1.2 CY person months (12 X 0.10 = 1.2)
- 25% of 9 mo AY appointment= 2.25 AY person months (9 X .25 = 2.25)
- 35% of 3 mo summer appointment= 1.05 SM (3 X .35 = 1.05)
Person Months and Budgets
For an institution with a 9 month academic year, if the PI will devote 9 months at 30% time/effort and 3 months summer term at 30% time/effort to the project, then 2.7 academic months and .9 summer months should be listed in the academic and summer term blocks of the application (9 x 30% = 2.7 person months; 3 x 30%= 0.9)
12 mo appointment, $120,000 base salary, devoting 30% effort
$120,000 X 30% = $36,000
12 mos X 0.3 = 3.6 person months
Salary Cap
Since Fiscal Year 1990 Congress has legislatively mandated a provision limiting the direct salary that an individual may receive under an NIH grant.
The FY 2008, Public Law 110-161: Consolidated Appropriation Act, 2008 restricts the amount of direct salary of an individual under an NIH grant or cooperative agreement to Executive Level I of the Federal Executive pay scale.
Effective 1/1/2008, that level increased to $191,300.
Equipment is defined as…
…having a useful life of more than one year and an acquisition cost which equals or exceeds the lesser of the capitalization level established by the organization for financial statement purposes or $5,000.
Equipment costs analyzed…
- Is it allocable to this research?
- Particularly for expensive pieces of equipment, is use exclusive to this project?
- Does this lab need this equipment?
Supplies
- Itemize in categories
- Not necessary to itemize amounts less than $1,000
- Provide species and number of any animals
Travel
May include…
- travel to professional meetings (includes registration)
- travel to work with collaborators
- site visits for data collection
Other Direct Costs
May include…
- Animal care costs
- Patient travel
- Participant fees
- Publication costs
- Computer charges
Just-in-Time Information
Just-in-time information (JIT) is information that NCCAM requests if your application scored roughly within the top 15 percent. Though you may not get funded, you should prepare your just-in-time information anyway.
- Other support information
- Human subjects
- Assurance number.
- Certification of institutional review board approval of Research Plan.
- Certification of human subjects education.
- Animals
- Animal welfare assurance number.
- Certification of institutional animal care and use committee approval.
- Consortium and contractual agreements.
- Product Integrity Information
- When it's due. Your institution's business official should submit other support and human subjects training information within two weeks of receiving a just-in-time notice. You don't need to sign this information.
- Since institutional review board and institutional animal care and use committee certifications may take more than two weeks, your business official may submit these approvals at the earliest date possible.
- NIH prefers that your institution submit the documentation through the NIH Commons.
- Prepare your JIT information early but don’t send it until it is requested.
Notice of Grant Award
Overview of Terms and Conditions
- Includes those terms and conditions that apply, in general, to NIH awards
- These terms and conditions are not intended to be all-inclusive
- In addition to the requirements in the NIH Grants Policy Statement (NIHGPS), some of which repeat or highlight requirements found in the following:
- Authorizing program legislation
- Program regulations, including those in 42 CFR Part 52
- Other statutory requirements, such as those included in appropriations acts
- HHS requirements in 45 CFR Part 74 or 45 CFR Part 92, as appropriate for the type of recipient organization and the type of activity (e.g., research).
- Notice of requirements not specified in the NIHGPS generally will be provided in the NoA, but such notice is not required for the award to be subject to the requirements of pertinent statutes and regulations
- An individual award also may contain award-specific terms and conditions. For example, the GMO may include terms or conditions necessary to address concerns about an applicant's management systems
- Program and administrative policies and the terms and conditions of individual awards are intended to supplement, rather than substitute for, governing statutory and regulatory requirements
- If there is a perceived conflict between or among these categories of requirements—statutory and regulatory requirements, the terms and conditions in the NIHGPS, and award-specific terms and conditions—or if the grantee has other questions concerning award terms and conditions, the grantee should request written clarification from the GMO.
- If the inclusion of the term or condition would cause the grantee not to accept the award or to be unable to comply, the question should be raised before funds are requested from the HHS payment system.
- By drawing funds from the HHS payment system, the grantee agrees to the terms and conditions of the award.
Division of Extramural Activities (DEA)